Abstract
A one-pot, three-step reaction of N-tosylhydrazones, p-bromobenzeneboronic acid, and arylboronic acids is established for the synthesis of 9-biarylfluorenes. Sequential formation of Csp3–Csp2 bond and Csp2–Csp2 bonds using readily available starting materials leads to the 9-biarylfluorene products in good yields. The present transformation features a broad substrate scope and good functional group tolerance.

Introduction
Biaryl units are important building blocks that have been found in natural products, pharmaceutical intermediates, conducting polymers, pesticides, and liquid crystals. 1 The palladium-catalyzed Suzuki–Miyaura reaction has become one of the most powerful methods for the formation of Caryl–Caryl bonds because organoboranes are air- and moisture-stable and show relatively low toxicity.2,3 In addition, 9-arylfluorenes have attracted considerable attention due to their important physical properties, which can be utilized in organic light-emitting materials, thin-film transistors, photovoltaic cells, and so on.4–10 Recently, 9-biarylfluorenes have been employed in organic chemical reactions and in fluorescent materials applications.11–14 However, the preparation of such compounds typically requires complex catalytic systems, non-readily available substrates, and harsh reaction conditions. Thus, it is highly desirable to develop efficient synthetic routes to 9-biarylfluorenes.
Structural analysis of the 9-biarylfluorene skeleton (Scheme 1) reveals that both the Csp2–Csp2 and Csp2–Csp3 bonds are constructed separately. As is known, the Csp2–Csp2 bond can be formed by a palladium-catalyzed Suzuki reaction of an aryl halide with an arylboronic acid (Scheme 1(a)), and the Csp2–Csp3 bond can be easily achieved via our previously reported metal-free coupling reaction of an N-tosylhydrazone with an aryl boronic acid (Scheme 1(b)). 15 Hence, if we use p-bromobenzeneboronic acid as a bifunctional substrate, it is possible to easily achieve access to 9-biarylfluorenes via a one-pot, multi-step reaction. Based on the above analysis and other research,16–21 we herein report an efficient approach for the synthesis of 9-biarylfluorenes via the sequential formation of one Csp2–Csp3 bond and one Csp2–Csp2 bond by one-pot, three-step reactions of N-tosylhydrazones, p-bromobenzeneboronic acid, and arylboronic acids (Scheme 1(c)).

Construction of Csp2–Csp2 and Csp2–Csp3 bonds. (a) Classical coupling reaction, (b) our previous work, and (c) this work: one-pot, multi-step reaction.
Results and discussion
First, N-tosylhydrazone was prepared by treating 9-fluorenone (
Optimization of the reaction conditions. a

