Abstract
2,4-Dichlorophenylacetic acid is synthesized in high yield via the carbonylation of 2,4-dichlorobenzyl chloride, and various experimental conditions are evaluated. Xylene, bistriphenylphosphine palladium dichloride, tetraethylammonium chloride and sodium hydroxide in solution are added to the reaction system and held at 80 °C under a CO atmosphere. 2,4-Dichlorophenylacetic acid is obtained in a maximum yield of 95%, and a mechanism for 2,4-dichlorobenzyl chloride carbonylation is proposed. The reaction system provides a mild, effective and novel means by which to prepare phenylacetic acid derivatives from their corresponding benzyl chloride derivatives.
Keywords

Introduction
Phenylacetic acid and its derivatives comprise an important class of organic carboxylic acids. They are fundamental building blocks of many pharmaceutical intermediates such as ibuprofen, carbenicillin, diclofenac and spirodiclofen.1,2 Carbonylation and carboxylation are the most widely investigated reactions for the synthesis of phenylacetic acid derivatives. Benzyl halides and their derivatives are used as raw materials, and either sodium cyanide (NaCN) or CO2 is utilized as a C1 source.3–9 However, NaCN is a nitrile and is highly toxic to humans, while the experimental process involving CO2 places a relatively high demand on the electrochemical reactor. The synthesis of phenylacetic acid via direct, metal-catalysed hydrocarboxylation of styrene derivatives has recently been reported.10–12 The disadvantage of using highly reactive metal compounds like Grignard reagents is the need for an anhydrous environment. Currently, the most promising carbonylation method for industrial applications combines the use of a transition metal and phase-transfer catalysis (TM-PTC) chemistry. 13 Herein, a new carbonylation process for the synthesis of phenylacetic acid derivatives from their corresponding benzyl chloride derivatives is reported. A water-soluble palladium catalyst was employed in the presence of a base. The method was facile, mild, and provided carbonylated products in good yields (Scheme 1). Carbonylation with a small amount of CO and a tetraalkylammonium (TAA) salt produced a higher yield of 2,4-dichlorophenylacetic acid (2,4-DCPA) than previously reported methods.

Synthesis of phenylacetic acid derivatives.
Results and discussion
Effect of the solvents
Various solvents were evaluated as the reaction medium for the synthesis of 2,4-DCPA via carbonylation of 2,4-dichlorobenzyl chloride (2,4-DCBC) using the bistriphenylphosphine palladium dichloride (Pd(PPh3)2Cl2) and tetraethylammonium chloride (TEAC) catalytic system. The results are summarized in Table 1. Polar N,N-dimethylformamide (DMF) was not an effective solvent for carbonylation, as 2,4-DCPA was obtained in a yield of only 35% (Table 1, entry 5). However, the reaction produced 2,4-DCPA in maximum yields of up to 95% in a non-polar xylene (DMB) solvent system (Table 1, entry 1). 14 As the results in Table 1 show, less polar solvents lead to better conversions and yields of the carbonylation product (Table 1, entries 1–5).
Effect of the solvents. a

