Abstract
Glycol dicarbonate is successfully synthesized via transesterification of glycol with dimethyl carbonate. A catalyst screening is performed by studying the effect of acidity and alkalinity on the catalytic performance. The results indicate that compared with acidic catalysts, high conversions and yields are obtained with basic catalysts. Ca(OH)2, with medium-strength alkalinity, exhibits excellent catalytic activity with glycol dicarbonate yields of 95%-99% when using a fractionation reaction system.

Reaction conditions: reaction temperature of 90°C, column temperature of 60°C, n(DMC)/n(OEG) = 35:1, Ca(OH)2 (10 wt%), 1 h.
Introduction
Given the rapid increase in CO2 emission, especially from power plants, there is a constant need for technologies that capture and sequestrate post-combustion CO2.1,2 Current technologies involving physical absorbents such as methanol, 3 sulfolane, propylene carbonate, polyethyleneglycol dimethyl ethers, and N-methylpyrrolidone 4 for CO2 capture have relatively low absorption capacities and can be sources of corrosion problems in equipment.5,6 Although dimethyl carbonate (DMC) has a high CO2 solubility, non-corrosiveness, and low cost, it has its own drawbacks centered around its low boiling point and the fact that it is relatively volatile.7,8 Recently, the results of experiments and quantum chemistry calculations indicated that CO2 physical absorbents with carbonyl groups or ether bonds in linear chain structures exhibited excellent performance in CO2 capture. For example, the CO2 solubility is in the order: propylene carbonate (ring) < DMC (chain) < diethyl carbonate (one carbonyl) < diethyl succinate (two carbonyls) and n-hexane (no ether bond) < diglyme (three ether bonds), which coordinated with the quantum chemistry calculation results.9–14 Inspired by these investigations, and extending with DMC as the basic unit, glycol dicarbonates (DCn) containing both ether bonds and ester bonds with the structure MeOC(O)O[C2H4O] n C(O)OMe were designed to further improve the solubility of CO2.
DCn as an organic carbonate is also widely used in the fields of cosmetics, perfumes, electrolytes, polymers, and biomedicines15–17 and is synthesized by the following three routes: (1) the phosgene route, (2) the CO2 route, and (3) the transesterification route. The phosgene route is the conventional route for synthesizing organic carbonates, but is a highly toxic process. 15 Although the direct formation from aliphatic diols with CO2 was an attractive synthetic route, there were some drawbacks, such as operational safety under high pressure and a low yield of the product.18–21 Transesterification with DMC as a green transesterification reagent is an alternative route to produce dicarbonates under mild conditions.22–24 Therefore, a process involving route of transesterification of glycol and DMC was chosen to synthesize DCn (Scheme 1). As shown, the process is a consecutive and reversible reaction, with MCn being the intermediate product. This kind of transesterification reaction has scarcely been studied.

Synthesis of DCn via transesterification.
Kim et al. 25 successfully synthesized pentane-3-oxy-1, 5-diyl dimethyl dicarbonate (DC2) with the structure of MeOC(O)O[C2H4O]2C(O)OMe via transesterification of diethylene glycol (DGL) and DMC catalyzed by NaOH. However, the selectivity of DC2 was only 34%, and the homogeneous basic catalyst, NaOH, runs counter to green and sustainable chemistry.
In terms of easy separation, reusability, and nontoxicity, heterogeneous catalysts are superior to homogeneous ones. 26 In this article, DGL was employed as a model substrate, and a series of cheap and readily available heterogeneous solid acids and solid bases were used to catalyze the synthesis of pentane-3-oxy-1,5-diyl dimethyl dicarbonate (DC2) (Scheme 2). 3-Oxa-5-hydroxypentyl methyl carbonate (MC2) is the intermediate product. The effect of acidity and alkalinity on the catalytic performance was investigated. The reaction conditions were optimized to enhance the yield of target product DC2. Then, a series of DCns were also obtained in high yields under the optimal conditions.

Synthesis of pentane-3-oxy-1,5-diyl dimethyl dicarbonate (DC2).
Results and discussion
Catalyst screening
As the transesterification of DGL and DMC was a consecutive and reversible reaction, DMC as both the solvent and raw material was required in sufficient quantity to ensure DGL being surrounded by DMC to form DC2 as soon as possible. Therefore, the transesterification of DGL with DMC to synthesize DC2 was performed in a reflux reaction system, and the catalytic performances for the synthesis of DC2 via the transesterification of DGL with DMC over different solid acids and bases are listed in Table 1. In the presence of a large amount of DMC, the products for all catalysts were MC2 and DC2 with a trace of polymers.
The catalytic performance of solid acid and basic catalysts for the synthesis of DC2 under reflux reaction system.

