Abstract
On the basis of 2-benzyl-6-hydroxypyridazin-3(2H)-one, a series of its novel O-substituted (including 6-(1,3,5-triazin-2-yl)oxy) derivatives is prepared. It is proven that the substitution reactions in the initial compound occur mainly at the oxygen atom of the hydroxy group. On the basis of the obtained oxy-aceto(propane)hydrazides, the corresponding azides and anilides are synthesized. A series of 2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N′-(substituted benzylidene)aceto(propane)hydrazides is obtained via the reaction of various aromatic aldehydes with the same hydrazides. Heterocyclization of the latter affords compounds with a combination of pyridazine and 1,3,4-oxadiazole rings in the molecule. The reaction of oxy-acetohydrazide with potassium thiocyanate and a mixture of CS2/KOH leads to potassium salts of 2-{[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]methyl}hydrazine-1-carbothioamide and 2-{2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetyl}hydrazine-1-carbodithioic acid, respectively. Acid hydrolysis of the latter affords 2-benzyl-6-[(5-thioxo-4,5-dihydro-1,3,4-thiadiazol-2-yl)methoxy]pyridazin-3(2H)-one.
Introduction
In medical and agricultural practice, a range of medicines and plant protection chemicals created on the basis of azines and azoles are used. Pyridazine derivatives represent one of the most active classes of organic compounds possessing a broad spectrum of biological activity. Pyridazin-3-ones, a form of pyridazine with a carbonyl group, also exhibit a diverse range of pharmaceutical properties. A variety of drugs (e.g. chloridazon, emorfazone, zardaverine, pyridaphenthion, minaprine, gabazine, hydralazine, levosimendan, amipizone, indolidan, imazodan, pimobedan) are widely used in medical therapy. 1 The arsenal of pesticides based on pyridazine and pyridazinone includes mainly herbicides (credazine, pyridafol, pyridate, brompyrazon, chloridazon, dimidazon, flufenpyr, metflurazon, norflurazon, oxapyrazon, pydanon). 2 Because of the significant interest in these heterocyclic derivatives, over the last decades, studies on pyridazine derivatives have continued to lead to new compounds with fungicidal,3–6 herbicidal,7,8 and insecticidal 9 activities.
Among the five-membered heterocycles with three heteroatoms, one of the most applicable is 1,2,4-triazole derivatives, which are widely used in medicine, agriculture, and industry. A number of compounds synthesized on the basis of this heterocyclic ring are used in agriculture as plant protection chemicals: herbicides (amitrole, cafenstrole, epronaz, flupoxam, amicarbazone, bencarbazone, carfentrazone, flucarbazone, ipfencarbazone, propoxycarbazone, sulfentrazone, thiencarbazone, and a series of triazolopyrimidines), fungicides (amisulbrom, bitertanol, fluotrimazole, triazbutil, and a large number of conazole fungicides), and organothiophosphate insecticides (isazofos, triazophos). 2
The spectrum of the pesticidal activity of 1,3,4-oxadiazole derivatives is more limited. However, in the recent years, derivatives of this heterocycle have been the subject of many studies in terms of searching for new biologically active compounds.
The increase of environmental requirements and the fact that harmful organisms сan acquire resistance against the chemical means of plant protection make it necessary to systematically replenish the arsenal of pesticides with new and more environmentally friendly preparations having different mechanisms of action. In this regard, the targeted synthesis of new compounds with a combination of the above mentioned pharmacophore heterocycles in the same molecule could lead to new biologically active derivatives, with respect to which resistance has not yet emerged. The purpose of this study was to develop accessible and effective methods for the synthesis of novel pyridazine derivatives, as well as compounds in which the pyridazine moiety is linked with azines or azoles, and to study their biological activity in terms of searching for new environmentally friendly pesticides or plant growth regulators.
