Abstract
A series of nickel complexes containing bispyridylamine ligands is synthesized and characterized by UV-Vis, FTIR, 1H NMR, ESI-MS, and elemental analysis. All nickel complexes activated with methylaluminoxane exhibited moderate activity for ethylene oligomerization (5.30 × 104 to 2.54 × 105 g/(mol·Ni·h)), producing oligomers in the range C4–C18. The effects of reaction temperature, reaction pressure, Al/Ni molar ratio, and solvent type on the catalytic activity and product selectivity are also investigated. The resulting products are mainly low-carbon olefins (C4 and C6). Under the optimized conditions, the complex with R1 = C4H9 formed by the reaction of the corresponding ligand R1 = C4H9 with NiCl2·6H2O showed a higher catalytic activity of 2.54 × 105 g/(mol·Ni·h) compared to the complex with R2 = C6H13 and R3 = C6H11 using toluene as the solvent. Moreover, the selectivity for the C4 and C6 oligomers can reach up to 82%.
Keywords

Introduction
Linear α-olefins (LAOs) are important commodity chemicals with a variety of applications in industrial processes, ranging from co-monomers for the production of polyolefins to intermediates for synthetic lubricants, plasticizers, and surfactants.1–4 1-Butene is used as a co-monomer in the production of high-density polyethylene (HDPE) and linear low-density polyethylene (LLDPE). Therefore, the production of 1-butene directly influences the market for both HDPE and LLDPE. Several manufactures such as Shell, Idemitsu, and Chevron Phillips Chemical Company use metal-catalyzed multi-step ethylene oligomerization processes to produce LAOs. Most of these processes generate a wide distribution of LAO carbon chain lengths from C4 (1-butene) to C20 (1-eicosene).5,6 In this context, high-carbon olefins (C8–C12) are also particularly valuable, imparting polymers with good tear resistance and other desirable properties. 7 For certain applications, high purity of α-olefins is essential because internal olefins are unreactive and thus build up during co-polymerizations. Preventing formation of unwanted polymeric materials is a major task, influencing the profitability of unselective or selective catalytic systems designed to produce LAOs. 8
Numerous studies on ethylene polymerizations using nickel catalysts have established that the branching density and distribution strongly depend on the nature of the ligand.9,10 An effective approach to controlling the branching distribution of polyethylene products is the modification of ligand frameworks. For example, α-diamine nickel catalysts with modified ortho-substituents afforded methyl-branched polyethylene by controlling the chain propagation reaction. 11 In addition, hyperbranched macromolecules are well-defined, highly branched, three-dimensional macromolecules with characteristic globular structures, 12 imparting the catalyst with good solubility, low viscosity, and high chemical reactivity. In this article, a series of novel hyperbranched nickel complexes with different alkyl groups has been designed for the preparation of LAOs via ethylene oligomerization.13,14 According to studies, the P and N donors have a strong potential to facilitate the selective ethylene oligomerization.15–20 At the same time, we investigated the effects of the reaction conditions and the solvent on ethylene oligomerization.
Results and discussion
Synthesis and characterization of the hyperbranched bispyridylamine ligands and nickel complexes
The hyperbranched bispyridylamine ligands

Synthetic routes of the bispyridylamine ligands and nickel complexes.
The reactions of NiCl2·6H2O with ligands
Evaluation of the nickel complexes as catalysts for ethylene oligomerization reaction
These complexes
Effects of solvent and ligand type on the catalytic activity and the product selectivity
Considering the effect of the solvent on the catalytic activity and product selectivity, 22 the catalytic systems were studied with toluene, methylcyclohexane, and cyclohexane and the results are listed in Table 1. The highest activity was observed in toluene. This may be due to the greater polarity of toluene and the poorer solubility of the nickel complexes in methylcyclohexane and cyclohexane. However, the oligomers were mainly low-carbon olefins in this solvent and we chose the toluene as the best solvent.
