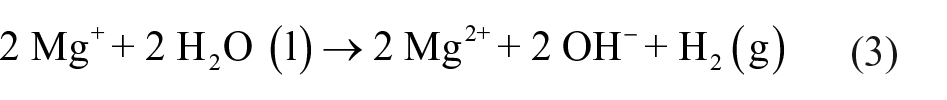Abstract
Calcium phosphate coatings are used in orthopedics due to their excellent bioactivity, which improves the bonding between the metal implant and the bone. The use of self-assembling monolayers of long-chain organic acids can induce calcium phosphate growth. In this article, the self-assembling monolayers of stearic acid and octadecylphosphonic acid formed on the Mg alloy surface were additionally modified with electrodeposited hydroxyapatite coating to increase the bioactivity and biocompatibility of the Mg alloy in a physiological solution. Hydroxyapatite coating was prepared by a two-step reaction: hydrogen phosphate formed by electrodeposition at constant potential was converted into hydroxyapatite coating through an acid–base reaction. The results obtained by voltammetry and electrochemical impedance spectroscopy have shown a beneficial effect of organic acid self-assembling monolayer and especially of organic acid self-assembling monolayer modification by hydroxyapatite electrodeposition on the corrosion properties of Mg alloy in physiological solution. Fourier transform infrared spectroscopy and scanning electron microscopy were used to verify the existence of the organic acid SAM|HAp film on the Mg alloy surface and their morphology.
Keywords

Introduction
As the potential metallic materials for degradable implants, magnesium (Mg) and its alloys are recognized due to their eligible mechanical properties, minimal toxic responses, degradability, and good biocompatibility. 1 Compared with titanium, magnesium-based biomaterials are sensitive and not inert, and they integrate bioactivity and biodegradability and can foster several cellular responses at the molecular level, for example, dissolved Mg ions may promote bone cell attachment, tissue growth at the implants, and so can be applied in surgical as well as in cardiovascular treatments. 2
Magnesium and its alloys are not without their disadvantages, such as high strength-to-weight ratios and poor corrosion resistance, that lead to fast biodegradation at physiological pH (7.2–7.4) or in physiological media containing high concentrations of aggressive chloride ions (~104 mmol L−1) 3 and ultimately affect the sustainability of increased magnesium use. Also, degradation cavities as a consequence of the gas produced during biodegradation, even in pure magnesium, lead to losses of mechanical integrity before tissues have sufficient time to heal.4,5 In the last decade, there have been several critical reviews focusing on magnesium materials: their applications as biodegradable materials, 6 overviews of in vitro/in vivo investigations and brief histories, 7 for medical/orthopedic applications, 8 and all point out the need and methods of improving their properties. As a replacement for orthopedic biomaterials to the formation of new bone, or for uses in cardiovascular treatments as stents, it is necessary to have materials resistant to corrosion (safely degradable) for several months. The present researched magnesium alloy exhibits a too fast corrosion rate and further improvement of corrosion resistance is strongly needed. Therefore, to improve the corrosion resistance of metallic magnesium or its alloys, different suitable pretreatment methods can be applied.9–11 However, besides the improved lifetime of implants, controlling the corrosion rate of alloys in simulated body fluid plays an important role because the concentration of degradation products should be within the body’s acceptable absorption levels.12,13
According to some authors, calcium phosphate formed on Mg materials14–16 is a very promising way to prevent fast corrosion and premature degradation. Hydroxyapatite (HAp) is particularly interesting due to its similarity to biological apatite, in both crystallographic structure and Ca/P, 17 and excellent bioactivity, good biocompatibility, osteoconductivity, all of which ultimately improve binding of metal implants and bone. 18
