Abstract
Four series of novel 1,3-thiazin(thiazol)-4-one derivatives were synthesized by Suzuki coupling. Preliminary bioassays showed that most of the synthesized compounds exhibited good inhibitory activity in vivo against root-knot nematodes, Meloidogyne spp. at 20 mg L−1. Among the tested compounds, we found that two compounds displayed 46.4% and 41.4% inhibitory activity even at 1 mg L−1, respectively.

Four series of novel 1, 3-thiazin(thiazol)-4-one derivatives were designed and synthesized. Preliminary bioassays showed that most of the synthesized compounds exhibited good inhibitory activity in vivo against Meloidogyne incognitaita at 20 mg L-1. This investigation suggested that this 1, 3-thiazin(thiazol)-4-one could be further optimized to explore novel, high-bioactivity nematicidal leads.
Introduction
Plant-parasitic nematodes (PPNs) cause approximately US$157 billion of annual crop losses globally 1 . RKNs, Meloidogyne spp., are considered as the most damaging nematode group in the world as they result in approximately 5% of global crop loss to most cultivated plant species such as tomato, pepper, watermelons, and onions.2–4 It is difficult to control RKNs because they spend their lives in the soil or plant roots. Different methods are used in the field to control nematodes, including crop rotation, conventional chemical, botanical nematicides, biological controls, 5 soil solarization, and the use of resistant crop varieties. Among them, chemical nematicides have played a prominent role in the management of nematodes, 6 which are efficacious and easy to apply, with rapid onset. 7
For decades, the use of chemical nematicides is somewhat reduced, and many products have actually been phased-out from the market because their indiscriminate use affected nontarget organisms with consequential problems for the environment.8,9 Such as methyl bromide, the most widely used fumigant, has prohibited to use in 2015 because of its ozone depletion and human health concern in most countries.10,11 Although recently newly developed nematicides fluensulfone and tioxazafen were released to the market,12–16 compared with the numbers of new insecticides, it is not enough to control various nematodes with serious resistance and meet the increasingly stringent regulatory requirements for protecting the environment and ensuring food safety. Thereby, the research and development of novel nematicides with higher safety and efficiency are necessary and pressing. 17
Thiazinone ring structure is very popular in the field of pesticide and pharmaceutical applications, and compounds with a thiazinone ring are used to have antifungal activity (

Application of thiazinone and thiazolidinone structure in pharmacy and pesticide.
Thiazolidinone, a class of heterocycle including nitrogen and sulfur atom, have been reported to have anticonvulsant activity, cardiovascular effects, antibacterial activity, anticancer activity, and anti-inflammatory activity.24–27 Especially, Wang et al. reported that thiazolidinone compound
Biphenyl group is a polycyclic aromatic hydrocarbon which consists of two independent benzene rings, and combined with its unique chemical structure, it has some special physiological activity and is widely used in pharmaceuticals and pesticides, 28 such as bifonazole, felbinacethyl, brequinar, bifenazate, boscalid, and fluxapyroxad (Figure 2). It is well known that biphenyl ring is stable, which is not easy to be degraded in the soil. Another purpose of the introduction of the biphenyl structure is to imitate the ternary ring compound tioxazafen of Monsanto.

Application of biphenyl structure in pharmacy and pesticide.
Integrating the structure characteristic, we designed four series of novel 1,3-thiazin(thiazol)-4-one derivatives (Figure 3(A)–(D)). Their in vivo biological activities against M. incognita were measured. In order to investigate the relationship between structure and nematicidal activity, two parts of contents were analyzed as follows: (a) first, different substituents were introduced into phenyl ring to explore the influence of substituents at different positions and (b) biphenyl group was introduced to replace the phenyl ring of lead structure and their effects on inhibitory activities were evaluated.

The design strategy of title compounds.
Results and discussion
Synthesis
We used Suzuki coupling reaction to synthesize benzidine according to the literature. 29 Phenylboronic acid and 2-fluoro-4-iodoaniline reacted through the combination of oxidation addition, transfer metallization, and reduction elimination in one pot, catalyzed by Pd(dba)2 [bis(dibenzylideneacetone) palladium] (Scheme 1).

Synthetic route of 4-aminobiphenyls.
