Abstract
Three new aromatic compounds named fistuloates:

Introduction
Cassia is a genus of 600 species of flowering plants (angiosperm) in the family Fabaceae, which is also recognized as Leguminosae. It exists in South American countries, Mexico, South Asia, Sri Lanka and China. 1 Cassia fistula is 10 to 20 m in height and alternates leaves with five to eight pairs of leaflets, 6–15 cm in length, while the lower surface is waxy white. Flowers have hanging axillary racemes which are 80 cm long. The flowers have five yellow-coloured petals. The fruits are narrow, cylindrical pods 30–60 cm in length. The pods are sticky and pulpy, and the pulp is dark brown in colour, sweet in taste and filled with seeds.
The plant extracts of different organs of C. fistula are important sources of secondary metabolites, mainly phenolic compounds. 2 Alkaloids and triterpenes are reported from its flowers, 3 and anthraquinone and dihydroxyanthraquinone are reported from its bark. 4 Aromatic compounds, rhein, steroids and wax have also been isolated from this plant in addition to anthraquinone-bearing skeletons, isovanillic acid (an aromatic acid), stigmasterols, flavon-3-ol derivatives, reducing sugars, saponin and 2,4-dihydroxybenzaldehyde. 5 From its seeds, octacosan-5,8-diol, (+)-catechin, (–)-epicatechin, rhein, glycerides, palmitic acids and stearic acid have been isolated.
Medicinally, different extracts of Cassia species display antimicrobial properties,6,7 for example, its betulinic acid constituent has strong antimicrobial potential and anti-HIV infection properties. It also possesses antipyretic, anti-inflammatory, antitumor, analgesic and antioxidant potential.8–10 Phytotherapeutic studies of the genus Cassia indicated that it has been largely used in folk medicine as an antiseptic, a purgative laxative, a diuretic, and to treat leprosy and different types of ulcers and jaundice. 