DMF: N,N-dimethylformamide.
Reaction conditions: 2,4-DCBC (0.01 mol), solvent (10 mL), NaOH (4 M, 8 mL), Pd(PPh3)2Cl2 (0.13 mmol), TEAC (0.18 mmol), CO (1.5 MPa), 80°C, 20 h.
Effect of temperature
The reaction yield depended greatly on temperature. As the reaction temperature increased, the extent of conversion clearly increased. However, the catalyst and TAA salt decomposed rapidly at high temperatures, which was detrimental to carbonylation. Therefore, it was necessary to investigate the effect of temperature on the reaction.
The results are shown in Table 2. At 60 °C, 2,4-DCPA was obtained in a moderate yield of 79% (Table 2, entry 1). When the temperature was increased from 60 °C to 80 °C, both the conversion of 2,4-DCBC and the yield of 2,4-DCPA increased (Table 2, entries 1–3). This was due to the higher probability of collision between the reactant molecules. As the reaction temperature increased from 80 °C to 120 °C, the yield decreased from 95% to 36%. This was attributed to the decomposition of Pd(PPh3)2Cl2 and denaturation of TEAC (Table 2, entries 4–7). Thus, the optimal reaction temperature was 80 °C.
Effect of temperature. a
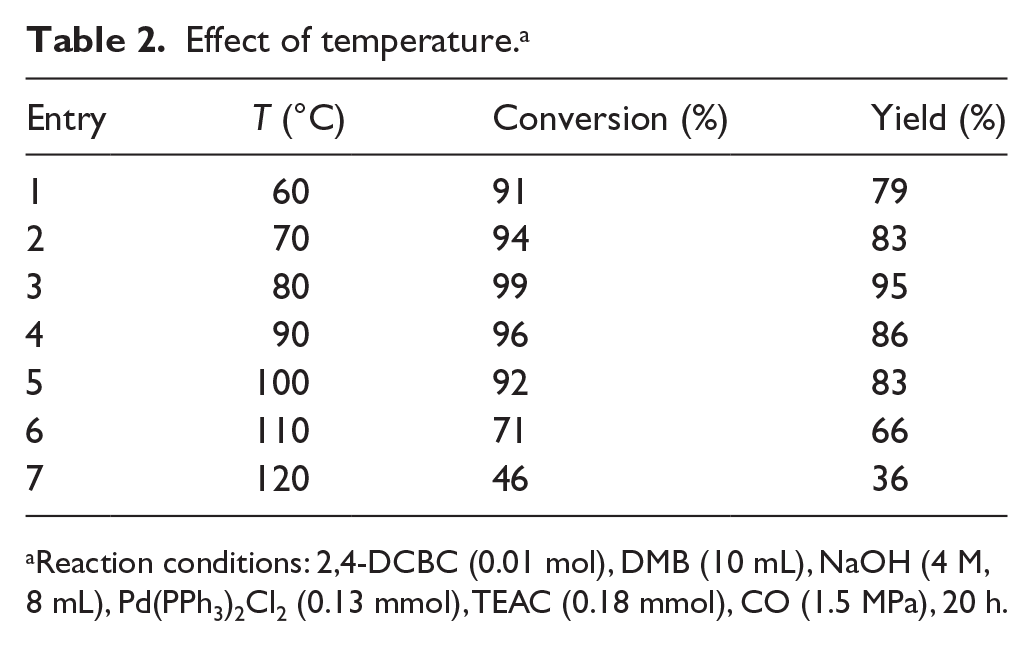
Reaction conditions: 2,4-DCBC (0.01 mol), DMB (10 mL), NaOH (4 M, 8 mL), Pd(PPh3)2Cl2 (0.13 mmol), TEAC (0.18 mmol), CO (1.5 MPa), 20 h.
Effect of the catalysts
The Pd(PPh3)2Cl2 and TEAC catalytic system exhibited the highest activity for 2,4-DCBC carbonylation. The palladium complex contained phosphine ligands, and its catalytic activity in the carbonylation reaction was moderate to high (Table 3, entries 1–5). Under the CO atmosphere, Pd(II) was readily reduced to Pd(0). Thus, only a moderate yield of 2,4-DCPA was obtained from the reaction performed with ligand-free palladium chloride (Table 3, entries 6 and 7).
Effect of the catalysts. a
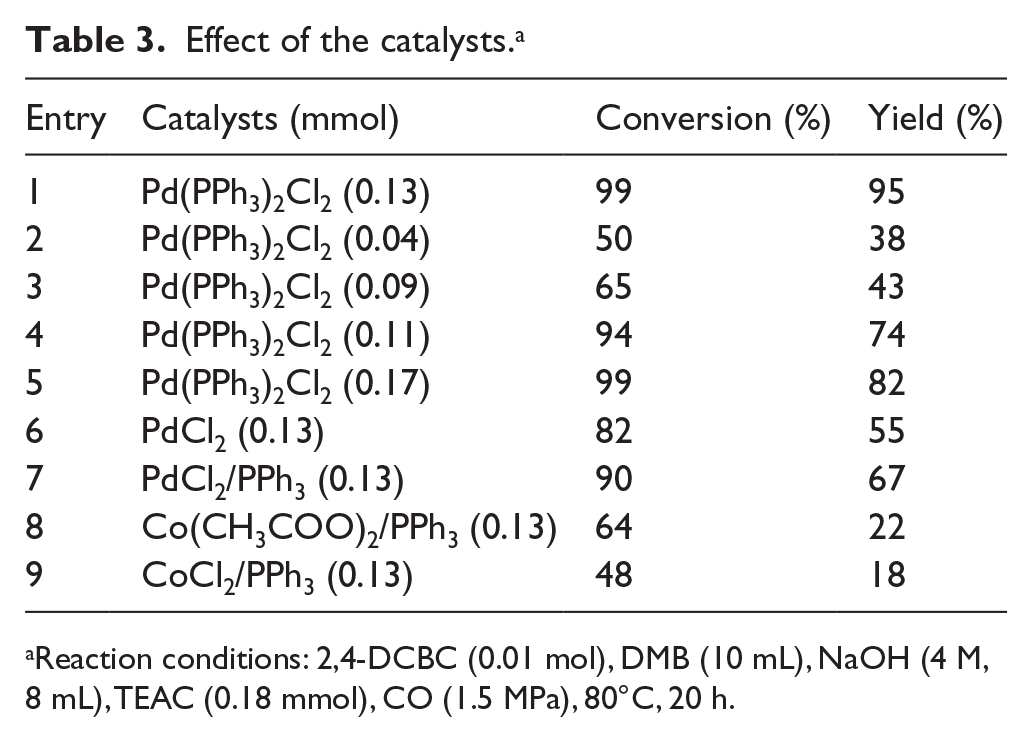
Reaction conditions: 2,4-DCBC (0.01 mol), DMB (10 mL), NaOH (4 M, 8 mL), TEAC (0.18 mmol), CO (1.5 MPa), 80°C, 20 h.
As reported previously, nanoscale Pd colloids can be stabilized by quaternary ammonium salts, which prevent undesired agglomeration and promotes carbonylation.15–22 In this work, Pd ions played the role of a Pd colloid. The activities of Co(CH3COO)2/PPh3 (0.13 mmol) and CoCl2/PPh3 (0.13 mmol) were evaluated for comparison. 2,4-DCPA yields of only 22% and 18% were obtained with Co(CH3COO)2/PPh3 and CoCl2/PPh3, respectively (Table 3, entries 8 and 9). The results indicated that the catalytic effect of Pd catalysts was superior to that of Co catalysts in the carbonylation reaction, and Pd(PPh3)2Cl2 had the highest catalytic activity.
Effect of the TAA salts
TAA salts played an important role in the organic reaction, 23 and the effect of the salt (R4N+X−) on carbonylation was investigated. The results are shown in Table 4. The short-chain TAA salts had higher catalytic efficiencies than the long-chain TAA salts. For example, 2,4-DCPA was obtained in a 95% yield with TEAC, a short-chain TAA salt (Table 4, entry 1). In the presence of hexadecyltrimethylammonium chloride (HTMAC), a long-chain TAA salt, 2,4-DCPA was obtained in a yield of only 58% (Table 4, entry 7). This was because the long-chain TAA salts could be carbonylated by the Pd catalyst. Carbonylation of the long-chain TAA salts competed with 2,4-DCBC carbonylation and reduced the yield of 2,4-DCPA. 24 In addition, the catalytic activity of chloride salts in the carbonylation reaction was higher than that of the bromide salts (Table 4, entries 1 and 2). Most importantly, these results demonstrated that 2,4-DCPA could be produced in excellent yield via carbonylation in the presence of TEAC.
Effect of the TAA salts. a

