Abstract
Seven hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione compounds were synthesized by a catalyst-free 1,3-dipolar cycloaddition reaction in a one-pot three-component system containing 3,5-diarylidene-1-benzylpiperidin-4-one, isatin and

Natural nitrogenous heterocyclic compounds have attracted great attention of chemical and pharmaceutical researchers for their various structures and extensive bioactivities.1,2 The synthesis of these compounds or new creations from these skeletons has become an important research direction,3–5 and many new molecules with novel structure6,7 or high bioactivity6,8 have been constructed. Among them, spiro-oxindole compounds are an important family with diverse biological activities such as anti-tumour (

Potential synthetic bioactive spiro-oxindole compounds.
The research and development of anti-neoplastic drugs has been the focus of pharmaceutical researchers in recent years. Facing the large number of chemical monomers and extracts, the brine shrimp lethality test is often used for the preliminary evaluation of cytotoxicity.13,14
Based on our strong interest in the bioactivities of spiro-oxindole heterocyclic compounds and the further study of this type of compounds, 15 we synthesized a new group of hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione compounds through 1,3-dipolar cycloaddition reaction in a one-pot three-component system (Scheme 1). By screening on the brine shrimp bioassay, these compounds exhibited shrimp lethality effect with LC50 valves ranging from 7.27 to 174.59 µg/mL. The most potential compound had an LC50 of 7.27 g/mL. The LC50 value of positive control podophyllotoxin is 2.16 µg/mL. The strong shrimp lethality suggests it has promising cytotoxicity.
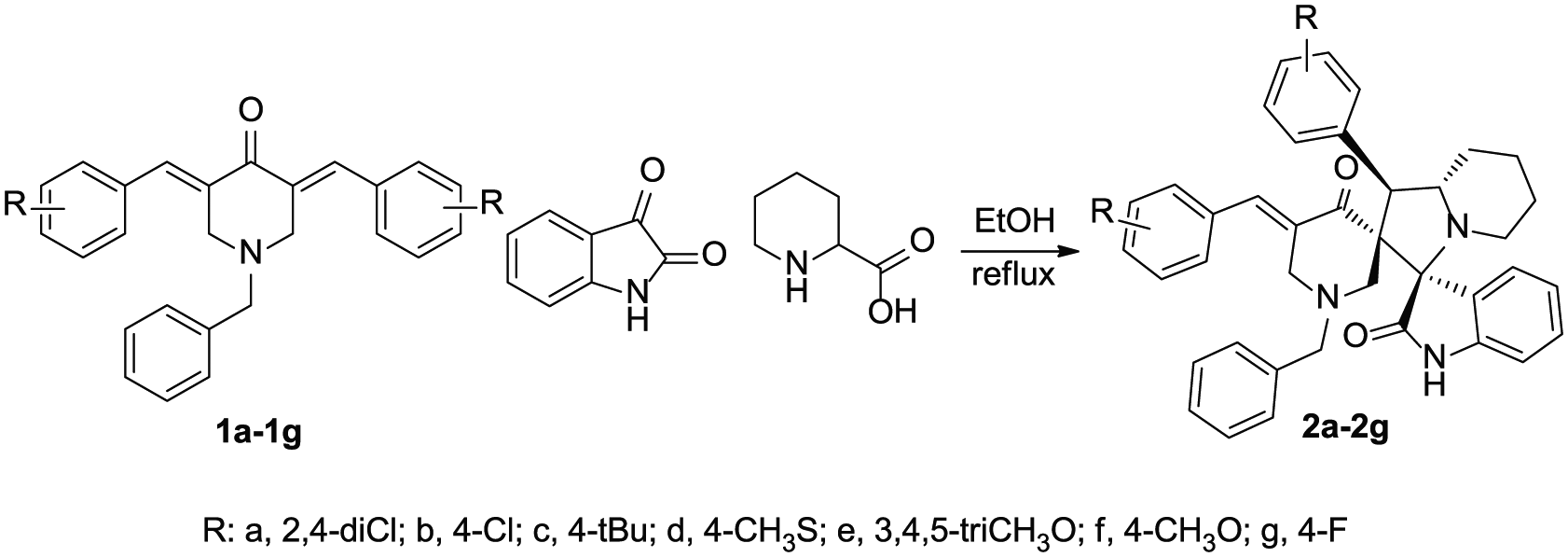
Synthesis of hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione compounds.
