Abstract
A series of 1,3,4-oxadiazole-fused and piperazine-fused quinazoline derivatives is synthesized and evaluated as antibacterial agents. The synthetic protocol involves an efficient Suzuki C–C cross-coupling reaction on the quinazoline ring followed by formation of 1,3,4-oxadiazole intermediates. These are further treated with piperazine bases in the presence of formalin in methanol to furnish the final N-Mannich products. The majority of these compounds show potent antibacterial activities against several different strains of Gram-positive bacteria including multidrug-resistant clinical isolates. The cytotoxic activity assay demonstrates that the synthesized compounds do not affect cell viability on Human cervical (HeLa) cells at their minimum inhibitory concentrations. The newly synthesized compounds are characterized by infrared spectroscopy, 1H NMR, 13C NMR, mass spectrometry, and elemental analysis.

Introduction
The investigation of new heterocycles that demonstrate potency toward multiple biological targets remains an intriguing scientific endeavor. There has been a growing demand for the development of more sustainable antibiotics in recent decades, particularly those targeting the expansion of multidrug resistance in pathogenic microbes. In view of this, we have combined different moieties, such as quinazoline, piperazine, and oxadiazole, in a single molecule. Compounds bearing a quinazoline moiety have been reported to possess a wide spectrum of biological and therapeutic properties, such as anti-inflammatory,1–3 antiplasmodial, 4 antibacterial, 5 antitumor, 6 antimicrobial,7,8 anthelmintic, 9 and anticonvulsant properties. 10 Expecting an enhancement of biological activity, we have incorporated a 1,3,4-oxadiazole moiety as a potential bioactive site in our systems. The large number of synthetic compounds with a 1,3,4-oxadiazole nucleus plays a vital role in medicinal chemistry. They are known to possess various biological activities such as antimicrobial,11–13 antituberculosis,14,15 anti-inflammatory, 16 antimalarial, 17 and anticancer activities. 18 Moreover, the literature survey revealed that analogs with inserted N-substituted phenyl piperazine bases have been reported to demonstrate a wide range of pharmacological activities.19–23
In continuation of our efforts to find new promising pharmacologically active molecules, we report here the synthesis and in vitro antimicrobial activity of quinazoline analogs by applying an efficient palladium-catalyzed C–C Suzuki coupling. The synthesized compounds were tested in vitro with various antibacterial strains, including multidrug-resistant clinical isolates such as methicillin-resistant Staphylococcus aureus (MRSA CCARM 3167 and MRSA CCARM 3506) and quinolone-resistant Staphylococcus aureus (QRSA CCARM 3505 and QRSA CCARM 3519) by recording the minimum inhibitory concentrations (MICs).
Results and discussion
Chemistry
The synthetic strategy to prepare the target compounds is illustrated in Scheme 1. The initial analog, 4-[(2-chloroquinazolin-4-yl)oxy]benzonitrile (

Synthetic protocol for the preparation of analogs
Antibacterial activity
The in vitro antimicrobial activities (Table 1) of the synthesized analogs were evaluated against three Gram-positive bacteria (Staphylococcus aureus RN4220, KCTC 503, and KCTC 209) and one Gram-negative bacterium (Escherichia coli CCARM 1356). It was observed that many of the synthesized analogs displayed excellent efficacy against all the three Gram-positive bacteria, specifically S. aureus RN4220 and S. aureus KCTC503 (MIC values of ⩽64 μg mL−1) as compared to the control drugs. The introduction of fluoro, trifluoromethyl, hydroxy, and methoxy functional groups on the phenyl ring attached to the piperazine moiety significantly enhanced the antibacterial activity. The trifluoromethyl-piperazine-fused quinazoline derivative
Inhibitory activities (MIC, µg mL−1) of compounds

Staphylococcus aureus RN4220.
Staphylococcus aureus KCTC503.
Staphylococcus aureus KCTC209.
Escherichia coli CCARM 1356.