A wide variety of coating methods for HAp and materials are available for different coating applications with a common purpose. Electrodeposition17,19,20 has received much attention due to its universality, controlled chemical composition, simple setup, ability to control the thickness and chemical composition of HAp by varying the conditions employed for deposition and cost-effectiveness, compared to other methods with higher operation costs, such as sputtering, 21 incorporation of Mg ions in HAp by sol–gel and hydrothermal methods, 22 plasma spraying, 23 pulsed-laser melting, 24 electrophoresis. 25
Electrodeposition as a method for forming nano-particulate HAp on a substrate is very simple, but according to the literature it is insufficiently explored. The possibility to control size, morphology, adherence, and crystal growth orientation of the HAp is very important in determining the physicochemical properties of the material for biomedical applications. The properties of HAp formed by electrodeposition are pronouncedly influenced by many parameters, such as surface pretreatment (mechanical/chemical/electrochemical), bath composition, oxygen level, pH, stirring, the applied potential (or current density), temperature, and cell geometry. 17
In this sense, it is essential to have well-defined and reproducible surfaces which are relatively easy to form by self-assembled monolayers (SAMs) of biomolecules like alkylcarboxylic acids26,27 and alkylphosphonic acids.27–29 It has been shown that surfaces modified by SAMs bearing various functional end groups have a significant effect on the chemical precipitation and nucleation of CaP.30–33 Some functional groups (
In this study, HAp coating formation was studied on well-defined surfaces using organic SAMs (stearic acid (SA) or octadecylphosphonic acid (ODPA)) on Mg alloy (Mg93Al6Zn1 alloy). The Ca/P coating was prepared by electrodeposition from a solution containing Ca2+ and
Results and discussion
Polarization measurements
Anodic potentiodynamic polarization curves for unmodified and modified Mg alloy were recorded at a scan rate of 1 mV s−1 after an immersion period of 1 h in order to examine the influence of Mg alloy surface modification with HAp film formed on the SA SAM or ODPA SAM on the potential–current (E/j) profiles. The influence of the surface treatment on the anodic current densities is illustrated in Figure 1, which presents anodic potentiodynamic curves recorded with the unmodified and modified Mg alloy specimens. It can be observed that a much higher current density was recorded with the unmodified Mg alloy in comparison with modified Mg alloy specimens, that is, both surface treatments significantly decrease the anodic current densities and the start of anodic dissolution shifted to more anodic potential values (shift of 0.2 V to a more noble potential) in comparison to the unmodified sample. The anodic current density values registered at −1.37 V on Mg alloy|Anodized|SA|HAp and Mg alloy|TT|ODPA|HAp samples were equal to 0.19 and 0.02 mA cm−2, respectively, and illustrate the influence of different surface treatments on the polarization behavior, because for the Mg alloy|TT|ODPA|HAp sample a rapid increase of the current density is observed at less noble anodic potential values. For modified samples, a steep increase in current points to an accelerated degradation/desorption of the surface film under the influence of anodic polarization, appearing on higher anode potentials compared to an unmodified surface. The anodic portion of the polarization curves prior to the active metal dissolution corresponds to magnesium oxide hydroxide (MgO/Mg(OH)2) on the electrode surface. The current density increases with increasing potential due to magnesium dissolution, and chloride ions can transform MgO/Mg(OH)2 into more soluble magnesium chloride (MgCl2).37,38 Thus, the presence of the surface film on the electrode surfaces significantly suppresses the anodic dissolution of both samples investigated.
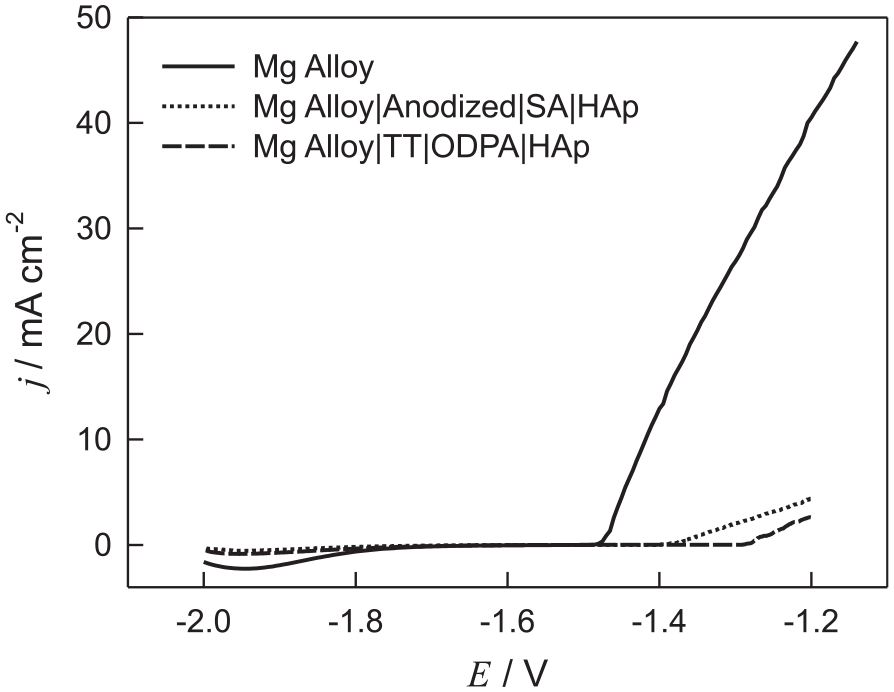
The potentiodynamic polarization curves for (. . . .) unmodified Mg alloy, (─) anodized Mg alloy, the SA SAM of which was additionally modified with HAp, and (—) thermally treated Mg alloy, the ODPA SAM of which was additionally modified with HAp recorded in Hanks’ solution at 37°C, v = 1 mV s−1.