Reaction conditions: (i) 9-fluorenone (
Isolated yields.
Pd(PPh3)4 (5 mol%).
Using the optimized conditions, we further investigated the scope of arylboronic acids in this one-pot, three-step reaction. As shown in Scheme 2, 4-substituted phenylboronic acids bearing electron-donating groups afforded the desired products
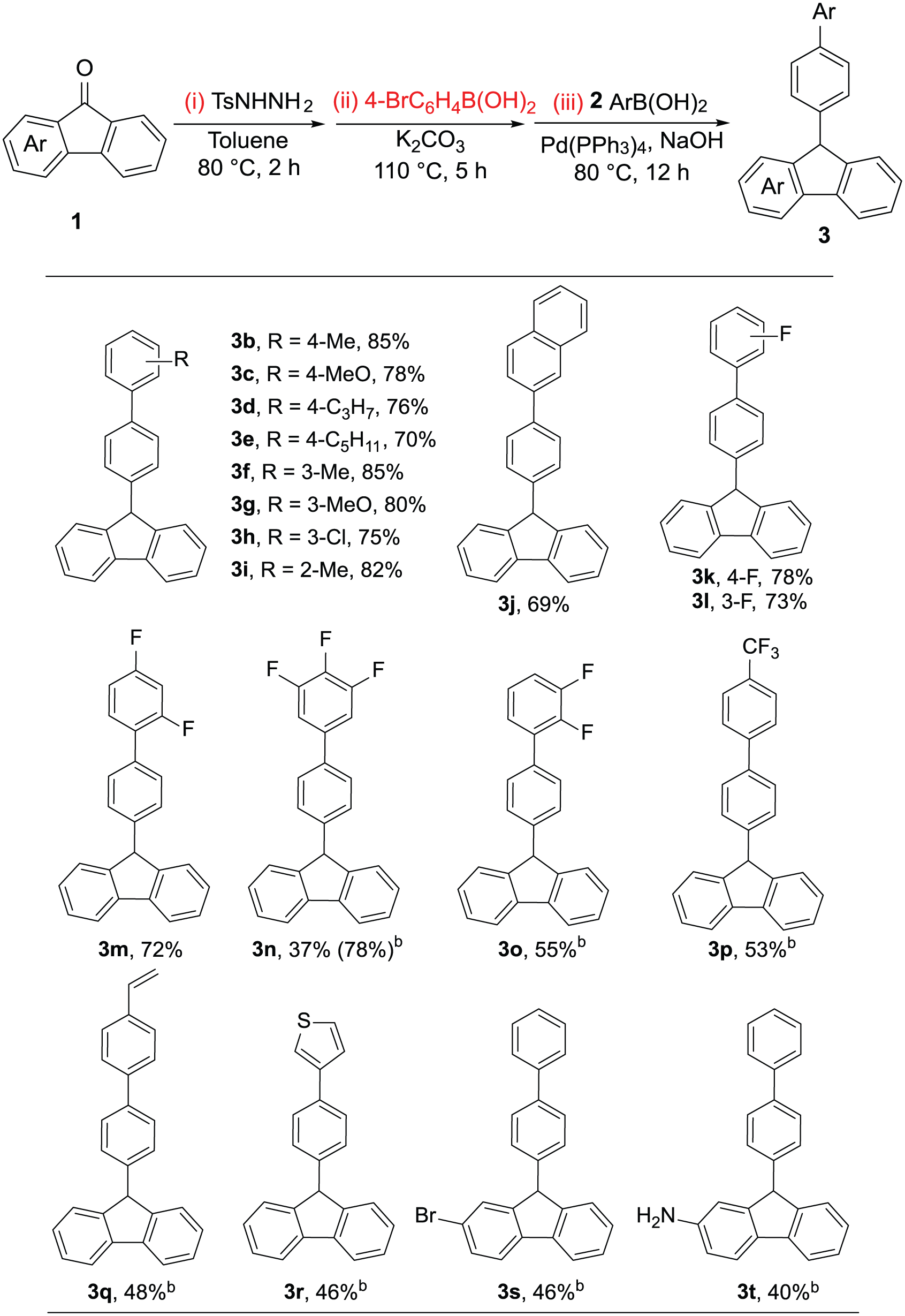
One-pot reductive coupling of substituted 9-fluorenones with boronic acids.a
Conclusion
In summary, we have developed an efficient procedure for synthesizing 9-biarylfluorenes in moderate to good yields, via one-pot, three-step reactions of N-tosylhydrazones of 9-fluorenone, p-bromobenzeneboronic acid, and arylboronic acids. The process involves sequential formation of Csp3–Csp2 and Csp2–Csp2 bonds. Importantly, the system is capable of using readily available starting materials and the intermediates do not need to be separated, and it has a wide substrate scope and good functional group tolerance.
Experimental
General materials and instruments
Unless otherwise noted, all reactions were performed under an argon atmosphere in Schlenk tubes. Melting points were determined with a fusiometer and are not corrected. Materials obtained from commercial suppliers were used without further purification. For chromatography, Qingdao Ocean Chemical 200–300 mesh silica gel was employed. 1H NMR and 13C NMR spectra were recorded on Bruker Avance III HD 400 MHz spectrometer in CDCl3 or dimethyl sulfoxide (DMSO)-d6 solution and the chemical shifts are reported in ppm (δ) relative to the internal standard tetramethylsilane (TMS) (0 ppm). High-resolution mass spectrometry (HRMS) was performed on a Thermo Scientific LTQ or bitrap XL mass spectrometer. Please see the supplemental material for the NMR spectra of all compounds.
General procedure for the one-pot, three-step reaction of N-tosylhydrazones, p-bromobenzeneboronic acid, and arylboronic acids
(i) To a Schlenk tube (25 mL) was added 9-fluorenone (
9-([1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(4′-methyl-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(4′-methoxy-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(4′-propyl-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(4′-pentyl-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(3′-methyl-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(3′-methoxy-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(3′-chloro-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(2′-methyl-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(4-(naphthalen-2-yl)phenyl)-9H-fluorene (
9-(4′-fluoro-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(3′-fluoro-[1,1′-biphenyl)-4-yl)-9H-fluorene (
9-(2′,4′-difluoro-(1,1′-biphenyl) (
9-(3′,4,′5′-trifluoro-(1,1′-biphenyl) (
9-(2′,3′-difluoro-(1,1′-biphenyl) (
9-(4′-(trifluoromethyl)-[1,1′-biphenyl]-4-yl)-9H-fluorene (
9-(4′-vinyl-[1,1′-biphenyl)-4-yl)-9H-fluorene (
3-(4-(9H-fluoren-9-yl)phenyl)thiophene (
9-([1,1′-biphenyl)-4-yl)-2-bromo-9H-fluorene (
9-([1,1′-biphenyl)-4-yl)-9H-fluoren-2-amine (
Supplemental Material
HJ-20190220-SI – Supplemental material for Synthesis of 9-biarylfluorenes by one-pot, three-step reactions of N-tosylhydrazones, p-bromobenzeneboronic acid, and arylboronic acids
Supplemental material, HJ-20190220-SI for Synthesis of 9-biarylfluorenes by one-pot, three-step reactions of N-tosylhydrazones, p-bromobenzeneboronic acid, and arylboronic acids by Jing He, Jie Zhang, Bin Dai and Ping Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 21563025), the Program for Changjiang Scholars and Innovative Research Team in University (No. IRT_15R46), and Yangtze River Scholar Research Project of Shihezi University (No. CJXZ201601).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