Results and discussion
The initial 2-benzyl-6-hydroxypyridazin-3(2H)-one (
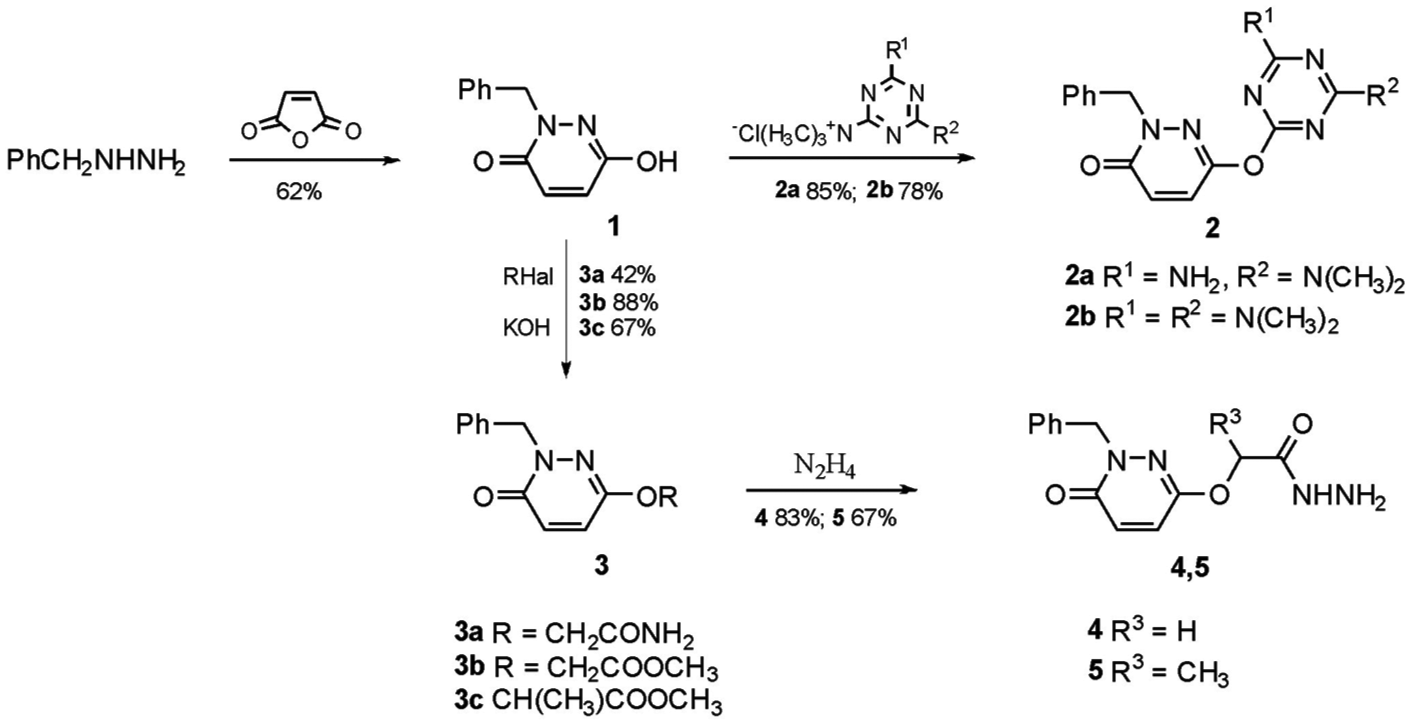
Synthesis and substitution reactions of 2-benzyl-6-hydroxypyridazin-3(2H)-one (
Further syntheses were carried out on the basis of hydrazides
The reactions of hydrazides
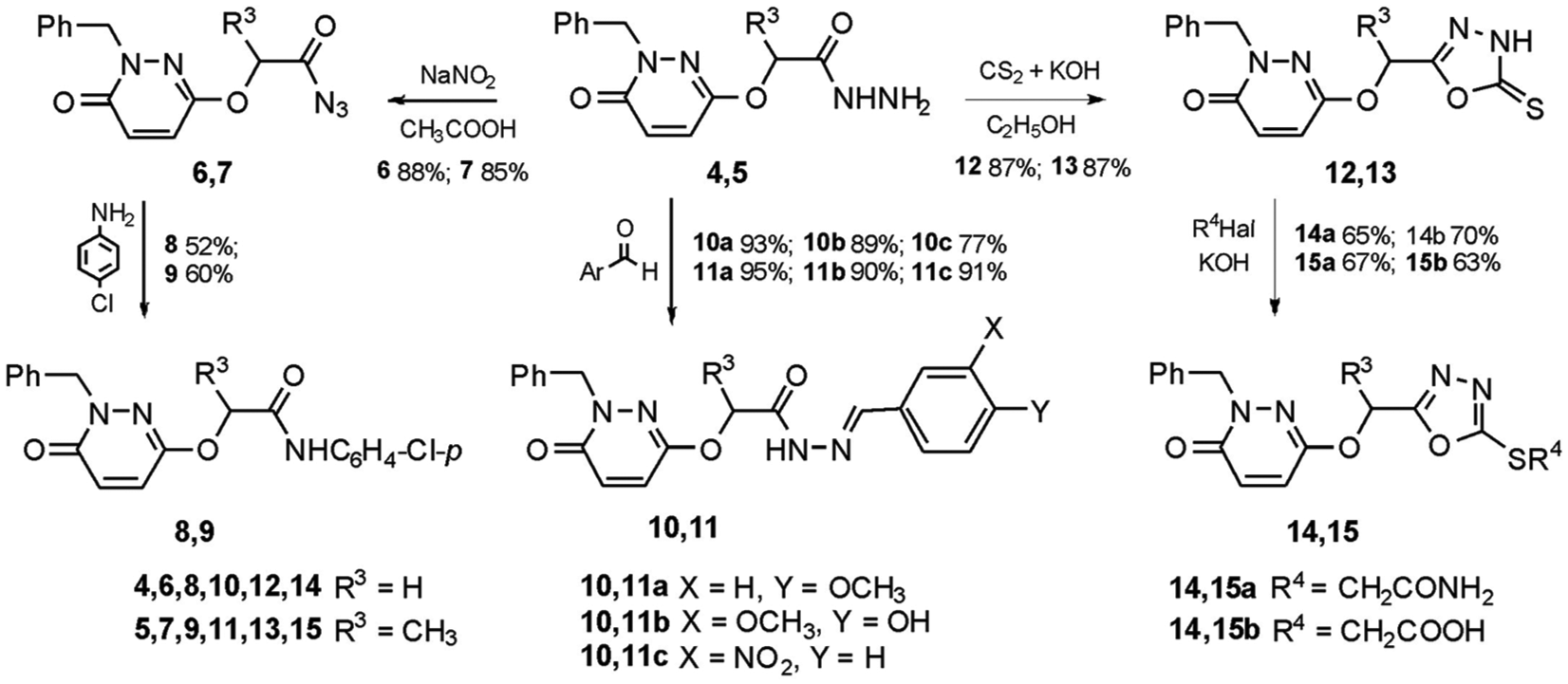
Transformations based on hydrazides (
In order to synthesize compounds with a combination of pyridazine ring and other azoles (1,3,4-thiadiazole or 1,2,4-triazole) in the molecule, potassium 2-{2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetyl}hydrazine-1-carbodithioate (

Synthesis and transformations of potassium salts of hydrazine-1-carbodithioic acid
The acid hydrolysis of compound
Biological properties
Preliminary screening of the pesticidal and growth regulatory activities of the novel synthesized compounds was studied. None of the preparations demonstrated any noticeable herbicidal or fungicidal properties, but they showed pronounced growth stimulation activity.
For the evaluation of growth regulatory properties, the action of aqueous emulsions (25 and 50 mg L−1) of the compounds synthesized on the germination, growth, and survivability of seeds and seedlings of dicotyledonous bean (Phaseolus vulgaris L.) were studied and compared with that of heteroauxin (IAA). Two series of bean seeds were incubated for 24 h in appropriate mediums in the dark at 25 °C. Then, the seeds were transplanted into soil and watered daily. The experimental data calculations were produced in 20–25 days. The number of plant roots of each series, their length and weight in moist and dry forms, and their average values were calculated. The results were compared with similar data of plants placed in IAA solutions, and the activities of preparations in comparison with IAA (in %) were determined.