Aryl isothiocyanate was synthesized according to the literature. 30 First, in the presence of triethylene diamine, aryl anilines were treated with carbon disulfide to give corresponding salts of dithiocarbamic acid, and then, the corresponding salts of dithiocarbamic acid were dissolved in chloroform and a solution of bis(trichloromethyl)carbonate (BTC) in chloroform was added dropwise at 0 °C to obtain the compound (Scheme 2). This procedure efficiently yields (71%–95%). But it is difficult to synthesize aromatic isothiocyanates containing strongly electron-withdrawing groups such as nitro-trifluoromethyl and two or more halogen groups. 30

Synthetic route to aryl isothiocyanate.
The title compounds

Synthetic route of title compounds.
Mohareb had reported that the final products
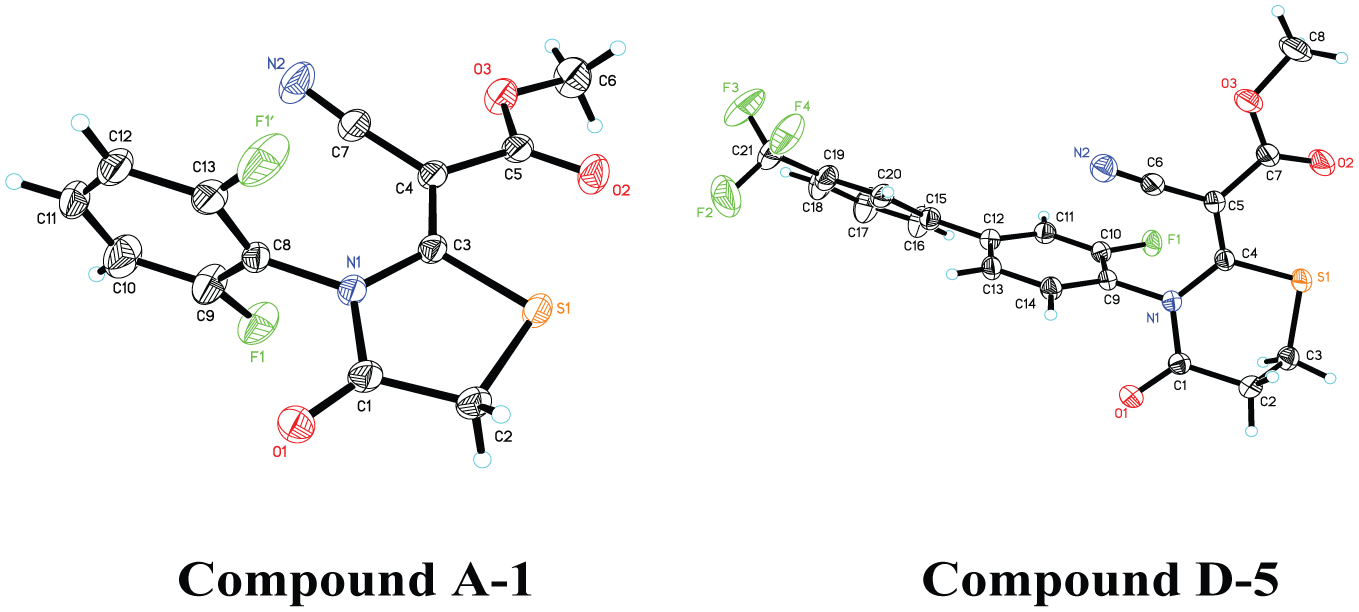
The monocrystalline of compounds
Nematicidal activity
The nematicidal activities of compound
Structure of the target compound.

In conclusion, 33 1,3-thiazin(thiazol)-4-one derivatives were synthesized and their nematicidal activities against M. incognita in vivo were evaluated. Some of the tested compounds showed good nematicidal activity against M. incognita at 20 mg L−1. Among them, compounds
Experimental
Chemistry
All reagents and solvents were obtained from commercial suppliers and used without further purification. All reactions were carried out without nitrogen protection unless otherwise noted. All melting points were obtained with a Büchi Melting Point B540 and are uncorrected. Nuclear Magnetic 1H, 13C, and 19F nuclear magnetic resonance (NMR) spectra were recorded in dimethyl sulfoxide (DMSO)-d6 on a Bruker AM-400 (400 MHz) spectrometer at ambient temperature and TMS as the internal standard. Chemical shifts were reported in parts per million (δ). Coupling constants (J) were reported in hertz (Hz). High-resolution electron mass spectra (electrospray ionization time-of-flight (ESI-TOF)) were performed on a Micromass liquid chromatography time-of-flight (LC-TOF) spectrometer using electrospray (ES) ionization modes (positive or negative). Analytical thin-layer chromatography (TLC) was carried out on precoated plates (silica gel 60 F254), and spots were visualized with ultraviolet (UV) light. X-ray crystallography was carried out on Bruker CMOS. All infrared (IR) spectra were obtained with Nicolet Magna-IR 550 infrared spectrometer.