Magnesium corrosion in aqueous electrolyte can be described by the following reactions
The electrochemical hydrogen evolution reaction, that is, the cathodic reaction, is shown in equation (1). The reaction of magnesium dissolution, that is, the anode reaction, is shown in equation (2). Equation (3) represents the evolution of hydrogen in the reaction of magnesium and water. In the total reaction (equation (4)), H+ ions are consumed and OH− ions are formed, that is, the pH increases which results in magnesium hydroxide film formation by a deposition reaction (equation (5)).38,39
Linear polarization curves in Tafel representation recorded with unmodified and modified Mg alloy electrodes are shown in Figure 2. The measurements were performed in Hanks’ solution and obtained after 60 min of stabilization at open-circuit potential, starting from −2.0 V versus Ag/AgCl/3M KCl in anodic direction at a sweep rate of 1 mV s−1. The presence of organic acid SAMs modified with HAp on the Mg alloy surface reduces the values of the cathodic and anodic current density and the corrosion potential is shifted in an anodic direction. Anodic current densities of the modified Mg alloys are characterized by a gradual increase in current density to the film breakdown potential, indicating that the surface layer suppresses to a certain extent the reaction of the anodic oxidation.26,40 In anode branch, the film breakdown potential of the Mg alloy, the carboxylate SAM of which was further modified with HAp film, is −1.4 V, whereas that of the Mg alloy electrode, the phosphonate SAM of which was further modified with HAp film, is −1.3 V. In the cathodic branch, a linear Tafel region with slope values ranging from −157 to −189 mV decade−1 is noticeable, indicating that the hydrogen reduction reaction takes place on the electrodes covered with a surface film.26,28,41,42
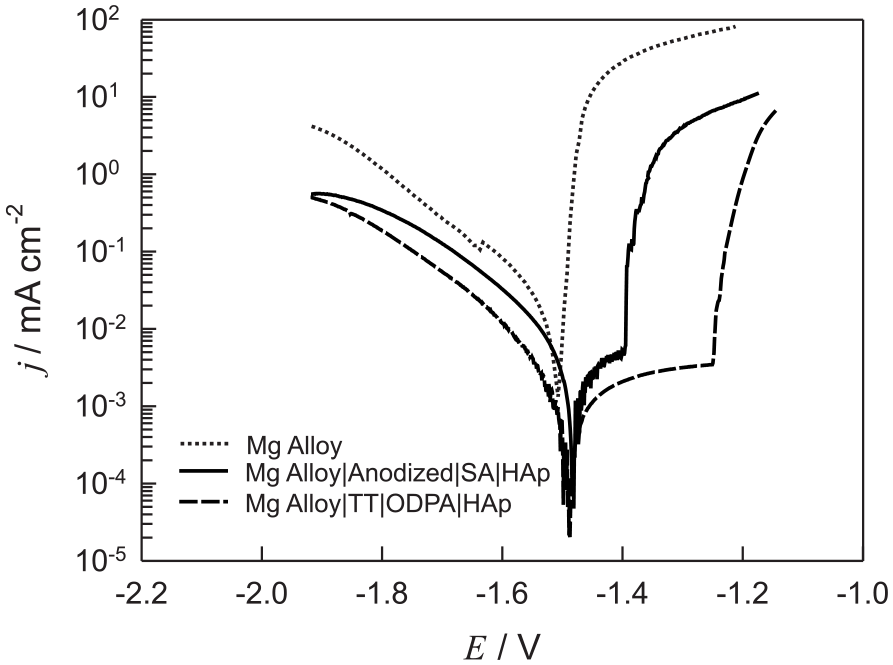
The Tafel plots for (. . . .) unmodified Mg alloy, (─) anodized Mg alloy, the SA SAM of which was additionally modified with HAp, and (—) thermally treated Mg alloy, the ODPA SAM of which was additionally modified with HAp recorded in Hanks’ solution at 37°C, v = 1 mV s−1.
The values of the cathodic and anodic Tafel slope, corrosion current density, and corrosion potential were determined by the Tafel extrapolation method. The obtained values are listed in Table 1 together with the values for the coverage of the Mg alloy surface with organic acid SAM additionally modified with HAp film. The coverage is calculated according to the expression
where jcorr. and jcorr.SAM|HAp are the corrosion current density values of unmodified Mg alloy and Mg alloy coated with organic acid SAM modified with HAp film, respectively. The numerical values of the corrosion current densities for Mg alloy specimens are 39.2 μA cm−2 for unmodified Mg alloy, 8.8 μA cm−2 for Mg alloy|Anodized|SA|HAp, and 3.4 μA cm−2 for Mg alloy|TT|ODPA|HAp.
The corrosion kinetic parameters for unmodified Mg alloy and modified Mg alloy recorded in the Hanks’ solution, pH = 6.67 at 37°C, v = 1 mV s−1.

SA: stearic acid; ODPA: octadecylphosphonic acid; HAp: hydroxyapatite.
Electrochemical impedance behavior
The protective behavior against corrosion in Hanks’ solution of the HAp film formed on the surface of Mg alloy modified with organic acid SAM was also tested by EIS measurements. The impedance spectra of the Mg alloy whose potentiostatically formed oxide film were modified with SA SAM, as shown in Figure 3.
