Abstract
This work presents a simple and practical strategy for the synthesis of 2-substituted-3,4-dihydroquinazolines in which the ring closure reaction of 2-aminobenzylamine with corresponding iminoester hydrochlorides via microwave irradiation allows the formation of the target compounds. Also, the 3,4-quinazoline derivatives were synthesized by conventional heating procedures for comparison in terms of reaction time and yield.

Introduction
The presence of nitrogen-containing heterocycles has contributed significantly to the rapid growth of the field of medicinal chemistry for decades. Among these heterocycles, quinazoline derivatives have attracted attention due to their various pharmacological properties such as antitumor, antimicrobial, anti-inflammatory, anticonvulsant, antihypertensive, analgesic, anti-viral, and anti-urease.1–8 Quinazoline compounds are also active ingredients of many commercially available drugs such as Prazosin, Gefitinib, Erlotinib, Alfuzosin, Trimetrexate, Bunazosin, and Vandetanib. 9 Among quinazoline derivatives, the structure of 3,4-dihydroquinazoline is among those of well-known alkaloids and demonstrates different and important biological activities such as antimicrobial activity, 10 antifungal activity, 11 Type-T calcium channel blocking activity, 12 anticancer activity, 13 inhibition of trypanothione reductase (TryR), 14 anticholinesterase activity, 15 antidepressant, 16 and anticonvulsant activities. 17
Over the last few decades, a vast number of synthetic methods have been developed for the synthesis of new, original, and functionalized quinazolines to provide more effective medicines and to design synthetic medicines. 18 Some of these methods can be listed as follows: Niementowski’s synthesis; Grimmel, Guinther, and Morgan’s synthesis; from isatoic anhydride; from 1,3,4-benoxazones (acylanthranils) and amines; from ethyl 2-acetamido-5-nitrobenzoate; Sen and Ray’s synthesis; from anthranilic acid and urea; from o-ureidobenzoic acid; from o-ethoxy carbonylaminobenzoic esters or amides; from phthalic acid derivatives; from isatins; from 2-aminobenzylamine; and from 2-azido-4-chlorobenzoic acid. 19 The majority of these methods are associated with the synthesis of quinazolinone derivatives. Besides these methods, there are certain methods of synthesis of 3,4-dihydroquinazolines in the literature: solid-phase synthesis; 20 reduction or oxidation of quinazolines, tetrahydroquinazolines, or quinazolinones; 21 synthesis from N-acylation and cesium carbonate–mediated N-alkylation reactions; 21 synthesis from o-iodoxybenzoic acid by mediated tandem reactions; 22 synthesis from sequential Ugi/Staudinger/aza-Wittig reactions; 23 and synthesis from para- and meta-substituted anilines. 24 In most of these methods, 2-aminobenzylamine or N-functionalization of the aliphatic amino group of 2-aminobenzylamine is used, which reacts with the aldehydes, carboxylic acids, amidines, and orthoformates, which require long-time reactions, harsher conditions, and the use of specific reactants and catalysts. Therefore, the present study provides a more suitable and effective alternative method for the synthesis of 3,4-dihydroquinazoline compounds than those methods that are currently available.
