Abstract
A green novel synthesis of ZnO@CuO@Iron ore nanocomposites as a novel and magnetic heterogeneous nanocatalyst is used for the one-pot synthesis of

Introduction
Nature as a source of biological system has great potential to produce nanomaterials using biomimetic approaches. The approaches are safe and biocompatible due to the coating of biological molecules on the surface of nanoparticles (NPs). Nowadays, nanomaterials, especially those consisting of metal, metal oxide, and mixed alloy nanoparticles have been used to make significant progress in science and technology; mainly, for providing a very large surface area as a principal site of thermodynamical reactions.1–4
In fact, the large surface area of nanosized systems (nanocatalysts) covers a huge number of active sites per unit area of the catalyst which leads to a considerable improvement of their catalytic ability.5,6
Currently, the attention of scientists is focused on preventing or decreasing agglomeration and aggregation side processes and increasing the stability of NPs when they are used as potential nanocatalysts in various applications. In recent years, natural supporting materials have attracted the attention of researchers as they prevent agglomeration, over-stoichiometric use of metal reagents, and also increase the stability of the produced nanostructures by altering their sensitivity to oxygen, water, and other chemical entities.7–10
α-Aminophosphonates are used widely and their synthesis, in a more efficient and safe way, has become a focus of synthetic organic chemistry and medicinal chemistry.11–13 The addition reaction of nucleophilic phosphorous compounds to imines catalyzed by Lewis acids is a useful synthetic method for their preparation. However, the mentioned process does not progress in one pot from a carbonyl compound, an amine and a phosphite as the produced water during the process degrades or deactivates the Lewis acids.14,15 This drawback has been overcome by some recent methods using lanthanide triflates/magnesium sulfate, 16 InCl3, 17 TaCl5–SiO2, 18 ZrCl4, 19 Sc(DS)3, 20 H3PW12O40, 21 BiNO3·5H2O, 22 Mg(ClO4)2, 23 and β-cyclodextrine. 24 As these methods require long reaction times and demonstrate low efficiencies, more efficient, economic catalytic processes for the synthesis of α-aminophosphonates are highly desirable.
Nowadays, solvent-free reactions benefit remarkably from shorter reaction times, higher yields, easier workups, and follow green approaches. Besides the selection of ecofriendly types of reactions, the employment of safe and green heterogeneous catalysts in chemical transformations leads to the better efficiency with evident economic and ecological advantages especially for industrial processes.
25
Until now, there have been no reported applications of green ZnO@CuO@Iron ore nanocomposites (NCs) as a multi-mineral acidic nanocatalysts for the synthesis of

Solventless synthesis of
Results and discussion
The catalytic reaction of aniline and 4-methylbenzaldehyde was studied as a model reaction in various solvents media and also solvent-free conditions. Different amounts of the green catalyst were detected at various temperatures, and also under solventless conditions. According to Table 1, the solventless system (entries 10, 11, 12, 14, and 15) gave higher yields and shorter reaction times compared to the solvent-containing systems. Furthermore, among the solventless conditions, the best result was obtained using 15 mg of the nanocatalyst at 100 °C, thus these reaction conditions were selected for the synthesis of the α-aminophosphonates. The optimum amount of ZnO@CuO@Iron ore NCs was found to be 15 mg in the presence of 4-methylbenzaldehyde (1 mmol), aniline (1 mmol), and triethyl phosphite (1 mmol). Besides the large surface area of the nanocatalyst which provides a suitable substrate for the reaction fractions in a high concentration, the multi-mineral system of iron ore substrate composed of a homogeneous crystalline structure involving magnetite, hematite, quartz, and chromite constituents as active acid sites caused a considerable increase in the yield and rate of the reaction. The ZnO@CuO@Iron ore NCs exhibited good catalytic activity in which the turnover number (TON) was calculated for the model reaction according to the following equation

Optimization of the conditions for the synthesis of α-aminophosphonates a .
