Abstract
The kinetics and mechanism of the osmium(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III) in aqueous alkaline medium is studied. The role of the osmium(VIII) catalyst is delineated to account for the experimental observations. A plausible reaction mechanism is suggested. Activation parameters such as the energy and entropy of activation are evaluated by employing the Eyring equation and are found to be 36.833 kJ mol−1 and −141.518 J K−1 mol−1, respectively.
Introduction
The use of hexacyanoferrate(III) in acidic medium results in certain complications due to complexation of the oxidant by one of its reduced products, hexacyanoferrate(II). However, reactions in alkaline medium1–8 are not fast probably due to the reduced oxidation potential of the [Fe(CN)6]3−/[Fe(CN)6]4− redox couple. 9 It is for this reason that a large number of transition metal ions such as Os(VIII),10–13 Ru(III),14–17 Ru(VI), 18 Ru(IV), 19 Rh(III), 20 Ir(III), 21 Ru(VIII) 22 and Pd(II)23,24 have been employed as catalysts in alkaline medium. A large number of Os(VIII)-catalysed oxidations of both organic25–30 and inorganic31–33 compounds with hexacyanoferrate(III) in alkaline media have been reported.
Most of the kinetic studies on the oxidation of unsaturated alcohols have been made in the absence 34 and presence of heterogeneous catalysts.35,36 Also transition metal ions such as Ru(III), 37 Os(VIII), Pd(III) 38 and Ru(II) have been employed as homogeneous catalysts. There is an ambiguity regarding the oxidation product of these unsaturated alcohols with metal ion oxidants in solution.
Since fewer kinetics studies of the oxidation of unsaturated alcohols have been reported, the title reaction is one such reaction in which Os(VIII) has been employed as a catalyst to understand the reactivity pattern of unsaturated alcohols.
Results and discussion
Hexacyanoferrate(III) dependence
The concentration of hexacyanoferrate(III) was varied in the range 0.3–1.0 × 10−3 mol dm−3 keeping fixed concentrations of the other reagents at 45°C in the presence of [EDTA] = 1 × 10−2 mol dm−3. Pseudo first-order plots were made (Figure 1), and the evaluated first-order rate constants (k’, s−1) were found to be independent of the initial concentrations of the oxidant, thereby confirming the order with respect to the oxidant to be one (Table 1).
Pseudo first-order plots for the osmium(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III).
k(obs) and k(cal) rate constants of hydroxide ion variation in the osmium(VIII) catalysed oxidation of crotyl alcohol with potassium hexacyanoferrate(III) in alkaline aqueous medium.[HCF(III)] = 5 × 10−4 mol dm−3, [CrA] = 0.01 mol dm−3, [Os(VIII)] = 1 × 10−6 mol dm−3, [EDTA] = 0.01 mol dm−3.

Crotyl alcohol dependence
The concentration of crotyl alcohol was varied from 5× 10−3 to 5 × 10−2 mol dm−3 at fixed concentrations of other reagents, namely [HCF(III)] = 5 × 10−4 mol dm−3; [OH]− = 5 × 10−2 mol dm−3; [EDTA] = 1 × 10−2 mol dm−3 and [Os(VIII)] = 1 × 10−6 mol dm−3 at 45°C. First-order rate constants were calculated and a plot of the first-order rate constants versus [CrA] yielded a straight line passing through the origin (Figure 2), indicating the order to be one with respect to the alcohol (Table 1).

Variation of crotyl alcohol in the osmium(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III).
Osmium(VIII) dependence
The concentration of Os(VIII) was varied from 1× 10−6 to 1 × 10−5 mol dm−3 keeping constant concentrations of the other reagents, namely [HCF(III)] = 5 × 10−4 mol dm−3; [CrA] = 1 × 10−2 mol dm−3; [OH]- = 5 × 10−2 mol dm−3 and [EDTA] = 1 × 10−2 mol dm−3 at 45°C. A plot of the first-order rate constant versus [Os(VIII)] yielded a straight line passing through the origin (Figure 3), exhibiting first-order dependence with respect to the catalyst.