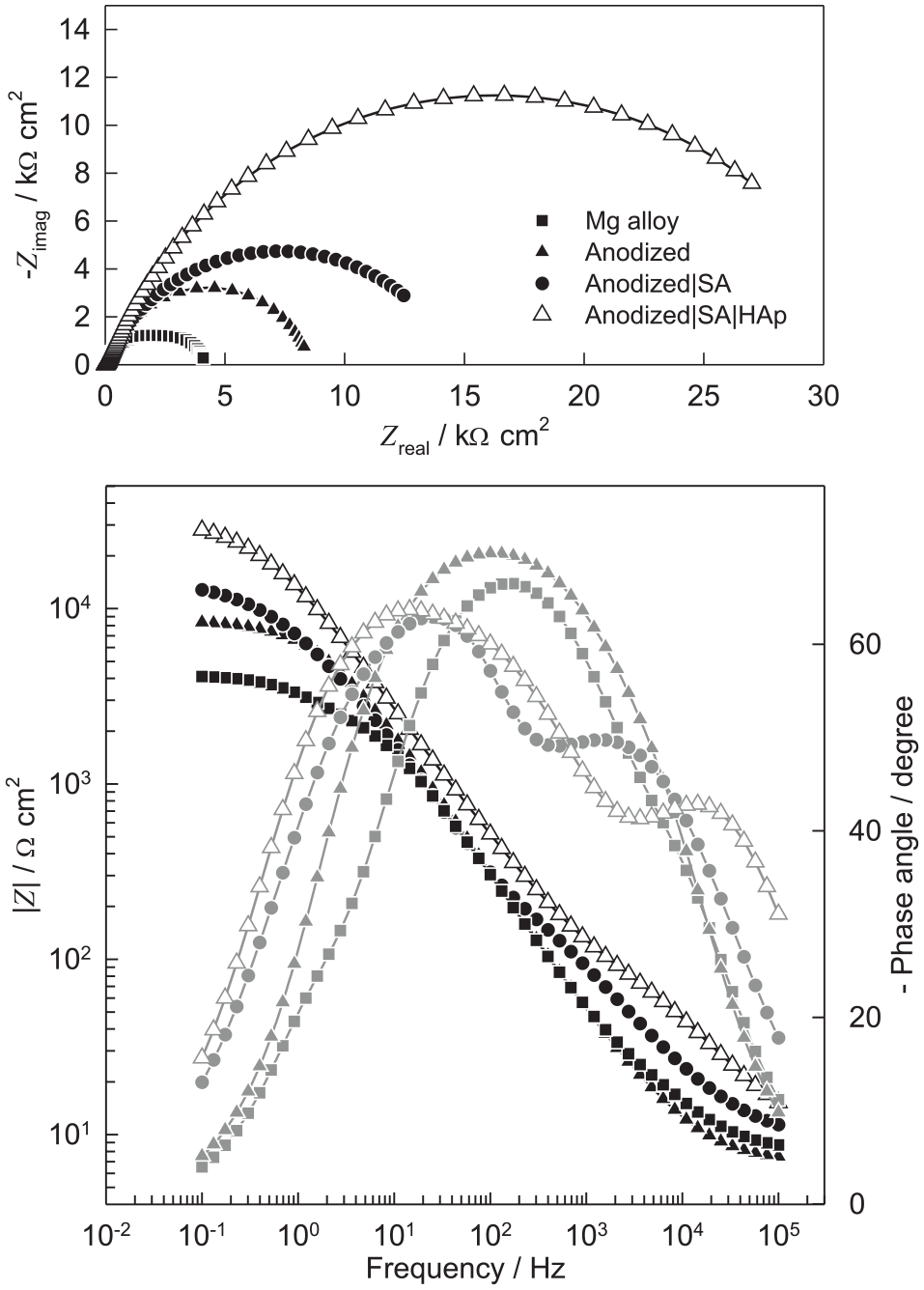
Nyquist and Bode plots of impedance spectra for unmodified Mg alloy, anodized Mg alloy, anodized Mg alloy coated with SA SAM, and anodized Mg alloy whose SA SAM was additionally modified with HAp recorded in Hanks’ solution at 37°C at open-circuit potential. Carboxylate SAM was deposited from the SA alcoholic solutions of 1.0 mmol dm−3 concentration and self-assembling time was 24 h. Electrodeposition was performed at a potential of −2.0 V for 3 h at room temperature.
Figure 3 also shows the Nyquist and Bode plots of the impedance spectra of the Mg alloy whose SA SAM was additionally modified by HAp film. For comparison, the impedance spectra of the electrode with the natural oxide film (unmodified Mg alloy) and the impedance spectra of the electrode with the potentiostatically formed oxide film are also shown. For the electrode of the unmodified Mg alloy in the Nyquist plot, a flattened capacitive semicircle with a center below the real axis is noticeable. In the Nyquist plot, it should also be noted that the SA SAM causes an increase of the capacitive semicircle diameter, that is, the polarization resistance of the electrode modified with potentiostatically formed oxide film increases. The polarization resistance (Rp), which represents corrosion resistance, is equal to the sum of resistance R1 and R2, and for the electrode of the unmodified Mg alloy is equal to 4.0 kΩ cm2. In the Bode plot of the Mg alloy electrode whose potentiostatically formed oxide film was modified with SA SAM (Figure 3), two time constants can be observed.
The impedance data are modeled by an electrical equivalent circuit (EEC, shown in Figure 4) analysis and the EEC parameters obtained are listed in Table 2. In this equivalent circuit, a constant phase element, CPE1, is parallel with the resistor R1, which is in series with a parallel combination of a constant phase element CPE2 and a resistor R2. The meaning of EEC elements is as follows: Rel is the electrolyte resistance. The first R1–CPE1 combination represents the film|solution interface (i.e. the charge transfer resistance and the double layer capacitance). The second R2–CPE2 combination represents the surface film itself (i.e. its resistance and capacitance).

EEC used to fit the impedance data.