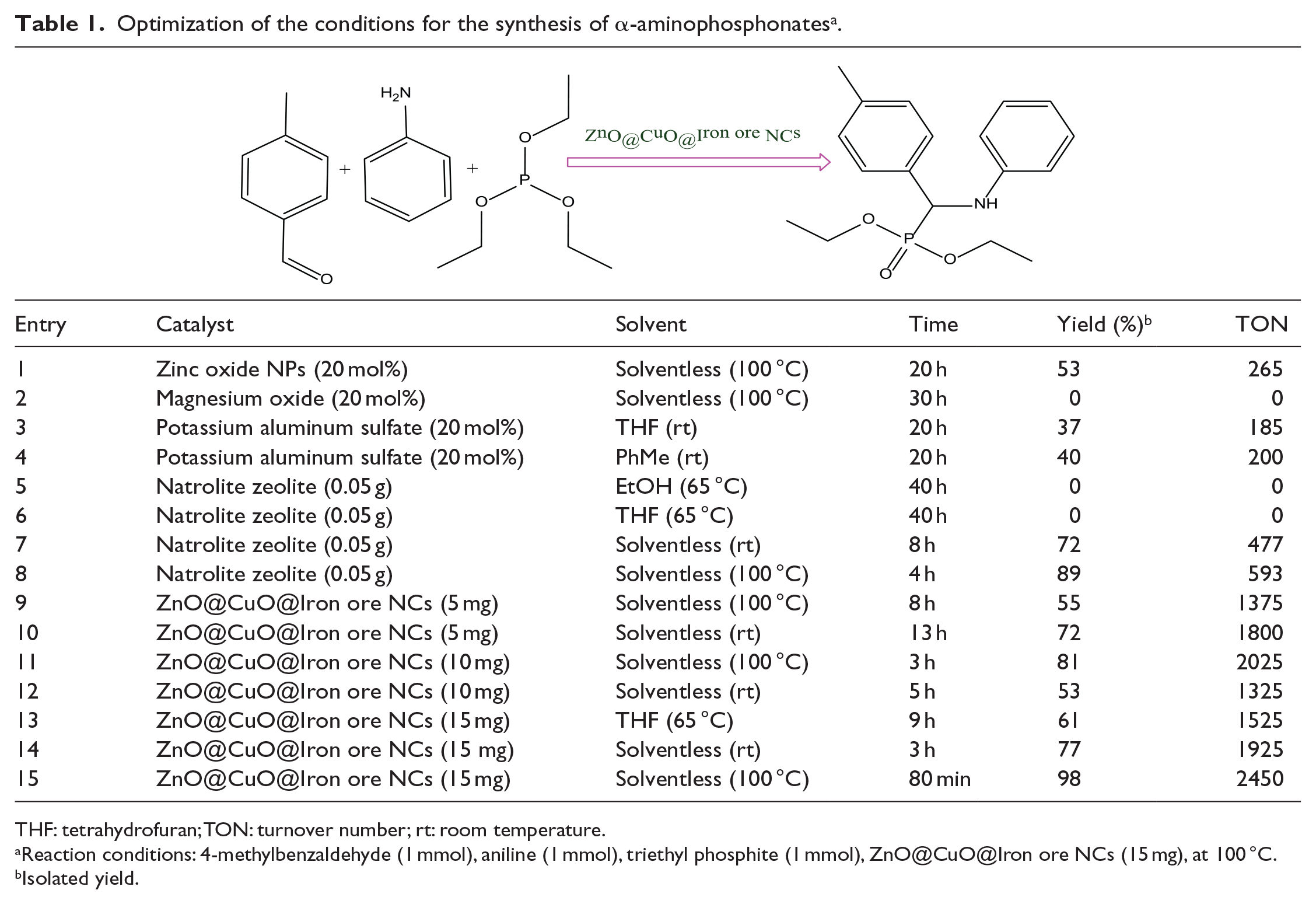
THF: tetrahydrofuran; TON: turnover number; rt: room temperature.