General procedure for compounds
General procedure for 4-aminobiphenyls 3 (carcinogenic)
Substituted 4-iodoaniline
General procedure for Aryl Isothiocyanates 6
A mixture of 1,4-diazabicyclo[2.2.2]octane (DABCO; 15 mmol), substituted aromatic amine
General procedure for target compounds (A-1 −A-10 , B-1 −B-6 , C-1 −C-5 , and D-1 −D-12 )
To a cold suspension of finally divided KOH (4 mmol) in dry DMF (5 mL) was added the methyl cyanoacetate
Methyl 2-cyano-2-[3-(2-fluorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-1)
Yield: 73%. m.p. 169.3–169.5 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.65–7.55 (m, 2H), 7.43 (t, J = 9.0 Hz, 1H), 7.36 (t, J = 7.6 Hz, 1H), 4.20 (ABq, Jgem = 18.8 Hz, 2H), 3.73 (s, 3H). 19 F NMR (376 MHz, DMSO-d6): δ −122.55 to −122.67 (m). High-resolution mass spectrometry (HRMS; ESI) calcd for C13H9FN2O3S (M + Na)+, 315.0216; found: 315.0218. IR (KBr): ν = 2938, 2219, 1752, 1690, 1505, 1460, and 1395 cm−1.
Methyl 2-cyano-2-[3-(3-fluorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-2)
Yield: 65%. m.p. 179.3–179.5 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.59–7.53 (m, 1H), 7.41 (t, J = 8.0 Hz, 2H), 7.30 (d, J = 7.6 Hz, 1H), 4.13–3.99 (m, 2H), 3.71 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ −111.83 (dt, J1 = 10.2 Hz, J2 = 2.6 Hz). HRMS (ESI) calcd for C13H9FN2O3S (M + Na)+, 315.0216; found: 315.0217. IR (KBr): ν = 2987, 2223, 1741, 1681, 1585, 1499, 1357, and 1233 cm−1.
Methyl 2-cyano-2-[3-(4-fluorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-3)
Yield: 69%. m.p. 188.4–188.8 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.48 (dd, J1 = 8.0 Hz, J2 = 4.0 Hz, 2H), 7.34 (t, J = 8.0 Hz, 2H), 4.05 (s, 2H), 3.71 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ 110.70–110.77 (m). HRMS (ESI) calcd for C13H9FN2O3S (M + Na)+, 315.0216; found: 315.0216. IR (KBr): ν = 2988, 2219, 1738, 1696, 1597, 1507, 1386, and 1229 cm−1.
Methyl 2-cyano-2-[3-(2,4-difluorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-4)
Yield: 74%. m.p. 171.0–171.4 °C; 1H NMR (400 MHz, DMSO-d6): δ 7.72 (dd, J1 = 17.3 Hz, J2 = 8.4 Hz, 1H), 7.44 (dt, J1 = 20.2 Hz, J2 = 6.5 Hz, 2H), 4.22 (ABq, Jgem = 11.0 Hz, 2H), 3.74 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 173.22, 171.97, 165.42, 150.44, 147.57, 127.37, 125.68, 124.10, 120.87, 112.58, 76.52, 53.05, 32.51; 19F NMR (376 MHz, DMSO-d6): δ −137.38 (ddd, J1 = 14.8, J2 = 9.7, J3 = 4.2 Hz), −145.86 (dt, J1 = 21.9, J2 = 6.2 Hz); HRMS (EI) calcd for C13H8F2N2O3S(M + Na)+, 333.0122; found: 333.0127. IR (KBr): ν = 2997, 2217, 1745, 1683, 1588, 1497, 1386, and 1211 cm−1.
Methyl 2-cyano-2-[3-(2,6-difluorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-5)
Yield: 77%. m.p. 175.3–175.8 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.78–7.70 (m, 1H), 7.39 (t, J = 8.8 Hz, 2H), 4.37 (s, 2H), 3.75 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ 119.03 (t, J = 8.3 Hz). HRMS (ESI) calcd for C13H8F2N2O3S (M + Na)+, 333.0122; found: 333.0129. IR (KBr): ν = 2997, 2220, 1744, 1695, 1591, 1491, 1352, 1299 cm−1.
