Abstract
N,N,N’,N’-tetramethyl-N,N’-bis(sulfo)ethane-1,2-diaminium mesylate ([TMBSED][OMs]2) was employed for the synthesis of piperidines and dihydropyrrol-2-ones via one-pot multi-component reactions in simple and green processes. This pseudo five-component reaction of aromatic aldehydes, anilines and alkyl acetoacetates was carried out under reflux conditions in ethanol to afford substituted piperidines. Also, dihydropyrrol-2-one derivatives were synthesized by means of four-component reactions of various amines, dialkyl acetylenedicarboxylates and formaldehyde in ethanol at room temperature. The present approaches have several advantages such as good yields, easy work-ups, short reaction times, and utilize mild and clean reaction conditions.

Introduction
In recent years, ionic liquids (ILs) have been extensively used and have attracted remarkable research interest as catalysts for the green synthesis of the organic compounds due to their tunable physical and chemical properties. 1 ILs have also been employed because of their advantages, including high thermal and chemical stability, low vapor pressure, non-flammability, and good ionic conductivity, as well as ability to catalyze numerous types of organic reactions, their ability to be used in solvent-free or solution conditions, the ease of changing the cation or anion in their structures to modify their physical and chemical properties, and their ability to dissolve in many organic and inorganic materials.2–5
Piperidine derivatives, as a valuable class of nitrogen-containing heterocycles, have received much attention owing to their properties such as anti-hypertensive, 6 neurotoxic activity, 7 anti-bacterial, 8 treatment of Alzheimer’s diseases, 9 anticonvulsant, anti-inflammatory, 10 anticancer, 11 and antimalarial. 12 Recently, multicomponent reactions (MCRs) have been employed for the synthesis of substituted piperidines.13–22
In addition, dihydropyrrol-2-ones are important in organic synthesis due to their wide spectrum of activity which includes anti-microbiological,23,24 antitumor,25,26 anti-inflammatory, 27 antimalarial, 28 antifungal, 29 antidiabetics, 30 and antibiotic properties. 31 Therefore, many methodologies have been developed to synthesize these practical heterocycles.32–38 However, some of the above-mentioned methods for the synthesis of both those types heterocycles have disadvantages such as long reaction times, low or moderate yields, and environmental pollution. Hence, the development of beneficial and environmentally friendly methods for the preparation of these compounds is still in demand.
In this work, N,N,N’,N’-tetramethyl-N, N’-bis(sulfo)ethane-1,2-diaminium mesylate ([TMBSED][OMs]2) was used as an effective dual-functional IL for the synthesis of substituted piperidines and dihydropyrrol-2-one derivatives via one-pot MCRs in ethanol as a green solvent (Scheme 1).

