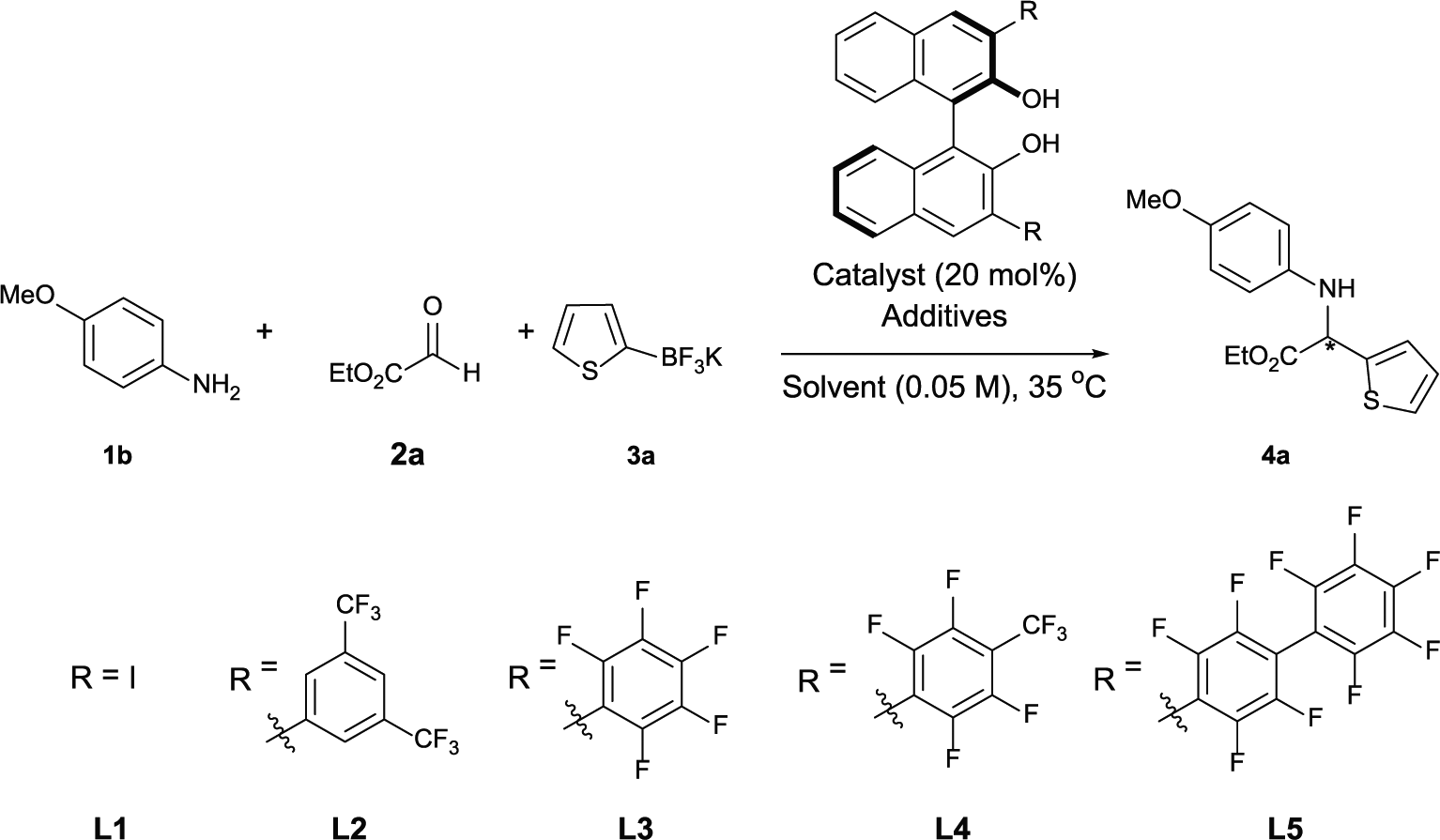
Abstract
Enantioselective synthesis of α-amino esters have been achieved through the Petasis borono-Mannich multicomponent reaction using (R)-BINOL-derived catalysts with stable heteroaryl and alkenyl trifluoroborate salts under mild conditions. The reaction provides direct access to optically active α-amino esters with moderate to good yields and enantioselectivities.