Methyl 2-cyano-2-[3-(2,3,5,6-tetrafluorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-6)
Yield: 80%. m.p. 202.2–202.8 °C; 1H NMR (400 MHz, DMSO-d6): δ 8.44–8.37 (m, 1H), 4.40 (s, 2H), 3.77 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 172.28, 170.50, 164.82, 150.76, 147.11, 112.51, 111.04, 98.46, 76.88, 53.32, 32.26; 19F NMR (376 MHz, DMSO-d6): δ −117.47 (dd, J1 = 15.7 Hz, J2 = 9.1 Hz), −135.00 to −141.03 (m), −142.23 to −146.76 (m), −156.09 (d, J = 15.9 Hz); HRMS (EI) calcd for C13H6F4N2O3S(M)+, 346.0034; found: 346.0035. IR (KBr): ν = 2219, 1760, 1685, 1519, 1440, 1299, and 1260 cm−1.
Methyl 2-cyano-2-[3-(2-chlorophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-7)
Yield: 67%. m.p. 158.2–158.3 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.67–7.57 (m, 3H), 7.50 (td, J1 = 7.6 Hz, J2 = 1.2 Hz, 1H), 4.22 (ABq, Jgem = 18.8 Hz, 2H), 3.72 (s, 3H). 13C NMR (100 MHz, DMSO-d6): δ 173.19, 171.63, 165.62, 133.31, 133.24, 132.41, 132.18, 130.28, 128.85, 112.28, 76.40, 53.02, 32.28. HRMS (ESI) calcd for C13H9ClN2O3S (M + Na)+, 330.9920; found: 330.9927. IR (KBr): ν = 3010, 2222, 1740, 1684, 1602, 1507, 1391, and 1214 cm−1.
Methyl 2-cyano-2-[3-(4-methoxyphenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-8)
Yield: 74%. m.p. 128.3–129.7 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.30 (d, J = 8.8 Hz, 2H), 7.02 (d, J = 8.8 Hz, 2H), 4.04 (s, 2H), 3.80 (s, 3H), and 3.70 (s, 3H). 13C NMR (100 MHz, DMSO-d6): δ 174.01, 173.67, 166.02, 161.09, 131.00, 127.81, 114.91, 112.72, 76.25, 55.91, 52.81, and 32.51. HRMS (ESI) calcd for C14H13N2O4S (M + H)+, 305.0597; found: 305.0597. IR (KBr): ν = 2955, 2213, 1755, 1682, 1603, 1434, 1398, and 1288 cm−1.
Methyl 2-cyano-2-[3-(4-nitrophenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-9)
Yield: 78%. m.p. 189.7–190.5 °C. 1H NMR (400 MHz, DMSO-d6): δ 8.40 (d, J = 8.8 Hz, 2H), 7.79 (d, J = 8.8 Hz, 2H), 4.09 (s, 2H), 3.72 (s, 3H). 13C NMR (100 MHz, DMSO-d6): δ 173.68, 172.68, 165.67, 148.91, 140.96, 131.79, 125.00, 113.09, 76.23, 52.95, and 32.89. HRMS (ESI) calcd for C13H9N3O5S (M + Na)+, 342.0161; found: 342.0165. IR (KBr): ν = 2219, 1735, 1696, 1527, 1505, 1350, and 1297 cm−1.
Methyl 2-cyano-2-[3-(2-fluoro-4-methoxyphenyl)]-4-oxo-1,3-thiazol-2-ylidene]acetate A-10)
Yield: 83%. m.p. 145.6–145.9 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.44 (t, J = 8.8 Hz, 1H), 7.05 (dd, J1 = 12.0 Hz, J2 = 2.8 Hz, 1H), 6.91 (dd, J1 = 8.4 Hz, J2 = 2.4 Hz, 1H), 4.17 (ABq, Jgem = 18.8 Hz, 2H), 3.83 (s, 3H), 3.73 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ −120.26 (t, J = 11.6 Hz). HRMS(ESI) calcd for C14H11FN2O4S (M + Na)+, 345.0322; found: 345.0321. IR (KBr): ν = 2989, 2224, 1751, 1682, 1599, 1487, 1386, and 1228 cm−1.
Methyl 2-cyano-2-[3-(2-fluorophenyl)]-4-oxo-1,3-thiazin-2-ylidene]acetate B-1)
Yield: 84%. m.p. 222.0–222.5 °C. 1H NMR (400 MHz, CDCl3): δ 7.46 (dd, J1 = 13.2 Hz, J2 = 7.2 Hz, 1H), 7.36 (t, J = 7.2 Hz, 1H), 7.31–7.19 (m, 2H), 3.80 (s, 3H), 3.23–3.17 (m, 2H), 3.15–3.10 (m, 2H). 19F NMR (376 MHz, CDCl3): δ −116.15 to −116.13 (m). HRMS (ESI) calcd for C14H12N2O3FS (M + H)+, 307.0553; found: 307.0554. Vmax (KBr)/cm−1 2207 (–CN), 16991 (–CON–), and 1748 (–COOMe).