Reaction conditions: 4-methylbenzaldehyde (1 mmol), aniline (1 mmol), triethyl phosphite (1 mmol), ZnO@CuO@Iron ore NCs (15 mg), at 100 °C.
Isolated yield.
Based on the above equation, the TON obtained from the optimum conditions of the model reaction (Table 1, entry 15) was calculated as 2450, which clearly indicates that the method is useful for the synthesis of α-aminophosphonates.
To study the scope of the optimized reaction process, the solventless reactions of several aldehydes, amines (primary and secondary), and trimethyl or triethyl phosphites were assessed in the presence of 15 mg of ZnO@CuO@Iron ore NCs at 100 °C. Benzaldehyde and electron-deficient aromatic aldehydes reacted with aromatic and aliphatic amines to give the corresponding α-aminophosphonates in excellent yields. No promoter was required for this method (Table 2). In the case of benzaldehyde, aniline and trimethyl phosphite, the reaction did not occur in the absence of the green catalyst. Besides the selected aldehydes, several ketones were also screened in this three-component coupling under solvent-free conditions.
Synthesis of α-aminophosphonates using ZnO@CuO@Iron ore NCs (15 mg) under solventless conditions at 100 °C.

Yield of pure isolated product.
Table 3 compares the potential of our green catalyst–based method with other reports on the same reaction catalyzed by different catalysts. From the results, the ZnO@CuO@Iron ore NCs demonstrate a better performance in terms of the reaction rate and obtained higher yields of the products.
Effect of various catalysts on the synthesis of diethyl (4-methoxybenzaldehyde) (phenylamino)methylphosphonate by the reaction of 4-methoxybenzaldehyde, aniline, and alkyl phosphite.

THF: tetrahydrofuran; NCs: nanocomposites.
In contrast to heterogeneous catalysts, the homogeneous ones show problems on large-scale operation such as recycling and economic aspects. ZnO@CuO@Iron ore nanocatalyst shows a high potential for commercialization because of its ability to solve recycle problems and to give excellent product yields, even when it used in a very low amount.
In fact, besides the multi-mineral system of the iron ore substrate acting as a Lewis acid and the nonostructural properties of the ZnO@CuO@Iron ore nanocatalyst, coating the plant phytochemicals on the surface of the catalyst as capping agents resulted in a high concentration of reagents on the large surface of the nanocatalyst and also activated the carbonyl compounds considerably to enhance the reaction rate and yield (Figure 1). Finally, after removal of the solvent and purification, the products were characterized by infrared (IR), 1H NMR, 13C NMR, and from their melting points.
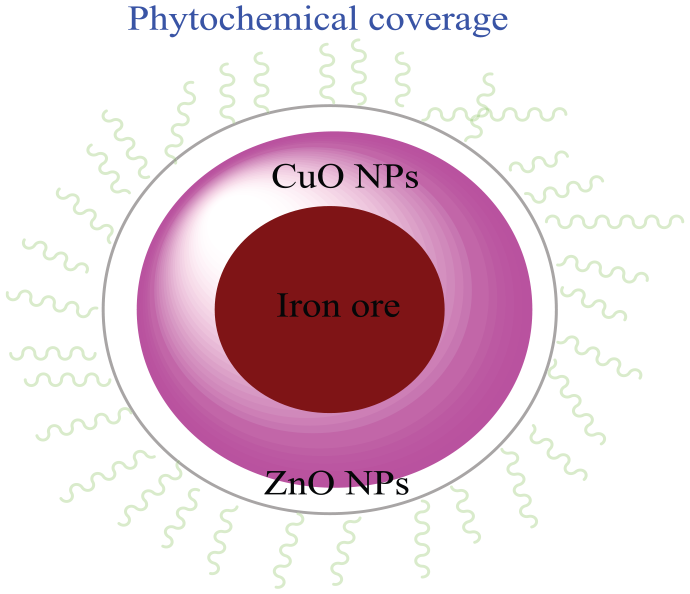
The ZnO@CuO@Iron ore nanocatalyst consisting of the natural iron ore substrate, CuO NPs, ZnO NPs, and a covering of plant phytochemicals on its surface.