Keywords

Introduction
α-Amino acids are widely found in bioactive natural products and medicinal compounds.1,2 Since α-amino esters were recognized as main precursors of α-amino acid derivatives, much efforts have been devoted to the synthesis of α-amino esters including organometallics addition3–5 or metal-catalyzed addition reactions,6–9 Friedel–Crafts reactions,10–13 Mannich reactions,14,15 Michael additions, 16 hydrogenations,17,18 [4+2] cycloadditions,19–22 and so on. Among them, Petasis borono-Mannich (PBM) reaction highlighted the prominence toward the facile synthesis of α-amino esters in which highly functionalized products were obtained from simple aldehydes, amines, and boronic acid precursors. 23
Besides excellent functional group compatibility with the great tolerance of air and moisture, PBM multicomponent reaction usually requires aldehydes with an adjacent heteroatom directing group to form a boron “ate” complex where the boronic acid nucleophiles were likely activated for further addition. 24 Recently, Carrera’s group 25 reported acid-promoted addition of organotrifluoroborate salts to imines and enamines with the elimination of pendant heteroatom on the iminium intermediate (Scheme 1(a)). Pioneered by Lou and Schaus, 26 catalytic asymmetric PBM reaction provides direct access to induce the enantioenriched α-amino esters by employing alkenyl boronates and biphenol-derived catalysts (Scheme 1(b)). Muncipinto et al. 27 established catalytic diastereoselective PBM reaction of α-hydroxyaldehydes with secondary amines. Although several groups including Yamaoka et al., 28 Han et al., 29 and Au and Pyne 30 have been involved in the development of catalytic asymmetric PBM reaction, poor activity of these reaction systems with modest enantioselectivity was often observed between boronic acids or their esters with corresponding reaction partners.
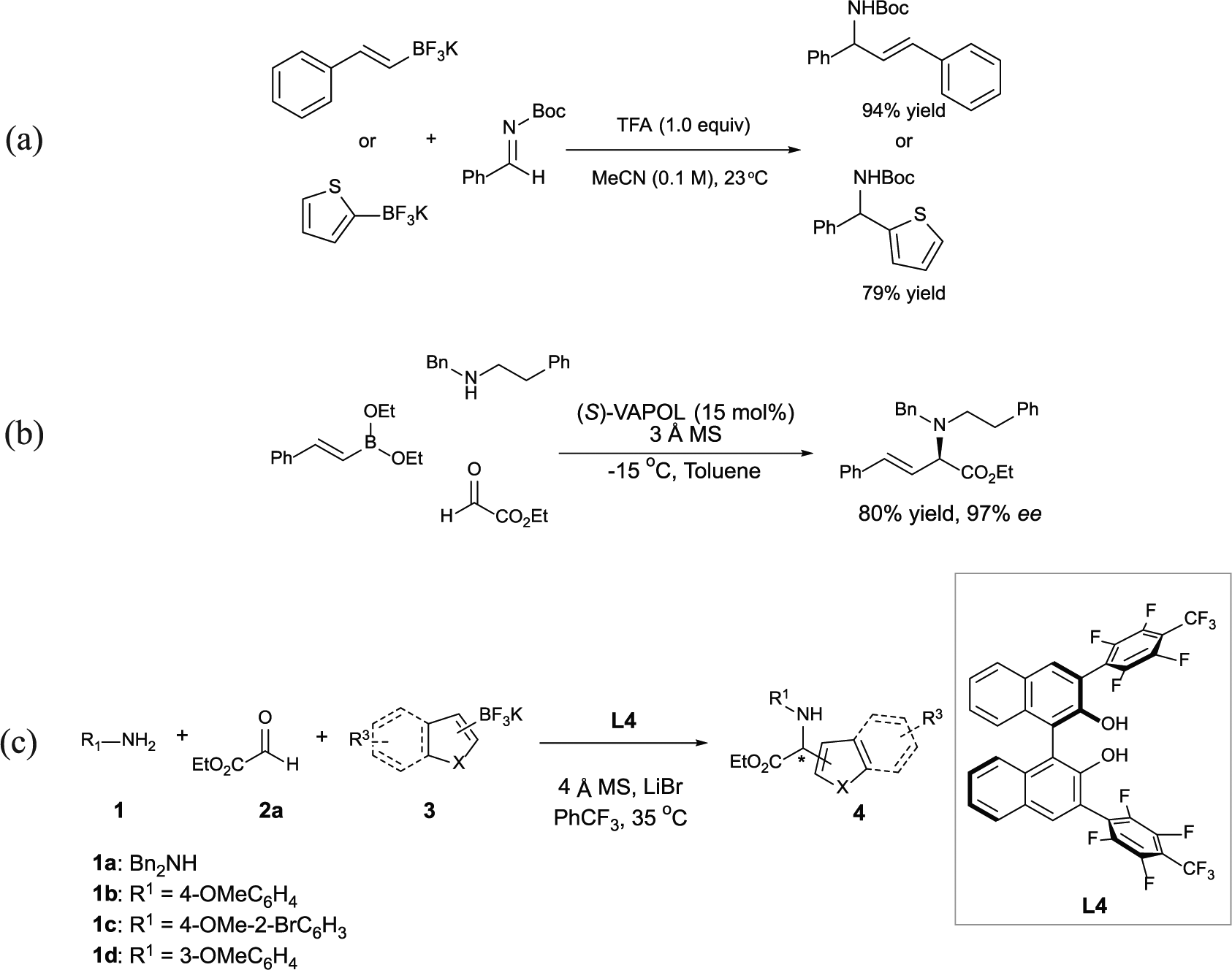
(a) Acid-promoted Petasis borono-Mannich reactions of trifluoroborate salts. (b) Catalytic asymmetric Petasis borono-Mannich reactions of boronate esters. (c) This work: enantioselective synthesis of α-amino esters via Petasis borono-Mannich reaction of trifluoroborate salts.