Variation of Os(VIII) in the Os(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III).
Effect of hydroxide ions
The effect of hydroxide ions was studied from 0.05 to 0.5 mol dm−3 at fixed concentrations of other reagents, namely [HCF(III)] = 5 × 10−4 mol dm−3; [CrA] = 1 × 10−2 mol dm−3; [EDTA] = 1 × 10−2 mol dm−3; [Os(VIII)] = 1 × 10−6 mol dm−3 and [I] = 0.5 mol dm−3 at 40, 45 and 50°C, respectively. The rate initially increases and then tends towards a limiting rate at higher hydroxide ion concentrations. Such a trend indicates a complex dependence on the hydroxide ion concentration.
Effect of ionic strength
The effect of the ionic strength was studied by employing sodium nitrate from 0.05 to 0.5 mol dm−3 at fixed concentrations of other reagents, namely [HCF(III)] = 5 × 10−4 mol dm−3; [CrA] = 1 × 10−2 mol dm−3; [OH]− = 5 × 10−2 mol dm−3; [EDTA] = 1 × 10−2 mol dm−3and [Os(VIII)] = 1 × 10−6 mol dm−3at 45°C. The rate increases with increasing ionic strength (Figure 4).

Variation of the ionic strength on the osmium(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III).
Effect of temperature
The effect of the temperature was studied at 30, 35, 40, 45, and 50°C, respectively (Figure 5), keeping constant concentrations of the other reagents at [I] = 0.5 M. The activation parameters such as energy and entropy of activation were evaluated, by employing Eyring plots, to be 36.833 kJ mol−1 and −141.518 J K−1 mol−1, respectively.

Variation of the temperature in the osmium(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III).
Discussion
The osmium(VIII)-catalysed oxidation of crotyl alcohol by HCF(III) in alkaline medium shows first-order dependence with respect to the oxidant, substrate and catalyst, respectively. However, the hydroxide ion dependence is complex. Since the reaction does not exhibit reproducibility of the kinetics, the addition of EDTA restores reproducible kinetics. It appears that trace metal ions interfere, which on complexation with EDTA are masked. Osmium tetraoxide in strong alkaline solution is red in colour, which is assigned 39 to be [OsO4(OH)2]2−. This species converts into [OsO3(OH)3]− on dilution, in agreement with earlier results reported by Sauerbrum and Sandell. 40
Considering all such experimental observations, a reaction mechanism consisting of steps, equations (1)–(7) can be envisaged as follows


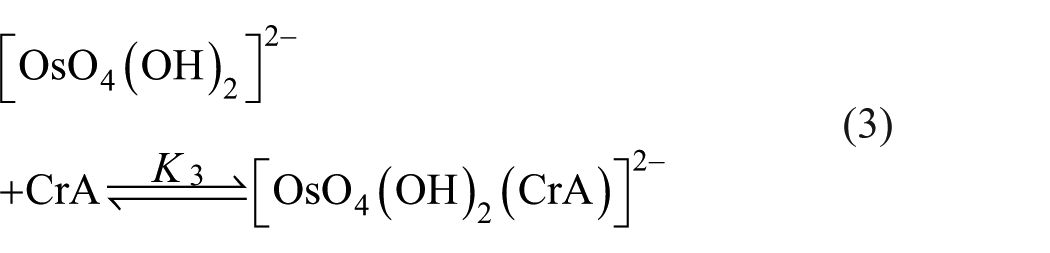




Such a mechanism leads to the rate law (8)

Since the order with respect to crotyl alcohol has been observed to be one, neither kinetic nor spectral evidence for complex formation between the oxidant and the substrate was found, even for longer ranges of substrate concentrations. Therefore, inequalities 1 >>K3 [CrA] and 1 >>K2 (CrA) are valid and reduce the rate law (8) to (9)

With further rate increases with increasing hydroxide ion concentration, the catalyst species OsO4(OH)22− is more reactive than [OsO3(OH)3]−, the term k1K3 can be further neglected as compared with k2K1K2, reducing the rate law (9) to (10) or (11).

where [Fe(CN)6]3−, [CrA] and [Os(VIII)] are the gross analytical concentrations of HCF(III), crotyl alcohol and osmium(VIII), respectively. [OH−] is the free equilibrium concentration of hydroxide ions

where ‘k’ is observed third order rate constant.
Taking the double reciprocal of equation (11), equation (12) is obtained

A plot of 1/k versus 1/[OH−] was made from equation (12) that yielded a straight line with a non-zero intercept (Figure 6). ‘K1’ was evaluated from the ratio of the intercept and slope to be 12.1 ± 0.05, 15.1 ± 0.05 and 17.5 ± 0.1 dm3 mol−1 at 40, 45 and 50°C, respectively, and I = 0.5 mol dm−3. k2K2 was evaluated from the intercept to be 10.1 ± 1.0 × 108, 11.6 ± 1.0 × 108 and 12.9 ± 2.0 × 108 dm6 mol−2s−1 at 40, 45 and 50°C, respectively, and I = 0.5 mol dm−3. These values of K1 and k2K2 were further substituted in rate equation (11), with the values of k(obs) and k(cal) being in close agreement despite the system being complex in nature.