Results and discussion
Chemistry
The 1-benzyl-3,5-diarylmethylidene-piperidin-4-one (
The possible way of the azomethine ylide approaching the dipolarophile

A plausible mechanism for the formation of hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione compounds.
In heteronuclear multiple bond correlation (HMBC), the correlations from characteristic H-4 to C2′ (δ 57.81), C4′, C2 and C5 (δ 30.66) can be easily observed. The HMBC showed the key correlations from H-2′ (δ 3.23 and 1.87) to C6′ (δ 54.29), C4′ and spiro carbons C3 and C2. Protons of –NH (δ 7.92) and H-4″ (δ 7.11) correlate with spiro carbon C2. Additional HMBC correlations from the olefin proton (δ 7.55) of 2,4-dichlorobenzylidene moiety to C4′ and C6′ and from the CH2 protons (δ 3.53 and 3.03) of N-benzyl group to C2′ and C6′ are observed. All the HMBCs supported the dispiro heterocycle structure of

Partial HMBC correlations of

ORTEP diagram of
Bioassay
All the synthesized hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione compounds were evaluated for their cytotoxicity (brine shrimp bioassay).
14
Five products showed different cytotoxic effects against Artemia salina. In particular, compound
Experimental
All the chemical reagents are commercially available without further purification. All of the solvents were dried and redistilled before use. The TLC analysis was performed with silica gel GF254 plates. The column chromatography was carried out using silica gel (200–300 mesh). All NMR spectra were recorded on a Bruker AV-II 500 MHz NMR spectrometer – 1H at 500 MHz and 13C at 125 MHz. Transcranial magnetic stimulation (TMS) was used as an internal reference for 1H and 13C chemical shifts, and CDCl3 was used as a solvent. All mass spectra were obtained using a Waters ACQUITY UPLC XEVO Q-TOF mass spectrometer. IR spectra were recorded on a Perkin-Elmer spectrometer (Spectrum One). Melting points were measured with a Yanaco MP500 melting point apparatus and are uncorrected. X-ray analysis was performed on a Bruker Apex-II CCD diffractometer. The crystallographic data for compound
Crystallographic data for

General procedure for the synthesis of hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]dione compounds (2a–g)
A mixture of (3E,5E)-1-benzyl-3,5-bis(2,4-dichlorobenzylidene)piperidin-4-one (
(1′RS,2′SR,3′RS,8a′SR)-1″-benzyl-5″-(2,4-dichlorobenzylidene)-1′-(2,4-dichlorophenyl)-1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2a)
White solid, yield 81%; m.p.: 237–239 °C; IR (KBr) (v cm−1): 3421, 1691; 1H NMR (500 MHz, CDCl3) δ 7.92 (s, 1H), 7.87 (d, J = 8.5 Hz, 1H), 7.56 (s, 1H), 7.38 (d, J = 1.6 Hz, 1H), 7.32 (d, J = 9.1 Hz, 2H), 7.16 (m, 4H), 7.11 (d, J = 7.5 Hz, 1H), 6.96 (d, J = 8.4 Hz, 1H), 6.92 (t, J = 7.6 Hz, 1H), 6.89 (m, 2H), 6.75 (d, J = 7.6 Hz, 1H), 6.60 (d, J = 8.4 Hz, 1H), 4.64 (d, J = 9.4 Hz, 1H), 3.86 (t, J = 9.5 Hz, 1H), 3.53 (d, J = 13.5 Hz, 1H), 3.23 (d, J = 13.1 Hz, 1H), 3.20 (d, J = 16.1 Hz, 1H), 3.03 (d, J = 13.5 Hz, 1H), 2.70 (d, J = 15.2 Hz, 1H), 2.29 (d, J = 10.0 Hz, 1H), 2.10 (td, J = 10.5, 3.2 Hz, 1H), 1.87 (d, J = 12.6 Hz, 1H), 1.71 (m, 2H), 1.42 (m, 2H), 1.22 (m, 2H). 13C NMR (125 MHz, CDCl3) δ 196.44, 177.20, 142.03, 136.97, 135.69, 135.15, 134.78, 134.13, 133.60, 132.95, 131.93, 131.65, 130.30, 129.59, 129.15, 128.68, 128.60, 128.31 × 2, 128.11, 128.07 × 2, 127.04, 127.00, 126.73, 126.52, 122.79, 108.76, 77.17, 63.41, 62.70, 61.80, 57.81, 54.29, 49.00, 45.71, 30.66, 25.25, 24.05. ESI-HRMS calc. for [C39H34Cl4N3O2]+ ([M+H]+): 716.1400, Found: 716.1402.
