Abstract
Fifteen 7,4’-O-modified genistein amino acid derivatives are synthesized through nucleophilic substitution and hydrolysis, followed by condensation with diverse amino acid esters. The antiproliferative activity of all the synthesized compounds is evaluated against three cancer cell lines (MGC-803, HeLa, HCT-116) and one normal cell line (HUVEC) using 5-fluorouracil (5-Fu) as the positive control. The results show that methyl [2-({5-hydroxy-3-[4-(2-{[3-(4-hydroxyphenyl)-1-methoxy-1-oxopropan-2-yl]amino}-2-oxoethoxy)phenyl]-4-oxo-4H-chromen-7-yl}oxy)acetyl] tyrosinate exhibits significant antiproliferative activity against the MGC-803 cell line with an IC50 value of 8.52 µM, and its inhibitory effects on HeLa and HCT-116 cancer cells are stronger than that of the positive control drug 5-Fu.

Introduction
Genistein, also known as 5,7-dihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-one, is a type of isoflavone widely found in leguminous plants. 1 A large number of studies have reported that genistein possesses a wide spectrum of pharmacological activities such as estrogenic, anti-inflammatory, antitumor, antihypertension, antidiabetic activity, and so on.2–6 The extensive antitumor activities of genistein have received significant attention.7–9 Growing evidence suggests that genistein exhibits inhibitory effects on human prostate cancer cells, 10 colon cancer cells, 11 and human ovarian cancer cells, 12 and the mechanism of its action may be associated with its effect on the cell cycle, 13 cellular invasion and metastasis, 14 and apoptosis. 15 However, due to its poor solubility and bioavailability, genistein has not been widely used in the medical field.16,17 In order to overcome this shortcoming and to enhance the anticancer activity of genistein, structural modification based on genistein is required.
Amino acids, as the basic components of proteins, are characterized by diverse structures, good safety, stability and water solubility, as well as improved sustained release and enhanced drug targeting property.18,19 Studies have shown that amino acids have been widely used in prodrug design and can improve oral availability of parent drugs with poor solubility and permeability.20–22 For instance, Lee et al. 23 reported that introducing 11 amino acids into the structure of the antitumor compound DW22822 improved its water solubility, and compounds conjugated with L-valine and L-leucine showed significant antiproliferative activity against human colon cancer cells (SW620). Mattarei et al. 24 showed that the metabolic stability of resveratrol derivatives linked with isoleucine was improved, because the introduction of amino acids could shield them from the first-pass effect through the liver.