To address the continued lack of such methods, we envisioned the application of chiral biphenol catalysts and trifluoroborate salts would overcome the above-mentioned limitations by taking advantage of the multicomponent strategy of the PBM reaction. The use of trifluoroborate salts would be preferred to boronic acids or boronate esters from a practical standpoint for ease of use and long-term stability. Here, we describe the successful implementation of this concept for the synthesis of optical active α-amino esters by the one-pot strategy of the PBM reaction in the presence of trifluoroborate salts, ethyl glyoxylates in combination with aromatic or aliphatic amines.
Results and discussion
Encouraged by Lou et al.
31
and Jiang et al.’s work,
32
our research was initiated with amine
Optimization of the reaction conditions. a
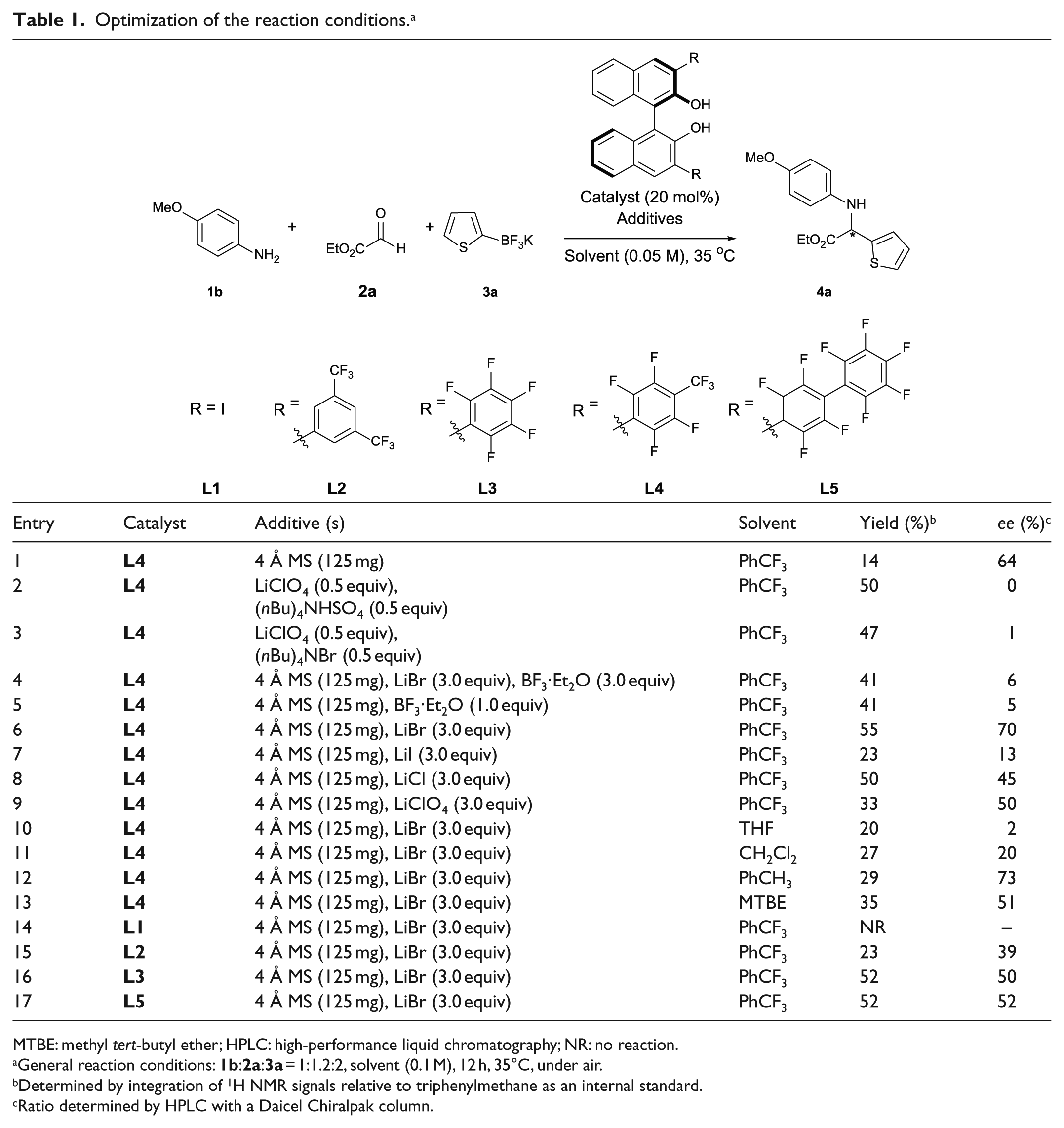
MTBE: methyl tert-butyl ether; HPLC: high-performance liquid chromatography; NR: no reaction.
General reaction conditions:
Determined by integration of 1H NMR signals relative to triphenylmethane as an internal standard.