The synthesized analogs were further evaluated for their inhibitory activity against selected methicillin-resistant S. aureus (MRSA) and quinolone-resistant S. aureus (QRSA) (Table 2). It is clear from the data that all of the compounds exhibited higher levels of inhibitory activity than oxacillin against MRSA 3167 (methicillin-resistant S. aureus CCARM 3167) and MRSA 3506 (methicillin-resistant S. aureus CCARM 3506). The synthesized analogs,
Inhibitory activities (MIC, µg mL−1) of compounds
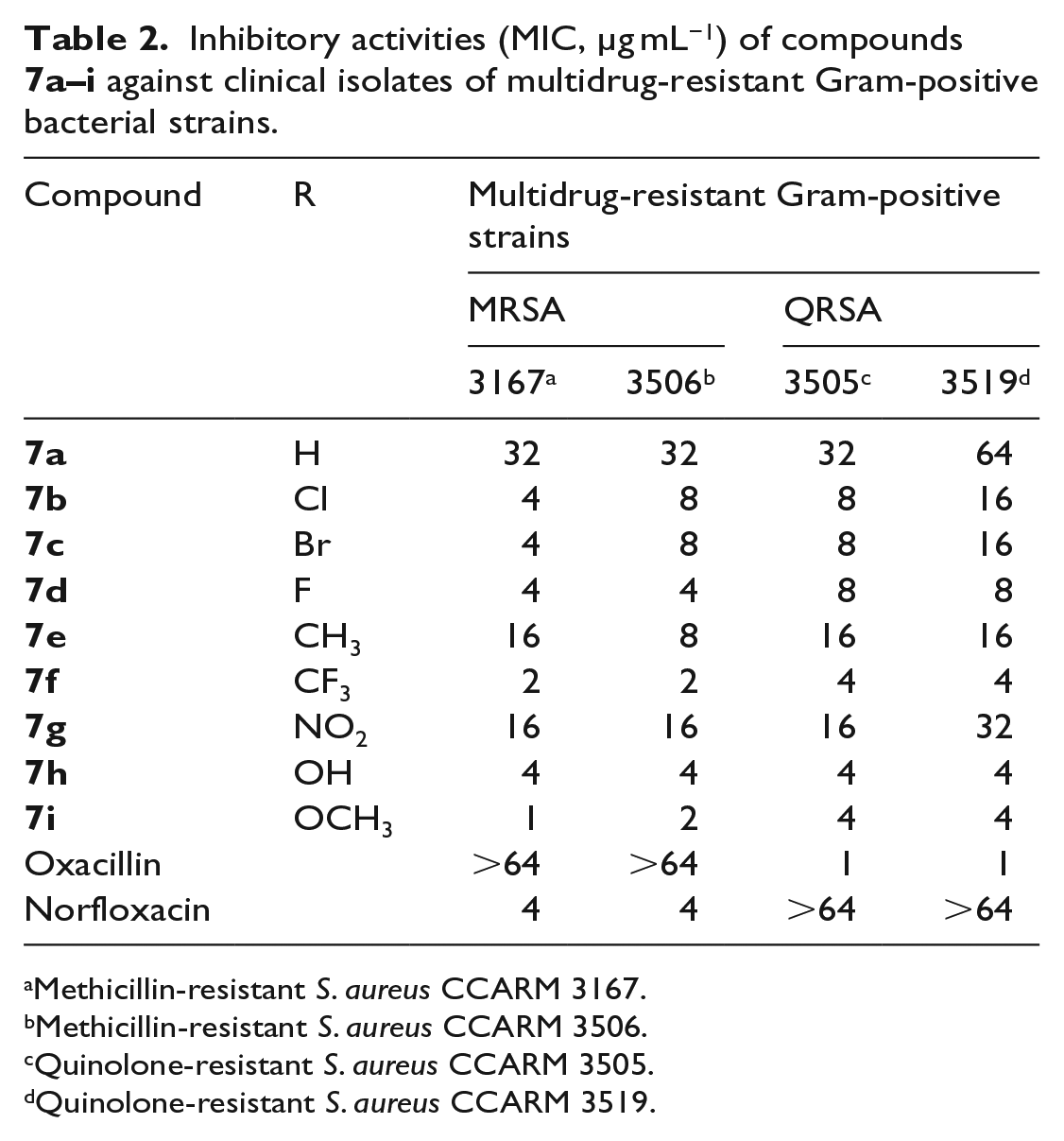
Methicillin-resistant S. aureus CCARM 3167.