In our previous work, we discovered that genistein bearing an alanine chain at the C7 position showed better antiproliferative activity than the parent scaffold genistein against the MGC-803 cell line. 25 In order to further explore the structure-activity relationship, fifteen genistein derivatives with amino acid chains at both C7 and C4’ were synthesized and evaluated for their cytotoxicity against three cancer cell lines (MGC-803, HeLa, HCT-116) and one normal cell line (HUVEC) by using the MTT assay.
Results and discussion
The general synthetic route for 7,4’-O-modified genistein amino acid derivatives

Synthetic route to 7,4’-O-modified genistein amino acid derivatives
All the synthesized compounds were screened for their antitumor activity against three cancer cell lines (MGC-803, HeLa, HCT-116) and one normal cell line (HUVEC) by the MTT assay with 5-fluorouracil (5-Fu) as the positive control,27–30 and the results are showed in Table 1. From Table 1, the following conclusions can be made: (1) genistein modified with an alanine group (
Antiproliferative activity of 7,4’-O-modified genistein amino acid derivatives.
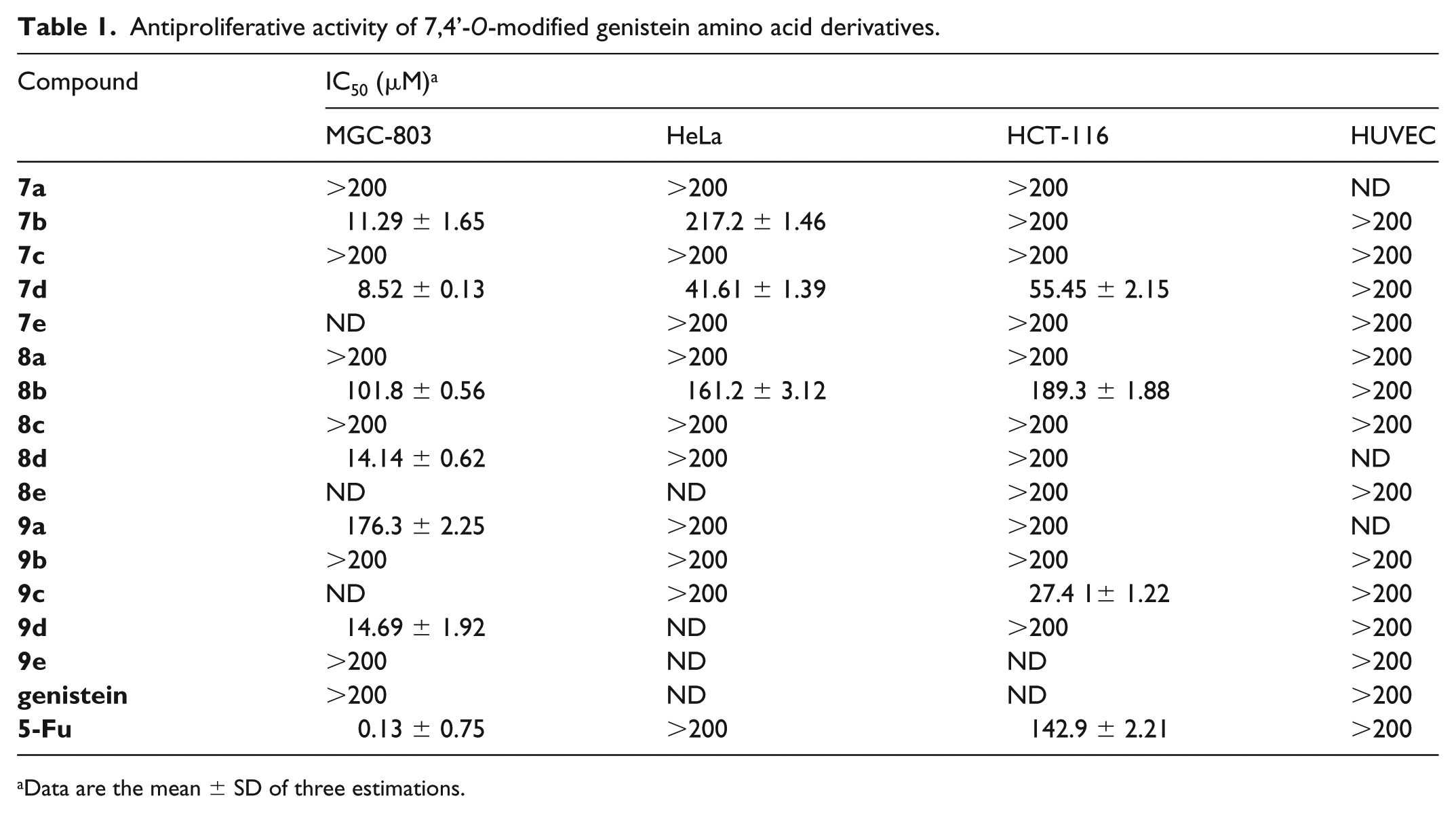
Data are the mean ± SD of three estimations.
In conclusion, 15 novel 7,4’-O-modified genistein amino acid ester derivatives have been synthesized and evaluated for their antiproliferative activity by MTT assays. Genistein bearing a tyrosine group (
Experimental
Commercially available chemicals were obtained from commercial suppliers and used without further purification unless otherwise stated. Proton NMR ( 1 H) and carbon ( 13 C) NMR spectra were recorded on a Bruker Avance III 400 MHz (Ascend TM 500 MHz) NMR spectrometer. The following abbreviations are used for the multiplicities: s: singlet, d: doublet, t: triplet, q: quartet, m: multiplet, br s: broad singlet for proton spectra. Coupling constants (J) are reported in Hertz (Hz). High-resolution mass spectra (HRMS) were recorded on a BRUKER VPEXII spectrometer with electron ionization (EI) and electrospray ionization (ESI) mode unless otherwise stated. Melting points (uncorrected) were determined on an automatic melting point apparatus (ZRD-1) from Tianjin optical instrument factory. Analytical thin-layer chromatography was performed on Polygram SIL G/UV254 plates. Visualization was accomplished with shortwave UV light, or by KMnO4 staining followed by heating. Flash column chromatography was performed using silica gel (200–300 mesh) from Qingdao Haiyang Chemical Co., Ltd, with solvents distilled prior to use.