Ratio determined by HPLC with a Daicel Chiralpak column.
With the optimized reaction conditions in hand, the scope of the heteroaryl trifluoroborate salts50–53 was then examined (Table 2). Electron-rich heteroaryl trifluoroborate salts such as thiophene, furan, pyrrole, and indole underwent the direct addition, providing the products with good levels of enantioselectivities. It was found that the 5-substituted-2-heteroaryl trifluoroborate salts resulted in products
Scope of the heteroaryl trifluoroborate salts. a

HPLC: high-performance liquid chromatography.
General reaction conditions:
Isolated yield.
Ratio determined by HPLC using a Daicel Chiralpak column.
A diverse and representative scope of amines with heteroaryl and alkenyl trifluoroborate salts was well tolerated for this process (Table 3). Good yields and enantioselectivities were obtained when the alkenyl trifluoroborate salts were employed (Table 3, entries 1 and 2). The functional group tolerance of primary amines was also investigated in which oxygen-, nitrogen-, and sulfur-containing heterocyclic trifluoroborates served as nucleophiles and exhibited similar yields and different enantioselectivities (Table 3, entries 3–5). Notably, only trace amount of desired product
Scope of various amines with heteroaryl or alkenyl trifluoroborate salts. a

HPLC: high-performance liquid chromatography.
General reaction conditions:
Isolated yield.
Ratio determined by HPLC with a Daicel Chiralpak column.
Conclusion
In conclusion, α-amino esters as precursors of unnatural amino acid derivatives play a pivotal role in both medicinal chemistry and chemical biology. We developed an efficient method toward the enantioselective synthesis of heterocyclic-derived α-amino esters through BINOL-catalyzed PBM multicomponent reaction under mild conditions. The reaction performs well for both stable alkenyl and heteroaryl trifluoroborate salts, in addition with a series of aromatic and aliphatic amines. The synthetic applications of such method will be further explored in our laboratory.
Experimental
General
Melting points were determined using a Büchi B-540 capillary melting point apparatus. NMR data including 1H NMR, 13C NMR, and 19F NMR spectra were recorded on Bruker 400 MHz or 600 MHz. All of the 13C NMR spectra were broad band proton-decoupled. 1H NMR Chemical shifts were reported in ppm relative to residual signals of the solvents (CDCl3: 7.26 ppm; (CD3)2SO: 2.50 ppm). 13C NMR chemical shifts were reported in ppm relative to the solvent (CDCl3: 77.16 ppm; (CD3)2SO: 39.52 ppm). Hexafluor-obenzene (δ = –164.9 ppm) was employed as an external standard in 19F NMR spectra. Coupling constants J are given in Hertz (Hz). Multiplicities are reported as follows: s = singlet, d = doublet, t = triplet, q = quartet, p = pentet, m = multiplet or as a combination of them. High-resolution mass spectra (HRMS) were recorded on an Agilent 6210 TOF LC/MS using electron spray ionization (ESI) as ion source. Optical rotations were determined using an AUTOPOL V automatic polarimeter. Enantiomeric excess (ee) was determined by high-performance liquid chromatography (HPLC) analysis using Agilent 1100 HPLC equipped with Daicel Chiralpak IA, IB, IC, IF, IG, and AS-H columns.
General procedure for the enantioselective synthesis of α-amino ester derivatives (4a-p ) through PBM reaction
To a 25-mL vial equipped with a stir bar, 4 Å powdered molecular sieves (125 mg) was added. The amine
Ethyl 2-((4-methoxyphenyl)amino)-2-(thiophen-2-yl)acetate (4a)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 50% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.22 (dd, J = 5.1, 1.2 Hz, 1H), 7.14–7.10 (m, 1H), 6.96 (dd, J = 5.1, 3.5 Hz, 1H), 6.74 (d, J = 8.9 Hz, 2H), 6.61 (d, J = 8.9 Hz, 2H), 5.26 (s, 1H), 4.33–4.14 (m, 2H), 3.70 (s, 3H), 1.25 (t, J = 7.1 Hz, 3H) (Supplemental material, S24). 13C NMR (101 MHz, CDCl3): δ 171.2, 153.0, 141.7, 140.1, 127.1, 125.6, 125.5, 115.3, 114.9, 62.1, 58.0, 55.7, 14.1 ppm. HRMS (ESI) m/z calcd. for C15H18NO3S [M+H]+: 292.1102; found 292.1104 (Supplemental material, S24). [α]D20 = −5.0 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 70% (n-Hexane/ethanol = 70/30, flow rate 1 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 14.212 min, tr (minor) = 16.317 min).