(1′RS,2′SR,3′RS,8a′SR)-1″-benzyl-5″-(4-chlorobenzylidene)-1′-(4-chlorophenyl)- 1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2b)
White solid, yield 68 %; m.p.: 213–215 °C; IR (KBr) (v cm−1): 3220, 1694; 1H NMR (500 MHz, CDCl3) 1H NMR (500 MHz, CDCl3) δ 8.20 (s, 1H), 7.33–7.30 (m, 2H), 7.29 (m, 2H), 7.23–7.16 (m, 3H), 7.13 (d, J = 8.1 Hz, 2H), 7.07 (d, J = 8.2 Hz, 2H), 7.01 (d, J = 7.3 Hz, 2H), 6.95–6.90 (m, 2H), 6.77 (d, J = 8.2 Hz, 2H), 6.67 (d, J = 7.6 Hz, 1H), 4.25 (d, J = 10.4 Hz, 1H), 3.79 (t, J = 8.8 Hz, 1H), 3.68 (d, J = 13.3 Hz, 1H), 3.55 (d, J = 12.3 Hz, 1H), 3.33 (d, J = 14.6 Hz, 1H), 3.15 (d, J = 13.2 Hz, 1H), 2.65 (dd, J = 14.5, 1.9 Hz, 1H), 2.34 (t, J = 6.6 Hz, 2H), 2.25 (d, J = 6.6 Hz, 2H), 1.86 (dt, J = 12.5, 6.2 Hz, 2H), 1.78 (d, J = 12.5 Hz, 1H), 1.37 (m, 2H). 13C NMR (125 MHz, CDCl3) δ 198.52, 177.82, 141.95, 136.60, 136.49, 136.12, 134.49, 133.83, 133.37, 132.57, 131.66, 130.91 × 2, 128.72 × 2, 128.44 × 2, 128.41 × 2, 128.31, 128.12 × 2, 127.84, 127.75, 127.12, 121.96 × 2, 108.65, 75.62, 64.74, 62.01, 61.38, 56.97, 53.52, 52.31, 45.39, 30.78, 25.55, 23.76. ESI-HRMS calc. for [C39H36Cl2N3O2]+ ([M+H]+): 648.2179, Found: 648.2180.