The protection efficiency (η %) of modified surface layers and optimal values of the equivalent circuit parameters with the fitting of the impedance spectra (Figure 1) of the unmodified Mg alloy and modified Mg alloy obtained in Hanks’ solution, pH 6.67 (37°C) at Eocp.

SA: stearic acid; HAp: hydroxyapatite.
Rel = 8 Ω cm2.
The proposed physical meaning of the elements of EEC with two time constants is in accordance with those reported in the literature used to describe the impedance data obtained for Mg and its alloys modified with a surface film in electrolyte solutions.28,41 In the Bode plot, it can also be seen that the modification of the Mg alloy surface increases the value of log|Z| at low frequencies at which polarization resistance dominates.
Figure 5 shows the impedance spectra of thermally treated (TT; 24 h at 120°C) modified Mg alloy. For comparison, the impedance spectra of the Mg alloy with thermally untreated and TT natural oxide film without the presence of ODPA SAM are also shown. From the value of the impedance parameters, it results that the thermal treatment at 120°C of the electrode increases the resistance of the film R2 and the polarization resistance of the Mg alloy system. The main reason for the increase of Rp values of octadecylphosphonate SAM for the modified Mg alloy could be a change in the type of interaction of the surface SAM from physical bonds (hydrogen bonds) to chemical bonds. 42 Also, the contribution to increasing the oxide layer polarization resistance due to the increase of the oxide layer compactness and the reduction of the pore in the outer part of the oxide film caused by thermal treatment should not be neglected. 43 From the Nyquist plot, it is evident that ODPA increases the polarization resistance, while the Bode plot clearly indicates the existence of two time constants.

Nyquist and Bode plots of impedance spectra for unmodified Mg alloy, thermally treated (24 h at 120°C) Mg alloy, thermally treated Mg alloy coated with ODPA SAM, and thermally treated Mg alloy whose ODPA SAM was additionally modified with HAp recorded in Hanks’ solution at 37°C at open-circuit potential. Phosphonate SAM was deposited from the ODPA alcoholic solutions of 0.7 mmol dm−3 concentration and self-assembling time was 1 h. Electrodeposition was performed at a potential of −2.0 V for 3 h at room temperature.
Since the impedance spectra shown in Figure 5 are very similar and have the same shape as those shown in Figure 3, the obtained results are mathematically modeled with the same EEC. The numerical values obtained by mathematical modeling are shown in Table 3. Decreasing of the parameter of CPE2, that is, Q2, which represents the capacitive behavior of the surface film, indicates that film capacity during the SAM formation decreases. Since the film capacity C is inversely proportional to the film thickness, it can be considered that the film capacity decrease is caused by an increase in the film thickness as well as structural changes within the film by forming a more compact film. From Figures 3 and 5 and Tables 2 and 3, it can be seen that the modification of the Mg alloy surface increases the corrosion resistance of the alloy electrode to Hanks’ solution. From the Nyquist plots shown in Figures 1 and 2, it is evident that the electrodes additionally modified with HAp have the highest polarization resistance, that is, the overall impedance. In Bode plots of HAp additionally modified electrodes, three time constants can be seen, that is, at low frequencies, an additional time constant attributed to the diffusion processes and described by the Warburg element WS appears. Therefore, for the mathematical modeling of the EIS results for HAp additionally modified Mg alloy electrodes, an EEC with WS which is in series with a parallel combination of a constant phase element CPE2 and a resistor R2 was used (Figure 4). The values of these EEC elements are also shown in Tables 2 and 3. The polarization resistance is equal to the sum of the charge transfer resistance R1, the resistance of the ions traveling through the film R2, and Warburg resistance, WS-R.5,28 From the impedance parameter values (as illustrated in Figures 3 and 5 and Tables 2 and 3), it can be seen that HAp modification increases the polarization resistance of organic acid SAM (Figure 3) from ~15 to ~33 kΩ cm2 and (Figure 5) from ~25 to ~58 kΩ cm2, respectively. The numerical values of the CPE2 exponent (n2) for the unmodified Mg alloy and Mg alloy whose organic acid SAM was additionally modified with HAp indicate the diffusion process of the magnesium ions through the surface layer. 26 The efficiency of the magnesium corrosion inhibition (η %) by barrier organic acid SAM additionally modified with HAp was calculated according to the expression η = (Rp(SAM|HAp) − Rp)/Rp(SAM|HAp), and the efficiency for Mg alloy|Anodized|SA|HAp is 88% and that for Mg alloy|TT|ODPA|HAp is 93%, which indicates a good corrosion resistance of modified Mg alloy in Hanks’ solution. For the Mg alloy|TT|ODPA|HAp system (Figure 5 and Table 3), a significant increase of the polarization resistance (40%–50%) was observed indicating a stronger bond between phosphonate SAM and the Mg alloy surface compared to the bond between carboxylate SAM and the surface of the Mg alloy. Also, self-assembling of carboxylic acids is a slow process because this SAM was self-assembled during a longer period of 24 h. The stronger bond makes the phosphonate SAM more stable during the process of electrochemical formation of the HAp film.