Ethyl 2-(5-bromothiophen-2-yl)-2-((4-methoxyphenyl)amino)acetate (4b)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 54% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 6.95–6.86 (m, 2H), 6.76 (d, J = 8.9 Hz, 2H), 6.61 (d, J = 8.9 Hz, 2H), 5.14 (s, 1H), 4.31–4.17 (m, 2H), 3.73 (s, 3H), 1.28 (t, J = 7.2 Hz, 3H) (Supplemental material, S25). 13C NMR (101 MHz, CDCl3): δ 170.5, 153.3, 143.6, 139.7, 130.1, 125.9, 115.4, 115.0, 112.3, 62.5, 58.3, 55.8, 14.2 ppm (Supplemental material, S25). HRMS (ESI) m/z calcd. for C15H17BrNO3S [M+H]+: 370.0107; found 370.0091. [α]D20 = −1.4 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 8% (n-Hexane/ethanol = 80/20, flow rate 0.8 mL min−1, λ = 254 nm, T = 20°C, tr (major) = 21.206 min, tr (minor) = 23.235 min).
Ethyl 2-(furan-2-yl)-2-((4-methoxyphenyl)amino)acetate (4c)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 69% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.39 (dd, J = 1.8, 0.9 Hz, 1H), 6.83–6.70 (m, 2H), 6.69–6.59 (m, 2H), 6.40–6.26 (m, 2H), 5.13 (s, 1H), 4.29–4.13 (m, 2H), 3.73 (s, 3H), 1.24 (t, J = 7.1 Hz, 3H) (Supplemental material, S26). 13C NMR (101 MHz, CDCl3): δ 170.3, 153.1, 150.5, 142.8, 140.0, 115.5, 114.9, 110.7, 108.2, 62.1, 56.4, 55.8, 14.2 ppm (Supplemental material, S26). HRMS (ESI) m/z calcd. for C15H17NNaO4 [M+Na]+: 298.1050; found 298.1060. [α]D20 = −37.6 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IF column, ee = 52% (n-Hexane/ethanol = 95/5, flow rate 0.7 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 25.185 min, tr (minor) = 23.940 min).
Ethyl 2-((4-methoxyphenyl)amino)-2-(5-methylfuran-2-yl)acetate (4d)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 52% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 6.82–6.69 (m, 2H), 6.68–6.59 (m, 2H), 6.22 (d, J = 3.1 Hz, 1H), 5.91 (m, 1H), 5.06 (s, 1H), 4.34–4.06 (m, 2H), 3.72 (s, 3H), 2.27 (s, 3H), 1.24 (t, J = 7.1 Hz, 3H) (Supplemental material, S27). 13C NMR (101 MHz, CDCl3): δ 170.6, 153.0, 152.6, 148.4, 140.2, 115.5, 114.9, 109.0, 106.6, 61.9, 56.5, 55.7, 14.2, 13.7 ppm (Supplemental material, S27). HRMS (ESI) m/z calcd. for C16H20NO4 [M+H]+: 290.1387; found 290.1385. [α]D20 = −1.8 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 3% (n-Hexane/ethanol = 80/20, flow rate 0.8 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 18.173 min, tr (minor) = 19.782 min).
tert-Butyl-2-(2-ethoxy-1-((4-methoxyphenyl)amino)-2-oxoethyl)-1H-pyrrole-1-carboxylate (4e)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 60% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.24–7.18 (m, 1H), 6.78–6.71 (m, 2H), 6.71–6.63 (m, 2H), 6.28 (dd, J = 3.5, 1.8 Hz, 1H), 6.09 (t, J = 3.4 Hz, 1H), 5.67 (s, 1H), 4.19 (q, J = 7.1 Hz, 2H), 3.72 (s, 3H), 1.58 (s, 9H), 1.26 – 1.17 (m, 3H) (Supplemental material, S28). 13C NMR (101 MHz, CDCl3): δ 171.4, 152.8, 149.4, 140.7, 131.1, 122.6, 115.7, 114.8, 114.2, 110.2, 84.3, 77.4, 61.7, 56.1, 55.7, 28.1, 14.3 ppm (Supplemental material, S28). HRMS (ESI) m/z calcd. for C20H27N2O5 [M+H]+: 375.1914; found 375.1903. [α]D20 = −11.6 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IF column, ee = 5% (n-Hexane/ethanol = 80/20, flow rate 0.8 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 8.238 min, tr (minor) = 9.124 min).
Ethyl 2-((4-methoxyphenyl)amino)-2-(1-(phenylsulfonyl)-1H-pyrrol-2-yl)acetate (4f)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 20:1) to afford the yellow oil in 54% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.78 (dd, J = 8.6, 1.3 Hz, 2H), 7.56–7.50 (m, 1H), 7.40–7.34 (m, 3H), 6.78–6.72 (m, 2H), 6.55 (d, J = 8.9 Hz, 2H), 6.29–6.24 (m, 2H), 5.71 (s, 1H), 4.14 (m, J = 7.1, 3.2 Hz, 2H), 3.75 (s, 3H), 1.19 (t, J = 7.1 Hz, 3H) (Supplemental material, S29). 13C NMR (101 MHz, CDCl3): δ 171.6, 153.2, 140.1, 139.3, 133.8, 131.2, 129.1, 127.4, 124.4, 115.7, 114.8, 114.6, 111.6, 61.8, 55.8, 54.4, 14.2 ppm (Supplemental material, S29). HRMS (ESI) m/z calcd. for C21H22N2NaO5S [M+Na]+: 437.1142; found 437.1132. [α]D20 = −2.2 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IF column, ee = 0% (n-Hexane/ethanol = 90/10, flow rate 1 mL min−1, λ = 254 nm, T = 20°C, tr = 16.789 min, tr = 19.107 min).