TEAC: tetraethylammonium chloride; TMAB: tetramethylammonium bromide; TPAB: tetrapropylammonium bromide; DMBAC: benzyltrimethylammonium chloride; TEBAC: benzyltriethylammonium chloride; DTAC: dodecyltrimethylammonium chloride; HTMAC: hexadecyltrimethylammonium chloride.
Reaction conditions: 2,4-DCBC (0.01 mol), DMB (10 mL), NaOH (4 M, 8 mL), Pd(PPh3)2Cl2 (0.13 mmol), TAA salts (0.18 mmol), CO (1.5 MPa), 80°C, 20 h.
Effect of the base
As shown in Table 5, the yield of 2,4-DCPA was 27% at a NaOH concentration of 3 M. When the NaOH concentration was increased to 4 M, the yield of 2,4-DCPA increased sharply to 95% (Table 5, entries 1 and 2). Increasing the NaOH concentration further to 6 M decreased the yield of 2,4-DCPA (Table 5, entries 3 and 4). Potassium hydroxide was also effective, as indicated by a 67% yield of 2,4-DCPA. However, sodium carbonate and potassium carbonate were not efficient bases for carbonylation. Thus, 2,4-DCPA was obtained in low yields of 19% and 30% with Na2CO3 and K2CO3, respectively (Table 5, entries 6 and 7). An excess of base was needed for the carbonylation reaction. When the molar ratio of 2,4-DCBC and NaOH was greater than 1:2, 1:3.2 in particular, a portion of the OH− provided by NaOH was consumed in the synthesis of the products. The remaining OH− reacted with the TAA salt and produced intermediates for the carbonylation reaction. 25
Effect of the base. a

Reaction conditions: 2,4-DCBC (0.01 mol), DMB (10 mL), base (8 mL), Pd(PPh3)2Cl2 (0.13 mmol), TEAC (0.18 mmol), CO (1.5 MPa), 80°C, 20 h.
Evaluation of scope
Under the optimized conditions, the generality of the catalytic system was determined by carbonylating several different benzyl chlorides. The various benzyl chloride derivatives were converted to their corresponding phenylacetic acid derivatives in good yields (Table 6, entries 1–13, Supplemental material). It was noted that only the benzylic position was carbonylated, while the aryl chlorines remained in their original positions. The results indicated the carbonylation reaction was highly regioselective. This arose from the reactivity of the benzylic chlorine atom, which was higher than that of chlorine substituents on the benzene ring.
Evaluation of scope. a