At the end of the reaction, the green nanocatalyst was efficiently isolated from the reaction medium with a simple magnet then washed with ethanol and dried; this process can be continued for several consequent times without any loss of the catalyst activity, for example, the reaction of benzaldehyde (Table 2, entry 1) with aniline and triethyl phosphite has been repeated ten times using the same catalyst with consistently high yields of 93%–96%. The scanning electron microscope (SEM) micrograph of the recycled nanocatalyst after 10th cycle reveals its excellent stability, where its size, shape, and morphology were almost unchanged (Figure 2). Besides the structure of the catalyst and its natural origin, the phytochemicals coated on the surface of the nanocatalyst preserve it from the instability processes such as decomposition and deformation.

The SEM micrograph of the recycled nanocatalyst after the 10th cycle.
Conclusion
Due to the significance of α-aminophosphonates in medicinal and pharmaceutical applications, the development of novel, fast, and safe synthetic methodologies, especially using heterogeneous catalysts with important economic properties, is of great importance. This study has introduced a potent and green synthesized ZnO@CuO@Iron ore nanocatalyst which can be used for the synthesis of α-aminophosphonates by treatment of aldehydes or ketones with amines and trialkyl phosphites. The catalyst benefits from a large surface area, a phytochemical coating on its surface acting as a capping agent, and a multi-mineral system acting as a Lewis acid. Therefore, in view of its easy recovery, eco-friendliness, good reaction rates, high yields, and better performance compared to previously reported catalytic procedures for the synthesis of α-aminophosphonates, this method has significant advantages. In addition, the syntheses are solvent-free and involve short reaction times and a simple work-up procedure.
Experiment
All reagents were purchased from Merck and Sigma–Aldrich Ltd and were used without further purification. Products were characterized by spectroscopic data (IR, 1H NMR, and 13C NMR) and from melting points. The NMR spectra were recorded in CDCl3. 1H NMR spectra were recorded on a Bruker Avance DRX 500-MHz instrument. The chemical shifts (δ) are reported in parts per million (ppm) relative to the tetramethylsilane (TMS) as internal standard.
Magnetic iron ore collection
The iron ore deposit was collected in the north west of Choman, Iraqi Kurdistan (Figure 3). The sample was washed with hot water to remove possible impurities, then dried, and powdered to use as a natural magnetic substrate during this study.

A field image of iron ore deposit, north west of Choman, Iraqi Kurdistan.
Preparation of the Bryonia dioica root extract
An amount of 20.0 g of dried root powder of the plant (collected in Summer 2018 in the Haji Umran alpine region) was boiled in 150 mL of double distilled water for 30 min at 80 °C. The obtained extract was filtered and used for the green synthesis of the nanocatalyst.
Green synthesis of ZnO@CuO@Iron ore NCs
The ZnO@CuO@Iron ore NCs was synthesized according to our previous report with some modifications. 8 Briefly, 2 g iron ore powder was mixed with 100 mL of plant extract and the same volumes of 0.8% CuCl2·2H2O and 0.3% Zn(NO3)2 solutions in an alkaline medium adjusted using 0.1 M Na2CO3 while stirring at 80 °C. After the color of the reaction mixture had changed from light yellow to black, it was filtered and the obtained precipitate was dried and identified for use as a green nanocatalyst.
Catalyst characterization
The XRD pattern of the NCs (Figure 4) revealed the phase purity and crystal planes of numerous minerals in the texture of the iron ore substrate including ZnO and CuO NPs. This mineral matrix of the NCs indicates a valuable Lewis acid surface present in the catalyst system, which makes it an excellent catalyst for organic transformations.