(1′RS,2′SR,3′RS,8a′SR)-1″-benzyl-5″-(4-tert-butylbenzylidene)-1′-(4-tert-butylphenyl)-1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2c)
Pale yellow syrupy, yield 75%; IR (KBr) (v cm−1): 3264, 1696; 1H NMR (500 MHz, CDCl3) δ 7.82 (s, 1H), 7.29 (m, 3H), 7.21 (d, J = 8.3 Hz, 3H), 7.17 (d, J = 7.2 Hz, 2H), 7.15 (d, J = 6.6 Hz, 1H), 7.12 (d, J = 8.2 Hz, 1H), 7.08 (t, J = 7.7 Hz, 1H), 7.01 (d, J = 6.7 Hz, 3H), 6.94 (t, J = 7.5 Hz, 1H), 6.85 (d, J = 8.2 Hz, 2H), 6.64 (d, J = 7.7 Hz, 1H), 4.24 (d, J = 10.3 Hz, 1H), 3.83 (t, J = 8.5 Hz, 1H), 3.64 (d, J = 13.4 Hz, 1H), 3.53 (d, J = 12.7 Hz, 1H), 3.39 (d, J = 14.5 Hz, 1H), 3.27 (d, J = 13.4 Hz, 1H), 2.82 (d, J = 14.6 Hz, 1H), 2.34 (t, J = 6.6 Hz, 1H), 2.26 (t, J = 8.5 Hz, 1H), 1.86 (m, 1H), 1.81 (d, J = 12.5 Hz, 1H), 1.72 (m, 1H), 1.38 (m, 2H), 1.31 (s, 9H), 1.26 (m, 2H), 1.25 (s, 9H). 13C NMR (125 MHz, CDCl3) δ 198.86, 177.82, 151.77, 149.36, 141.80, 137.22, 136.56, 134.85, 132.90, 132.39, 129.90 × 2, 128.94 × 2, 128.53, 128.22, 127.97 × 2, 127.97, 126.90 × 2, 126.01, 125.16 × 2, 125.02 × 2, 122.02, 108.47, 75.67, 65.05, 61.78, 61.37, 56.42, 53.68, 52.81, 45.47, 34.68, 34.41, 31.40 × 3, 31.12 × 3, 30.93, 25.67, 23.86. ESI-HRMS calc. for [C47H54N3O2]+ ([M+H]+): 692.4211, Found: 692.4211.
(1′RS,2′SR,3′RS,8a′SR)-1″-benzyl-5″-(4-methylthiobenzylidene)-1′-(4-methylthiophenyl)-1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2d)
White solid, yield 83 %; m.p.: 222–224 °C; IR (KBr) (v cm−1): 3258, 1695; 1H NMR (500 MHz, CDCl3) δ 7.30 (m, 2H), 7.20 (d, J = 7.1 Hz, 4H), 7.18–7.14 (m, 1H), 7.09 (d, J = 7.9 Hz, 1H), 7.08–7.04 (m, 1H), 7.03–6.98 (m, 5H), 6.91 (t, J = 7.5 Hz, 1H), 6.81 (d, J = 8.2 Hz, 2H), 6.64 (d, J = 6.5 Hz, 1H), 4.24 (d, J = 10.3 Hz, 1H), 3.80 (t, J = 9.0 Hz, 1H), 3.68 (d, J = 13.3 Hz, 1H), 3.54 (d, J = 12.5 Hz, 1H), 3.38 (d, J = 14.6 Hz, 1H), 3.16 (d, J = 13.3 Hz, 1H), 2.76–2.69 (m, 1H), 2.48 (s, 3H), 2.41 (s, 3H), 2.24 (d, J = 6.5 Hz, 2H), 1.83 (d, J = 12.6 Hz, 1H), 1.77 (d, J = 8.4 Hz, 1H), 1.71 (s, 1H), 1.49–1.43 (m, 1H), 1.42–1.36 (m, 1H), 1.33 (d, J = 12.2 Hz, 1H), 1.26 (m, 1H). 13C NMR (125 MHz, CDCl3) δ 198.59, 177.67, 141.88, 139.96, 136.99, 136.88, 136.40, 135.01, 132.63, 131.53, 130.40 × 2, 128.71 × 2, 128.59, 128.08 × 2, 127.97, 127.81, 127.00 × 2, 126.57 × 2, 125.37 × 2, 123.61, 121.90, 108.47, 75.70, 64.71, 62.15, 61.41, 56.94, 53.93, 52.60, 45.43, 30.83, 25.60, 23.82, 15.93, 15.06. ESI-HRMS calc. for [C41H42N3O2S2]+ ([M+H]+): 672.2713, Found: 672.2708.
