Abstract
A new series of C2-glycosyl benzofuranylthiazole derivatives was synthesised by the further cyclization of glycosyl thiourea and 2-(bromoacetyl)-benzofuran via Hantzsch’s method. The corresponding 2-(bromoacetyl)-benzofuran derivatives were obtained by the reaction from various salicylaldehydes, and the glycosyl thiourea was prepared through a series of steps from D-Glucosamine. The acetylcholinesterase-inhibitory activities of the products were tested by Ellman’s method. The most active compounds were subsequently evaluated for the 50% inhibitory concentration values. N-(1,3,4,6-tetra-O-benzyl-2-deoxy-β-D-glucopyranosyl)-4-(5-methoxy-benzofuran-2-yl)-1,3-thiazole-2-amine possessed the best acetylcholinesterase-inhibition activity with a 50% inhibitory concentration of 2.03 ± 0.26 μM.
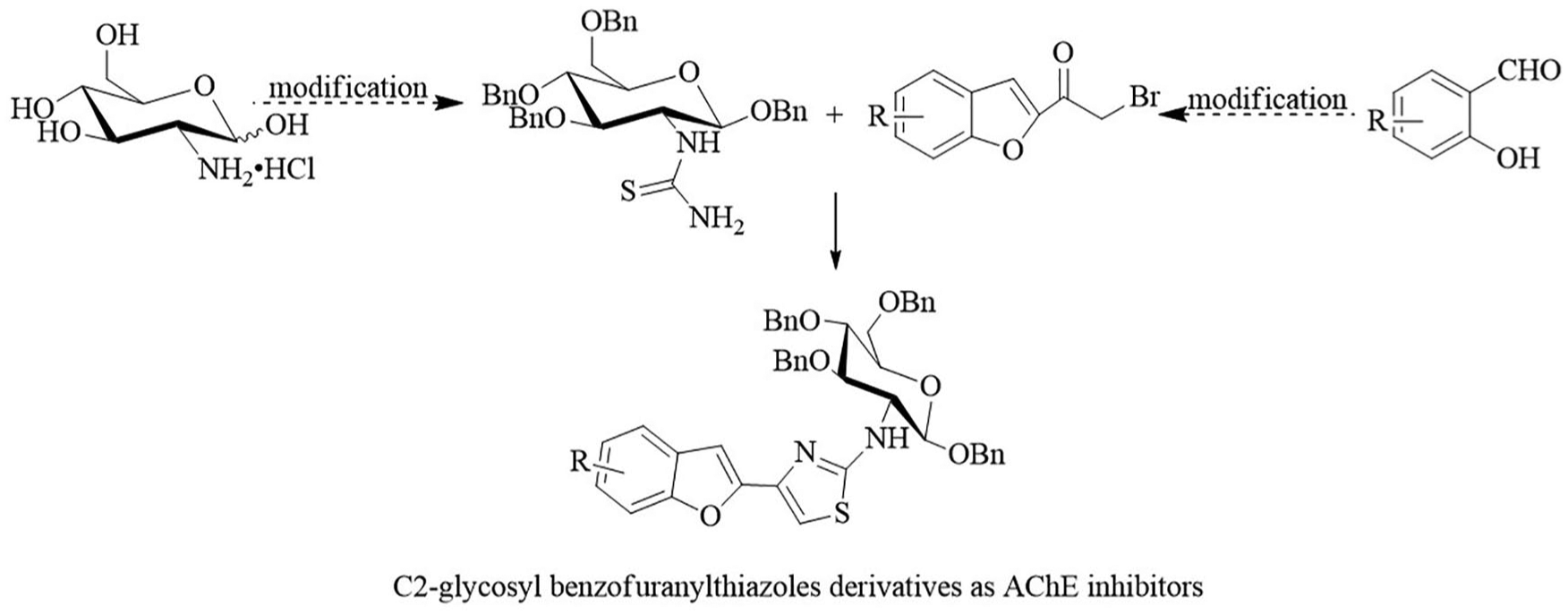
Introduction
Carbohydrates have long attracted the interest of chemists and biochemists due to their leading role in biological and industrial applications. 1 As an energy source and a feature of many metabolic processes, sugar exists in every part of the human body. 2 D-glucosamine is a naturally occurring amino sugar, 3 one of the most abundant monosaccharides and is widely used in the treatment of rheumatoid arthritis and osteoarthritis. 4 Furthermore, D-glucosamine exhibits a broad variety of bioactivities such as anti-inflammatory, anti-cancer, antibacterial and the suppression of tumour growth.5,6 Modified carbohydrates provide access to potential mimetics of naturally occurring amino sugars and represent targets for the development of antioxidant, anti-acetylcholinesterase (AChE), anti-proliferative, and other active agents.7–9
The emergence of heterocyclic compounds with novel structures may promote discovery of new drugs and treatment of refractory diseases. 10 Natural coumarones are oxygen heterocycles having anti-inflammatory, anti-cancer, anti-virus, anti-tuberculosis, and anti-senile dementia activities11–13 and provide important heterocyclic active molecules for the development of new drugs. Currently, the main clinical drugs containing the coumarone skeleton are antiarrhythmic amiodarone, antihypertensive bufurolol, and antifungal griseofulvin. 14 Moreover, recent studies have shown that a number of compounds containing the coumarone skeleton act as monoamine oxidase inhibitors for the treatment of Alzheimer’s disease (AD). 15
To date, researchers have been interested in molecular hybrid-based approaches to discover new compounds with potential biological activities. 16 Accordingly herein, we report the design and synthesis of novel D-glucosamine/benzofuranylthiazoles hybrids with the coumarone ring providing an important pharmacophore in the search to discover new potent AChE inhibitors. The synthesised derivatives were evaluated by Ellman’s method for AChE inhibitors and by exploring the influence of D-glucosamine hydrochlorides against AChE-inhibition activity.