The protection efficiency (η %) of modified surface layers and optimal values of the equivalent circuit parameters with the fitting of the impedance spectra (Figure 2) of the unmodified Mg alloy and modified Mg alloy obtained in Hanks’ solution, pH 6.67 (37°C) at Eocp.
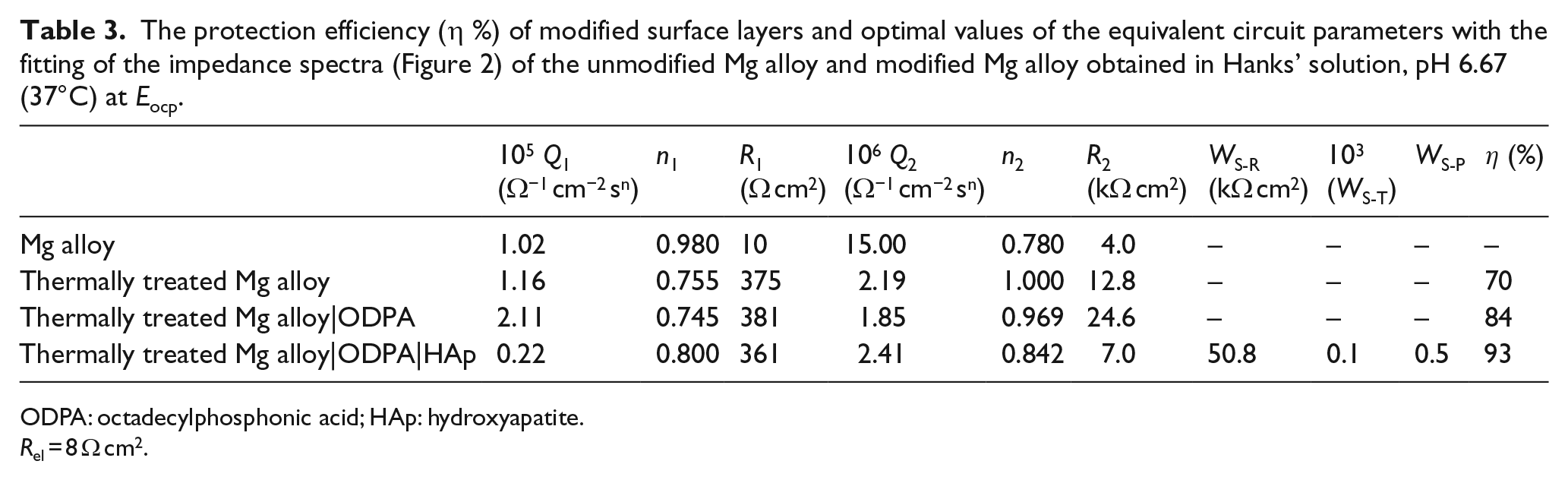
ODPA: octadecylphosphonic acid; HAp: hydroxyapatite.
Rel = 8 Ω cm2.
Scanning electron microscopy characterization
The morphologies of SA|HAp film on the Mg alloy surface are characterized by scanning electron microscopy (SEM). The analysis was performed on samples immediately after electrodeposition and alkali treatment (Figure 6) of the anodized Mg alloy modified with SA SAM. Carboxylate SAM was deposited from the SA alcoholic solutions of 1.0 mmol dm−3 concentration and self-assembling time was 24 h. Electrodeposition was performed at a potential of −2.0 V for 3 h at room temperature. The modification clearly leads to the formation of a crystalline compact dense coating that completely covered the surface. 44 The images also reveal that the surface coating exhibits a needle-like dendrite structure,14,45 that is, a regular flower-like structure diverging from the center toward the periphery.46,47 A needle-like shape has calcium phosphate crystals in the presence of magnesium or carbonate. 48

SEM images of Mg alloy coated with SA SAM|HAp film.
FTIR characterization
To verify the existence of an organic acid|HAp film on the Mg alloy surface, FTIR spectroscopy measurements were performed. The obtained spectra are presented in Figure 7. Several conclusions can be derived from the obtained spectra:
A well-organized monolayer consists of alkyl chains directed toward the surface at the same angle. Confirmation: stretching values ν(C–H)asym. ⩽ 2918 cm−1 and ν(C–H) ⩽ 2850 cm−1 indicate their existence. 49
The wavenumber separation between COO− antisymmetric and symmetric vibrations, Δν (=νas − νs) is 104 cm−1 and the frequency difference can be denoted to the bridging coordination character of the carboxylic group and enables the identification of the type of coordination of the carboxylate group to metal cations (Figure 8). Confirmation: the appearance of peaks for carboxylate asymmetric stretching νas(COO−) at 1560 and symmetric stretching νs(COO−) from the carboxylate group at 1456 cm−1 (Δν = 104 cm−1). From theory, two bands commonly noted in the dimeric form of a carboxylic acid due to the presence of carbonyl C=O (at 1699 cm−1) and hydroxy C–OH groups (at 1431 cm−1). 50
Phosphonic acid is on the Mg alloy surface also bonded through the head group. Confirmation: peaks at 1103 and 1017 cm−1 that can be attributed to the stretching vibration of the P–O bond. 51

FTIR spectra of the AZ91D alloy surface modified with (a) SA|HAp and (b) ODPA|HAp films. Carboxylate SAM was deposited from the SA alcoholic solutions of 1.0 mmol dm−3 concentration and self-assembling time was 24 h. Phosphonate SAM was deposited from the ODPA alcoholic solutions of 0.7 mmol dm−3 concentration and self-assembling time was 1 h. The electrodeposition was performed at −2.0 V for 3 h.