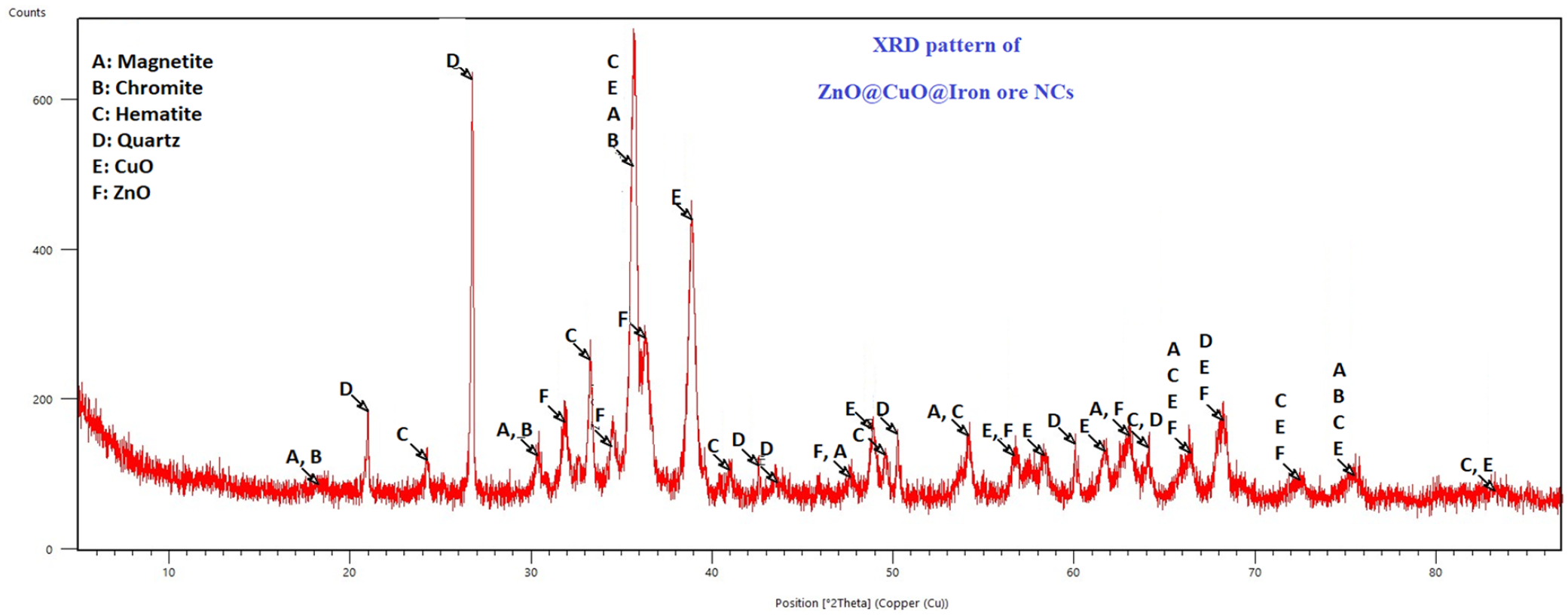
The XRD diffractogram of the ZnO@CuO@Iron ore NCs.
Furthermore, the SEM micrograph shown in Figure 5 demonstrates a nanosized system (mostly in the range of 50–70 nm) for the

SEM micrograph of the green synthesized
Also, the point analysis of a determined section of the nanocatalyst shows peaks related to the Fe, O, Zn, Si, and Cu, thereby confirming the successful anchoring of the CuO and ZnO NPs on the iron ore substrate and fabrication of the ZnO@CuO@Iron ore NCs (Figure 6).