Ethyl 2-(benzo[b]thiophen-2-yl)-2-((4-methoxyphenyl)amino)acetate (4g)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 15% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.84–7.66 (m, 1H), 7.39 (d, J = 0.9 Hz, 1H), 7.31 (pd, J = 7.2, 1.4 Hz, 1H), 6.77–6.71 (m, 2H), 6.69–6.62 (m, 2H), 5.32 (d, J = 1.0 Hz, 2H), 4.50–4.01 (m, 3H), 3.71 (s, 3H), 1.28 (t, J = 7.1 Hz, 3H) (Supplemental material, S30). 13C NMR (101 MHz, CDCl3): δ 170.7, 153.1, 142.8, 139.8, 139.7, 124.5, 124.5, 123.7, 122.5, 122.5, 115.4, 115.0, 62.5, 58.6, 55.8, 14.2, 1.2 ppm (Supplemental material, S30). HRMS (ESI) m/z calcd. for C19H20NO3S [M+H]+: 342.1158; found 342.1154. [α]D20 = −3.4 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 71% (n-Hexane/ethanol = 80/20, flow rate 0.8 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 47.504 min, tr (minor) = 51.387 min).
Ethyl 2-((4-methoxyphenyl)amino)-2-(5-methylfuran-2-yl)acetate (4h)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 52% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.50 (m, 2H), 7.29 (dd, J = 7.2, 1.3 Hz, 1H), 7.21 (td, J = 7.5, 1.1 Hz, 1H), 6.78–6.73 (m, 3H), 6.68 (d, J = 9.0 Hz, 2H), 5.25 (s, 1H), 4.38–4.15 (m, 2H), 3.72 (s, 3H), 1.25 (t, J = 7.1 Hz, 3H) (Supplemental material, S31). 13C NMR (101 MHz, CDCl3): δ 169.8, 155.1, 153.3, 153.2, 139.5, 128.1, 124.6, 123.1, 121.3, 115.7, 114.9, 111.6, 105.3, 62.5, 56.9, 55.8, 29.8, 14.2 ppm (Supplemental material, S31). HRMS (ESI) m/z calcd. for C19H19NNaO4 [M+Na]+: 348.1206; found 348.1198. [α]D20 = −15.6 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 71% (n-Hexane/ethanol = 80/20, flow rate 0.8 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 31.296 min, tr (minor) = 34.523 min).
tert-Butyl 3-(2-ethoxy-1-((4-methoxyphenyl)amino)-2-oxoethyl)-1H-indole-1-carboxylate (4i)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 20:1) to afford the yellow oil in 80% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 8.15 (d, J = 8.3 Hz, 1H), 7.77 (dd, J = 7.9, 1.2 Hz, 1H), 7.64 (s, 1H), 7.36–7.31 (m, 1H), 7.29–7.26 (m, 1H), 6.78–6.71 (m, 2H), 6.66–6.59 (m, 2H), 5.26 (s, 1H), 4.20 (ddq, J = 43.9, 10.7, 7.1 Hz, 2H), 3.72 (s, 3H), 1.65 (s, 9H), 1.21 (t, J = 7.1 Hz, 3H) (Supplemental material, S32). 13C NMR (101 MHz, CDCl3): δ 172.1, 152.8, 140.6, 136.4, 132.9, 128.7, 128.1, 126.8, 125.5, 121.5, 115.2, 115.0, 114.5, 61.9, 60.0, 55.9, 55.6, 29.8, 14.3 ppm (Supplemental material, S32). HRMS (ESI) m/z calcd. for C24H28N2O5 [M+H]+: 425.2071; found 425.2056. [α]D20 = −17.2 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IB column, ee = 44% (n-Hexane/ethanol = 95/5, flow rate 1 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 9.787 min, tr (minor) = 11.029 min).
