Abstract
A simple strategy to afford trifluoromethyl-containing bendamustine hydrochloride in 34% overall via nine simple steps including substitution, selective reduction, N-acylation, cyclization, esterification, nitro-reduction, N-dihydroxyethylation, chlorination, and acid-catalyzed hydrolysis from commercially available 2,4-dinitrochlorobenzene is described. The structures of the intermediates and target product are established on the basis of infrared, nuclear magnetic resonance, and high resolution mass spectrometer. Moreover, the structure of target product is also confirmed by X-ray crystal analysis, and further studies indicate that the existence of intermolecular O–H···Cl and N–H···Cl hydrogen bonds are effective in stabilization of the crystal structure.
Introduction
Bendamustine hydrochloride (Figure 1(a)) is a bifunctional molecule that combines the alkylating activity of a bis(2-chloroethyl)amine moiety and the antimetabolite activity of a benzimidazole group.1–5 It causes DNA damage that is thought to lead to cell death, including inhibition of mitotic checkpoints and induction of mittic catastrophe.6–8 In the 1960s, bendamustine hydrochloride was designed with the aim of creating a compound which possesses similar properties but less toxicity than nitrogen mustard agents. 9 This alkylating agent has been used against a number of malignancies since the 1970s in Germany. 10 It was approved by the US Food and Drug Administration (FDA) for the treatment of chronic lymphocytic leukemia in March 2008 and for rituximab-refractory indolent B-cell non-Hodgkin lymphoma (NHL) in October 2008.9,11,12

The chemical structures of (a) bendamustine hydrochloride and (b) trifluoromethylated bendamustine hydrochloride.
The small and highly electronegative fluorine atom can play a significant role in medicinal chemistry, biochemistry, and agrochemicals.13,14 Around 20%–30% of medicines contain at least one fluorine atom. 15 Among the various fluorine-containing groups, the trifluoromethyl group (CF3) has received significant attention as its introduction into pharmaceuticals has a major impact on the physical and chemical properties of the molecule such as lipophilicity, pharmacokinetics, and the binding properties of molecules.16–20 Currently, the late-stage introduction of CF3 in drug-like compounds is a well-known practice in medicinal chemistry and widely used in the treatment of diseases.21,22 For example, lansoprazole and omeprazole are popular proton pump inhibitors (PPIs) used as anti-ulcer drugs. 