(1′RS,2′SR,3′RS,8a′SR)-1″-benzyl-5″-(3,4,5-trimethoxybenzylidene)-1′-(3,4,5-trimethoxyphenyl)-1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2e)
White solid, yield 66 %; IR (KBr) (v cm−1): 3308, 1708; m.p.: 231–233 °C; 1H NMR (500 MHz, CDCl3) δ 7.68 (s, 1H), 7.29 (s, 1H), 7.18 (t, J = 7.3 Hz, 2H), 7.16 (s, 2H), 7.10 (t, J = 7.5 Hz, 1H), 7.08 (t, J = 7.5 Hz, 1H), 7.04 (d, J = 7.6 Hz, 2H), 7.00 (s, 1H), 6.93 (t, J = 7.5 Hz, 1H), 6.69 (d, J = 7.7 Hz, 1H), 6.11 (s, 2H), 4.18 (d, J = 9.8 Hz, 1H), 3.90 (s, 6H), 3.85 (s, 3H), 3.79 (s, 3H), 3.75 (d, J = 11.5 Hz, 1H), 3.71 (d, J = 6.9 Hz, 1H), 3.64 (s, 6H), 3.60 (d, J = 12.5 Hz, 1H), 3.42 (d, J = 14.7 Hz, 1H), 3.19 (d, J = 12.9 Hz, 1H), 2.76 (d, J = 14.6 Hz, 1H), 2.28 (d, J = 5.2 Hz, 2H), 1.89 (d, J = 12.6 Hz, 1H), 1.84 (d, J = 9.2 Hz, 1H), 1.73 (m, 2H), 1.51 (m, 1H), 1.33–1.28 (m, 2H). 13C NMR (125 MHz, CDCl3) δ 198.56, 177.46, 152.93 × 2, 152.67 × 2, 141.85, 138.50, 137.51, 136.87, 136.74, 136.31, 133.78, 132.80, 132.64, 130.50, 128.91 × 2, 128.50, 128.13 × 2, 127.88, 127.24, 121.97, 108.47 × 2, 107.28 × 2, 75.71, 64.58, 62.48, 61.97, 60.86 × 2, 56.32, 56.23, 55.86 × 2, 53.86, 53.46, 45.46, 30.94, 29.71, 25.61, 23.85. ESI-HRMS calc. for [C45H50N3O8]+ ([M+H]+): 760.3592, Found: 760.3591.
(1′RS,2′SR,3′RS,8a′SR)-1″-benzyl-5″-(4-methoxybenzylidene)-1′-(4-methoxyphenyl)-1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2f)
White solid, yield 63 %; m.p.: 225–227 °C; IR (KBr) (v cm−1): 3430, 1693; 1H NMR (500 MHz, CDCl3) δ 7.92 (s, 1H), 7.31 (m, 2H), 7.23–7.13 (m, 3H), 7.10 (d, J = 7.5 Hz, 1H), 7.06 (d, J = 7.9 Hz, 2H), 7.04 (d, J = 7.7 Hz, 2H), 6.91 (d, J = 7.6 Hz, 1H), 6.88 (d, J = 8.8 Hz, 2H), 6.85 (d, J = 8.2 Hz, 2H), 6.70 (d, J = 8.5 Hz, 2H), 6.64 (d, J = 7.7 Hz, 1H), 4.23 (d, J = 10.4 Hz, 1H), 3.80 (s, 3H), 3.74 (s, 3H), 3.70 (d, J = 13.3 Hz, 1H), 3.53 (d, J = 12.5 Hz, 1H), 3.38 (d, J = 14.5 Hz, 1H), 3.16 (d, J = 13.3 Hz, 1H), 2.94 (s, 1H), 2.88 (s, 1H), 2.74 (dd, J = 14.4, 1.9 Hz, 1H), 2.24 (d, J = 6.3 Hz, 2H), 1.84 (d, J = 12.6 Hz, 1H), 1.79–1.67 (m, 2H), 1.45 (d, J = 12.3 Hz, 1H), 1.34–1.28 (m, 2H). 13C NMR (125 MHz, CDCl3) δ 198.77, 177.66, 162.60, 159.85, 158.35, 141.91, 137.33, 137.11, 131.89 × 2, 131.38, 130.15, 128.75 × 2, 128.47, 128.14, 128.06 × 2, 127.84, 127.81, 126.93 × 2, 121.85, 113.69 × 2, 113.61 × 2, 108.40, 75.81, 64.61, 62.29, 61.67, 57.00, 55.22, 54.11, 52.42, 45.47, 30.87, 29.72, 25.66, 23.90. ESI-HRMS calc. for [C41H42N3O4]+ ([M+H]+): 640.3170, Found: 640.3173.