Synthesis of piperidines
Results and discussions
[TMBSED][OMs]2 was synthesized according to the our previously reported work.
39
To optimize the amount of the catalyst for the synthesis of substituted piperidines, the reaction of aniline (2 mmol), methyl acetoacetate (1 mmol), and benzaldehyde (2 mmol) was selected as a model system. Next, 5, 10, and 15 mol% of [TMBSED][OMs]2 were examined at ambient temperature, 50°C, and reflux conditions in EtOH. According to the results in Table 1, 10 mol% of the IL catalyst afforded methyl 1,2,5,6-tetrahydro-1,2,6-triphenyl-4-(phenylamino)pyridine-3-carboxylate (
Optimization of the reaction conditions for the synthesis of
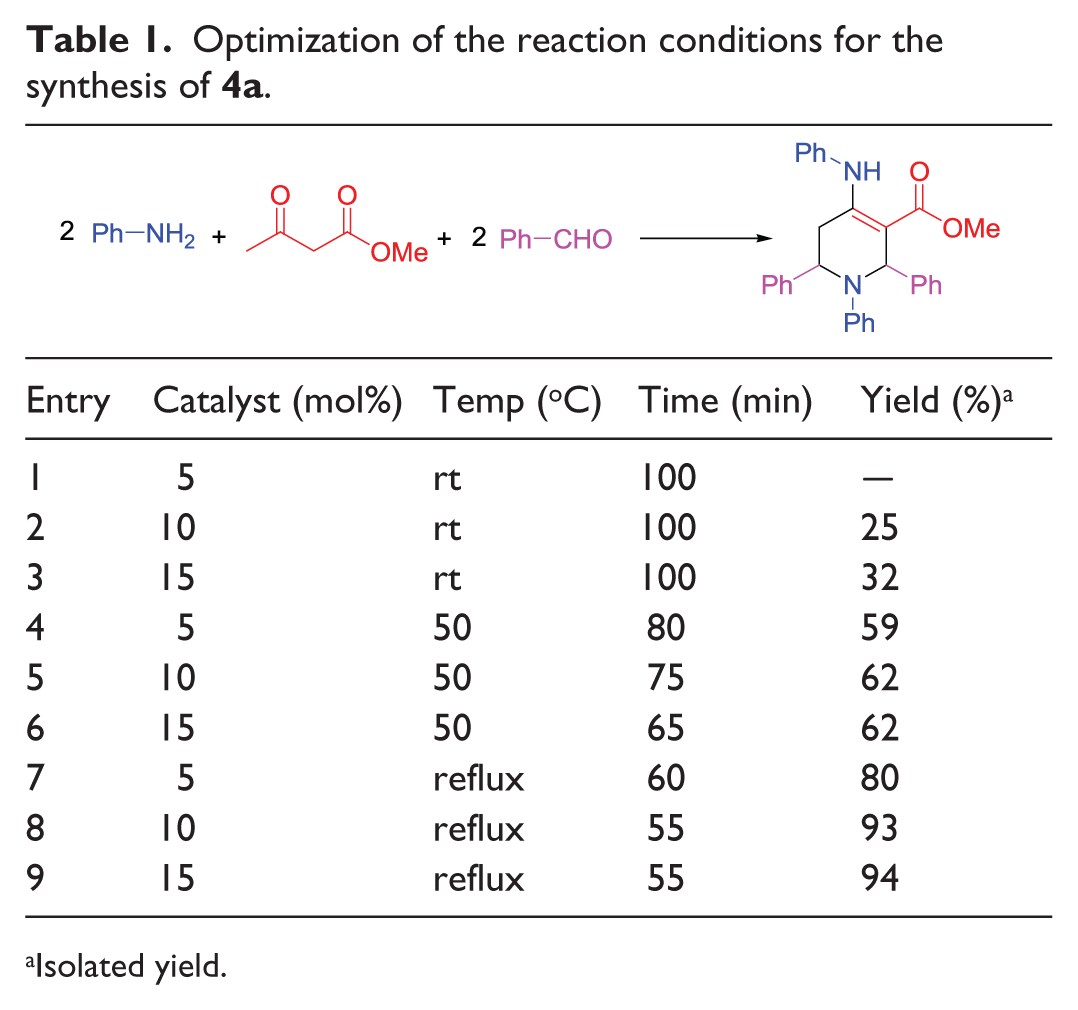
Isolated yield.
Next, the pseudo five-component reaction of anilines
Preparation of functionalized piperidines