tert-Butyl-5-bromo-3-(2-ethoxy-1-((4-methoxyphenyl)amino)-2-oxoethyl)-1H-indole-1-carboxylate (4j)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 20:1) to afford the white solid in 55% yield at 0.1 mmol scale. 1H NMR (400 MHz, DMSO-d6): δ 8.07 (d, J = 2.0 Hz, 1H), 7.99 (d, J = 8.8 Hz, 1H), 7.89 (s, 1H), 7.50 (dd, J = 8.9, 2.0 Hz, 1H), 6.72 (s, 3H), 6.06 (d, J = 8.9 Hz, 1H), 5.54–5.46 (m, 1H), 4.20–4.02 (m, 2H), 3.34 (s, 3H), 1.63 (s, 9H), 1.12 (t, J = 7.1 Hz, 3H) (Supplemental material, S33). 13C NMR (101 MHz, DMSO-d6): δ 172.1, 152.0, 149.1, 141.6, 134.1, 130.8, 127.7, 126.4, 123.4, 117.4, 117.1, 115.8, 114.9, 114.8, 85.1, 61.5, 55.7, 53.7, 28.1, 14.5 ppm (Supplemental material, S33). HRMS (ESI) m/z calcd. for C24H28BrN2O5 [M+H]+: 503.1176; found 503.1155. m.p.: 102.5–104.6°C. [α]D20 = −6.8 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IB column, ee = 38% (n-Hexane/ethanol = 95/5, flow rate 0.8 mL min−1, λ = 230 nm, T = 20°C, tr (major) = 10.045 min, tr (minor) = 11.153 min).
Ethyl (E)-2-(dibenzylamino)-4-phenylbut-3-enoate (4k)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the colorless oil in 65% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.46–7.39 (m, 4H), 7.42–7.34 (m, 2H), 7.31 (td, J = 7.4, 3.1 Hz, 6H), 7.23 (ddd, J = 7.4, 5.6, 1.4 Hz, 3H), 6.57 (dd, J = 16.1, 1.2 Hz, 1H), 6.37 (dd, J = 16.1, 7.0 Hz, 1H), 4.33–4.17 (m, 2H), 4.10 (dd, J = 7.1, 1.3 Hz, 1H), 3.90–3.69 (m, 4H), 1.33 (t, J = 7.1 Hz, 3H) (Supplemental material, S34). 13C NMR (101 MHz, CDCl3): δ 172.1, 139.7, 136.7, 134.3, 128.8, 128.7, 128.4, 128.0, 127.1, 126.7, 124.6, 64.0, 60.8, 54.8, 14.6 ppm (Supplemental material, S34). HRMS (ESI) m/z calcd. for C26H28NO2 [M+H]+: 386.2115; found 386.2129. [α]D20 = −68.6 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 82% (n-Hexane/ethanol = 70/30, flow rate 1 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 4.640 min, tr (minor) = 5.640 min).
Ethyl (E)-2-((4-methoxyphenyl)amino)-4-phenylbut-3-enoate (4l)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 60% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): 7.40–7.35 (m, 2H), 7.34–7.28 (m, 2H), 6.82–6.74 (m, 3H), 6.64 (d, J = 8.9 Hz, 2H), 6.29 (dd, J = 15.9, 5.9 Hz, 1H), 4.65 (dd, J = 5.9, 1.5 Hz, 1H), 4.24 (p, J = 7.2 Hz, 2H), 3.73 (s, 3H), 1.29 (t, J = 7.1 Hz, 3H) (Supplemental material, S35). 13C NMR (101 MHz, CDCl3): δ 172.1, 152.8, 140.6, 136.4, 132.9, 128.7, 128.1, 126.8, 125.5, 115.2, 115.0, 61.9, 60.0, 55.9, 14.3 ppm (Supplemental material, S35). HRMS (ESI) m/z calcd. for C19H22NO3 [M+H]+: 312.1594; found 312.1604. [α]D20 = −14.8 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 80% (n-Hexane/Ethanol = 70/30, flow rate 1 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 21.573 min, tr (minor) = 31.569 min).
Ethyl 2-((2-bromo-4-methoxyphenyl)amino)-2-(thiophen-2-yl)acetate (4m)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 82% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.24 (dd, J = 5.1, 1.2 Hz, 1H), 7.14 (dt, J = 3.5, 0.9 Hz, 1H), 7.07 (d, J = 2.8 Hz, 1H), 6.97 (dd, J = 5.1, 3.6 Hz, 1H), 6.69 (dd, J = 8.9, 2.8 Hz, 1H), 6.49 (d, J = 8.9 Hz, 1H), 5.28 (s, 1H), 4.33–4.14 (m, 2H), 3.69 (s, 3H), 1.26 (t, J = 7.1 Hz, 3H) (Supplemental material, S36). 13C NMR (101 MHz, CDCl3): δ 170.5, 152.5, 141.1, 137.4, 127.2, 125.8, 125.7, 118.5, 114.4, 113.4, 110.9, 62.3, 57.7, 55.9, 14.1 ppm (Supplemental material, S36). HRMS (ESI) m/z calcd. for C15H16BrNNaO3S [M+Na]+: 391.9926; found 391.9910. [α]D20 = −4.8 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IB column, ee = 69% (n-Hexane/ethanol = 95/5, flow rate 0.8 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 9.029 min, tr (minor) = 7.730 min).