23 The difference between these drugs is that lansoprazole contains a trifluoromethyl group, which can significantly improve drug stability and help to enhance its inhibitory effect on gastric acid secretion mechanism, thereby significantly improving the effect of clinical disease control.24,25
In this context, in view of the importance of bendamustine hydrochloride as an anti-tumor agent as well as the positive effect of CF3 in organic molecules, it would be meaningful to introduce a CF3 group on bendamustine hydrochloride (Figure 1(b)). In our continuing efforts for preparing important bioactive compounds,26 herein, using 2,4-dinitrochlorobenzene as a raw material, trifluoromethyl-containing bendamustine hydrochloride has been designed and synthesized by a series of organic synthesis steps including substitution, selective reduction, N-acylation, cyclization, esterification, nitro-reduction, N-dihydroxyethylation, chlorination, and acid-catalyzed hydrolysis (Scheme 1). This synthetic route involves simple operations and mild reaction conditions affording a 34% total yield.
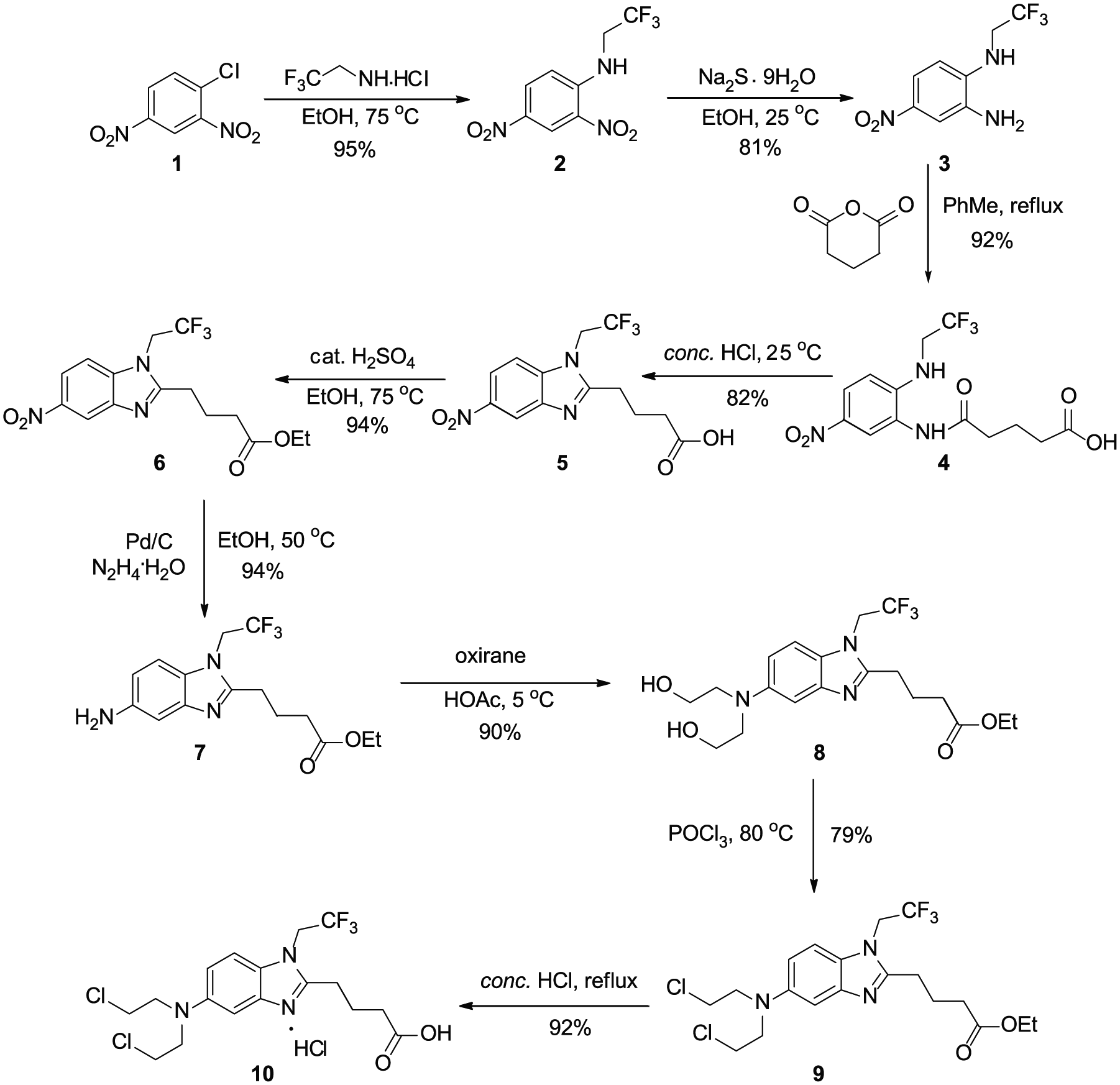
The synthetic pathway to trifluoromethylated bendamustine hydrochloride
Results and discussion
In order to introduce a trifluoromethyl group into bendamustine hydrochloride, our investigation began with the conversion of 2,4-dinitrochlorobenzene (
With the desired product
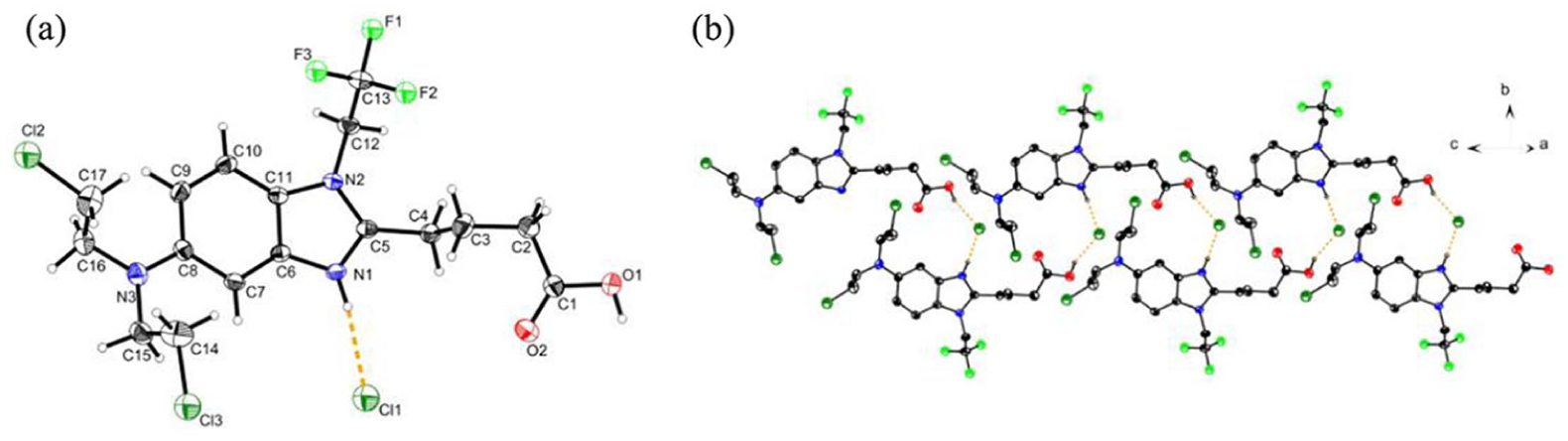
(a) The molecular structure of trifluoromethylated bendamustine hydrochloride
Compound 10 crystallizes in the P21/c space group and its molecular structure contains one protonated trifluoromethyl-containing bendamustine cation and one free chlorine anion. Furthermore, the dihedral angle between the benzene ring and imidazole ring is 3.4(86)°, indicating that these rings are basically coplanar. Intermolecular O(1)–H(1O)···Cl(1) and N(1)–H(1N)···Cl(1) hydrogen bonds (Table 1) are formed between the free chlorine anions and trifluoromethyl-containing bendamustine and carboxyl carboxylic cations, and link the molecules forming a zig-zag chain along the c-axis, which is effective in stabilization of the crystal structure (Figure 2(b)).