Methicillin-resistant S. aureus CCARM 3506.
Quinolone-resistant S. aureus CCARM 3505.
Quinolone-resistant S. aureus CCARM 3519.
The cytotoxic properties of compounds
Cytotoxic activity of compounds

HeLa cell (Human cervical) monolayers were used as an in vitro model of cervicovaginal epithelium for testing the cytotoxicity of the new compounds.
IC50 is defined as the concentration at which 50% growth inhibition is observed.
These compounds did not affect cell viability of Human cervical (HeLa) cells at their MICs, but showed cytotoxicity at much higher concentrations. Hence, the inconsistency between the antibacterial activity and cytotoxicity of compounds
Conclusion
In conclusion, a series of oxadiazole- and piperazine-integrated quinazoline derivatives has been synthesized through an efficient Suzuki C–C cross-coupling reaction and the product tested for their antibacterial efficacy. The results demonstrated that most of the synthesized analogs demonstrate good levels of antibacterial activity against Gram-positive bacteria, including multidrug-resistant strains of clinical isolates. The presence of trifluoromethyl, hydroxy, and methoxy groups on the piperazine ring is essential for the compound to show high antibacterial activity between 2 and 8 μg mL−1 (
Materials and methods
General
All reactions were carried out under a nitrogen atmosphere. Air- and moisture-sensitive solvents and solutions were transferred via syringe or stainless steel cannula. All chemicals including 1-(4-substituted phenyl)piperazine bases were purchased from Sigma-Aldrich, Merck, and Fluka. Solvents used were of analytical grade. Anhydrous potassium carbonate was stored in a nitrogen-filled glovebox, ground, and was taken out in small quantities and stored in a desiccator. All reactions were routinely checked by TLC. TLC was performed on aluminum-backed silica gel plates (silica gel 60 F254 grade, Merck DC) with spots visualized by UV light. Column chromatography was performed on silica gel LC 60A (70–200 µm). Melting points were determined in open capillaries on a Veego electronic apparatus VMP-D (Veego Instrument Corporation, Mumbai, India) and are uncorrected. 1H NMR and 13C NMR spectra were recorded on a Bruker 400 MHz model spectrometer using DMSO-d6 as the solvent and tetramethylsilane (TMS) as the internal standard with a 1H resonance frequency of 400 MHz and a 13C resonance frequency of 100 MHz. 1H NMR and 13C NMR chemical shifts are reported as parts per million (ppm) downfield from TMS (Me4Si). The splitting patterns are designated as follows: s, singlet; d, doublet; dd, doublet of doublets; t, triplet; m, multiplet. The mass spectra were measured using a Waters Micromass Q-Tof Micro instrument (time of flight (TOF) mass spectrometer). Column chromatography was performed on a 1½ foot (2.5 cm diameter) glass column using silica gel LC 60A (70–200 µm). Elemental analysis was carried out using a Heraeus Carlo Erba 1180 C,H,N,S analyzer (Hanau, Germany).