Methyl 2-cyano-2-[3-(4-methylphenyl)]-4-oxo-1,3-thiazin-2-ylidene]acetate B-2)
Yield: 78%. m.p. 232.9–233.3 °C; 1H NMR (400 MHz, CDCl3): δ 7.27 (d, J = 8.8 Hz, 2H), 7.42 (d, J = 7.6 Hz, 2H), 3.80 (s, 3H), 3.20–3.14 (m, 2H), 3.11–3.04 (m, 2H), 2.40 (s, 3H). HRMS (ESI) calcd for C15H15N2O3S (M + H)+, 303.0803; found: 303.0804. Vmax (KBr)/cm−1 2214 (–CN), 16,993 (–CON–), 1716 (–COOMe).
Methyl 2-cyano-2-[3-(4-bromophenylphenyl)]-4-oxo-1,3-thiazin-2-ylidene]acetate B-3)
Yield: 71%. m.p. 232.4–233.0 °C. 1H NMR (400 MHz, CDCl3): δ 7.59 (dt, J1 = 8.8 Hz, J2 = 2.4 Hz, 2H), 7.21 (dt, J1 = 8.8 Hz, J2 = 2.4 Hz, 2H), 3.81 (s, 3H), 3.20–3.16 (m, 2H), 3.10–3.06 (m, 2H). HRMS (ESI) calcd for C14H12N2O3S79Br (M + H)+, 366.9752; found: 366.9758; calcd for C14H12N2O3S81Br (M + H)+, 368.9732; found: 368.9746. Vmax (KBr)/cm−1 2216 (–CN), 16,995 (–CON–), and 1712 (–COOMe).
Methyl 2-cyano-2-[3-(2,3-difluorophenyl)]-4-oxo-1,3-thiazin-2-ylidene]acetate B-4)
Yield: 67%. m.p. 210.0–210.7 °C; 1H NMR (400 MHz, CDCl3): δ 7.32–7.25 (m, 1H), 7.23–7.17 (m, 1H), 7.12 (t, J = 7.2 Hz, 1H), 3.81 (s, 3H), 3.22–3.18 (m, 2H), 3.16–3.13 (m, 2H). 19F NMR (376 MHz, CDCl3): δ −135.37 to −135.44 (m, 1F), −138.62 (dt, J1 = 22.0 Hz, J2 = 6.8 Hz, 1F). HRMS (ESI) calcd for C14H9N2O3F2S (M − H)−, 323.0302; found: 323.0305. Vmax (KBr)/cm−1 2211 (–CN), 16,991 (–CON–), and 1712 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-4-methylphenyl)]-4-oxo-1,3-thiazin-2-ylidene]acetate B-5)
Yield: 72%. m.p. 214.7–215.0 °C. 1H NMR (400 MHz, CDCl3): δ 7.21 (t, J = 7.6 Hz, 1H), 7.05 (t, J = 10.4 Hz, 2H), 3.80 (s, 3H), 3.21–3.16 (m, 2H), 3.14–3.10 (m, 2H), 2.41 (s, 3H). 19 F NMR (376 MHz, CDCl3): δ −117.45 (dd, J1 = 10.8 Hz, J2 = 8.4 Hz). HRMS (ESI) calcd for C15H14N2O3FS (M + H)+, 321.0709; found: 321.0710. Vmax (KBr)/cm−1 2210 (–CN), 16,989(–CON–), and 1708 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-4-methoxyphenyl)]-4-oxo-1,3-thiazin-2-ylidene]acetate B-6)
Yield: 69%. m.p. 228.8–229.0 °C. 1H NMR (400 MHz, CDCl3): δ 7.22 (t, J = 8.8 Hz, 1H), 6.81–6.72 (m, 2H), 3.84 (s, 3H), 3.80 (s, 3H), 3.20–3.15 (m, 2H), 3.13–3.08 (m, 2H). 19F NMR (376 MHz, CDCl3): δ −114.19 (dd, J1 = 12.0 Hz, J2 = 9.2 Hz). HRMS (ESI) calcd for C15H14N2O4FS (M + H)+, 337.0658; found: 337.0660. Vmax (KBr)/cm−1 2216 (–CN), 16,984 (–CON–), and 1721 (–COOMe).
