Abstract
Mauveine chromophores and a family of phenosafranins have been extracted from the Colour Experience Museum black precipitate with boiling ethanol and separated by chromatography. Phenosafranin (287.1295), monomethylphenylsafranin (301.1452) and a dimethylphenosafranin (315.1609) were observed. Tentative evidence for small quantities of N-tert-butylmauveine A and N-tert-butylmauveine B has been found by comparison of the data to standard retention times and accurate mass data. A synthetic method which reproduces the composition of mauveine extracted from the black precipitate, which is rich in mauveine A, is described.
A liquid chromatography–mass spectrometry (LC-MS) chart of the dyes extracted from the Colour Experience Museum black precipitate reveals mauveine chromophores, small quantities of phenosafranins and traces of higher molecular weight mauveine chromophores.
Introduction
Mauveine was the first coal-tar or aniline dye to be commercialised by William Henry Perkin (WHP), 1 but many more chemists played a pivotal role in developing the British dye industry.2,3 Hofmann, who founded the Royal College of Chemistry in 1845, now known as Imperial College, trained many chemists such as Mansfield, Nicholson, Medlock, Price, Griess, Martius, Greville Williams and WHP.2–4 They owed much to Hofmann’s inspired leadership and teaching and to the personal friendship borne out of the cultural interest nurtured in them by their tutor. After the success of WHP with mauveine, colouring matters became of particular interest and much of Hofmann’s later work in London centred on dyes and their structure. One dye known as Hofmann’s violet is named after him. WHP’s success in showing the technical value of mauveine led to the world’s first industrial production of aniline dyes. Within a decade, the environs of London and the Thames valley had dye-making factories at Greenford Green near Harrow, Locksfields at Walworth, Atlas Dye Works at Hackney Wick and Star Chemical Works at Brentford. All of these were essentially founded by pupils of Hofmann. The original WHP work on mauveine had seeded or inspired an important group of dye-making companies which have influenced dye-making up to the present day.2–4 Figure S1 in the Supplemental appendix is a chart illustrating this and the fate of Perkin & Sons. Twelve flow charts are available, which include Levinstein, Holliday, Roberts, Dale & Co and Bedford, which illustrate the foundation, mergers and progress of British dye companies with dates from as far back as 1820 in Leeds. 2
WHP mauveine has attracted much historical interest in part because the aniline used to prepare it was not pure but contained variable amounts of o-toluidine and p-toluidine.
5
The mauveine stored in a number of museums6–11 is a mixture of chromophores, typically rich in mauveine A and B, which is not what was expected because mauveine made by the WHP patented method of 1856,
1
with aniline rich in toluidines, shows a distinct pattern of chromophores each time and is rich in mauveine A, B2, B and C.12–24 Figure 1 shows some key mauveine chromophores

Drawings of mauveine A, B, B2, C, C25b and Pseudo-mauveine.

