Abstract
A practical synthetic route to pimavanserin tartrate, in which the target compound was obtained with 99.84% purity and in 46% total yield via a 5-step synthesis starting from 4-hydroxybenzaldehyde and (4-fluorophenyl)methanamine, is reported. The main advantages of the route include inexpensive starting materials, mild reaction conditions and an acceptable overall yield.
Introduction
Pimavanserin has been administered as its tartrate salt (
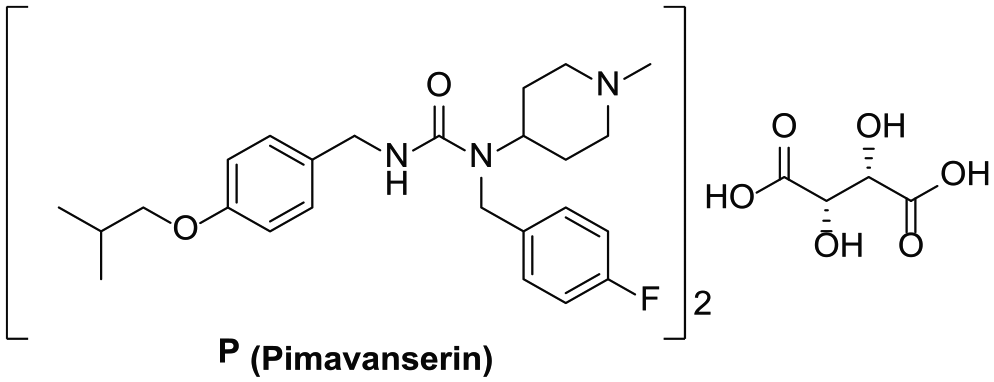
Chemical structure of pimavanserin tartrate (
The reported routes to

Synthetic routes toward pimavanserin tartrate or pimavanserin.
Accordingly, we discuss our attempts to develop a safe and scalable process for the synthesis of
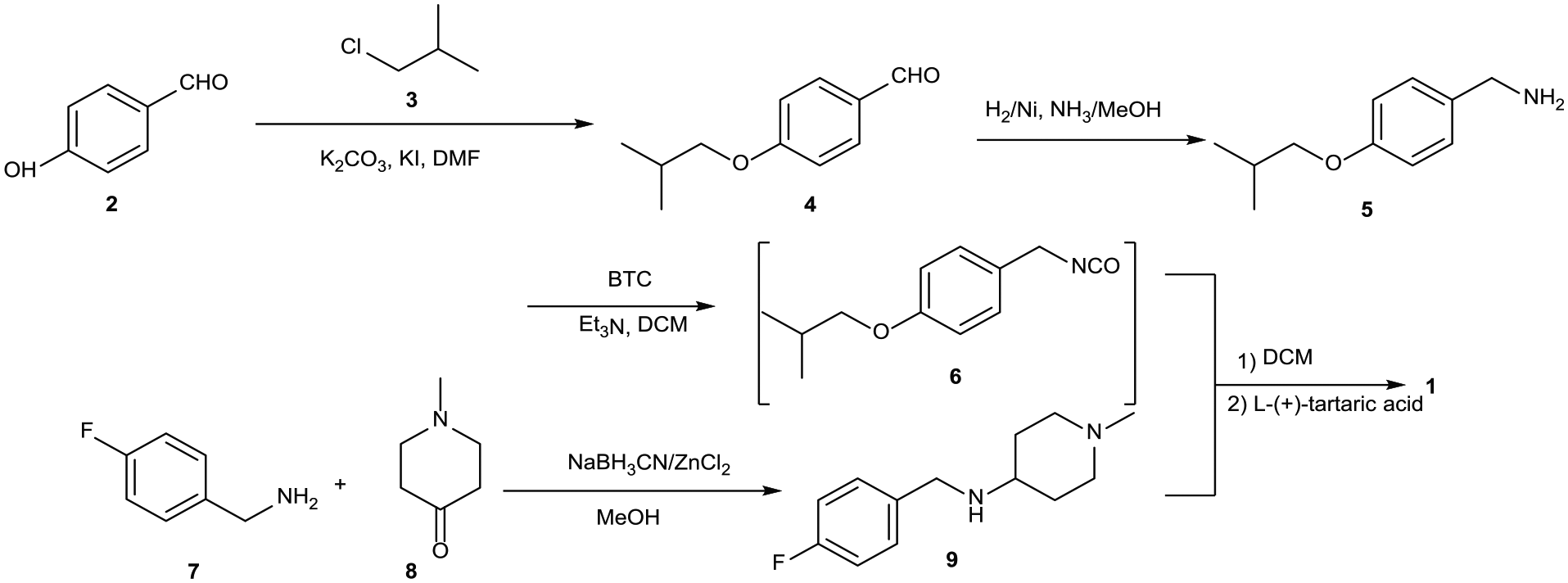
Our an alternative synthetic route to pimavanserin tartrate (
Results and discussion
During the process for the assembly of
Optimization of the reduction reaction a .