A plot of (k)− versus [OH−] in the osmium(VIII)-catalysed oxidation of crotyl alcohol by hexacyanoferrate(III).
The assumption made earlier as to the negligible contribution of the first term in the numerator of rate law (11) is justified in light of the agreement between these calculated and observed values.
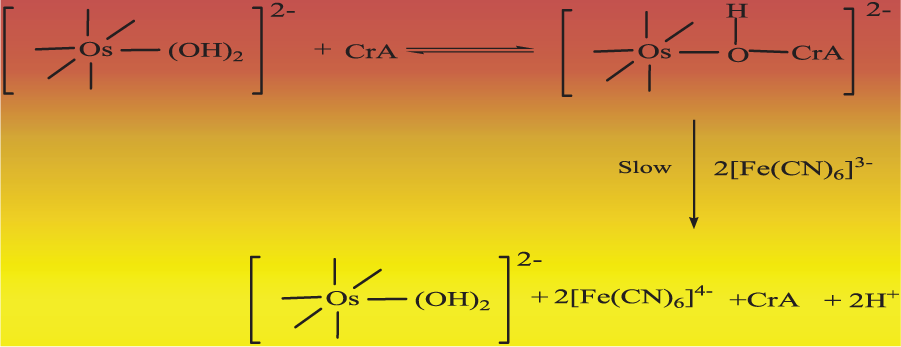
As far as the mode of electron transfer from the substrate to the oxidant is concerned, Scheme 1 adequately accounts for the reaction events.
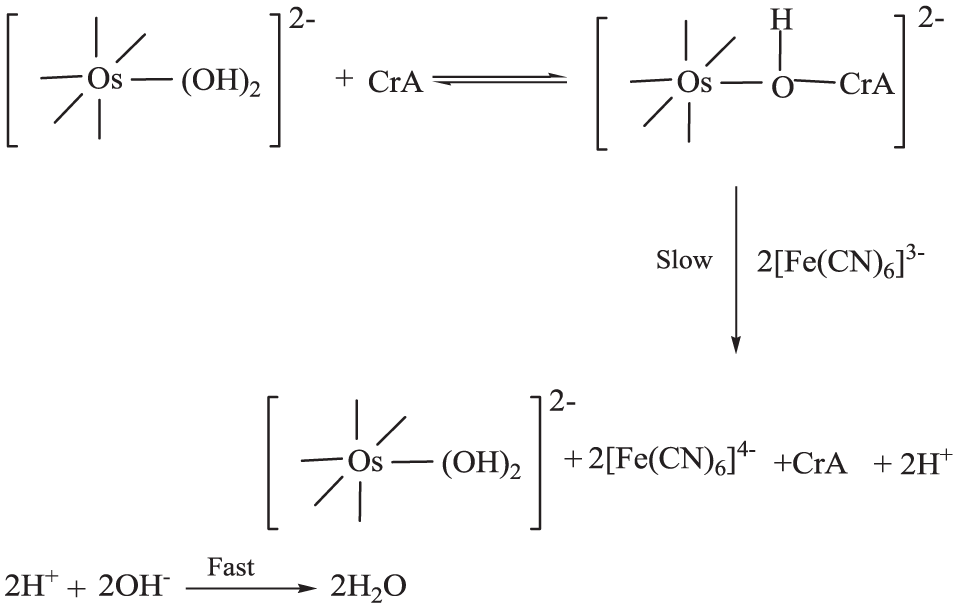
Reaction Events in the reaction between Crotyle Alcohol and Hexacyanoferrate(III).
Neither kinetic nor spectral evidence was obtained for complex formation between osmium(VIII) and the substrate. However, the broad λmax value observed for the osmium(VIII) solution was slightly affected by the addition of crotyl alcohol, which cannot be taken to be conclusive evidence for complexation. Nevertheless, transfer of an electron from the substrate to the catalyst is energetically, via complex formation, a more facile pathway than a direct encounter. Further hexacyanoferrate(III) is inert to substitution, which is further negated by the fact that the rate is not affected by hexacyanoferrate(II). Therefore, any probability of forming a complex with the oxidant is completely ruled out.
Conclusion
It is worth mentioning that the reaction has been carried out in the presence of EDTA, as the reaction is non-stoichiometric with irreproducible kinetic results. Trace metal ions are known to catalyse a large number of oxidation reactions by hexacyanoferrate(III). Probably, the presence of EDTA masked such trace metal ions and the reproducibility was returned to the reaction. Since EDTA also forms a complex with osmium(VIII), this is probably the reason that complexation between osmium(VIII) and the alcohol was efficiently checked by blocking of the co-ordination sites of the metal centre. Had this not been the situation, crotyl alcohol would have kinetically shown a complex rate dependence instead of an observed simple first-order dependence.
Furthermore, the proposed reaction mechanism is also justified in view of the effect of ionic strength on the rate of the reaction. The activation parameters such as the entropy of activation being more negative also supports rate-controlling steps in the reaction mechanism due to loss of certain degrees of vibrational modes. The energy of activation is closer to the value usually observed for a bimolecular reaction.
Experimental
Materials and method