LC-MS chart of mauveine from the Bradford Colour Experience Museum. 23 UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 2.
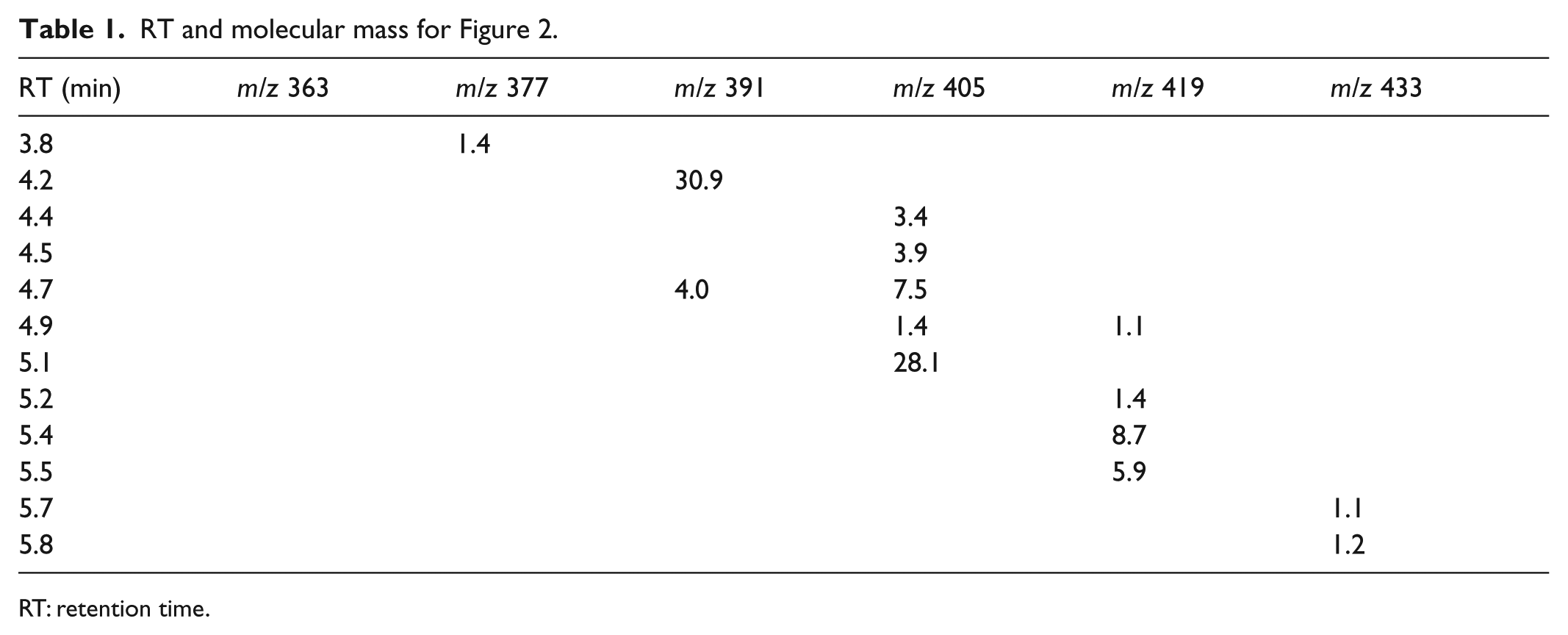
RT: retention time.
The composition data arise from the peak areas in the LC-MS charts. The numbers by the short lines are the molecular weights of the mauveine chromophores which have been separated down a reversed-phase silica column. The wavelength of detection is 550 nm and mAU stands for milli absorption units. ESMS stands for electrospray mass spectrometry counts and is shown by the coloured charts.
Discussion
A sample of mauveine and black precipitate for analysis were kindly supplied from the Bradford Colour Experience Museum. 9 The results of the mauveine analysis have been reported previously and are presented in the introduction here.23,24 The so-called black precipitate is most likely the by-product from the oxidation of aromatic amines with potassium dichromate. The addition of MeOH immediately gives a deep coloured solution which can be shown by TLC to be purified mauveine. The precipitate was boiled with EtOH three times for 10 min which extracts most of the purple chromophores. Much insoluble precipitate remains. The combined extracts were concentrated and then analysed by LC-MS without any further treatment (Figure 3 and Table 2). The chart shows a predominance of mauveine A along with some mauveine B and other mauveine chromophores. The pattern of mauveine chromophores is different from the typical dominant mixture of both mauveine A and B found in WHP mauveine. 24

UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 3.

RT: retention time.
A sample of the mauveine extracted from the black precipitate with boiling EtOH was purified by chromatography on silica gel eluting with aqNH3/EtOH (20:80). This separated the mauveine from a fainter more polar red band whose LC-MS analysis is shown (Figure 4 and Tables 3 and 4). The mauveine chromophores are not absent but are very weak.

Top: LC-MS analysis of phenosafranins and residual mauveine chromophores in the more polar red fraction separated from mauveine by chromatography eluting with aqNH3/EtOH (20:80); Bottom: An expansion of the top chart. UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 4 Top.

RT: retention time. Some peaks from 3.5–7.0 min are below 1% so the total is below 100%.
RT and molecular mass for Figure 4 Bottom.