Isolated yield.
Based on the literature,14–16 the proposed mechanism for this pseudo five-component reaction is displayed in Scheme 2. It was assumed that the aniline
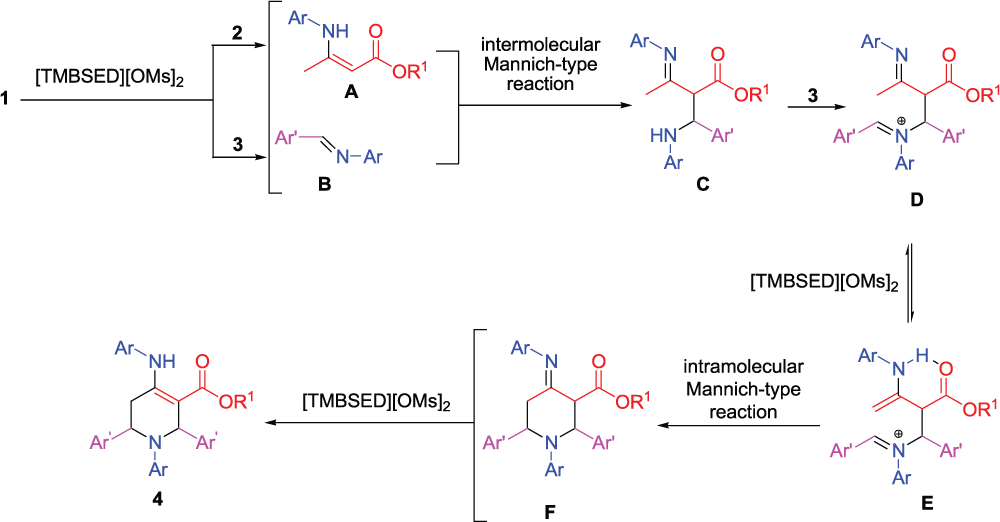
The proposed mechanism for the preparation of piperidine derivatives.
To demonstrate the applicability of the presented work, it can be compared with several reported results in the literature. The results show that the reactions were performed in short times and gave excellent yields in the presence of [TMBSED][OMs]2 when compared to other catalysts (Table 3).
Comparison the results of [TMBSED][OMs]2 with other catalysts in the synthesis of compound

Yields refer to isolated pure products based on the reaction of aniline (2 mmol), methyl acetoacetate (1 mmol) and benzaldehyde (2 mmol).
We next synthesized dihydropyrrol-2-ones
Preparation of dihydropyrrol-2-one derivatives in the presence of [TMBSED][OMs]2..

Isolated yield.
A possible reaction mechanism for the preparation of dihydropyrrol-2-ones
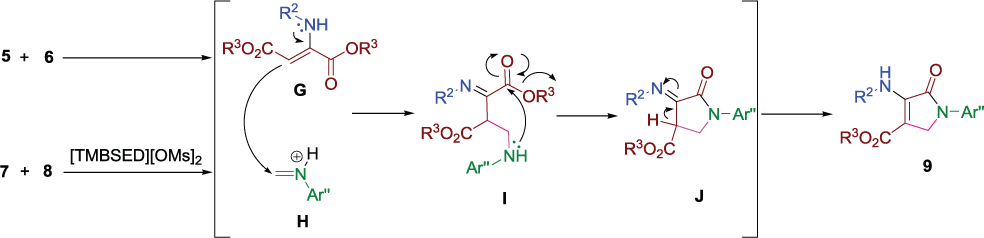
The proposed mechanism for the synthesis of dihydropyrrol-2-ones 9.
A comparison the results obtained with [TMBSED][OMs]2 and some reported catalysts for the synthesis of product
Comparison the results of [TMBSED][OMs]2 with other catalysts in the synthesis of compound

Yields refer to isolated pure products.
Conclusion
In summary, we have developed efficient one-pot procedures for the synthesis of functionalized piperidines and dihydropyrrol-2-one derivatives using [TMBSED][OMs]2 as a catalyst. These methods offered several advantages such as short reaction times, good to high yields, simplicity in operation, no need for column chromatography, and a green aspect by using ethanol as the solvent.
Experimental
Materials
All chemicals were obtained from Merck, Fluka and Sigma-Aldrich and were used without subsequent purification. The nuclear magnetic resonance (NMR) spectra were recorded with a Bruker DRX-400 AVANCE instrument (400 MHz for 1H, 100 MHz for 13C) in dimethyl sulfoxide (DMSO)-d6 as solvent. Melting points were measured on an Electro thermal 9100 apparatus. Please see the supplemental material for the spectral data of some selected compounds.
General procedure for the synthesis of piperidines 4
A solution of aromatic amine
General procedure for the preparation of dihydropyrrol-2-ones 9
A mixture of amine
Supplemental Material
Supplemental_Material – Supplemental material for Multi-component synthesis of piperidines and dihydropyrrol-2-one derivatives catalyzed by a dual-functional ionic liquid
Supplemental material, Supplemental_Material for Multi-component synthesis of piperidines and dihydropyrrol-2-one derivatives catalyzed by a dual-functional ionic liquid by Narjes Basirat, Seyed Sajad Sajadikhah and Abdolkarim Zare in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Research Council of Payame Noor University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



