Abstract
A simple and efficient one-pot multicomponent approach for the synthesis of tertiary arylamines bearing N,N-bis(isoxazol-5-yl)methyl groups is developed through reactions including sequential diprop-3-ynylation of primary amines with propargyl bromide in the presence of calcium hydride in N,N-dimethylformamide, and 1,3-dipolar cycloaddition with nitrile oxides generated in situ from hydroximyl chlorides in DMF-Et3N. This protocol provides advantages such as high regioselectivity, easy operation, and moderate-to-good product yields with a wide substrate scope under mild conditions.
Introduction
Amines and isoxazoles are examples of nitrogen-containing compounds occupying a significant position in many fields such as organic chemistry, natural products, materials science, and pharmaceuticals. A secondary or tertiary amino group is often embedded as a structural motif in various biologically active compounds, which are important intermediates in the synthesis of pharmaceutically active substances, dyes, and fine chemicals. 1 The formation of C–N bonds between primary amines and organic halides has been applied as the major method to prepare secondary and tertiary amines.2–4 Isoxazoles are important five-membered heteroaromatic molecules because of their wide applications in organic synthesis, pharmacy, chemistry, biologically active molecules, and advanced organic materials.5,6 Among various synthetic methods for the synthesis of isoxazoles, the major routes typically involve 1,3-dipolar cycloadditions of alkenes/alkynes and nitrile oxides.7–11 Introduction of the isoxazole scaffold into amine molecules represents a meaningful research area for the exploration of biologically active compounds and pharmaceutical agents. As far as we know, several synthetic methods have been developed for the construction of isoxazolyl-containing primary,12–16 secondary, and tertiary amines.17–21 However, there are very few reports on the synthesis of tertiary arylamines with isoxazolyl groups. 21 Therefore, the development of simple, efficient, and economical protocols for the synthesis of isoxazole-containing tertiary arylamines using readily accessible starting material will be of great use. It is well known that multicomponent reactions (MCRs) are very useful synthetic methods because they allow rapid and convergent construction of complex molecules without the isolation of intermediates.22–24 Moreover, this process is especially useful when the reaction intermediates are unstable and difficult to isolate. Obviously, in order to construct isoxazole scaffolds, the generation of terminal alkynes in situ from suitable precursors, followed by reaction with nitrile oxides in a one-pot process, would avoid the difficulties associated with the volatile nature of terminal alkynes. Herein, we report a highly regioselective one-pot MCR protocol for the facile synthesis of N,N-bis(isoxazol-5-yl)methyl tertiary arylamines involving in situ generation of terminal alkynes, N,N-di(prop-2-yn-1-yl) arylamines via diprop-3-ynylation of primary amines with propargyl bromide and 1,3-dipolar cycloaddition with nitrile oxides under mild reaction conditions.
Results and discussion
Initially, the diprop-3-ynylation conditions, such as the base and solvent, for the reaction of aniline (
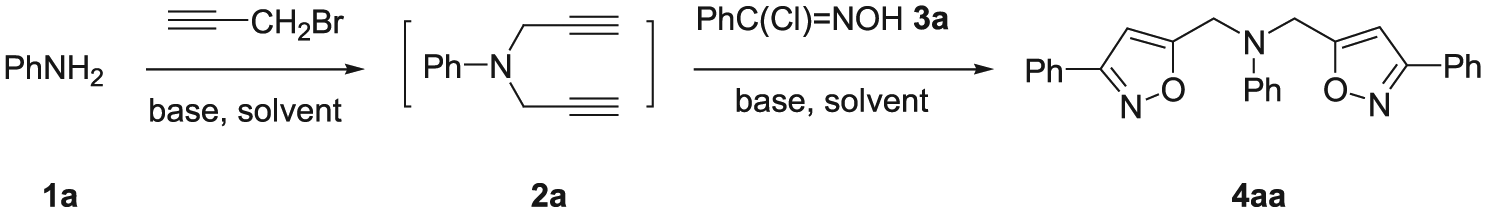
Optimization of the one-pot reaction from aniline
Optimization of the diprop-2-ynylation conditions. a

DMF: N,N-dimethylformamide; DMSO: dimethylsulfoxide; THF: tetrahydrofuran; DIPEA: N,N-diisopropylethylamine.
Unless otherwise noted, all reactions were performed with 1.0 mmol of aniline, 4.0 mmol of propargyl bromide, and 5 mL of solvent at room temperature.
Isolated yield after column chromatography, based on aniline.