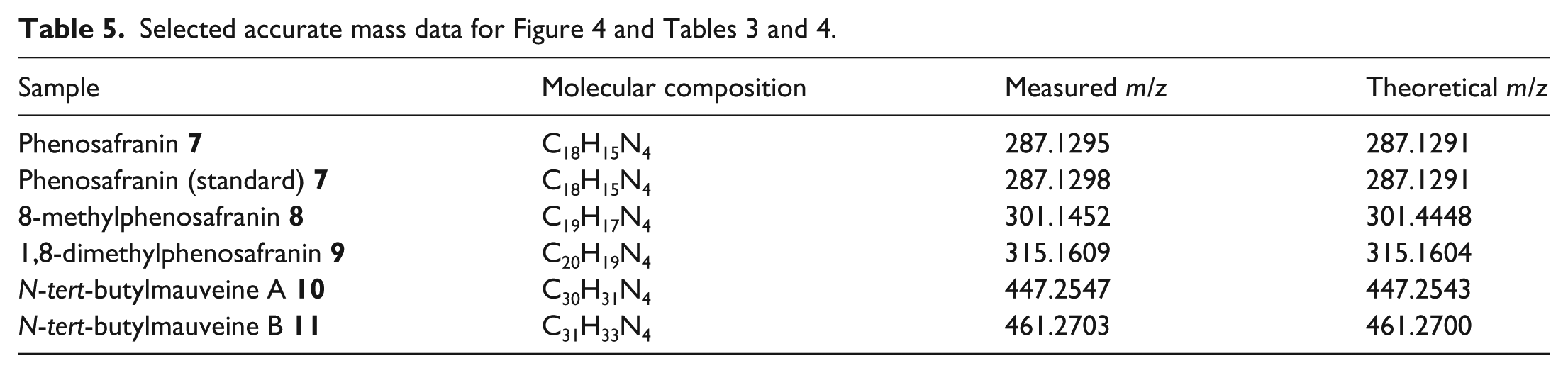
The main components eluting ahead of the mauveine chromophores are peaks at 287 (C18H15N4), 301 (C19H17N4) and 315 (C20H19N4) (Figure 4 Top). These correspond to phenosafranins which WHP reported3–5 as forming in the mother liquor in mauveine syntheses (Figure 5 and Table 5). Mauveine was also oxidised to safranin or aniline pink using lead peroxide, PbO2, as a commercial synthesis.
5
The phenosafranins presumably form from the oxidative degradation of mauveine chromophores during the synthesis. These phenosafranins are proposed to be compounds
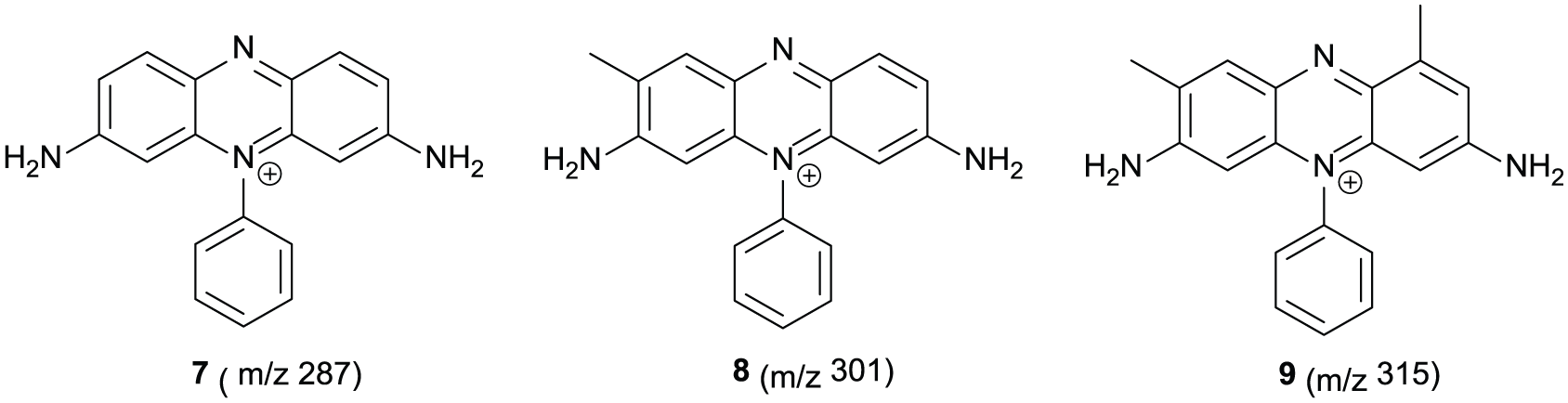
Proposed structures of the phenosafranins isolated from the Colour Experience Museum black precipitate.
The retention time for these compounds is correct when compared to a standard of phenosafranin (Figure S2 and Table T1 in the Supplemental appendix). In Figure 4, we also noticed two peaks of longer retention time where N-tert-butylmauveine chromophores normally run. An expansion is shown (Figure 4 Bottom). Two weak peaks in the region 5.9–6.6 min have the correct retention time and molecular mass for N-tert-butylmauveine A