DGL: diethylene glycol; DMC: dimethyl carbonate.
Reaction conditions: 90°C, n(DMC)/n(DGL) = 35:1, catalysts 7 wt% (the mass ratio of catalyst to DMC and DGL), 4 h.
From Table 1, it can be seen that the reaction without catalyst did not occur. Common acidic catalysts, such as AlCl3 and 4 Å molecular sieves, showed extremely low catalytic activity. For the amphoteric oxide ZnO, the conversion of DGL was less than 5%, and the reaction mainly stayed at the first step with the major product being MC2. For the weakly basic acetates (CH3COO)2Mg, CH3COONa, and CH3COOK, the increase in basicity enhanced the conversions of DGL and the yields of DC2, but overall, the conversions and yields of DC2 were less than 70% and 10%, respectively. This phenomenon may be attributed to the weak alkalinity of the acetate catalysts.
In contrast, strong bases exhibited better catalytic performances with conversions of DGL above 90%. With the alkalinity enhancement, 27 the conversion of DGL increased to 100%, and the yields of DC2 were more than 50%. However, when the alkalinity was too strong, the yield of DC2 decreased. Ca(OH)2, with medium-strength alkalinity, showed the best catalytic activity with a DC2 yield of 77%. Therefore, Ca(OH)2 was selected for further investigations on the synthesis of DC2 via transesterification of DGL with DMC.
Effect of reaction conditions on the synthesis of DC2 over Ca(OH)2
Effect of the molar ratio of DMC to DGL
Theoretically, the molar ratio of DMC to DGL is 2:1 for the synthesis of DC2. However, DMC as both the raw material and reaction solvent was expected to be present in excess to enhance the yield of DC2. So, the effect of the molar ratio of DMC to DGL over the Ca(OH)2 catalyst for the synthesis of DC2 was studied ranging from 15 to 45 (Figure 1).

Effect of the molar ratio of DMC to DGL with the Ca(OH)2 catalyst for the synthesis of DC2 in the reflux reaction system.
Upon increasing the molar ratio of DMC to DGL, the conversion of DGL was stable at 100%, the yield of DC2 increased and reached a maximum of 84% at a molar ratio of 35 and was stable thereafter. The yield of MC2 first decreased upon increasing the molar ratio of DMC to DGL from 15 to 35 and then remained at 10% with further increases in the molar ratio. The results suggest that increasing the molar ratio of DMC to DGL was beneficial to transform MC2 into DC2. Thus, the optimum molar ratio of DMC to DGL was 35.
Effect of the amount of Ca(OH)2
Under conditions of 90°C, and a molar ratio of DMC to DGL of 35, the effect of the amount of Ca(OH)2 (the mass ratio of Ca(OH)2 to DMC and DGL) on the synthesis of DC2 was investigated (Figure 2).

Effect of the amount of Ca(OH)2 on the synthesis of DC2 in the reflux reaction system.
It was found that upon increasing the amount of Ca(OH)2, the conversion of DGL increased to 100% at 4 wt% and tended to be stable. The yield of DC2 first increased and reached a maximum of 84% at 10 wt% and slightly decreased thereafter. However, the yield of MC2 first increased, then decreased and eventually increased slightly. The aforementioned results indicate that the effect of the increase in the amount of alkali on the synthesis of DC2 was positive. But excessive Ca(OH)2 may lead to the incomplete exposure of the active sites of Ca(OH)2 and cause a decrease in the yield of DC2. Thus, 10 wt% of Ca(OH)2 showed the best promoting effect on the synthesis of DC2.
Effect of reaction time
Under conditions of 90°C, a molar ratio of DMC to DGL of 35, and 10 wt% of Ca(OH)2, the effect of the reaction time on the synthesis of DC2 was examined (Figure 3).