Effects of the solvent and ligand type on the catalytic activity and the product selectivity. a

Reaction conditions: catalyst (5 µmol), reaction temperature (55°C), reaction pressure (4 MPa), Al/Ni (700), solvent (total volume: 50 mL), and reaction time (30 min).
From Table 1, we can also see that the ligand type had a great influence on the catalytic activity and product selectivity. Changes in the ligand structure by substituting the butylamine with N-hexylamine and cyclohexylamine led to a decrease of activity (
Effects of the reaction temperature on the catalytic activity and the product selectivity
The effect of the reaction temperature on the catalytic activity and the product selectivity is shown in Figure 1. The results of the oligomerization performed at temperatures between 25°C and 65°C are considered first. On elevating the reaction temperature from 25°C to 65°C, the catalytic activity increases initially and then decreases. The catalytic activity reaches a maximum (2.54 × 105 g/(mol·Ni·h)) at 55°C, but a higher temperature leads to a decrease in the catalytic activity because of the decreasing ethylene solubility in toluene and the catalytic species decomposing at a higher temperature.24–26 Different reaction temperatures also have an effect on the product distribution. Increasing the reaction temperature from 25°C to 65°C results in a gradual increase in C4 and C6 oligomers at the expense of high-carbon olefins. This shows that the rate of chain transfer increases more than the rate of chain propagation.
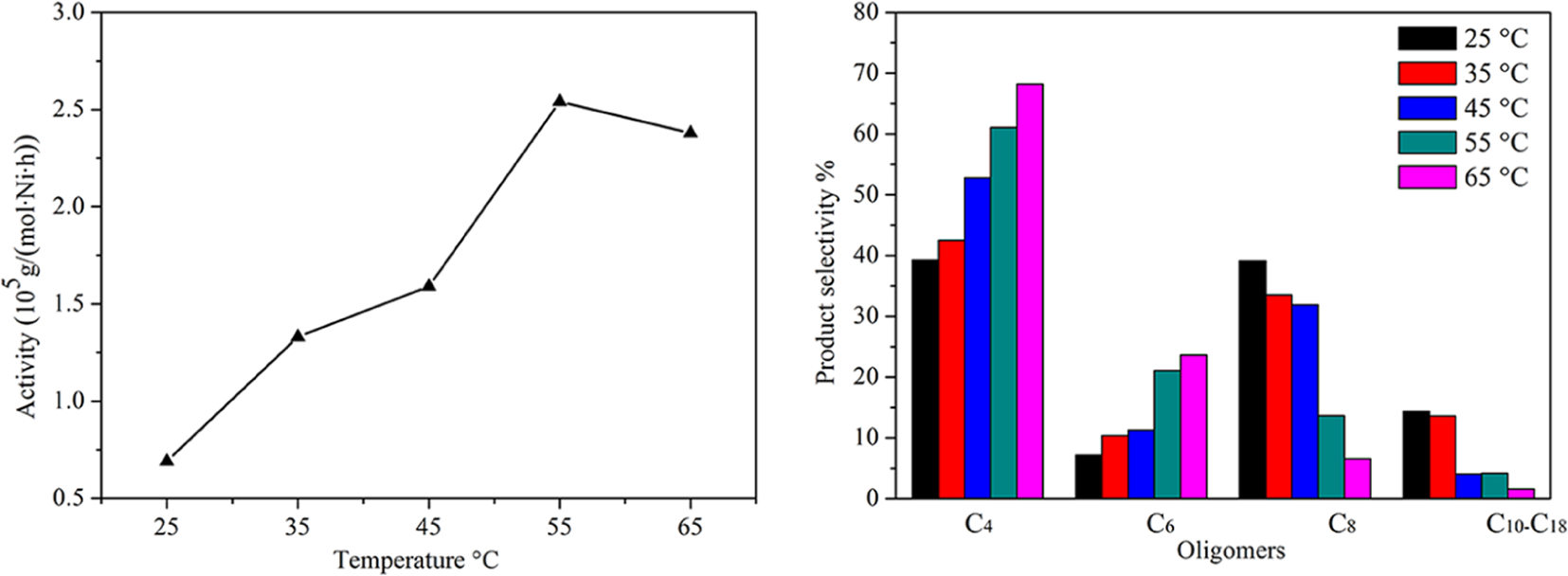
Effects of the reaction temperature on the catalytic activity and the product selectivity. Reaction conditions: catalyst (5 μmol), reaction pressure (4 MPa), Al/Ni (700), toluene (50 mL), and reaction time (30 min).