4-[(2-Chloroquinazolin-4-yl)oxy]benzonitrile (2 )
An oven-dried Schlenk flask was charged with a magnetic stir bar, 2,4-dichloroquinazoline (
White solid, yield: 2.14 g, 76%; m.p. 167–171°C; IR (KBr, cm−1): 2850 (C–H), 2220 (C≡N), 1238 (C–O–C), 779 (C–Cl); 1H NMR (400 MHz, DMSO-d6): δ 8.15 (dd, J = 6.8, 1.8 Hz, 1H, quinazoline), 7.95–7.78 (m, 3H, quinazoline), 7.77–7.46 (m, 4H, ArH); 13C NMR (100 MHz, DMSO-d6): δ 167.3, 164.9, 146.4, 139.4, 135.0, 132.5, 129.4, 127.4, 123.6, 122.4, 121.2 (Ar-C), 114.1 (
Ethyl 4-[4-(4-cyanophenoxy)quinazolin-2-yl]benzoate (3 )
In an oven-dried round-bottomed flask, a mixture of 4-[(2-chloroquinazolin-4-yl)oxy]benzonitrile (
White solid, yield: 3.20 g, 81%; m.p. 219–221°C; IR (KBr, cm−1): 2949 (C–H), 2220 (C≡N), 1236 (C–O–C); 1H NMR (400 MHz, DMSO-d6): δ 8.45 (dd, J = 6.8, 1.8 Hz, 1H, quinazoline), 8.27–7.87 (m, 3H, quinazoline), 7.72–7.21 (m, 8H, ArH); 4.07 (q, J = 5.9 Hz, 2H, OC
4-[4-(4-Cyanophenoxy)quinazolin-2-yl]benzohydrazide (4 )
An oven-dried Schlenk tube was charged with a magnetic stir bar, ethyl 4-[4-(4-cyanophenoxy)quinazolin-2-yl]benzoate (
Pale yellow solid, yield: 2.82 g, 74%, m.p. 263–265°C; IR (KBr, cm−1): 3319 (N–H), 2993 (C–H), 2220 (C≡N), 1176 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 9.88–9.71 (m, 1H, CON
Potassium 2-{4-[4-(4-cyanophenoxy)quinazolin-2-yl]benzoyl}hydrazinecarbodithioate (5 )
An oven-dried Schlenk tube was charged with a magnetic stir bar, 4-[4-(4-cyanophenoxy)quinazolin-2-yl]benzohydrazide (
4-({2-[4-(5-Thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (6 )
Using an oven-dried Schlenk tube, charged with a magnetic stir bar, potassium 2-{4-[4-(4-cyanophenoxy)quinazolin-2-yl]benzoyl}hydrazinecarbodithioate (
White solid, yield: 2.87 g, 68%, m.p. 211–215°C; IR (KBr, cm−1): 3319 (N–H), 2993 (C–H), 2220 (C≡N), 1612 (C=N of oxadiazole), 1247 (C=S), 1176 (N–N, oxadiazole), 1141 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.44 (s, 1H, NH), 8.18 (dd, J = 6.2 Hz, 1H, quinazoline), 7.84–7.54 (m, 3H, quinazoline), 7.44–6.36 (m, 8H, ArH). 13C NMR (100 MHz, DMSO-d6): δ 182.1 (C=S), 170.7, 169.1, 159.6, 150.2, 142.4, 140.2, 134.4, 131.0, 129.7, 129.0, 127.5, 126.3, 121.4, 117.2, 115.4 (Ar-C), 108.4 (
General synthetic procedure for preparation of analogs 7a–i
An oven-dried Schlenk tube was charged with a magnetic stir bar, compound
4-{[2-(4-{4-[(4-Phenylpiperazin-1-yl)methyl]-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl}phenyl)quinazolin-4-yl]oxy}benzonitrile (7a )
White solid, yield: 4.30 g, 72%, m.p. 194–195°C; IR (KBr, cm−1): 3209 (C–H), 2218 (C≡N), 1608 (C=N of oxadiazole), 1257 (C=S), 1174 (N–N), 1112 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.51 (dd, J = 6.4 Hz, 1H, quinazoline), 8.21–7.79 (m, 3H, quinazoline), 7.57–7.25 (m, 13H, ArH), 4.92 (s, 2H, N–C
4-({2-[4-(4-{[4-(4-Chlorophenyl)piperazin-1-yl]methyl}-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7b )
White solid, yield: 5.31 g, 84%, m.p. 283–286°C; IR (KBr, cm−1): 3134 (C–H), 2230 (C≡N), 1612 (C=N of oxadiazole), 1237 (C=S), 1198 (N–N), 1103 (C–O–C), 747 (C–Cl). 1H NMR (400 MHz, DMSO-d6): δ 8.47 (dd, J = 6.8 Hz, 1H, quinazoline), 7.92–7.78 (m, 3H, quinazoline), 7.63–7.04 (m, 12H, ArH), 4.83 (s, 2H, N–C
4-({2-[4-(4-{[4-(4-Bromophenyl)piperazin-1-yl]methyl}-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7c )