Hexacyanoferrate(III) (E Merck) was employed as received without any further treatment and its aqueous solution was prepared by dissolving requisite amount in doubly distilled water. Osmium tetraoxide (Johnson Matthey) was employed as received for the preparation of its solution in 0.3 mol dm−3NaOH. If the concentration of NaOH is less than 0.3 mol dm−3, the solution of Os(VIII) is not stable. However, it is quite stable 41 at [NaOH] > 0.3 mol dm−3and its stability is further enhanced if it is stored in brown-coloured bottles at refrigerator temperature 42 (~5°C). The solution of osmium(VIII) was standardized iodometrically.43,44 Other reagents were of analytical grade quality and were used as supplied. Crotyl alcohol was distilled under reduced pressure and was kept in glass vessels (painted black from the outside) to prevent decomposition by light. Doubly distilled water was employed throughout the study; the second distillation was from alkaline permanganate solution in an all glass apparatus.
Kinetic procedure
The reactions were carried out using all reagents, except hexacyanoferrate(III), in glass-stoppered and black-coated Erlenmeyer flask. These flasks were suspended in a water bath at ± 0.1°C unless specified otherwise. The requisite volume of hexacyanoferrate(III) solution was then transferred to the reaction mixture via pipette. The kinetics were monitored periodically by withdrawing an aliquot (5 cm3) from the reaction mixture and quenching it in ice-cold water. The absorbance 45 of hexacyanoferrate(III) was measured at 420 nm (ε = 1020 mol dm−3 cm−1) by employing a UV-visible spectrophotometer. The absorbance was converted into concentration by taking into account the dilution. The kinetics were also studied under pseudo-first-order conditions with a sufficiently large excess concentration of crotyl alcohol (CrA) over that of hexacyanoferrate(III) [HCF (III)]. Pseudo-first-order plots were made and the first-order rate constants were calculated. The rates in triplicate were reproducible to within ±5%.
Stoichiometry and product analysis
The stoichiometry of the reaction was determined by taking excess [HCF(III)] with respect to that of [CrA] keeping [OH−] at 0.05 mol dm−3, [EDTA] = 0.01 mol dm−3 and [Os(VIII)] = 1 × 10−6 mol dm−3, allowing them to react in a thermostated water bath at 45°C ± 0.1°C for ca. 6 h. The excess hexacyanoferrate(III), after completion of the reaction, was determined spectrophotometrically. These stoichiometric results correspond to the reaction of two moles of hexacyanoferrate(III) with one mole of crotyl alcohol as represented by equation (13).
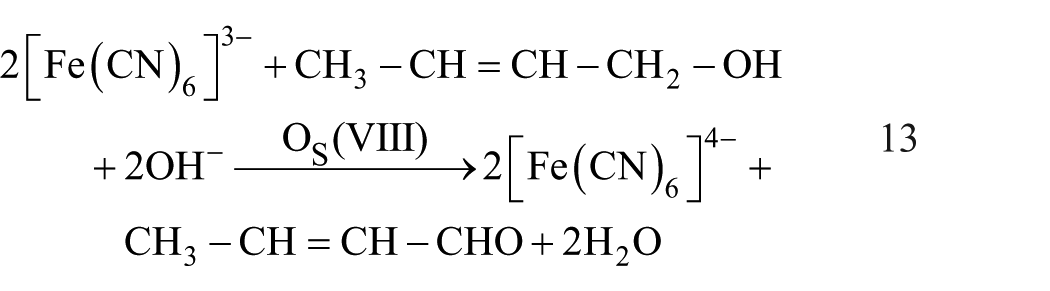
The oxidation product of the alcohol, crotonaldehyde was tested positive using Tollen’s reagent. The product was also identified and established spectroscopically by preparing the 2,4-dinitrophenyl hydrazone derivative of the oxidation product.
Footnotes
Acknowledgements
Acknowledgement is made to the co-authors and Head of Department of Chemistry, UOR, Jaipur, Rajasthan, India. Authors also want to dedicate this work to Prof. (Late) P. D. Sharma, Department of Chemistry, UOR, Jaipur, Rajasthan, India.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported in part by the University Grant Commission, New Delhi, India through a start-up grant (BSR).