Reaction conditions: material (0.01 mol), DMB (10 mL), NaOH (4 M, 8 mL), Pd(PPh3)2Cl2 (0.13 mmol), TEAC (0.18 mmol), CO (1.5 MPa), 80°C, 20 h.
Reaction mechanism
The mechanism of palladium-catalysed carbonylation of benzyl chlorides is shown in Scheme 2. RX was first transformed into R–Pd–X (A) via the insertion of the Pd complex. R–CO–Pd–X (B) was readily produced from A through oxidative addition in the CO atmosphere. Under basic conditions, ion exchange between X− in

Mechanism of palladium-catalysed carbonylation.
Conclusion
A mild, effective and novel method for the synthesis of 2,4-DCPA was developed using 2,4-DCBC as the starting material, CO as the carbon source and Pd(PPh3)2Cl2 as the catalyst. Results obtained under various experimental conditions showed that carbonylation at 80 °C using xylene (DMB) as the solvent and sodium hydroxide as the base with a Pd(PPh3)2Cl2 dosage of 0.13 mmol produced the highest yield of 2,4-DCPA, which reached 95%. The mechanism demonstrated that R–CO–Pd–X played an important role in the carbonylation process. In summary, this carbonylation system enables the carbonylation of substituted benzyl chlorides and provides a new alternative for preparing the corresponding substituted phenylacetic acids.
Experimental
Materials and instruments
All reagents were used without further purification. Toluene, DMF, isoamyl alcohol, butyraldehyde, diphenyl ether and DMB were obtained from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). All reagents were of analytical reagent (AR)-grade. Catalysts (Reagent Grade [RG]) and PPh3 (RG) in Table 3 were purchased from Xi’an Kaili Catalyst and New Materials Co., Ltd (Xi’an, China). TAA salts (98%) listed in Table 4 were purchased from Shanghai Aladdin Bio-Chem Technology Co., Ltd (Shanghai, China). CO (99.99%) was purchased from a local manufacturer. AR-grade bases (Table 5) and benzyl chlorides (Table 6) were obtained from Sinopharm Chemical Reagent Co., Ltd.
The reaction mixtures were analysed qualitatively via gas chromatography (GC) and compared with standard samples. The analyses were performed on a 7890A GC (Agilent) equipped with a flame ionization detector (FID) and an Agilent DB-1ms capillary column (30 m × 0.25 mm × 0.25 μm). Nitrogen was used as the carrier gas at a flow rate of 0.9 mL min−1. High performance liquid chromatography (HPLC) was used to quantitatively analyse the reaction mixtures. Analysis was carried out with a Prominence LC-20A HPLC (Shimadzu) equipped with a Shimadzu Shim-pack VP-ODS C18 column (250 mm × 4.6 mm, 5 μm) and ultraviolet detector (UVD) using o-dichlorobenzene as an internal standard. Fourier transform infrared (FTIR) measurements were conducted with a Nicolet IS10 spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) using the KBr pellet technique. The spectra were recorded with a resolution of 16 cm−1. Nuclear magnetic resonance (NMR) spectroscopy was conducted with an AV400 NMR spectrometer (Bruker, Billerica, MA, USA). The samples were dissolved in dimethyl sulfoxide-d6 (DMSO-d6), and tetramethylsilane (TMS) was used as the internal standard.
Catalytic experiments
The carbonylation reaction was performed in a 150 mL polytetrafluoroethylene (PTFE)-lined autoclave equipped with a magnetic stir bar. In a typical experiment, the substituted benzyl chloride (0.01 mol), Pd(PPh3)2Cl2 (0.13 mmol), TEAC (0.18 mmol), NaOH (4 M, 8 mL) and DMB (10 mL) were placed in the autoclave. The autoclave was purged three times with N2 and three times with CO, and then heated to 80 °C. During the reaction, the CO pressure was maintained at 1.5 MPa. When the reaction was complete, the autoclave was cooled to room temperature in ice water, and the CO was discharged to atmospheric pressure. The mixture was then adjusted to pH 2 with HCl (12 M), and the solid was collected via filtration and air-dried. The crude product was recrystallised in MeOH/H2O (1:1, v/v) to obtain 2,4-DCPA. The remaining commercially available catalysts were evaluated under the same conditions. The percent conversion and yield were quantified by the method reported by Lei et al. 27
2,4-Dichlorophenylacetic acid (
Supplemental Material
supplementary_material – Supplemental material for An improved method for the synthesis of phenylacetic acid derivatives via carbonylation
Supplemental material, supplementary_material for An improved method for the synthesis of phenylacetic acid derivatives via carbonylation by He Li, Yijun Zhang, Dinghua Liu and Xiaoqin Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Analytical data, FTIR, 1H NMR and 13C NMR spectra.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