Light brown solid, yield: 4.73 g, 70%, m.p. 240–241°C; IR (KBr, cm−1): 3089 (C–-H), 2225 (C≡N), 1617 (C=N of oxadiazole), 1240 (C=S), 1150 (N–N), 1123 (C–O–C), 938 (C-Br). 1H NMR (400 MHz, DMSO-d6): δ 8.21 (dd, J = 7.0 Hz, 1H, quinazoline), 8.04–7.93 (m, 3H, quinazoline), 7.79–7.30 (m, 12H, ArH), 4.85 (s, 2H, N–C
4-({2-[4-(4-{[4-(4-Fluorophenyl)piperazin-1-yl]methyl}-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7d )
White solid, yield: 4.80 g, 78%, m.p. 267–269°C; IR (KBr, cm−1): 3125 (C–H), 2220 (C≡N), 1597 (C=N of oxadiazole), 1345 (C–F), 1232 (C=S), 1186 (N–N), 1120 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.41 (dd, J = 7.0 Hz, 1H, quinazoline), 8.17–8.02 (m, 3H, quinazoline), 7.95–7.47 (m, 12H, ArH), 4.78 (s, 2H, N–C
4-({2-[4-(5-Thioxo-4-{[4-(p-tolyl)piperazin-1-yl]methyl}-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7e )
Pale White solid, yield: 4.22 g, 69%, m.p. 255–256°C; IR (KBr, cm−1): 3089 (C–H), 2218 (C≡N), 1617 (C=N of oxadiazole), 1244 (C=S), 1159 (N–N), 1132 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.50 (dd, J = 6.2 Hz, 1H, quinazoline), 8.09–7.93 (m, 3H, quinazoline), 7.66–7.25 (m, 12H, ArH), 5.02 (s, 2H, N–C
4-[(2-{4-[5-Thioxo-4-({4-[4-(trifluoromethyl)phenyl]piperazin-1-yl}methyl)-4,5-dihydro-1,3,4-oxadiazol-2-yl]phenyl}quinazolin-4-yl)oxy]benzonitrile (7f )
White solid, yield: 4.19 g, 63%, m.p. 289–293°C; IR (KBr, cm−1): 3111 (C–H), 2222 (C≡N), 1589 (C=N of oxadiazole), 1239 (C=S), 1185 (N–N), 1119 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.68 (dd, J = 7.2 Hz, 1H, quinazoline), 8.23–8.01 (m, 3H, quinazoline), 7.82–7.45 (m, 12H, ArH), 4.90 (s, 2H, N–C
4-({2-[4-(4-{[4-(4-Nitrophenyl)piperazin-1-yl]methyl}-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7g )
Light yellow solid, yield: 4.76 g, 74%, m.p. 247–249°C; IR (KBr, cm−1): 3044 (C–H), 2230 (C≡N), 1602 (C=N of oxadiazole), 1255 (C=S), 1180 (N–N), 1134 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.32 (dd, J = 6.8 Hz, 1H, quinazoline), 8.05–7.88 (m, 3H, quinazoline), 7.66–7.29 (m, 12H, ArH), 4.88 (s, 2H, N–C
4-({2-[4-(4-{[4-(4-Hydroxyphenyl)piperazin-1-yl]methyl}-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7h )
White solid, yield: 4.59 g, 75%, m.p. 189–192°C; IR (KBr, cm−1): 3057 (C–H), 2220 (C≡N), 1615 (C=N of oxadiazole), 1231 (C=S), 1189 (N–N), 1129 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.72 (dd, J = 7.2 Hz, 1H, quinazoline), 8.44–8.17 (m, 3H, quinazoline), 7.94–7.36 (m, 12H, ArH), 5.27 (s, 1H, OH), 4.03 (s, 2H, N–C
4-({2-[4-(4-{[4-(4-Methoxyphenyl)piperazin-1-yl]methyl}-5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)phenyl]quinazolin-4-yl}oxy)benzonitrile (7i )
White solid, yield: 4.46 g, 71%, m.p. 199–203°C; IR (KBr, cm−1): 3089 (C–H), 2224 (C≡N), 1590 (C=N of oxadiazole), 1224 (C=S), 1168 (N–N), 1123 (C–O–C). 1H NMR (400 MHz, DMSO-d6): δ 8.45 (dd, J = 6.8 Hz, 1H, quinazoline), 8.25–7.91 (m, 3H, quinazoline), 7.77–7.31 (m, 12H, ArH), 4.26 (s, 3H, OCH3), 3.98 (s, 2H, N–C
Pharmacology