Cited in Ji et al. 25
Second, after completion of the diprop-3-ynylation, the excess propargyl bromide was removed by vacuum distillation. Without further isolation and purification of intermediate
The structure of
Finally, the optimized reaction conditions were extended to a variety of primary aromatic amines and hydroximyl chlorides with different substituents (Scheme 2), and the corresponding results were summarized in Table 2. As observed in Table 2, for most of the examined substrates, the reactions were performed smoothly and the corresponding tertiary arylamines containing N,N-bis(isoxazol-5-yl)methyl groups were obtained. It is noteworthy that no significant difference in reactivity was observed for the examined arylamines substituted with 3-Me, 2-Me, and 4-Cl groups on the benzene rings (Table 2, entries 5–15). In general, aromatic nitrile oxides bearing electron-withdrawing groups such as chloro, bromo, and nitro (

Access to N,N-bis(isoxazol-5-yl)methyl tertiary arylamines
Sequential transformation of various arylamines and chlorooximes into N,N-bis(isoxazol-5-yl)methyl tertiary arylamines

All reactions were performed in 5 mL of DMF with 1.0 mmol of primary aromatic amine, 4.0 mmol of propargyl bromide, and 3.0 mmol of the chlorooxime.
Isolated yield based on the primary aromatic amine after column chromatography.
Conclusion
In summary, a facile and efficient, one-pot method for the preparation of N,N-bis(isoxazol-5-yl)methyl tertiary arylamines from primary amines, propargyl bromide, and hydroximyl chlorides has been developed. The procedure does not require isolation of the N,N-dipropargylated arylamine intermediates, and has considerable advantages in terms of its use of easily available substrates, its mild reaction conditions, its simple operation, and the moderate to good yields obtained.
Experimental
Melting points were measured with a Beijing-Taike X-4 apparatus and were uncorrected. 1H NMR and 13C NMR spectra were recorded on an Avance-Bruker 400 MHz NMR spectrometer, operating at 400 and 100 MHz, respectively. Chemical shifts are reported in ppm relative to TMS or the deuterated solvent as the internal reference. Fourier transform infrared (FTIR) analyses were performed with a PerkinElmer SP One FTIR spectrophotometer. Microanalyses were performed with a Carlo Erba 1106 Elemental Analyzer. Hydroximyl chlorides
Preparation of N,N-bis(isoxazol-5-yl)methyl tertiary arylamines 4aa–di ; general procedure
To a stirred solution of primary aromatic amine
N,N-Bis[(3-phenylisoxazol-5-yl)methyl]aniline (4aa )
Yellow solid, m.p. = 112–113 °C. 1H NMR (400 MHz, CDCl3): δ 7.84–7.82 (m, 5H), 7.50–7.48 (m, 8H), 7.33 (s, 2H), 6.95 (s, 2H), 4.61 (s, 4H). 13C NMR (100 MHz, DMSO-d6): δ 169.85, 162.17, 144.93, 132.19, 130.67, 129.32, 128.90, 128.17, 127.05, 126.08, 102.34, 48.23. IR (KBr): ν 3050, 2965, 1607, 1466, 1368, 1269, 1136, 1065, 742 cm−1. Anal. calcd for C26H21N3O2: C, 76.64; H, 5.19; N, 10.31; found: C, 76.72; H, 5.29; N, 10.35%.
N,N-Bis[(3-(o-tolyl)isoxazol-5-yl)methyl]aniline (4ab )
Yellow solid, m.p. = 53–55 °C. 1H NMR (400 MHz, CDCl3): δ 7.44–7.34 (m, 4H), 7.26–7.13 (m, 7H), 7.06 (dd, J = 8.6, 2.4 Hz, 1H), 6.94 (d, J = 8.6 Hz, 1H), 6.23 (s, 2H), 4.44 (s, 4H), 2.33 (s, 6H). 13C NMR (100 MHz, CDCl3): δ 168.29, 163.05, 144.29, 136.87, 131.28, 131.13, 130.61, 129.42, 128.49, 127.72, 126.04, 125.14, 104.26, 48.46, 21.10. IR (KBr): ν 3035, 2972, 2869, 1602, 1488, 1384, 1291, 1125, 751 cm−1. Anal. calcd for C28H25N3O2: C, 77.25; H, 5.75; N, 9.65; found: C, 77.32; H, 5.89; N, 9.73%.