This study describes condensation of 2-aminobenzylamine with the corresponding iminoester hydrochlorides (
Results and discussions
Our previous work has shown that nitrogen-containing heterocyclic compounds such as benzimidazoles, pyrimidines, and quinazolinones can be synthesized by condensation of compounds containing diamino groups in a suitable position with iminoester hydrochlorides by using a simple and efficient method.25–28 From this point of view, we have synthesized some 3,4-dihydroquinazolines (
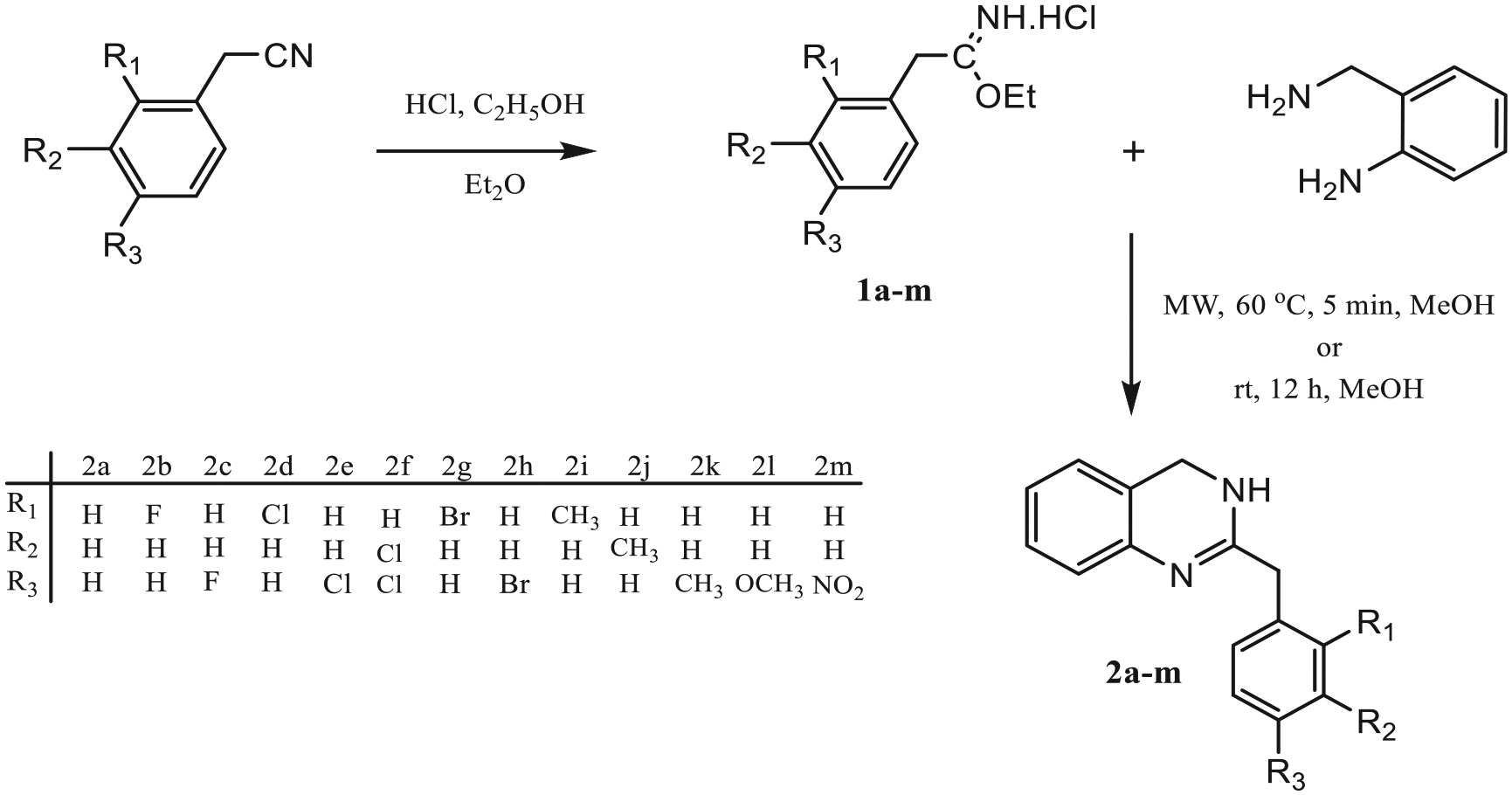
Synthetic route for the compounds
Comparison of conventional and microwave heating methods in terms of reaction time and yield for the synthesis of compounds

First, the iminoester hydrochlorides (
Then, the synthesis of 3,4-dihydroquinazoline compounds (
Conclusion
In this study, we have developed a convenient microwave-assisted method for the synthesis of 3,4-dihydroquinazoline derivatives, which provides a suitable way of synthesizing compounds of possible bioactive interest. Shorter reaction times, a simple work-up procedure, and no need for a catalyst are the most obvious advantages of this protocol.
Experimental
All the chemicals were supplied from Sigma-Aldrich, Merck, and Fluka. The progress of reactions was determined using thin-layer chromatography (TLC) plates (silica gel 60F 2.54–0.2 mm thickness). Melting points were determined in capillary tubes in the Stuart SMP30 melting point apparatus and were uncorrected. 1H NMR and 13C NMR (attached proton test (APT)) spectra were performed on the Varian-Mercury 400 MHz spectrometer in dimethyl sulfoxide-d6 (DMSO-d6) or CDCl3 using tetramethylsilane as internal standard and chemical shifts (δ values) are given in ppm. The microwave experiments were carried out in a monomod CEM Discover Microwave apparatus. The microwave radiation was adjusted so that the reaction mixture in the microwave process vials was kept at a constant temperature of 60 °C which was monitored by a computer. The elemental compositions were determined on a Carlo Erba 1106 CHN analyzer. The composition values were in agreement (±0.4%) with the calculated ones.
General procedure for synthesis of compounds 2a–m
Microwave method
A mixture of the iminoester hydrochlorides (
Conventional method
A mixture of the iminoester hydrochlorides (
2-Benzyl-3,4-dihydroquinazoline (
2-(2-Fluorobenzyl)-3,4-dihydroquinazoline (
2-(4-Fluorobenzyl)-3,4-dihydroquinazoline (
2-(2-Chlorobenzyl)-3,4-dihydroquinazoline (
2-(4-Chlorobenzyl)-3,4-dihydroquinazoline (
2-(3,4-Dichlorobenzyl)-3,4-dihydroquinazoline (
2-(2-Bromobenzyl)-3,4-dihydroquinazoline (
2-(4-Bromobenzyl)-3,4-dihydroquinazoline (
2-(2-Methylbenzyl)-3,4-dihydroquinazoline (
2-(3-Methylbenzyl)-3,4-dihydroquinazoline (
2-(4-Methylbenzyl)-3,4-dihydroquinazoline (
2-(4-Methoxybenzyl)-3,4-dihydroquinazoline (
2-(4-Nitrobenzyl)-3,4-dihydroquinazoline (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