The in vitro antimicrobial activity was performed using the broth microdilution method and the MIC with different strains, including multidrug-resistant clinical isolates. The newly prepared compounds were screened for their antibacterial activity against Gram-positive bacteria, Staphylococcus aureus (RN4220, KCTC 503 and KCTC 209) and a Gram-negative bacterium, Escherichia coli CCARM 1356. The strains of multidrug-resistant clinical isolates were multidrug-resistant Staphylococcus aureus (MRSA CCARM 3167 and MRSA CCARM 3506) and quinolone-resistant Staphylococcus aureus (QRSA CCARM 3505 and QRSA CCARM 3519). Clinical isolates were collected from various patients hospitalized in several clinics. Test susceptible microbacteria were grown to mid-log phase in Mueller-Hinton broth (MHB) and diluted 1000-fold in the same medium. The stock solutions of the synthesized compounds in DMSO were poured into 96-well plates and used to obtain final concentrations of 64–0.5 µg mL−1 by the twofold serial dilution technique. Oxacillin and Norfloxacin were used as positive controls for bacteria. Suspensions of microbacteria were prepared to contain approximate 105 CFU mL−1 and applied to 96-well plates with serially diluted compounds to be tested and incubated at 37°C for 24 h. The MIC (expressed in µg mL−1) was the lowest concentration of the test substance that completely inhibited growth of the microbacteria. The microbacteria growth was measured by the absorption at 630 nm using a microtiter enzyme-linked immunosorbent assay (ELISA) reader. All experiments were carried out three times. 27
In vitro cytotoxicity
Cytotoxicity activity assay: Human cervical (HeLa) cell monolayers were used as an in vitro model of cervicovaginal epithelium for testing the cytotoxicity of the new compounds. HeLa cells were grown in Dulbecco’s modified Eagle medium (DMEM) supplemented with fetal bovine serum (FBS; 10%) and antibiotics (penicillin–streptomycin mixture (100 U mL−1)). Cells at 80%–90% confluence were split by trypsin (0.25% in phosphate-buffered saline (PBS); pH 7.4), and the medium was changed at 24 h intervals. The cells were cultured at 37°C in a 5% CO2 incubator. The cells were grown to three passages and approximately 1 × 104 cells were seeded into each well of a 96-well plate and allowed to incubate overnight to allow cells to attach to the substrate. After 24 h, the medium was replaced with DMEM supplemented with 10% FBS containing various concentrations of test compounds and incubated for 48 h. Then, 10 µL of MTT solution (5 mg mL−1 in PBS) was added to each well. After incubation for 4 h, the medium was removed and the resulting formazan crystals were dissolved with 100 µL of DMSO. After shaking for 10 min, the optical density was measured at 570 nm using a microtiter ELISA reader. The assay was conducted four times. The IC50 values were defined as the concentrations inhibiting 50% of cell growth. 28
Supplemental Material
Supplementary_Infomation – Supplemental material for Investigation of the antibacterial activity of new quinazoline derivatives against methicillin and quinolone resistant Staphylococcus aureus
Supplemental material, Supplementary_Infomation for Investigation of the antibacterial activity of new quinazoline derivatives against methicillin and quinolone resistant Staphylococcus aureus by Amit B Patel in Journal of Chemical Research
Footnotes
Acknowledgements
The author is thankful to the Administration of Daman & Diu, Principal and Vice Principal, Government College, Daman for encouragement and facilities. He is also thankful to SAIF, Panjab University and Centre of Excellence, Vapi, India, for spectral analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