N,N-Bis[(3-(m-tolyl)isoxazol-5-yl)methyl]aniline (4ac )
Yellow solid, m.p. = 83–84 °C. 1H NMR (400 MHz, CDCl3): δ 7.57–7.51 (m, 4H), 7.30–7.20 (m, 6H), 6.87–6.82 (m, 3H), 6.43 (s, 2H), 4.72 (s, 4H), 2.35 (s, 6H). 13C NMR (100 MHz, CDCl3): δ 169.86, 162.68, 147.13, 138.68, 130.90, 129.60, 128.84, 128.68, 127.47, 124.00, 119.09, 113.59, 100.79, 47.48, 21.37. IR (KBr): ν 3033, 2975, 2872, 1605, 1490, 1386, 1290, 1127, 757, 698 cm−1. Anal. calcd for C28H25N3O2: C, 77.25; H, 5.75; N, 9.65; found: C, 77.38; H, 5.90; N, 9.71%.
N,N-Bis{[3-(3-methoxyphenyl)isoxazol-5-yl]methyl}aniline (4ad )
Yellow solid, m.p. = 65–67 °C. 1H NMR (400 MHz, CDCl3): δ 7.29–7.04 (m, 8H), 6.83–6.73 (m, 5H), 6.55 (s, 2H), 4.69 (s, 4 H), 3.70 (s, 6H). 13C NMR (100 MHz, CDCl3): δ 169.19, 161.21, 158.34, 147.18, 131.19, 129.53, 128.86, 124.16, 119.25, 117.54, 115.30, 113.84, 104.10, 55.68, 47.50. IR (KBr): ν 3430, 3045, 2927, 2848, 1605, 1508, 1248, 1178, 1030, 897, 752, 694 cm−1. Anal. calcd for C28H25N3O4: C, 71.96; H, 5.35; N, 8.99; found: C, 71.85; H, 5.46; N, 8.92%.
N,N-Bis[(3-phenylisoxazol-5-yl)methyl]-3-methylaniline (4ba )
Yellow solid, m.p. = 145–146 °C. 1H NMR (400 MHz, CDCl3): δ 7.78–7.75 (m, 5H), 7.44–7.43 (m, 9H), 6.47 (s, 2H), 4.50 (s, 4H), 2.52 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 169.43, 162.42, 146.23, 135.69, 133.91, 130.83, 130.11, 128.90, 127.59, 126.83, 125.88, 122.05, 101.53, 48.52, 19.21. IR (KBr): ν 3031, 2972, 2870, 1604, 1495, 1385, 1288, 1125, 862, 765, 700 cm−1. Anal. calcd for C27H23N3O2: C, 76.94; H, 5.50; N, 9.97; found: C, 76.82; H, 5.64; N, 9.91%.
N,N-Bis{[(3-(o-tolyl)isoxazol-5-yl)]methyl}-3-methylaniline (4bb )
Yellow solid, m.p. = 62–63 °C. 1H NMR (400 MHz, CDCl3): δ 7.73–7.70 (m, 2H), 7.32–7.20 (m, 8H), 6.78 (s, 2H), 6.33 (s, 2H), 4.66 (s, 4H), 2.44 (s, 6H), 2.34 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 169.04, 163.12, 146.21, 136.80, 133.47, 131.10, 130.93, 129.53, 129.42, 128.86, 126.00, 125.05, 117.12, 113.57, 103.51, 47.65, 20.56, 19.21. IR (KBr): ν 3040, 2978, 2875, 1603, 1489, 1385, 1287, 1122, 857, 768 cm−1. Anal. calcd for C29H27N3O2: C, 77.48; H, 6.05; N, 9.35; found: C, 77.58; H, 6.16; N, 9.43%.
N,N-Bis{[(3-(m-tolyl)isoxazol-5-yl)methyl]-3-methylaniline (4bc )
Yellow solid, m.p. = 86–87 °C. 1H NMR (400 MHz, CDCl3): δ 7.51–7.45 (m, 5H), 7.25–7.07 (m, 6H), 6.86–6.78 (m, 1H), 6.37 (s, 2H), 4.38 (s, 4H), 2.43 (s, 3H), 2.30 (s, 6H). 13C NMR (100 MHz, CDCl3): δ 169.27, 162.52, 144.55, 138.67, 136.07, 131.43, 130.86, 128.81, 128.70, 127.58, 127.41, 126.23, 123.98, 122.07, 101.60, 48.49, 21.37. IR (KBr): ν 3037, 2974, 2883, 1606, 1491, 1382, 1285, 1126, 865, 772, 765 cm−1. Anal. calcd for C29H27N3O2: C, 77.48; H, 6.05; N, 9.35; found: C, 77.60; H, 6.14; N, 9.44%.