Effects of the reaction pressure on the catalytic activity and the product selectivity
The effect of reaction pressure on the catalytic activity and the product selectivity is shown in Figure 2. From Figure 2, it can be seen that the reaction pressure played a vital role in this catalytic system. When the reaction pressure increased from 1 to 5 MPa, the catalytic activity increases initially and then decreases. The catalytic activity reaches up a maximum (2.54 × 105 g/(mol·Ni·h)) at 4 MPa. This may be due to elevated ethylene pressure resulting in an increase in the ethylene concentration in the solvent, leading to an increase of activity. However, the excess of ethylene prevents the reaction between Ni(II) and MAO which is responsible for formation of the active species. In other words, ethylene acts as a catalyst poison. Moreover, the rate of ethylene diffusion into the active center is slowed down. A combination of the above two factors can account for the observed reaction pressure dependence of the catalytic activity. In addition, the reaction pressure has a significant effect on the product selectivity. The selectivity for high-carbon olefins increases initially and generally decreases. The resulting oligomers are mainly low-carbon olefins.

Effects of the reaction pressure on the catalytic activity and product selectivity. Reaction conditions: Catalyst (5 μmol), reaction temperature (55°C), Al/Ni (700), toluene (50 mL), and reaction time (30 min).
Effects of the Al/Ni molar ratio on the catalytic activity and the product selectivity
The effect of the Al/Ni molar ratio on the catalytic activity and the product selectivity is shown in Figure 3, with the Al/Ni molar ratio having an important impact on the catalytic performance. As the Al/Ni molar ratio increases from 100 to 900, the catalytic activity increases above and then decreases. The highest activity is 2.54 × 105 g/(mol·Ni·h) at a molar ratio of 700. Meanwhile, the selectivity for C4 and C6 was maximized. The increasing Al/Ni molar ratio led to more active sites and hence an increase in the catalytic activity. In contrast, further increases in the MAO concentration can also interfere with the formation of the over-reduced nickel species leading to deactivation of the active species and a decrease in the activity. 27 Furthermore, the oligomer distributions are not obviously changed on increasing the Al/Ni molar ratio, which may due to the non-linear relationship between the ratio of chain transfer and chain propagation.

Effects of Al/Ni molar ratio on catalytic activity and product selectivity. Reaction conditions: Catalyst (5 μmol), reaction temperature (55°C), reaction pressure (4 MPa), toluene (50 mL), and reaction time (30 min).