Effect of the reaction time on the synthesis of DC2 with Ca(OH)2 in the reflux reaction system.
From 0–3 h, both the conversion of DGL and the yield of DC2 increased, with 100% conversion of DGL being achieved at a reaction time of 3 h. In the next 3 h, the conversion of DGL stabilized at 100%. The yield of DC2 continuously increased and reached a maximum of 93% at 5 h and then tended to be stable. The intermediate product MC2 was the main product with a yield of 40% at 1 h, which then decreased considerably and reached 5% at 5 h and did not change much afterward. These results indicated that on prolonging the reaction time, MC2 synthesized from the transesterification of DGL and DMC was gradually transformed into DC2, and the reaction nearly achieved equilibrium within 5 h. Therefore, a reaction time of 5 h was sufficient for the transesterification of DGL with DMC to synthesize DC2 over Ca(OH)2.
Effect of the reaction technology
The reaction temperature of 90°C reached the boiling point of the DMC. Therefore, the aforementioned study was carried out in a reflux system to prevent the evaporation of DMC. However, it takes 5 h to achieve equilibrium. In order to accelerate the reaction rate, a fractionation reaction system was utilized to remove the co-product of methanol. In the fractionation reaction system, the distillation/reflux of methanol and DMC was susceptible to the temperature of the column. Thus, the effect of the column temperature on the synthesis of DC2 was investigated (Table 2).
Effect of the column temperature on the synthesis of DC2 over Ca(OH)2 in the fractionation reaction system.

DGL: diethylene glycol; DMC: dimethyl carbonate.
Reaction conditions: 90°C, n(DMC)/n(DGL) = 35:1, Ca(OH)2 (10 wt%), 1 h.
The structural formula of the dimer: 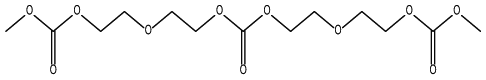
From Table 2, it was found that increasing the column temperature enhanced the volume of the distillate. Increasing the column temperature from 40°C to 60°C increased the yield of DC2, reaching a maximum of 98% at 60°C; the conversion of DGL remained at 100%. But when the column temperature was increased to 70°C, the conversion of DGL and the yield of DC2 instead declined, and the intermediate MC2 became the main product with a selectivity much higher than that of DC2. At the same time, 10% of a dimer was detected at a column temperature of 70°C.
These aforementioned phenomena were attributed to the fact that the higher column temperature facilitated the removal of DMC and methanol. Below 60°C, the amount of DMC in the reaction system was sufficient, so a higher column temperature promoted the conversion of DGL and the formation of DC2 by removing methanol from the reaction system to shift the reaction balance. When the column temperature was above 60°C, almost all DMC and methanol escaped from the reaction system. On one hand, the lack of DMC not only led to the incomplete transformation of DGL but also prevented the further transesterification of MC2 with DMC to form DC2. On the other hand, the complete removal of methanol accelerated the reaction rate so that DC2 further reacted to form a large amount of the dimer. Therefore, the optimum column temperature was 60°C.
Compared with the reflux reaction system, the time required for complete conversion of DGL in the fractionation reaction system was shortened to 1 h. Meanwhile, the yield of DC2 was 98%, which was higher than that obtained using the reflux reaction system. Compared with the Dean–Stark apparatus illustrated in the literature, 25 the controlled synthesis of DC2 could be realized by adjusting the column temperature in the fractionation device mentioned in this article, while the Dean–Stark apparatus was only suitable to obtain more dimer.
Possible reaction pathway for the transesterification of DMC and DGL
According to the aforementioned results, a possible reaction pathway for the transesterification of DMC and DGL is outlined in Scheme 3.