(1′RS,2′SR,3′RS,8a′SR)-1′-benzyl-5′-(4-fluorobenzylidene)-1′-(4-fluorophenyl)-1′,5′,6′,7′,8′,8a′-hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione (2g)
White solid, yield 85 %; m.p.: 243–245 °C; IR (KBr) (v cm−1): 3426, 1711; 1H NMR (500 MHz, CDCl3) δ 7.95 (s, 1H), 7.35 (m, 2H), 7.22–7.14 (m, 3H), 7.09 (t, J = 6.1 Hz, 2H), 7.00 (t, J = 8.0 Hz, 4H), 6.96 (s, 1H), 6.93 (t, J = 7.6 Hz, 1H), 6.87–6.80 (m, 4H), 6.66 (d, J = 7.8 Hz, 1H), 4.26 (d, J = 10.4 Hz, 1H), 3.79 (t, J = 8.8 Hz, 1H), 3.68 (d, J = 13.3 Hz, 1H), 3.54 (d, J = 12.5 Hz, 1H), 3.34 (d, J = 14.4 Hz, 1H), 3.16 (d, J = 13.3 Hz, 1H), 2.67 (dd, J = 14.5, 2.2 Hz, 1H), 2.24 (d, J = 6.5 Hz, 2H), 1.77 (d, J = 12.6 Hz, 4H), 1.44 (m, 1H), 1.35–1.28 (m, 2H). 13C NMR (125 MHz, CDCl3) δ 198.70, 177.68, 163.50, 162.82, 161.51, 160.87, 141.93, 136.70, 136.33, 133.65, 133.23, 131.63 × 2, 131.11, 128.74 × 2, 128.66, 128.09 × 2, 127.81, 127.09 × 2, 121.96, 115.28 × 2, 115.11 × 2, 108.55, 75.65, 64.72, 62.02, 61.60, 56.99, 53.58, 52.23, 45.42, 30.80, 25.59, 23.80. ESI-HRMS calc. for [C39H36F2N3O2]+ ([M+H]+): 616.2770, Found: 616.2770.
Cytotoxicity study protocol (brine shrimp assay)
Fifty milligrams of brine shrimp (Artemia salina Leach) eggs were hatched in a 500-mL flask filled with artificial seawater under constant aeration. After 48 h, the nauplii were collected by pipette from the lighted side. Ten nauplii were transferred to each well of 96-well plates. Each well was made up to 195 μL with artificial seawater. Sample solution was prepared with dimethyl sulfoxide (DMSO) at the initial concentration of 4.0 mg/mL. Being diluted with DMSO, sample solutions at five concentration gradients, 4.0, 2.0, 1.0, 0.5 and 0.25 mg/mL, were obtained. The initial concentration of positive control, podophyllotoxin, was 2.0 mg/mL, and six concentration gradients were prepared. From each sample, 5 μL of sample solution was transferred to each well, and each sample was repeated three times (LC50 is the mean of three values). For each sample, one well was kept as control by adding only 5 μL of DMSO. After 24 h, the number of survivors were counted and analysed by Bliss 17 method to determine the LC50 values. The brine shrimp bioassay results of hexahydrodispiro[indole-3,3′-indolizine-2′,3′′-piperidine]-2(1H),4″-dione compounds and positive control podophyllotoxin are listed in Table 2.
Brine shrimp bioassay data of dispiro heterocyclic compounds and podophyllotoxin.

Conclusion
Seven region-selective hexahydrodispiro[indole-3,3′-indolizine-2′,3″-piperidine]-2(1H),4″-dione compounds were synthesized by a catalyst-free one-pot 1,3-dipolar cycloaddition reaction in a three-component system containing 1-benzyl-3,5-diarylidenepiperidin-4-one, isatin and
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (No. 21172066), the Scientific Research Fund of Hunan Provincial Education Department (No. 15C0560) and the Open Project Programme of the Key Laboratory of Theoretical Chemistry and Molecular Simulation of the Ministry of Education (No. E21203).