Structures of N-tert-butylmauveine A
This result was so unusual that we requested a second batch of black precipitate from the Colour Experience Museum. The analysis was the same (Figure S3 and Tables T2 and T3 in the Supplemental appendix). The retention time is typical of N-alkylated mauveine chromophores. They are not extensively methylated mauveine chromophores because acid treatment decomposes these compounds (Figure 7 and Tables 6 and 7). This is the first tentative but direct evidence for our proposal that WHP developed a synthesis of mauveine using a modified method with N-tert-butyl-p-toluidine hydrochloride

Top: LC-MS analysis of phenosafranins and residual mauveine chromophores in the more polar red fraction separated from mauveine by chromatography eluting with aqNH3/EtOH (20:80), then treatment with aqHCl/MeOH; Bottom: An expansion of the top chart. UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 7 Top.

RT: retention time. Some peaks are below 1% so the total is below 100%.
RT and molecular mass for Figure 7 Bottom.

RT: retention time. Some peaks are below 1% so the total is below 100%.
We decided to try and find a way of reproducing the mauveine composition extracted from the black precipitate as it is different to the pseudo-mauveine/monomethylmauveine, A/B or A/B2/B/C compositions.23,24 Hundreds of experiments have been done by the author but unfortunately one good match for this particular composition was not present on file. However, some were on file in which the percentage of aniline in the reaction mixture was increased giving a reaction composition of N-tert-butyl-p-toluidine: o-toluidine: aniline of 1.0:1.5:3.0 (Figure 8 and Table 8). The reaction was purified by typical methods 24 and chromatographed on silica gel with aqNH3/MeOH (20:80) then acid treated to deprotect the protected or N-tert-butylmauveine chromophores.

LC-MS chart of mauveine made using a modification of the WHP method 1 in which p-toluidine is replaced with N-tert-butyl-p-toluidine hydrochloride. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:3.0). The product was acid treated after chromatography with aqNH3/H2O (20:80). UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 8.

RT: retention time.
Increasing the percentage of aniline in the reaction mixture increases the ratio of mauveine A to B and also increases the quantity of a monomethylmauveine chromophore (mol. wt 377) which is thought to be C25b by comparison with the retention time of authentic material. 24 This reaction was repeated but purified (without the acid treatment in the workup) with aqNH3/EtOH (20:80) on silica gel which separates deprotected from N-tert-butylated mauveine chromophores. The first band of deprotected mauveine chromophore was separated carefully and analysed by LC-MS (Figure 9 and Table 9). Previously this band was a mixture of mauveine chromophores dominated by mauveine A and B. 24 But this time, using the enriched aniline mixture, it was mainly mauveine A and showed resemblance to that seen in the extract of the black precipitate (Figure 3). Note that the front running monomethylmauveine chromophore (mol. wt 377) is largely separated and discarded using aqNH3/EtOH (20:80) as eluent.