Methyl 2-cyano-2-[3-([1,1′-biphenyl])-4-yl)-4-oxo-1,3-thiazol-2-ylidene]acetate C-1
Yield: 76%. m.p. 235.1–235.5 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.82 (d, J = 8.4 Hz, 2H), 7.75 (d, J = 7.2 Hz, 2H), 7.51 (t, J = 8.0 Hz, 4H), 7.43 (t, J = 7.2 Hz, 1H), 4.10 (s, 2H), 3.72 (s, 3H). HRMS (ESI) calcd for C19H14N2O3S (M + H)+, 351.0803; found: 351.0802. IR (KBr): ν = 2938, 2208, 1746, 1691, 1634, 1507, 1417, and 1276 cm−1.
Methyl 2-cyano-2-[3-(2-fluoro-2′-methyl[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazol-2-ylidene]acetate C-2
Yield: 70%. m.p. 198.6–198.9 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.63(t, J = 8.0 Hz, 1H), 7.44 (d, J = 10.8 Hz, 1H), 7.33–7.26 (m, 5H), 4.25 (ABq, Jgem = 18.8 Hz, 2H), 3.75 (s, 3H), 2.26 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ −122.72 (t, J = 9.4 Hz), HRMS (ESI) calcd for C20H15FN2O3S (M + H)+, 383.0866; found: 383.0868. IR (KBr): ν = 2967, 2212, 1751, 1688, 1619, 1526, 1434, and 1257 cm−1.
Methyl 2-cyano-2-[3-(2-fluoro-2′-cyano[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazol-2-ylidene]acetate C-3
Yield: 75%. m.p. 179.8–180.4 °C. 1H NMR (400 MHz, DMSO-d6): δ 8.01 (d, J = 7.6 Hz, 1H), 7.85 (t, J = 7.6 Hz, 1H), 7.75 (dd, J1 = 7.2 Hz, J2 = 2.4 Hz, 2H) 7.68–7.61 (m, 3H), 4.25 (ABq, Jgem = 18.8 Hz, 2H), 3.75 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ −121.43 (t, J = 11.5 Hz). HRMS (ESI) calcd for C20H12FN3O3S (M + H)+, 394.0662; found: 394.0655. IR (KBr): ν = 2979, 2215, 1755, 1639, 1597, 1529, 1449, and 1231 cm−1.
Methyl 2-cyano-2-[3-(2-fluoro-3′-trifluoromethyl[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazol-2-ylidene]acetate C-4
Yield: 67%. m.p. 194.5–194.7 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.89 (d, J = 7.6 Hz, 1H), 7.78 (t, J = 7.6 Hz, 1H), 7.70–7.64 (m, 2H), 7.46 (d, J = 12.0 Hz, 1H), 7.38 (d, J = 7.6 Hz, 1H), 7.33 (d, J = 8.0 Hz, 1H), 4.24(ABq, Jgem = 18.4 Hz, 2H), 3.75 (s, 3H), 3.36 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ −55.25 (s), −122.50 (t, J1 = 11.1 Hz, J2 = 2.2 Hz), HRMS (ESI) calcd for C20H12F4N2O3S (M + H)+, 437.0583; found: 437.0590. IR (KBr): ν = 2958, 2214, 1759, 1688, 1601, 1527, 1450, and 1279 cm−1.
Methyl 2-cyano-2-[3-(2-fluoro-4′-methoxy[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazol-2-ylidene]acetate C-5
Yield: 73%. m.p. 220.1–220.5 °C. 1H NMR (400 MHz, DMSO-d6): δ 7.59 (t, J = 8.0 Hz, 2H), 7.50 (dd, J1 = 8.4 Hz, J2 = 1.6 Hz, 1H), 7.43 (td, J1 = 8.0 Hz, J2 = 1.6 Hz, 1H), 7.37 (dd, J1 = 7.2 Hz, J2 = 1.6 Hz, 1H), 7.18 (d, J = 8.0 Hz, 1H), 7.08 (t, J = 7.4 Hz, 1H), 4.24 (ABq, Jgem = 18.8 Hz, 2H), 3.82 (s, 3H), and 3.75 (s, 3H). 19F NMR (376 MHz, DMSO-d6): δ −123.08 (dd, J1 = 12.8 Hz, J2 = 8.5 Hz), HRMS (ESI) calcd for C20H15FN2O4S (M + H)+, 399.0815; found: 399.0814. IR (KBr): ν = 2955, 2216, 1751, 1695, 1621, 1566, 1431, and 1281 cm−1.