Standard conditions:
Based on the quality of
Isolated yield after chromatography.
The main methods to synthesize isocyanates are as follow: amines reacting with phosgene,
3
using carboxylic acids in Curtius rearrangements in the presence of azide,
4
and utilizing N,N’-carbonyldiimidazole (CDI) as an activated amide.
7
Unfortunately, CDI readily formed the self-condensation byproduct

Chemical structures of
Interestingly, it was found that the order of addition had a significant influence on the reaction. When BTC was added to
On this basis, the effects of the amount of BTC, the solvent and the temperature were investigated. As shown in Table 2, the reaction temperature of the first step had a significant influence on the yield. It was found that slow, dropwise addition of a mixture of
Optimization of the amount of BTC, the solvent and the temperature on the yield of pimavanserin a .

BTC: bis(trichloromethyl)carbonate; DCM:dichloromethane; THF:tetrahydrofuran.
Standard conditions:
Temperature for the reaction of
Temperature for the reaction of
An attempt to increase the purity and yield by changing the recrystallization solvents proved successful. It was found that a yield of 70% could be obtained by recrystallization from ethyl acetate (Table 3).
Effect of the solvent on the yield of recrystallization a .

Standard conditions: Pimavanserin (93.6 mmol).
Finally, salt formation process was carried out in ethyl acetate, when ethyl acetate was used as solvent, the reaction mixture was stirred for 1 h at 50 oC, an acceptable yield of 89% could be obtained.
Conclusion
In summary, we have provided an alternative method for the production of pimavanserin tartrate. The modified route avoids several drawbacks of the original synthesis such as removal of highly toxic and expensive reagents. The method also reduces the cost and improves the yield. The procedure involves in five linear steps with an overall yield of 46% and a 99.84% high-performance liquid chromatography (HPLC) purity of the target compound.
Experimental
General
Reagents and solvents were obtained from commercial suppliers and used without further purification. Melting points were determined with a Yanaco apparatus and were uncorrected. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded using a Bruker ARX-400 apparatus (Billerica, MA, USA) with tetramethylsilane (TMS) as an internal standard. Electrospray ionization mass spectrometry (ESI-MS) data were obtained using an AlliAnce+QuattromicroTM Series liquid chromatography-tandem quadrupole mass spectrometer. The reactions were monitored by thin-layer chromatography (TLC; HG/T2354-92, GF254), and compounds were visualized on TLC with ultraviolet (UV) light. HPLC analyses were performed on a Shimadzu LC-20, column, Shimadzu Inertsil-SP C18 (5 µm; 250 mm × 4.6 mm).
Preparation of 4-isobutoxybenzaldehyde (
Preparation of (4-isobutoxyphenyl) methanamine (
Preparation of N-(4-fluorobenzyl)-1-methylpiperidin-4-amine (
Preparation of Pimavanserin tartrate (
Pimavanserin (20 g, 46.8 mmol) in EtOAC (200 mL) was stirred at 50 °C. Then L-(+)-tartaric acid (3.6 g, 24.0 mmol) was added to the mixture which was stirred at 50 oC for 1 h. The mixture was cooled to −15 °C, stirred for 0.5 h and then was filtered. The residue was washed with cold EtOAC (40 mL) and dried at 45–55 °C under vacuum to afford
Supplemental Material
Revised_Supplementary_information – Supplemental material for An improved process for the preparation of pimavanserin tartrate
Supplemental material, Revised_Supplementary_information for An improved process for the preparation of pimavanserin tartrate by Caijiao Wu, Qifan Zhou, Dake Song, Hui Li, Changshun Bao, Xuelong Liu, Xuefei Bao and Guoliang Chen in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
1H spectra, MS spectra, HPLC traces: This material can be found via the “Supplementary Content” section of this article’s web page.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