A possible reaction pathway for the transesterification of DMC and DGL.
First, DGL reacted with DMC via transesterification forming the intermediate MC2, which further reacted with DMC to form the desired product DC2. In addition, the dimer was formed by two routes. The first one involved DC2 reacting with DGL and DMC. The other proceeded via MC2 directly reacting with DC2.
Expanding the substrate of glycol to form DCns
To investigate the scope and limitations of the protocol, a series of DCns were synthesized via transesterification of various glycols and DMC with Ca(OH)2 under the optimized reaction conditions. The DCns were obtained with high yields of 95%–99%, and the results are listed in Table 3. The structural confirmation for the DCn products is presented in Supplemental material.
Synthesis of DCns in the fractionation reaction system.
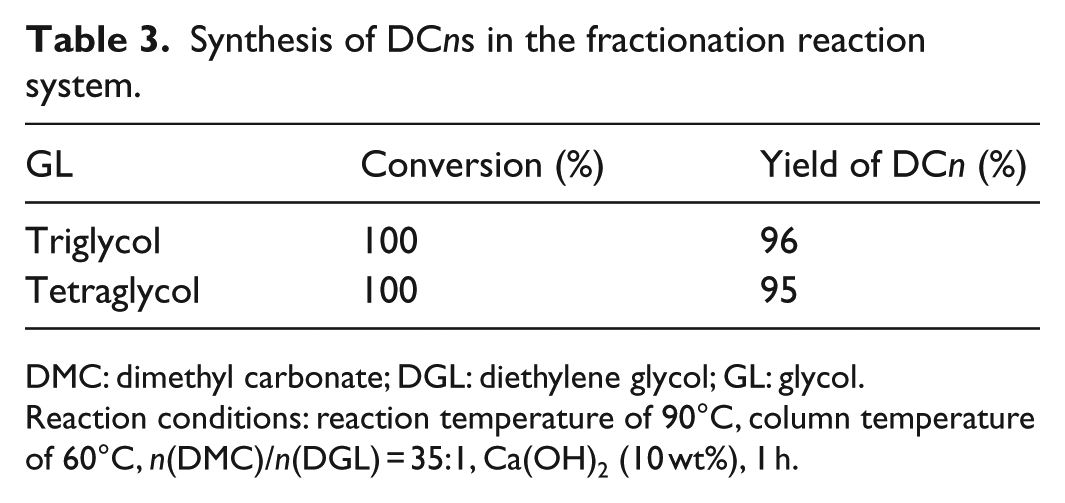
DMC: dimethyl carbonate; DGL: diethylene glycol; GL: glycol.
Reaction conditions: reaction temperature of 90°C, column temperature of 60°C, n(DMC)/n(DGL) = 35:1, Ca(OH)2 (10 wt%), 1 h.
Conclusion
The catalytic synthesis of glycol dicarbonate was successfully achieved via transesterification of DMC with glycol, and the structures of the products were confirmed by 1H NMR and 13C NMR spectroscopy. Compared with the acidic catalysts, the basic catalysts were more beneficial to the synthesis of DC2. Ca(OH)2 with medium-strength alkalinity was a cheap and efficient catalyst, which showed outstanding catalytic activity for the synthesis of DC2. Furthermore, the fractionation technology could accelerate the reaction rate and shift the reaction balance to produce DC2 by removing methanol timely. Thus, in the fractionation reaction system, under a reaction temperature of 90°C, a column temperature of 60°C, a molar ratio of DMC to glycol of 35, and a catalyst amount of 10 wt%, the yields of DCns reached 95%–99% within 1 h over Ca(OH)2.
Experimental
The transesterification reaction of DMC with glycol for the synthesis of DCn was carried out in a fractionation device, which was equipped with a two-necked flask with a magnetic stir bar, a thermometer, and a spurred fractionation column connected to a liquid dividing head. In a typical procedure, a certain amount of DGL, DMC, and catalyst were added into the 100 mL two-necked flask. When column temperature reached the preset value, the mixture was heated up to the boiling point of DMC (90°C) under vigorous stirring. During the reaction, a part of the steam was refluxed into the reaction system, while a part of the steam was fractionated into a liquid dividing head. After the reaction, liquid products were filtered for analysis.
The quantitative analyses of glycols, MC2, DC2, dimer, DC3, and DC4 were performed by Agilent 7820A gas chromatograph equipped with a DB-35 capillary column (30 m × 320 μm × 0.25 μm) and a flame ionization detector (FID) using internal standard. The internal standard substance was pentanol.
Finally, the target product of DCn and the by-products of MC2 and dimer were isolated by column chromatography.
Supplemental Material
Supporting_Information – Supplemental material for Catalytic synthesis of glycol dicarbonate from glycol and dimethyl carbonate by transesterification
Supplemental material, Supporting_Information for Catalytic synthesis of glycol dicarbonate from glycol and dimethyl carbonate by transesterification by Ruiyang Chen, Tong Chen, Xiaojia Hu, Youkun Fan and Gongying Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this work from the National Key Technology R&D Program of China (no. 2016YFB0301900) and the Science and Technology Plan Achievement Transformation Demonstration Program of Sichuan Province (no.18ZHSF0011) are gratefully acknowledged.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