LC-MS chart of mauveine made using a modification of the WHP method 1 in which p-toluidine is replaced with N-tert-butyl-p-toluidine hydrochloride. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:3.0). This is the first eluting band of deprotected mauveine on a silica column, separated from the none-deprotected mauveine, and it has not been acid treated. Eluent aqNH3/EtOH (20:80). UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 9.

RT: retention time.
If historic mauveine had been prepared using N-tert-butyl-p-toluidine: o-toluidine: aniline (1.0:1.5:3.0), purified by boiling with naphtha or some other means, extracted with MeOH or EtOH, then chromatographed on silica gel using aqNH3/EtOH (20:80) as eluent, then the first band or fraction would resemble Figure 3 or Figure 9 and traces of N-tert-butylated mauveine chromophore could appear as provenance of this method of synthesis.
Figure 10 (Table 10) shows the N-tert-butylated mauveine chromophores that are formed and can be separated from deprotected mauveine in this method of synthesis. The retention times of the A group and B group mauveine chromophores in Table 10 (5.6–6.4 min) are comparable to those in Table 4 for the proposed N-tert-butylated mauveine extracted from the black precipitate. Furthermore, the first eluting chromophore (m/z 433) in Figure 10 has a similar retention time to a mauveine chromophore (m/z 433) in Figure 4 believed to be N-tert-butylmauveine C25b (Rf = 5.6 min). It also deprotects with acid. The faster decomposition of a precursor to N-tert-butylated mauveine A compared to N-tert-butylated mauveine B has already been reported, 24 which explains why mauveine A predominates (Figure 9).

LC-MS chart of mauveine made using a modification of the WHP method 1 in which p-toluidine is replaced with N-tert-butyl-p-toluidine hydrochloride. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:3.0). This is the second eluting red band of N-tert-butylated mauveine chromophores. Eluent aqNH3/EtOH (20:80). UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 10.

RT: retention time.
WHP also made a commercial product called Dahlia 25 by the alkylation of mauveine with base and MeI or EtI but the pattern of molecular weights would be different from those studied here (Mauve A + CH2 = 405 and Mauve B + CH2 = 419) compared to N-tert-butylated mauveine A (447) and N-tert-butylated mauveine B (461). These compounds would also be stable to strong acid.
The WHP patent of 18561 reports the purification of mauveine by repeated extraction with boiling naphtha. The composition of the naphtha was not stated but it is assumed to be a mixture ranging from benzene to toluene or xylene. We have repeated this process many times and it is indeed effective at removing most of the unwanted by-products in the mauveine. However, the yield of mauveine is low (<1%) so a comparison was made of the ratio of deprotected mauveine chromophore to N-tert-butylated mauveine chromophore for mauveine purified by chromatography on silica gel or by multiple extractions with boiling toluene. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:1.5). Figure 11 (Table 11) shows the LC-MS chart of deprotected and N-tert-butylated mauveine chromophores for the reaction purified by chromatography on silica gel. The summed ratio of intensities for these in Table 11 is 47.3:52.7, respectively. However, Figure 12 (Table 12) shows the LC-MS chart for mauveine made the same way but purified by repeated extraction with boiling toluene. It is easy to see how there is much less deprotected mauveine compared to N-tert-butylated mauveine chromophore. The ratio is now 24.1:69.6, respectively, compared to that in the chart in Figure 11. The purification of this mixture of mauveine chromophore is effective by extractions with boiling toluene but deprotected mauveine appears to be lost in the process. The N-tert-butylated mauveine chromophores are more polar and must bind more effectively to the polyaniline reaction precipitate.

LC-MS chart of mauveine made using a modification of the WHP method 1 in which p-toluidine is replaced with N-tert-butyl-p-toluidine hydrochloride. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:1.5). The reaction precipitate was chromatographed with aqNH3/EtOH (20:80) giving a purple (eluting first) and a red (eluting second) band of deprotected and protected mauveine chromophore, respectively, which were both combined. UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 11.