2 This plant also serves in the treatment of diseases like liver disorders, tuberculosis, skin infections and diabetes.9–11 C. fistula has promising medicinal uses in China for the treatment of fungal diseases, ringworm, diarrhoea and skin infections.12,13 Similarly, plant parts are effective and are used against hypercholesterolaemia and nasal infections. 14 The leaf extract of C. fistula is also reported to possess anti-tussive and wound healing properties.15,16 It has been demonstrated that the C. fistula parts exhibited therapeutic properties against the treatment of hypercholesterolaemia partially due to their fibre and mucilage content. 17 C. fistula is one of the most commonly used plants in Unani and Ayurvedic medicines, and is suggested to be useful in the treatment of haematemesis, pruritus and leucoderm. 18
All these facts motivated us to further explore the hidden chemical constituents of the C. fistula plant. Here, we describe the isolation and characterization of three new compounds from C. fistula and the evaluation of their antioxidant potential.
Results and discussion
Extraction and structural characterization
The crude methanol extract was concentrated and the residue extracted using different solvents with increasing polarity: n-hexane, CH2Cl2, EtOAc (ethyl acetate) and methanol. The results of repeated column chromatography on EtOAc soluble fractions, and experiments performed for isolation showed the presence of three compounds as colourless solids. Their structural formulae were confirmed by different spectroscopic techniques as, infrared (IR), 1H and 13C NMR (nuclear magnetic resonance) spectroscopy, and mass spectrometry. The structures of the compounds are illustrated in Figure 1.
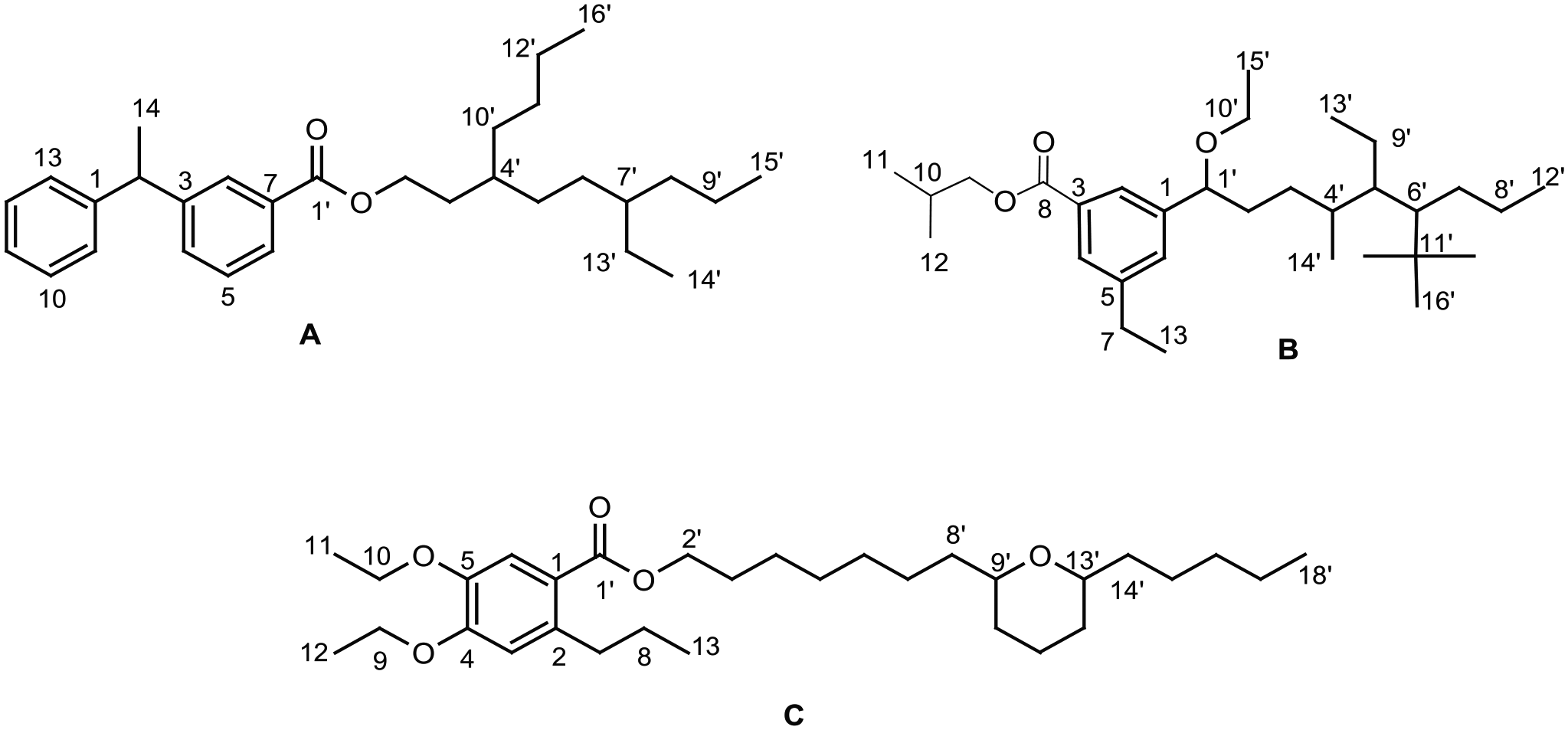
Structural formulae of compounds
Fistuloate
The 1H NMR spectrum recorded for fistuloate
Compound
The 1H NMR spectrum obtained for compound
Fistuloate
Considering the 1H NMR spectrum data for compound
1H and 13C NMR data of compounds