Distances and angles of possible weak interactions for compound

Symmetry codes: ax,–y + 3/2, z − 1/2.
Conclusion
In summary, using bendamustine hydrochloride as an original drug, trifluoromethyl-containing bendamustine hydrochloride was designed and synthesized. The target compound
Experimental
General information
Unless noted otherwise, all the reagents were commercial available and were used without further purification. Column chromatography was performed on EMD Silica Gel 60 (200–300 Mesh) using a forced flow of 0.5–1.0 bar. Melting points were measured with an X4-A microscopic melting point apparatus. Nuclear magnetic resonance (NMR) spectra were recorded on a 300 MHz spectrometer with chemical shifts reported in ppm (in CDCl3 or Dimethyl sulfoxide-d6 (DMSO-d6), with TMS as the internal standard). High resolution mass spectrometer (HRMS) were obtained on a 6540 Ultra High Definition (UHD) Accurate-Mass Quadrupole Time-of-Flight (Q-TOF) liquid chromatography/mass spectrometry (LC/MS). Infrared (IR) spectra were recorded using a Thermo fisher Nicolet IS50 spectrometer. X-Ray single crystal diffraction data were recorded on a Bruker Smart APEX Ⅱ CCD.
Preparation of 2,4-dinitro-N-(2,2,2-trifluoroethyl)aniline (2 )
A mixture of 2,4-dinitrochlorobenzene (compound
Preparation of 4-nitro-N1-(2,2,2-trifluoroethyl)benzene-1,2-diamine (3 )
A mixture of compound
Preparation of 5-({5-nitro-2-[(2,2,2-trifluoroethyl)amino]phenyl}amino)-5-oxopentanoic acid (4 )
Glutaric anhydride (12.55 g, 110 mmol) and compound
Preparation of 4-[5-nitro-1-(2,2,2-trifluoroethyl)-1H-benzo[d]imidazol-2-yl]butanoic acid (5 )
Compound
Preparation of ethyl 4-[5-nitro-1-(2,2,2-trifluoroethyl)-1H-benzo[d]imidazol-2-yl]butanoate (6 )
Compound
Preparation of ethyl 4-[5-amino-1-(2,2,2-trifluoroethyl)-1H-benzo[d]imidazol-2-yl]butanoate (7 )
A mixture of 10% dry palladium on carbon (0.80 g) and compound
Preparation of ethyl 4-{5-[bis(2-hydroxyethyl)amino]-1-(2,2,2-trifluoroethyl)-1H-benzo[d]imidazol-2-yl}butanoate (8 )
Compound
Preparation of ethyl 4-{5-[bis(2-chloroethyl)amino]-1-(2,2,2-trifluoroethyl)-1H-benzo[d]imidazol-2-yl}butanoate (9 )
A mixture of POCl3 (20 mL) and compound
Preparation of 4-{5-[bis(2-chloroethyl)amino]-1-(2,2,2-trifluoroethyl)-1H-benzo[d]imidazol-2-yl}butanoic acid hydrochloride (10 )
Compound
X-Ray structure determination
Single crystals suitable for X-ray diffraction were selected, glued on fiberglass, and mounted on a Bruker APEX II CCD diffractometer equipped with a graphite-monochromatic Mo-Kα radiation at 296 K (λ = 0.71073 Å). Data collection and reduction were performed using the APEX2 software suite. Empirical absorption corrections were carried out using SADABS. The structure was solved by direct methods and expanded by difference Fourier techniques. The non-hydrogen atoms were refined anisotropically and the hydrogen atoms were added according to the theoretical models with Uiso(H) = 1.2 (1.5 for methyl) Ueq(C). The structure was refined by full-matrix least-squares method on F2 with SHELXL-97. The crystallographic data for the compound
Crystallographic characteristics, experimental and refinement data for compound

Supplemental Material
Surporting_Information-20190716 – Supplemental material for Synthesis and crystal structure of trifluoromethyl-containing bendamustine hydrochloride
Supplemental material, Surporting_Information-20190716 for Synthesis and crystal structure of trifluoromethyl-containing bendamustine hydrochloride by Yue Yin, Huailiang Liu, Tangxin Xiao, Xiaoqiang Sun, Ke Yang and Zhengyi Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant nos. 21572026, 21702019, and 21702020), the Advanced Catalysis and Green Manufacturing Collaborative Innovation Center, and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Supplemental material
Supplemental material (1H, 13C and 19F NMR spectra of compound
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