Twelve of the obtained compounds
Experimental
IR spectra were obtained on an Avatar 330FT-IR (Thermo Nicolet) spectrometer, using the attenuated total reflectance (ATP) method. 1H and 13C NMR spectra were recorded at 30 °C on a Varian Mercury-300 (300 and 75 MHz appropriately) spectrometer with standard pulse sequences operating in a mixture of solvents (DMSO-d6 and CCl4 (1:3)), using tetramethylsilane (0.0 ppm) as internal standard. The NMR multiplicities br s, s, d, t, q, and m stand for broad singlet, singlet, doublet, triplet, quartet, and multiplet, respectively. The reaction progress and purity of the obtained substances was checked by thin-layer chromatography (TLC) on “Silufol UV-254” plates with acetone/hexane mixture (2:1) as the eluent. Elemental analysis was carried out on a Eurovector EA3000 elemental CHNS-O analyzer. All melting points were determined in open capillaries and are uncorrected.
Synthesis of 2-Benzyl-6-hydroxypyridazin-3(2H)-one (
Synthesis of compounds 2a,b ; general procedure
To a mixture of potassium salt of compound
6-{[4-Amino-6-(dimethylamino)-1,3,5-triazin-2-yl]oxy}-2-benzylpyridazin-3(2H)-one (
2-Benzyl-6-{[4,6-bis(dimethylamino)-1,3,5-triazin-2-yl]oxy}pyridazin-3(2H)-one (
Synthesis of compounds 3a–c ; general procedure
To a mixture of the potassium salt of compound
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetamide (
Methyl 2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetate (
Methyl 2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]propanoate (
Synthesis of compounds 4 , 5 ; general procedure
Safety warning: Appropriate precautions must be taken when using hydrazine hydrate or hydrazines due to their toxicity and possible explosive nature.
To compound
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetohydrazide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]propanehydrazide (
Synthesis of compounds 6, 7 ; general procedure
To a mixture of compound
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetyl azide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]propanoyl azide (
Synthesis of compounds 8 , 9 ; general procedure
To compound
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N-(4-chlorophenyl)acetamide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N-(4-chlorophenyl)propanamide (
Synthesis of compounds 10a–c , 11a–c ; general procedure
To compounds
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N′-(4-methoxybenzylidene)acetohydrazide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N′-(4-hydroxy-3-methoxybenzylidene)acetohydrazide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N′-(3-nitrobenzylidene)acetohydrazide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N′-(4-methoxybenzylidene)propanehydrazide (
2-((1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy)-N′-(4-hydroxy-3-methoxybenzylidene)propanehydrazide (
2-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]-N′-(3-nitrobenzylidene)propanehydrazide (
Synthesis of compounds 12 , 13 ; general procedure
2-Benzyl-6-[(5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)methoxy]pyridazin-3(2H)-one (
2-Benzyl-6-[1-(5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)ethoxy]pyridazin-3(2H)-one (
Synthesis of compounds 14a,b , 15a,b ; general procedure
To a mixture of compound
2-[(5-{[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]methyl}-1,3,4-oxadiazol-2-yl)thio]acetamide (
2-[(5-{[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]methyl}-1,3,4-oxadiazol-2-yl)thio]acetic acid (
2-[(5-{1-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]ethyl}-1,3,4-oxadiazol-2-yl)thio]acetamide (
2-[(5-{1-[(1-Benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]ethyl}-1,3,4-oxadiazol-2-yl)thio]acetic acid (
Synthesis of potassium 2-{2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetyl}hydrazine-1-carbodithioate (
Synthesis of 2-benzyl-6-[(5-thioxo-4,5-dihydro-1,3,4-thiadiazol-2-yl)methoxy]pyridazin-3(2H)-one (
Synthesis of potassium (2-{2-[(1-benzyl-6-oxo-1,6-dihydropyridazin-3-yl)oxy]acetyl}hydrazine-1-carbonothioyl)amide (
Supplemental Material
SUPPLEMENTARY_MATERIAL-Shainova – Supplemental material for Synthesis and biological evaluation of 3-O-substituted 1-benzyl-6-oxo-1,6-dihydropyridazine derivatives
Supplemental material, SUPPLEMENTARY_MATERIAL-Shainova for Synthesis and biological evaluation of 3-O-substituted 1-benzyl-6-oxo-1,6-dihydropyridazine derivatives by Roza S Shainova, Tiruhi A Gomktsyan, Armen V Karapetyan and Aleksandr P Yengoyan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Russian-Armenian University and the Ministry of Education and Science of the Russian Federation.
Supplemental material
The ESI (1H and 13C NMR spectra of compounds
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