Ethyl 2-((2-bromo-4-methoxyphenyl)amino)-2-(furan-2-yl)acetate (4n)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the colorless oil in 82% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.40 (dd, J = 1.8, 0.9 Hz, 1H), 7.07 (d, J = 2.8 Hz, 1H), 6.73 (dd, J = 8.9, 2.8 Hz, 1H), 6.52 (d, J = 8.9 Hz, 1H), 6.40–6.30 (m, 2H), 5.16 (s, 1H), 4.32–4.18 (m, 2H), 3.71 (s, 3H), 1.25 (t, J = 7.1 Hz, 3H) (Supplemental material, S37). 13C NMR (101 MHz, CDCl3): δ 169.7, 152.6, 150.0, 142.9, 137.4, 118.5, 114.4, 113.4, 111.0, 110.8, 108.3, 62.3, 56.1, 56.0, 14.2 ppm (Supplemental material, S37). HRMS (ESI) m/z calcd. for C15H16BrNNaO4 [M+Na]+: 376.0155; found 376.0147. [α]D20 = −8.0 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak AS-H column, ee = 17% (n-Hexane/ethanol = 90/10, flow rate 0.7 mL min−1, λ = 210 nm, T = 20°C, tr (major) = 10.669 min, tr (minor) = 9.456 min).
tert-Butyl-2-(1-((2-bromo-4-methoxyphenyl)amino)-2-ethoxy-2-oxoethyl)-1H-pyrrole-1-carboxylate (4o)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 54% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 7.21 (dd, J = 3.3, 1.8 Hz, 1H), 7.03 (d, J = 2.8 Hz, 1H), 6.74 (dd, J = 8.9, 2.7 Hz, 1H), 6.64 (d, J = 8.9 Hz, 1H), 6.28 (dd, J = 3.4, 1.8 Hz, 1H), 6.09 (t, J = 3.4 Hz, 1H), 5.71 (s, 1H), 4.21 (q, J = 7.1 Hz, 2H), 3.70 (s, 3H), 1.59 (s, 9H), 1.23 (t, J = 7.1 Hz, 3H) (Supplemental material, S38). 13C NMR (101 MHz, CDCl3): δ 170.8, 152.3, 149.4, 138.1, 130.6, 122.7, 118.3, 117.6, 116.8, 115.2, 114.5, 114.4, 113.8, 110.9, 110.3, 84.5, 61.8, 56.0, 55.8, 28.1, 14.3 ppm (Supplemental material, S38). HRMS (ESI) m/z calcd. for C20H25BrN2O5 [M+H]+: 453.1009; found 453.1020. [α]D20 = −4.0 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IF column, ee = 7% (n-Hexane/ethanol = 95/5, flow rate 0.8 mL min−1, λ = 254 nm, T = 20°C, tr (major) = 9.368 min, tr (minor) = 10.075 min).
tert-Butyl 3-(2-ethoxy-1-((3-methoxyphenyl)amino)-2-oxoethyl)-1H-indole-1-carboxylate (4p)
The product was purified via flash column chromatography (n-Hexane/EtOAc = 30:1) to afford the yellow oil in 6% yield at 0.1 mmol scale. 1H NMR (400 MHz, CDCl3): δ 8.15 (d, J = 8.2 Hz, 1H), 7.76 (d, J = 7.8 Hz, 1H), 7.34 (t, J = 7.8 Hz, 1H), 7.31–7.23 (m, 3H), 7.06 (t, J = 7.8 Hz, 1H), 6.29 (dd, J = 17.9, 8.2 Hz, 3H), 6.21 (s, 1H), 5.31 (s, 1H), 4.34–4.09 (m, 2H), 3.73 (d, J = 2.3 Hz, 3H), 1.65 (s, 9H), 1.24–1.20 (t, J = 7.1 Hz, 3H) (Supplemental material, S39). 13C NMR (101 MHz, CDCl3): δ 171.8, 160.8, 149.6, 147.8, 130.2, 128.6, 124.9, 124.5, 122.9, 119.9, 117.4, 115.5, 106.5, 103.8, 99.7, 84.2, 77.4, 62.1, 55.2, 54.1, 29.9, 28.3, 22.9, 14.2 ppm (Supplemental material, S39). HRMS (ESI) m/z calcd. for C24H28N2O5 [M+H]+: 425.2071; found 425.2051. [α]D20 = −5.0 (c 1.0, CHCl3). Enantiomeric excess was determined by chiral HPLC analysis using a Daicel Chiralpak IG column, ee = 54% (n-Hexane/ethanol = 70/30, flow rate 1 mL min−1, λ = 280 nm, T = 20°C, tr (major) = 8.595 min, tr (minor) = 9.883 min).
Supplemental Material
SUPPORTING_INFORMATION – Supplemental material for Enantioselective synthesis of α-amino esters through Petasis borono-Mannich multicomponent reaction of potassium trifluoroborate salts
Supplemental material, SUPPORTING_INFORMATION for Enantioselective synthesis of α-amino esters through Petasis borono-Mannich multicomponent reaction of potassium trifluoroborate salts by Mengnan Tong, Xiang Bai, Xin Meng, Jianfei Wang, Tao Wang, Xingyi Zhu and Bin Mao in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the National Natural Science Foundation of China (No. 21606200) and Zhejiang Provincial Natural Science Foundation of China (LY19B020010) for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.