N,N-Bis[(3-phenylisoxazol-5-yl)methyl]-2-methylaniline (4ca )
Yellow solid; m.p. = 127–128 °C. 1H NMR (400 MHz, CDCl3): δ 7.76–7.53 (m, 4H), 7.52–7.51 (m, 1H), 7.43–7.42 (m, 6H), 7.21 (d, J = 1.8 Hz, 1H), 7.08 (dd, J = 8.6, 2.0 Hz, 1H), 6.93 (d, J = 8.4 Hz, 1H), 6.38 (s, 2H), 4.33 (s, 4H), 2.41 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 169.42, 162.37, 146.34, 136.42, 131.27, 130.94, 130.41, 130.09, 128.90, 126.81, 126.60, 123.87, 101.55, 49.03, 17.96. IR (KBr): ν 3033, 2977, 2886, 1604, 1500, 1384, 1285, 1127, 745 cm−1. Anal. calcd for C27H23N3O2: C, 76.94; H, 5.50; N, 9.97; found: C, 76.85; H, 5.62; N, 8.90%.
N,N-Bis{[3-(m-tolyl)isoxazol-5-yl]methyl}-2-methylaniline (4cc )
Yellow solid, m.p. = 57–58 °C. 1H NMR (400 MHz, CDCl3): δ 7.73–7.70 (m, 2H), 7.55–7.52 (m, 3H), 7.32 (t, J = 7.6 Hz, 2H), 7.23 (dd, J = 9.9, 4.9 Hz, 3H), 7.08 (dd, J = 8.5, 2.4 Hz, 1H), 6.93 (d, J = 8.5 Hz, 1H), 6.37 (s, 2H), 4.33 (s, 4H), 2.41 (s, 3H), 2.39 (s, 6H). 13C NMR (100 MHz, CDCl3): δ 169.27, 162.47, 146.39, 138.67, 131.38, 131.25, 130.68, 129.23, 128.95, 128.86, 128.82, 127.39, 126.59, 123.95, 101.60, 49.02, 21.37, 19.20. IR (KBr): ν 3033, 2975, 2887, 1604, 1490, 1384, 1282, 1122, 862, 770, 745 cm−1. Anal. calcd for C29H27N3O2: C, 77.48; H, 6.05; N, 9.35; found: C, 77.61; H, 6.16; N, 9.47%.
N,N-Bis{[3-(3-methoxyphenyl)isoxazol-5-yl]methyl}-2-methylaniline (4cd )
Yellow solid, m.p. = 65–66 °C. 1H NMR (400 MHz, CDCl3): δ 7.32 (d, J = 8.9 Hz, 3H), 7.23–7.21 (m, 4H), 7.11 (d, J = 7.5 Hz, 1H), 7.04 (t, J = 7.1 Hz, 2H), 6.89 (dd, J = 8.9, 3.1 Hz, 2H), 6.56 (s, 2H), 4.39 (s, 4H), 3.78 (s, 6H), 2.43 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 169.17, 160.98, 158.33, 147.74, 134.46, 131.30, 130.94, 128.82, 126.62, 125.27, 124.17, 122.69, 117.44, 115.23, 104.78, 55.66, 49.14, 18.03. IR (KBr): ν 3028, 2951, 1602, 1467, 1238, 1031, 823, 647 cm−1. Anal. calcd for C29H27N3O4: C, 72.33; H, 5.65; N, 8.73; found: C, 72.16; H, 5.77; N, 8.85%.
N,N-Bis{[3-(4-chlorophenyl)isoxazol-5-yl]methyl}-2-methylaniline (4ce )
Yellow solid, m.p. = 123–124 °C. 1H NMR (400 MHz, CDCl3): δ 7.73–7.71 (m, 2H), 7.66 (d, J = 8.8 Hz, 4 H), 7.43–7.40 (m, 2H), 7.38 (d, J = 8.4 Hz, 4H), 6.34 (s, 2H), 4.36 (s, 4H), 2.45 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 170.18, 161.37, 147.90, 136.05, 131.54, 130.93, 129.14, 128.85, 128.04, 126.68, 125.33, 122.32, 101.25, 49.28, 19.20. IR (KBr): ν 3036, 2956, 2788, 1604, 1495, 1386, 1235, 1028, 905, 834, 760, 705 cm−1. Anal. calcd for C27H21N3Cl2O2: C, 66.13; H, 4.32; N, 8.57; found: C, 66.25; H, 4.41; N, 8.49%.