Methyl 2-cyano-2-[3-([1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene] acetate D-1
Yield: 81%. m.p. 185.8–186.3 °C; 1H NMR (400 MHz, CDCl3) δ 7.72–7.66 (m, J = 7.6 Hz, 2H), 7.63 (d, J = 8.0 Hz, 2H), 7.48–7.44 (m, 2H), 7.41–7.36 (m, 3H), 3.82 (s, 3H), 3.25–3.07 (m, 4H). 13C NMR (100 MHz, CDCl3): δ 171.27, 167.73, 164.73, 141.51, 139.92, 138.44, 128.80, 128.67, 127.74, 127.48, 127.25, 113.34, 91.73, 52.77, 35.67, and 23.54. HRMS (ESI) calcd for C20H17N2O3S (M + H)+, 365.0960; found: 365.0961. Vmax (KBr)/cm−1 2213 (–CN), 1700 (–CON–), and 1720 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-2
Yield: 83%. m.p. 198.6–199.1 °C; 1H NMR (400 MHz, CDCl3): δ 7.62 (d, J = 8.0 Hz, 2H), 7.52–7.36 (m, 6H), 3.82 (s, 3H), 3.27–3.11 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −116.11 to −116.12 (m, 1F). HRMS (ESI) calcd for C20H16N2O3FS (M + H)+, 383.0866; found: 383.0867. Vmax (KBr)/cm−1 2213 (–CN), 1701 (–CON–), and 1729 (–COOMe).
Methyl 2-cyano-2-[3-(2,6-difluoro[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-3
Yield: 77%. m.p. 207.1–208.0 °C; 1H NMR (400 MHz, CDCl3): δ 7.60 (d, J = 7.6 Hz, 2H), 7.46 (dt, J = 19.2 Hz, 6.8 Hz, 3H), 7.33–7.27 (m, 2H), 3.83 (s, 3H), and 3.20–3.14 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −114.16 (d, J = 9.8 Hz, 2F). HRMS (ESI) calcd for C20H15N2O3F2S (M + H)+, 401.0771; found: 401.0772. Vmax (KBr)/cm−1 2202 (–CN), 16996 (–CON–), and 1729 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-2′-trifluoromethyl[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-4
Yield: 74%. m.p. 188.9–199.5 °C; 1H NMR (400 MHz, CDCl3) δ 7.78 (d, J = 7.6 Hz, 1H), 7.60 (t, J = 7.6 Hz, 1H), 7.53 (t, J = 7.6 Hz, 1H), 7.43–7.36 (m, 2H), 7.22 (t, J = 8.4 Hz, 2H), 3.82 (s, 3H), 3.26–3.10 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −56.70 (s, 3F), −116.36 to −116.48 (m, 1F). HRMS (ESI) calcd for C21H15N2O3F4S (M + H)+, 451.0740; found: 451.0742. Vmax (KBr)/cm−1 2214 (–CN), 16994 (–CON–), and 1712 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-3′-trifluoromethyl[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-5
Yield: 73%. m.p. 177.9–188.2 °C; 1H NMR (400 MHz, CDCl3): δ 7.86 (s, 1H), 7.82–7.76 (m, 1H), 7.67 (d, J = 7.6 Hz, 1H), 7.60 (t, J = 7.6 Hz, 1H), 7.53–7.42 (m, 3H), 3.82 (s, 3H), 3.26–3.13 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −62.65 (s, 3F), −115.24 to −115.29 (m, 1F). HRMS (ESI) calcd for C21H15N2O3F4S (M + H)+, 451.0740; found: 451.0743. Vmax (KBr)/cm−1 2220 (–CN), 16,999 (–CON–), and 1715 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-4′-trifluoromethyl[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-6
Yield: 65%. m.p. 151.3–152.0 °C; 1H NMR (400 MHz, CDCl3): δ 7.76–7.68 (m, 4H), 7.51–7.44 (m, 3H), 3.82 (s, 3H), 3.23–3.14 (m, 4H). 19F NMR (376 MHz, CDCl3): δ 62.56 (s, 3F), −115.27 to −115.32 (m, 1F). HRMS (ESI) calcd for C21H15N2O3F4S (M + H)+, 451.0740; found: 451.0742. Vmax (KBr)/cm−1 2216 (–CN), 1708 (–CON–), and 1720 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-4′-methoxy[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-7