Q represents quaternary carbon.
1H and 13C NMR data of compound

Q represents quaternary carbon.
The mass fragmentation patterns for compounds

Mass spectral fragmentation patterns of compounds
In vitro antioxidant properties of compounds A–C
The results of the evaluation of the DPPH (2,2-diphenyl-1-picrylhydrazyl), ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) and superoxide anion radicals scavenging properties of compounds
Antioxidant activity of compounds

SEM: standard error of mean; DPPH: 2,2-diphenyl-1-picrylhydrazyl; ABTS: 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid).
Number of experiments conducted = 3.
Used as positive control.
It is a fact that oxygen is essential in aerobic life, but an excess of reactive oxygen species is responsible for infections such as inflammation, immune suppression, diabetes, atherosclerosis, neurodegenerative diseases, cancer and others. 19 To overcome such issues, antioxidants are promising agents, functioning through different mechanisms, that is, chelating transition metals, decomposition of peroxides, donation of hydrogen atoms to the free radicals and radical scavenging. The three assays mentioned are used to estimate the antioxidant agents and disclose their possible pathway of biochemical action. 20
Experiment
Plant material and chemicals
The plant material was obtained from Gomal University, Pakistan. Prof. Jamil Khan, Faculty of Agriculture, Gomal University, identified the plant and the voucher was submitted to herbarium of Gomal University. Aluminium thin-layer chromatography (TLC) plates (0.5 mm thick) pre-coated with silica gel 60 F254 (0.2 mm layer thickness; E. Merck, Darmstadt, Germany) were used for TLC to check the purity of the compounds. Column chromatography (CC) was carried out using silica gel of 70–230 mesh (E. Merck). Optical rotation was determined with Ogawa Seiki (OSK) polarimeter (ATAGO NO. 15533 Tokyo, Japan). The IR spectra (ν max, cm−1) were obtained through Jasco-320-A spectrophotometer in CHCl3. HRMS (EI) spectra were recorded using an LTQ-Orbitrap LC-MS spectrometer (Thermo Corporation, USA). NMR spectra were recorded with a Bruker Avance (400 MHz for 1H and 100 MHz for 13C spectrometer; Bruker Corporation, Switzerland). A kit for the antioxidant assay bearing catalogue number CS0790 (Sigma-Aldrich, USA), and the solvents and reagents including dimethyl sulfoxide, nitro blue tetrazolium and DPPH used in this research were of analytical grade (E. Merck).
Extraction from C. fistula and isolation of its constituents
The shade-dried (about 2 months) material of C. fistula was transformed into powdered material of about 3.1 kg, extracted with MeOH (3 × 3.5 L), and then, the solvent was removed through evaporation to afford a greenish gummy crude material of about 98.4 g. This methanolic extract was mixed with MeOH/H2O (dist.) and then distributed into 16.4 g of n-hexane (fraction F1), 18.2 g of CHCl3 (fraction F2), 22.2 g of EtOAc (fraction F3) and 16.6 g of n-butanol (fraction F4). Column, over silica gel, of the soluble fraction of ethyl acetate (22.2 g) and elution with n-hexane/EtOAc, EtOAc/MeOH and MeOH (following increasing polarity order) gave 10 fractions (Fa–Fj). The soluble fraction (Ff) obtained from mixtures of n-hexane/EtOAc at a polarity difference of 4:6 showed a prominent TLC spot in addition to some impurities. Further purification of this fraction through preparative TLC (silica gel) and a mobile phase system composed of n-hexane and EtOAc in a ratio of 2:8, which afforded 23 mg of fistuloate
Anti-oxidant assay (in vitro)
DPPH scavenging properties
This test was conducted by following the protocol described in the literature.21,22 Thus, to a 900-µL solution of DPPH containing the reaction mixture was added further to 100 µL of the samples where concentration of the test samples were 0.5–20 μM. Thorough mixing well of the reaction mixture and keeping under darkness for about 1 h at a temperature of 37 °C, was followed by measurement of the spectrophotometric absorbance at around 517 nm. As a reference/blank, MeOH (3%) was used. Ascorbic acid (Aa) was applied as a positive control. In the experiment, a 900-µL DPPH was employed as a negative control.
ABTS antioxidant activity
This antioxidant test was carried out using an antioxidant Assay Kit (bearing catalogue number #CS0790) in duplicate while using ultrapure H2O. A standard inhibitor such as Trolox, and an ABTS substrate working sample solution was made by addition of 3% of H2O2 (25 µL) solution to 10 mL of ABTS to achieve the reference curve. 10 µL of the standard mentioned previously was added to the wells, followed by addition of 20 µL of myoglobin solution in order to achieve a standard curve that was followed by adding 10 µL of the test sample in an amount of 0.5–20 μM and working solution (20 µL) to the wells of samples under examination. In the second step, to each well 150 µL of the substrate was added, then incubated at around 25 °C for 5 min, followed by adding a stop solution in amount of 100 µL (bearing catalogue S3446) into each well. The endpoint determination was carried out at 405 nm of absorbance using plate reader method. 23
Superoxide anion scavenging activity
The assay was conducted according to the method reported in Sabu and Ramadasan. 24 The reaction test mixture was composed of 0.5 mL of the solution of nitro blue tetrazolium (NBT) (1 M NBT in 100 mM buffer of phosphate, pH 7.4). Also, 0.3 mL of NADH (1 M NADH in phosphate buffer (100 mM), pH = 7.4) and 0.1 mL (0.5–20 μM) of test compounds and ascorbic acid (50 mM phosphate buffer pH 7.4) were mixed. The reaction was started by adding 100 µL of PMS (phosphomethazonesulfate) solution (60 µM PMS in 100 mM of phosphate buffer, pH = 7.4 to the mixture). The tubes were uniformly illuminated with an incandescent visible light for 15 min, and the optical density was measured at 530 nm before and after the illumination. The values (in percentage inhibition) obtained on account of superoxide generated in the control and test samples were measured.
Conclusion
Phytochemical studies of C. fistula were performed through chromatographic and spectral techniques. As a result, three new aromatic compounds were isolated from the ethyl acetate soluble fraction. The three new aromatic compounds, fistolates
Footnotes
Acknowledgements
The author is thankful to the Beijing University of Chemical Technology, Beijing, China, for spectral characterization.
Author’s Note
Masood Afzal is also affiliated to Department of chemistry, University Of Sargodha (Mianwali Campus), Pakistan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