RT: retention time.
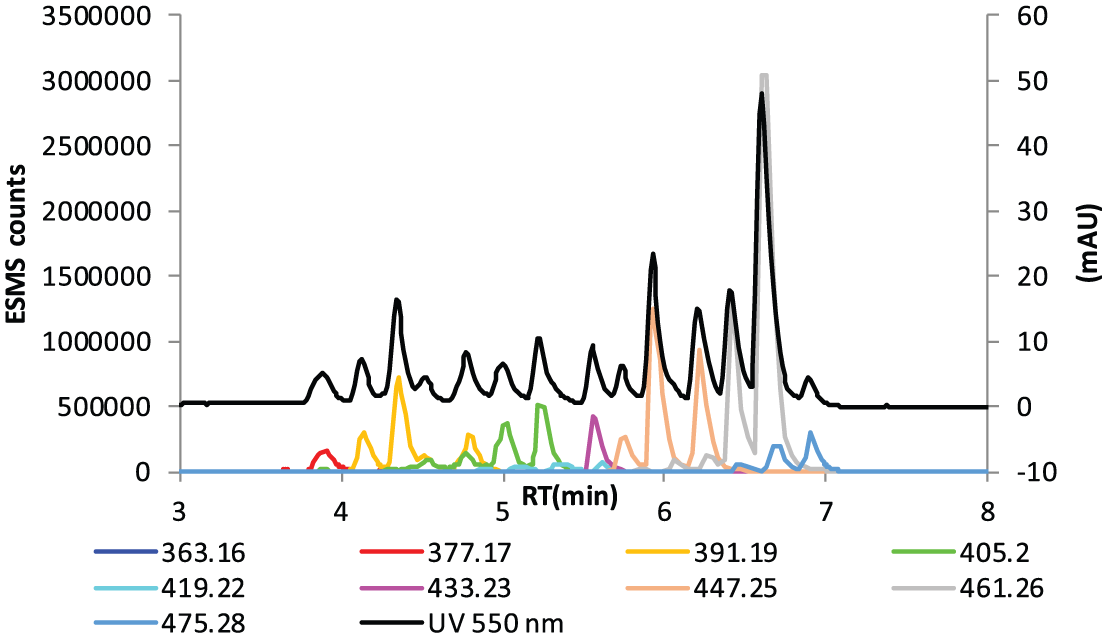
LC-MS chart of mauveine made using a modification of the WHP method 1 in which p-toluidine is replaced with N-tert-butyl-p-toluidine hydrochloride. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:1.5). The reaction precipitate was dried then purified by repeated extraction with boiling toluene. This was followed by chromatography with aqNH3/EtOH (20:80) giving a purple and a red band of deprotected and protected mauveine chromophore, respectively, which were both combined. UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 12.

RT: retention time.
The LC-MS chart in Figure 13 (Table 13) shows the mauveine chromophores which were formed after acid treatment of the mauveine chromophores shown in Figure 12 which were purified by extractions with boiling toluene. The result is quite typical in these studies showing a reduction in the amount of mauveine A relative to mauveine B. Nevertheless the dominant peaks are still mauveine A and mauveine B. Figure S4 in the Supplemental appendix shows a photograph of the silica column used to purify the mauveine chromophores, shown in Figure 12, produced by the extraction of dried reaction precipitate with boiling toluene. Notable is the pure white silica which can be reused or recycled for this chromatography by simply leaving the silica in the column. In our hands, purification of mauveine by boiling with toluene results in the loss of much mauveine though.

LC-MS chart of mauveine made using a modification of the WHP method 1 in which p-toluidine is replaced with N-tert-butyl-p-toluidine hydrochloride. The reaction composition was N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:1.5). The reaction precipitate was dried then purified by repeated extraction with boiling toluene followed by chromatography with aqNH3/EtOH (20:80) then deprotected with MeOH/aqHCl. UV at 550 nm and extracted ion chromatograms.
RT and molecular mass for Figure 13.