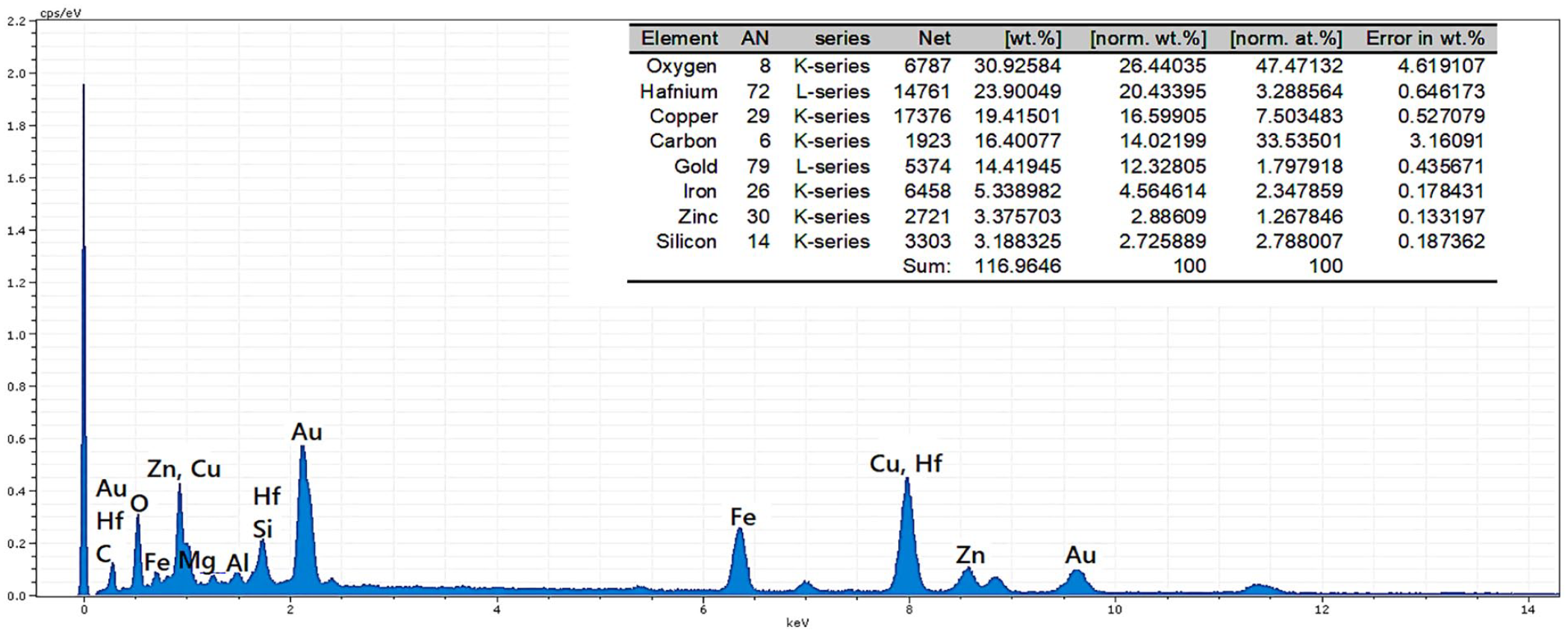
Point analysis of the ZnO@CuO@Iron ore NCs.
Preparation of α-aminophosphonates
A mixture of a carbonyl compound, an amine, and diethyl phosphite (each of 1 mmol) was added to the ZnO@CuO@Iron ore NCs (15 mg) at 100 °C. The progress of the reaction was monitored by thin-layer chromatography (TLC) (mobile phase EtOAc/n-hexane, 20:80). On completion of the reaction, EtOAc (2× 20 mL) was added and the catalyst was separated with a simple magnet. The organic solvent was removed under reduced pressure. After purification by chromatography on silica gel (EtOAc/n-hexane, 20:80) the α-aminophosphonate was obtained and characterized. All the products are known compounds and the spectral data and melting points were identical to those reported in the literature.21,23,26–30
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.