N,N-Bis{[3-(4-bromophenyl)isoxazol-5-yl]methyl}-2-methylaniline (4cf )
Yellow solid, m.p. = 132–133 °C. 1H NMR (400 MHz, CDCl3): δ 7.73–7.70 (m, 2H), 7.60–7.50 (m, 8H), 7.05 (dd, J = 8.0, 19.6 Hz, 2H), 6.34 (s, 2H), 4.35 (s, 4H), 2.45 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 170.21, 161.43, 147.90, 132.20, 131.54, 130.92, 128.56, 127.81, 126.68, 125.34, 124.32, 122.32, 101.21, 49.29, 19.20. IR (KBr): ν 3036, 2956, 2788, 1604, 1495, 1382, 1233, 1073, 905, 832, 757, 695 cm−1. Anal. calcd for C27H21N3Br2O2: C, 55.98; H, 3.65; N, 7.25; found: C, 55.85; H, 3.76; N, 7.16%.
N,N-Bis{[3-(4-nitrophenyl)isoxazol-5-yl]methyl}-2-methylaniline (4cg )
Yellow solid, m.p. = 153–154 °C. 1H NMR (400 MHz, CDCl3): δ 8.27 (d, J = 8.8 Hz, 4H), 7.73–7.68 (m, 4H), 7.43 (d, J = 8.8 Hz, 4H), 6.45 (s, 2H), 4.37 (s, 4H), 2.51 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 170.23, 162.33, 148.34, 147.93, 134.45, 131.58, 130.96, 128.85, 128.04, 126.69, 124.33, 122.35, 101.28, 49.31, 19.29. IR (KBr): ν 3072, 2935, 2855, 1604, 1510, 1495, 1445, 1382, 1345, 1135, 1108, 925, 850 cm−1. Anal. calcd for C27H21N5O6: C, 63.40; H, 4.14; N, 13.69; found: C, 63.25; H, 4.26; N, 13.57%.
4-Chloro-N,N-bis[(3-benzylisoxazol-5-yl)methyl]aniline (4dh )
Yellow solid, m.p. = 97–99 °C. δ 7.35–7.33 (m, 6H), 7.19–7.16 (m, 4H), 7.08 (d, J = 8.6 Hz, 2H), 6.75 (d, J = 8.6 Hz, 2H), 6.30 (s, 2H), 4.63 (s, 4H), 4.29 (s, 4H). 13C NMR (100 MHz, CDCl3): δ 170.27, 161.52, 147.96, 134.66, 131.15, 129.12, 128.73, 127.88, 123.00, 115.17, 101.22, 47.22, 32.42. IR (KBr): ν 3034, 2970, 2785, 1603, 1490, 1235, 1026, 906, 835, 760, 699 cm−1. Anal. calcd for C28H24ClN3O2: C, 71.56; H, 5.15; N, 8.94; found: C, 71.42; H, 5.24; N, 8.86%.
4-Chloro-N,N-bis[(3-propylisoxazol-5-yl)methyl]aniline (4di )
Yellow liquid. δ 7.56 (d, J = 8.8 Hz, 2H), 7.01 (d, J = 8.8 Hz, 2H), 6.26 (s, 2H), 4.38 (s, 4H), 2.37 (t, J = 7.2 Hz, 4H), 1.68–1.67 (m, 4H), 1.11 (t, J = 7.2 Hz, 6H). 13C NMR (100 MHz, CDCl3): δ 169.67, 160.92, 146.80, 132.46, 128.94, 115.22, 100.76, 47.02, 30.07, 19.78, 13.89. IR (film): ν 3034, 2962, 2932, 1608, 1455, 1016, 760, 698 cm−1. Anal. calcd for C18H24ClN3O2: C, 61.79; H, 6.91; N, 12.01; found: C, 61.93; H, 6.80; N, 12.11%.
Supplemental Material
supplemental_material – Supplemental material for One-pot synthesis of novel N,N-bis(isoxazol-5-yl)methyl tertiary arylamines via sequential diprop-3-ynylation and 1,3-dipolar cycloaddition from primary amines
Supplemental material, supplemental_material for One-pot synthesis of novel N,N-bis(isoxazol-5-yl)methyl tertiary arylamines via sequential diprop-3-ynylation and 1,3-dipolar cycloaddition from primary amines by Xiao-Lan Zhang, Mei-Hong Wei, Shou-Ri Sheng and Xiao-Ling Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to the Research Program of Jiangxi Province Department of Education (Nos GJJ170934, GJJ160289, and GJJ11380), the Opening Foundation of National Research Center for Carbohydrate Synthesis (No. GJDTZX-KF-201414), and the Opening Foundation of the Key Laboratory of Functional Small Organic Molecules of Ministry of Education (No. KLFS-KF-201411) for the financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