Conclusion
A series of novel types of bidentate bispyridylamine ligands
Experimental
Materials and instrumentation
All manipulations involving air- and water-sensitive compounds were carried out under an inert atmosphere in a glovebox or by using standard Schlenk techniques. Solvents were dried from the appropriate drying agents under nitrogen before use. Butylamine, N-hexylamine, cyclohexylamine, NiCl2·6H2O and 2-chloropyridine were purchased from Aladdin and used as received. Methyl acrylate, anhydrous ethylenediamine, triethylamine (Et3N) and toluene were obtained from Tianjin Damao Chemical Reagent. Methanol and THF were provided from Tianjin Kermel Chemical Reagent. MAO (10 wt% in toluene) was bought from Sigma-Aldrich. Polymerization grade ethylene was obtained from Daqing Summit Specially Gases (China). The 1.0 G hyperbranched macromolecules were synthesized according to the literature procedures described in our earlier study. 28
UV-Vis spectra were obtained using on a TU-1901 UV-Vis spectrophotometer with methanol as the solvent. FTIR spectroscopy was performed on a Bruker Vector 22 spectrophotometer from 4000 to 450 cm−1. The ESI-MS was obtained using a micrOTOF-QⅡ mass spectrometer. 1H NMR measurements were obtained on a Bruker 400 MHz NMR in CDCl3 solution. The 1H NMR spectra of all the compounds were recorded at ambient temperature. Gas chromatography (GC) analysis of the oligomers was conducted on a Fuli GC 9720 instrument equipped with an flame ionization detector (FID) and a 50 m (0.2 mm id, 0.5 μm film thickness) HP-PONA column, operating at 50°C for 5 min followed by heating at 10°C min−1 until 140°C, then heating at 5°C min−1 until 240°C, and then for 5 min, injecting the sample.
Synthesis of the hyperbranched bispyridylamine ligands
Synthesis of the hyperbranched bispyridylamine ligand with butylamine as the core, 3a
3,3′-(butylazanediyl)bis(N-(2-(di(pyridin-2-yl)amino)ethyl)propanamide)
Synthesis of the hyperbranched bispyridylamine ligand with N-hexylamine as the core, 3b
3, 3′-(hexylazanediyl)bis(N-(2-(di(pyridin-2-yl)amino)ethyl)propanamide)
Synthesis of the hyperbranched bispyridylamine ligand with cyclohexylamine as the core, 3c
3, 3′-(cyclohexylazanediyl)bis(N-(2-(di(pyridin-2-yl)amino)ethyl)propanamide)
Synthesis of the hyperbranched bispyridylamine nickel complexes
Synthesis of the hyperbranched bispyridylamine nickel complex with butylamine as the core, 4a
3, 3′-(butylazanediyl)bis(N-(2-(di(pyridin-2-yl)amino)ethyl)propanamide) nickel chloride
Synthesis of the hyperbranched bispyridylamine nickel complex with N-hexylamine as the core, 4b
3, 3′-(hexylazanediyl)bis(N-(2-(di(pyridin-2-yl)amino)ethyl)propanamide) nickel chloride
Synthesis of the hyperbranched bispyridylamine nickel complex with cyclohexylamine as the core, 4c
3, 3′-(cyclohexylazanediyl)bis(N-(2-(di(pyridin-2-yl)amino)ethyl)propanamide) nickel chloride
Ethylene oligomerization
All ethylene oligomerization tests were performed in a 250 mL stainless steel reactor equipped with mechanical stirring, internal temperature control, and a continuous feed of ethylene. The stainless steel reactor was dried in an oven at 140°C for 2 h prior to each run and then placed under vacuum for 30 min. The reactor was evacuated and flushed with N2 (three times) and ethylene (twice). A typical reaction was performed by introducing the solvent (30 mL), the appropriate amount of co-catalyst, and the catalyst precursor solution under a stream of ethylene. Then, the reactor was immediately pressurized. Ethylene was continuously fed in order to maintain the desired ethylene pressure. After 30 min, the reactor was cooled to 0°C and the reaction was quenched with 10 wt% HCl in ethanol. The distribution of the oligomerization product was analyzed by GC and compared with the standard authentic samples. The catalytic activity was calculated from the increase in product weight.
Supplemental Material
Supplement_Figure – Supplemental material for Nickel complexes based on hyperbranched bispyridylamine ligands as catalyst precursors for ethylene oligomerization
Supplemental material, Supplement_Figure for Nickel complexes based on hyperbranched bispyridylamine ligands as catalyst precursors for ethylene oligomerization by Jun Wang, Jinyi Liu, Liduo Chen, Tianyu Lan and Libo Wang in Journal of Chemical Research
Footnotes
Acknowledgements
The authors would like to thank the National Key R&D Program of China (No. 2017YFB0306701).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