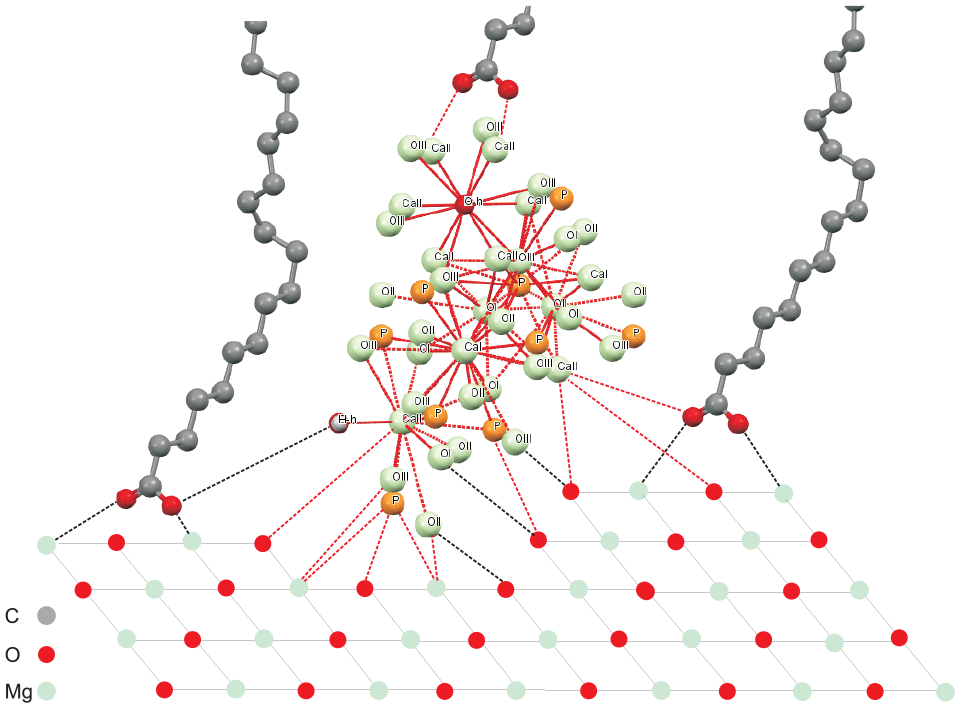
The schematic structure of SAM|HAp film on the Mg alloy surface.
In the FTIR spectrum of the Mg alloy surface modified with ODPA|HAp film, the peak of low intensity at about 2350 cm−1 confirms the presence of free P–OH groups and suggests the presence of several different binding modes of the phosphonic group.
51
Two fine-resolved ν3 bands at 1015 and 1086 cm−1 that can be attributed to the asymmetric P–O stretching39,45,52 indicate that the HAp coating is a crystalline.53,54 A wide peak of low intensity at about 3300 cm−1 was attributed to the O–H stretches in the HAp coating or in water.55–59 The peak at 867 cm−1, attributed to the
The schematic structure of SAM|HAp film on the Mg alloy surface is presented in Figure 8. A SAM can form through the chemical bond of the carboxy (phosphonyl) organic acid functional group and Mg atoms on the alloy surface. The organic acid SAM on the Mg alloy surface can induce CaP growth. The organic acid SAM most likely facilitates CaP growth by attracting Ca ions to the surface. 33
Biomineralization processes of apatite by developing controlled nanostructured assemblies predominantly occur when calcium ions adsorb and complex with negatively charged functional groups to form apatite. 33 In general, three aspects lead to specificity in nucleation are considered to be effective for accumulations of composites, constituted of biological (or organic) and inorganic compounds: electrostatic accumulation of cations, structural correspondence, and stereochemical requirements.
In our study, a stable organic/inorganic apatite coating on Mg alloy formed as a multilayer coating was induced rapidly, due to Ca2+ ions from the solution which are preferentially attracted by the –COOH or
Between two functional groups,
A stable organic/inorganic apatite coating is uniformly distributed over the surface and corrosion is successfully suppressed.
Such coatings could greatly improve the adhesion to the Mg substrate.
Electrodeposition as a method for deposition of HAp resulted in a more homogeneous and uniformly distributed coating.
Conclusion
In order to increase the bioactivity and biocompatibility of the Mg alloy modified with organic acid SAM, its surface was additionally modified with electrodeposited HAp. The corrosion resistance of Mg alloy|organic acid SAM|HAp coating was investigated in Hanks’ solution at 37°C using voltammetry and EIS methods. The protection efficiency of organic acid SAM is equal to 73% for SA SAM and 84% for ODPA SAM, respectively. The results verify that the formation of HAp coatings on the Mg alloy surface modified with organic acid SAM for increasing corrosion resistance is feasible. The bioceramic surface layer of HAp obtained by electrodeposition on the organic acid SAM has increased the barrier properties of Mg alloy to about 21% for SA and 11.3% for ODPA SAM, respectively.