Yield: 80%. m.p. 215.0–215.8 °C; 1H NMR (400 MHz, CDCl3): δ 7.55 (d, J = 8.4 Hz, 2H), 7.40–7.35 (m, 3H), 6.99 (d, J = 8.4 Hz, 2H), 3.87 (s, 3H), 3.81 (s, 3H), 3.24–3.11 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −116.32 to −116.37 (m, 1F). HRMS (ESI) calcd for C21H18N2O4FS (M + H)+, 413.0971; found: 413.0973. Vmax (KBr)/cm−1 2202 (–CN), 1704 (–CON–), and 1730 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-2′-methoxy[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-8
Yield: 83%. m.p. 219.8–220.5 °C; 1H NMR (400 MHz, CDCl3): δ 7.44 (t, J = 9.2 Hz, 2H), 7.37 (t, J = 8.0 Hz, 3H), 7.08–6.98 (m, 2H), 3.84 (s, 3H), 3.82 (s, 3H), 3.24–3.09 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −117.45 to −117.50 (m, 1F). HRMS (ESI) calcd for C21H18N2O4FS (M + H)+, 413.0971; found: 413.0973. Vmax (KBr)/cm−1 2213 (–CN), 16,994 (–CON–), and 1725 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-3′-methoxy[1,1′-biphenyl])-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-9
Yield: 78%. m.p. 180.9–181.0 °C; 1H NMR (400 MHz, CDCl3): δ 7.43–7.38 (m, 4H), 7.20 (d, J = 7.6 Hz, 1H), 7.13 (s, 1H), 6.96 (d, J = 8.0 Hz, 1H), 3.88 (s, 3H), 3.82 (s, 3H), and 3.25–3.09 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −116.07 to −116.12 (m, 1F). HRMS (ESI) calcd for C21H18N2O4FS (M + H)+, 413.0971; found: 413.0973. Vmax (KBr)/cm−1 2211 (–CN), 16,996 (–CON–), and 1727 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-3′-methyl[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-10
Yield: 76%. m.p. 199.1–192.2 °C; 1H NMR (400 MHz, CDCl3): δ 7.48–7.46 (m, 2H), 7.44–7.33 (m,4H), 7.24–7.21 (m, 1H), 3.81 (s, 3H), 3.25–3.09 (m, 4H), 2.44 (s, 3H). 19F NMR (376 MHz, CDCl3): δ −116.17 to −116.51 (m, 1F). HRMS (ESI) calcd for C21H18N2O3FS (M + H)+, 397.1022; found: 397.1033. Vmax (KBr)/cm−1 2206 (–CN), 1704 (–CON–), and 1728 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-3′-cyano[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-11
Yield: 79%. m.p. 217.7–218.4 °C; 1H NMR (400 MHz, CDCl3): δ 7.89 (s, 1H), 7.84 (d, J = 8.0 Hz, 1H), 7.70 (d, J = 7.6 Hz, 1H), 7.60 (t, J = 7.6 Hz, 1H), 7.50–7.39 (m, 3H), 3.82 (s, 3H), 3.27–3.16 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −114.80 to −114.83 (m, 1F). HRMS (ESI) calcd for C21H15N3O3FS (M + H)+, 408.0818; found: 408.0821. Vmax (KBr)/cm−1 2228, 2208 (–CN), 1704 (–CON–), and 1727 (–COOMe).
Methyl 2-cyano-2-[3-(2-fluoro-3′-nitro[1,1′-biphenyl]-4-yl)-4-oxo-1,3-thiazin-2-ylidene]acetate D-12
Yield: 81%. m.p. 209.1–210.7 °C; 1H NMR (400 MHz, CDCl3): δ 8.48 (t, J = 2.0 Hz, 1H), 8.27 (ddd, J = 8.0, 2.0, and 0.8 Hz, 1H), 7.94 (ddd, J = 8.0, 1.6, and 1.0 Hz, 1H), 7.67 (dd, J = 10.4 and 6.0 Hz, 1H), 7.57–7.46 (m, 3H), 3.82 (s, 3H), and 3.19 (m, 4H). 19F NMR (376 MHz, CDCl3): δ −114.72 to −114.77 (m, 1F). HRMS (ESI) calcd for C20H15N3O5FS (M + H)+, 428.0716; found: 428.0715. Vmax (KBr)/cm−1 2213 (–CN), 1710 (–CON–), and 1736 (–COOMe).
Footnotes
Acknowledgements
We are very thankful to Dr Bingli Gao and his co-workers in Huzhou Modern Agricultural Biotechnology Innovation Center, Chinese Academy of Sciences, China, for the discussion of nematicidal evaluation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (21672061) and National Key Research Program of China (2018YFD0200105). This work was also supported by the Fundamental Research Funds for the Central Universities (222201718004).