RT: retention time.
Summary
The extraction of black precipitate from the Colour Experience Museum 9 gives mauveine rich in mauveine A, some phenosafranins and trace amounts of higher molecular weight mauveine chromophores. These are assigned as N-tert-butylated mauveine chromophores. Retention times compared to standards and accurate mass data suggest that these are trace amounts of N-tert-butylmauveine A and N-tert-butylmauveine B which might be reminiscent of the modified method of Victorian synthesis of mauveine which we proposed.17–21,24 Mauveine made from a reaction composition of N-tert-butyl-p-toluidine hydrochloride: o-toluidine: aniline (1.0:1.5:3.0) followed by purification by chromatography on silica gel with aqNH3/EtOH (20:80) gives a front band of deprotected mauveine which is a good match to the mauveine extracted from the black precipitate, rich in mauveine A. Phenosafranins might arise as remnants of the WHP 1856 synthesis of mauveine1,3–5 but in our hands mauveine dominated by mauveine A is best made reproducibly by the modified method described here. We showed previously that the oxidation of different compositions of amines using the WHP method 1 of 1856, including an aniline rich mixture of amines, gave pseudo-mauveine and a monomethylmauveine, or a mixture of mauveine A/B2/B and C, but a mixture rich in mauveine A alone, or mauveine A and B alone, was not obtained. 23 Silica gel26–28 is remarkably clean if the mauveine is purified by boiling with toluene and can be reused or recycled in a column but in our hands much mauveine is lost by this method of purification with boiling toluene.
Experimental
WARNING: aqNH3 is concentrated aqNH3 and was always used in a fume hood with the fan on.
Extraction of black precipitate
A small vial of the precipitate (300 mg) was extracted with boiling EtOH for 10 min (80 ml ×3) in a 250 ml rb flask. The combined extracts were concentrated and analysed by LC-MS or purified by chromatography on silica gel with aqNH3/EtOH (20:80) as eluent.
Mauveine synthesis
The synthetic methods were reported in the previous paper in this series. 23 To purify dried reaction precipitate mimicking the WHP boiling naphtha treatment,1 dried precipitate was brought to the boil in toluene (60 ml) in a 250 ml flask then decanted while hot through a sinter. The boiling extraction and filtration was repeated 5 times. CAUTION: The vessel bumps if it is heated too quickly owing to the precipitate. The black precipitate was extracted with boiling MeOH (60 ml x 5), evaporated, and then chromatographed on silica gel as described previously in detail. 23 The silica column is left pristine white after elution of the mauveine chromophores (Figure S4 in the ESI)
Acid treatment of the purified phenosafranin/mauveine mixture. The mixture of chromophores was treated with 5 ml of cHCl/MeOH (50:50) in a beaker then evaporated to dryness on a hotplate. It was then re-analysed by LC-MS by dissolution in MeOH.
LC-MS
For analytical separation an Agilent 1290 Infinity HPLC system consisting of a quaternary HPLC pump, cooled auto sampler compartment, column compartment and diode-array UV-Vis detector was used. A Gold C-18 column (2.1 x 150 mm, Thermo Scientific, UK) was used for separation with a water/methanol gradient (both 0.1% v/v formic acid) from 40% MeOH to 100% in 7 min. The flow rate was 0.5 mlmin-1, column temperature 40 ºC and sample volume 5 μL. The mass spectrometer (ES-MS) used was a MAXIS II UHR-TOF LC-MS System (Bruker UK Ltd) with ESI source connected to the UV-Vis detector by a short length of Peek-tubing. The ES-MS was operated in positive ion mode with a capillary voltage of 4.5 kV using sodium formate clusters for calibration and methyl stearate as lock-mass. Mass spectra were recorded automatically.
Supplemental Material
Supplementary_Information – Supplemental material for Liquid chromatography–mass spectrometry analysis of mauveine and phenosafranins in black precipitate from the Bradford Colour Experience Museum
Supplemental material, Supplementary_Information for Liquid chromatography–mass spectrometry analysis of mauveine and phenosafranins in black precipitate from the Bradford Colour Experience Museum by M John Plater and Andrea Raab in Journal of Chemical Research
Footnotes
Acknowledgements
We are grateful to the Bradford Colour Experience Museum for black precipitate and to the EPSRC National Mass Spectrometry Foundation (NMSF) for mass spectra.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.