Chemical group characteristics of organic acid films, that is, HAp coating detected by FTIR spectroscopy on the modified Mg alloy, confirm the presence of organic acid|HAp film on the Mg alloy surface. The FTIR results also indicated that the organic acid is chemisorbed as carboxylate or phosphonate onto the Mg alloy surface, and that both oxygen atoms in the carboxylate are coordinated symmetrically to the Mg atoms. The SEM results confirmed the presence of a thick uniform layer of HAp needle-like structure and unique snowflake morphology. In this study, we suggest that organic acid SAM may facilitate CaP growth by attracting Ca2+ ions to the surface. Hence, –PO4H2 shows a stronger capability of nucleating calcium phosphate deposition than –COOH.
Experimental
Mg alloy (Mg93Al6Zn1) used as working electrodes (Goodfellow) and having a surface area of 0.5 cm2 was placed in the Princeton Applied Research (PAR) electrode holders. Before each measurement, the working electrodes were abraded with fine emery paper, polished with alumina powder down to 0.05 μm, degreased in ethanol in an ultrasonic bath, and rinsed with distilled water.
A self-assembling monolayer (SAM) of SA was formed on the Mg alloy covered with potentiostatically formed oxide film by a simple immersion method using a 1 mM ethanolic solution of SA at 25°C for 24 h. Anodization of the electrode was performed at 1.0 V for 5 min in sodium borate solution containing (g L−1): NaOH (50.0), H3BO3 (10.0), and Na2B4O7·10H2O (20.0). Alloy specimens with deposited SAMs were rinsed with distilled water and air-dried.
A SAM of ODPA was formed on the Mg alloy covered with a TT (30 min at 90°C) spontaneously formed oxide film by a simple immersion method using a 0.7 mM ethanolic solution of ODPA at 25°C for 1 h. The specimens were then heated in an air convection oven at 120°C for 24 h.
In order to improve the biocompatibility of the Mg alloy modified with organic acid SAM, its surface was additionally modified with a bioceramic HAp coating. The electrodeposition of Ca/P coating was carried out at constant potential values of −2.0 V for 3 h at room temperature in an electrodepositing solution (100 cm3) containing 50 cm3 of 0.1 mol dm−3 Ca(NO3)2 and 50 cm3 of 0.06 mol dm−3 NH4H2PO4. The pH value of the solution was 5 (adjusted with 0.5 mol dm−3 HNO3). During deposition, the solution was stirred with a magnetic stirrer. After deposition, the electrode was immersed in a 1.0 mol dm−3 solution of NaOH at 80°C for 1 h, rinsed with distilled water, and air-dried. Electrodeposition results in the formation of CaHPO4·2H2O (DCPD) film on the Mg alloy surface. The DCPD film, that is, the Ca/P coating by acid–base reaction (electrode immersion for 1 h in 1.0 mol dm−3 NaOH at 80°C), is converted into HAp. The experimental procedure is shown in Scheme 1. The barrier properties and electrochemical characterization of the prepared coatings were investigated at 37°C in Hanks’ solution, pH 6.67, of the following composition (g dm−3): NaCl (8.00), KCl (0.40), NaHCO3 (0.35), NaH2PO4·H2O (0.25), Na2HPO4·2H2O (0.06), CaCl2·2H2O (0.19), MgCl2 (0.19), MgSO4·7H2O (0.06), and glucose (1.00). All chemicals were of p.a. purity. All electrochemical experiments were carried out in a standard three-electrode cell containing 100 cm3 of Hanks’ solution. The counter electrode was a large area platinum electrode and the reference electrode, to which all potentials in the paper are referred, was Ag/AgCl/3M KCl (209 mV vs standard hydrogen electrode (SHE)). Before measurements, the electrodes were stabilized for 1 h in Hanks’ solution. Potentiodynamic measurements were performed at a sweep rate of 1 mV s−1. EIS measurements were performed at the open-circuit potential (EOCP) in the frequency range from 100 kHz to 0.1 Hz with an alternating current (AC) voltage amplitude of ±10 mV.

Experimental procedure scheme.
Measurements were carried out using a Solartron Frequency Response Analyzer SI 1255 and Solartron Electrochemical Interface 1287 controlled by a PC. Impedance data were fitted by a suitable EEC model, employing the complex non-linear least squares (CNLS) fit analysis 61 offered by the Solartron ZView® software. The fitting quality was evaluated by the chi-square and relative error values, which were in the order of 10−3–10−4 and below 5%, respectively, indicating that the agreement between the proposed EEC model and the experimental data was good. All prepared coatings were characterized by FTIR. The FTIR spectra were recorded in the region of 4000–650 cm−1 with a scan resolution of 4 cm−1 using the horizontal attenuated total reflectance (HATR) method on a PerkinElmer Spectrum One FTIR spectrometer. The microstructures and morphologies of the SA|HAp film on the Mg alloy substrates were determined by SEM (field emission SEM/energy-dispersive X-ray spectroscopy) using a Jeol Ltd. FE SEM, model JSM-7000F.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.