General procedure for the synthesis of compounds
1–3
To a solution of genistein (1 g, 3.70 mmol) in DMF (20 mL) was added KOH (829 mg, 14.78 mmol) and the mixture was stirred at 60 °C for 4 h. Next, ethyl bromoacetate, ethyl 4-bromobutyrate or ethyl 6-bromohexanoate (14.80 mmol) and KI (61.4 mg, 0.37 mmol) were added. After the reaction was complete (monitored by TLC), the reaction mixture was poured into distilled H2O (80 mL) and then extracted with EtOAc (3 × 30 mL). The combined organic phase was dried over anhydrous Na2SO4 and concentrated in vacuum. The residue was purified by column chromatography on silica gel (PE/EtOAc = 6:1, v/v) to afford the target compounds
Ethyl 2-{4-[7-(2-ethoxy-2-oxoethoxy)-5-hydroxy-4-oxo-4H-chromen-3-yl]phenoxy} acetate (
Ethyl 4-{4-[7-(4-ethoxy-4-oxobutoxy)-5-hydroxy-4-oxo-4H-chromen-3-yl]phenoxy} butanoate (
Ethyl 6-(4-{7-[(6-ethoxy-6-oxohexyl)oxy]H-chromen-3-yl}phenoxy) hexanoate (
General procedure for the synthesis of compounds
4–6
To a stirred solution of compound
General procedure for the synthesis of compounds
7a–9e
To a solution of compound
Methyl (2-{[5-hydroxy-3-(4-{2-[(2-methoxy-2-oxoethyl)amino]H-chromen-7-yl]oxy}acetyl)glycinate (
Methyl (2-{[5-hydroxy-3-(4-{2-[({S}-1-methoxy-1-oxopropan-2-yl)amino]H-chromen-7-yl]oxy}acetyl)-L-alaninate (
Methyl (2-{[5-hydroxy-3-(4-{2-[({S}-1-methoxy-1-oxo-3-phenylpropan-2-yl)amino]H-chromen-7-yl]oxy}acetyl)-L-phenylalaninate (
Methyl (2-{[5-hydroxy-3-(4-{2-[({S}-3-{4-hydroxyphenyl}-1-methoxy-1-oxopropan-2-yl)amino]H-chromen-7-yl]oxy}acetyl)-L-tyrosinate (
Methyl (2-{4-[7-(2-{[(S)-3-{1H-indol-3-yl}-1-methoxy-1-oxopropan-2-yl]amino}-2-oxoethoxy)-5-hydroxy-4-oxo-4H-chromen-3-yl]phenoxy}acetyl)-L-tryptophanate (
Methyl (4-{[5-hydroxy-3-(4-{4-[(2-methoxy-2-oxoethyl)amino]H-chromen-7-yl]oxy}butanoyl)glycinate (
Methyl (4-{[5-hydroxy-3-(4-{4-[({S}-1-methoxy-1-oxopropan-2-yl)amino]H-chromen-7-yl]oxy}butanoyl)-L-alaninate (
Methyl (4-{[5-hydroxy-3-(4-{4-[({S}-1-methoxy-1-oxo-3-phenylpropan-2-yl)amino]H-chromen-7-yl]oxy}butanoyl)-L-phenylalaninate (
Methyl (4-{[5-hydroxy-3-(4-{4-[({S}-3-{4-hydroxyphenyl}-1-methoxy-1-oxopropan-2-yl) amino]H-chromen-7-yl]oxy}butanoyl)-L-tyrosinate (
Methyl (4-{4-[7-(4-{[(S)-3-{1H-indol-3-yl}-1-methoxy-1-oxopropan-2-yl]amino}-4-oxobutoxy)-5-hydroxy-4-oxo-4H-chromen-3-yl]phenoxy}butanoyl)-L-tryptophanate (
Methyl (6-{[5-hydroxy-3-(4-{[6-({2-methoxy-2-oxoethyl}amino)-6-oxohexyl]H-chromen-7-yl]oxy}hexanoyl)glycinate (
Methyl (6-{[5-hydroxy-3-(4-{[6-({[S]-1-methoxy-1-oxopropan-2-yl}amino)-6-oxohexyl]H-chromen-7-yl]oxy}hexanoyl)-L-alaninate (
Methyl (6-{[5-hydroxy-3-(4-{[6-({[S]-1-methoxy-1-oxo-3-phenylpropan-2-yl}amino)-6-oxohexyl]H-chromen-7-yl]oxy}hexanoyl)-L-phenylalaninate (
Methyl (6-{[5-hydroxy-3-(4-{[6-({[S]-3-[4-hydroxyphenyl]-1-methoxy-1-oxopropan-2-yl}amino)-6-oxohexyl]H-chromen-7-yl]oxy}hexanoyl)-L-tyrosinate (
Methyl (6-{4-[7-({6-[({S}-3-[1H-indol-2-yl]-1-methoxy-1-oxopropan-2-yl)amino]-6-oxohexyl}oxy)-5-hydroxy-4-oxo-4H-chromen-3-yl]phenoxy}hexanoyl)-L-tryptophanate (
MTT assay
All the cells lines were seeded in 96-well plates at a volume of 100 μL/well (1 × 104 cells/well) and incubated under a humidified atmosphere of 5% CO2 at 37 °C for 24 h. Then different concentrations (0, 5, 10, 20, 40, 80, 160 μM) of tested compounds were added and incubated for 48 h. After 48 h, the supernatant was discarded, and then DMEM (90 μL) and MTT (10 μL, 5 mg/mL) were added. The plates were incubated for another 4 h. The suspension was discarded and DMSO (110 μL) was added to dissolve the crystal. The absorbance of the solution was measured at 490 nm by a microplate reader. All experiments were performed in triplicate. IC50 values were obtained using Prism software.
Supplemental Material
SI – Supplemental material for Synthesis and cytotoxic activity of 7,4’-O-modified genistein amino acid derivatives
Supplemental material, SI for Synthesis and cytotoxic activity of 7,4’-O-modified genistein amino acid derivatives by Yao-Fu Zeng, Yu-Qin Duan, Lanqing Liao, Xiaokang Long, Cheng Gao and Xianghao Wen in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the “Double First-Class” Discipline Construction Foundation of the University of South China, the Undergraduate Research Learning and Innovative Experiment Project (2018XJXZ191), and the Foundation of Hu’nan Educational Committee (09C830, 18C0476).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