Results and discussion
Chemistry
To develop a simple synthetic pathway to C2-glycosyl benzofuranylthiazole compounds, glycosyl thiourea was a critical intermediate in this reaction. 1,3,4,6-Tetra-O-benzyl-β-D-glucosamine hydrochloride (

Synthetic pathways of the C2-glycosylthiourea.
We then studied Compound
Optimising the conditions for the synthesis of
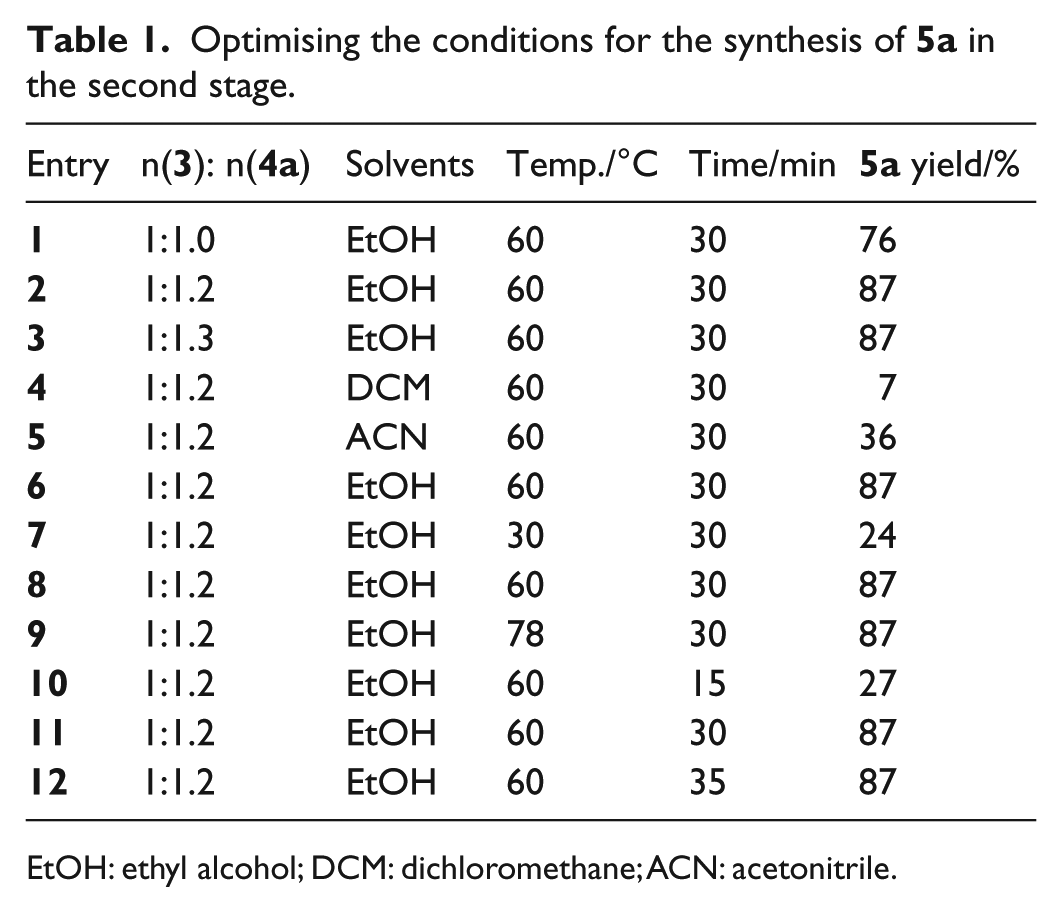
EtOH: ethyl alcohol; DCM: dichloromethane; ACN: acetonitrile.

Synthetic pathways of the C2-glycosyl benzofuranylthiazole derivatives.
Biological activity
The AChE-inhibition activities of the newly synthesised compounds were evaluated in vitro by Ellman’s method, 20 in which the AChE extracts from Electric eel were used. Their inhibitory potency was described by the inhibition rate at half of the maximal inhibitory concentration, IC50. The results are summarised in Table 2.
In vitro inhibitory activities of glycosyl benzofuranylthiazoles against AChE.
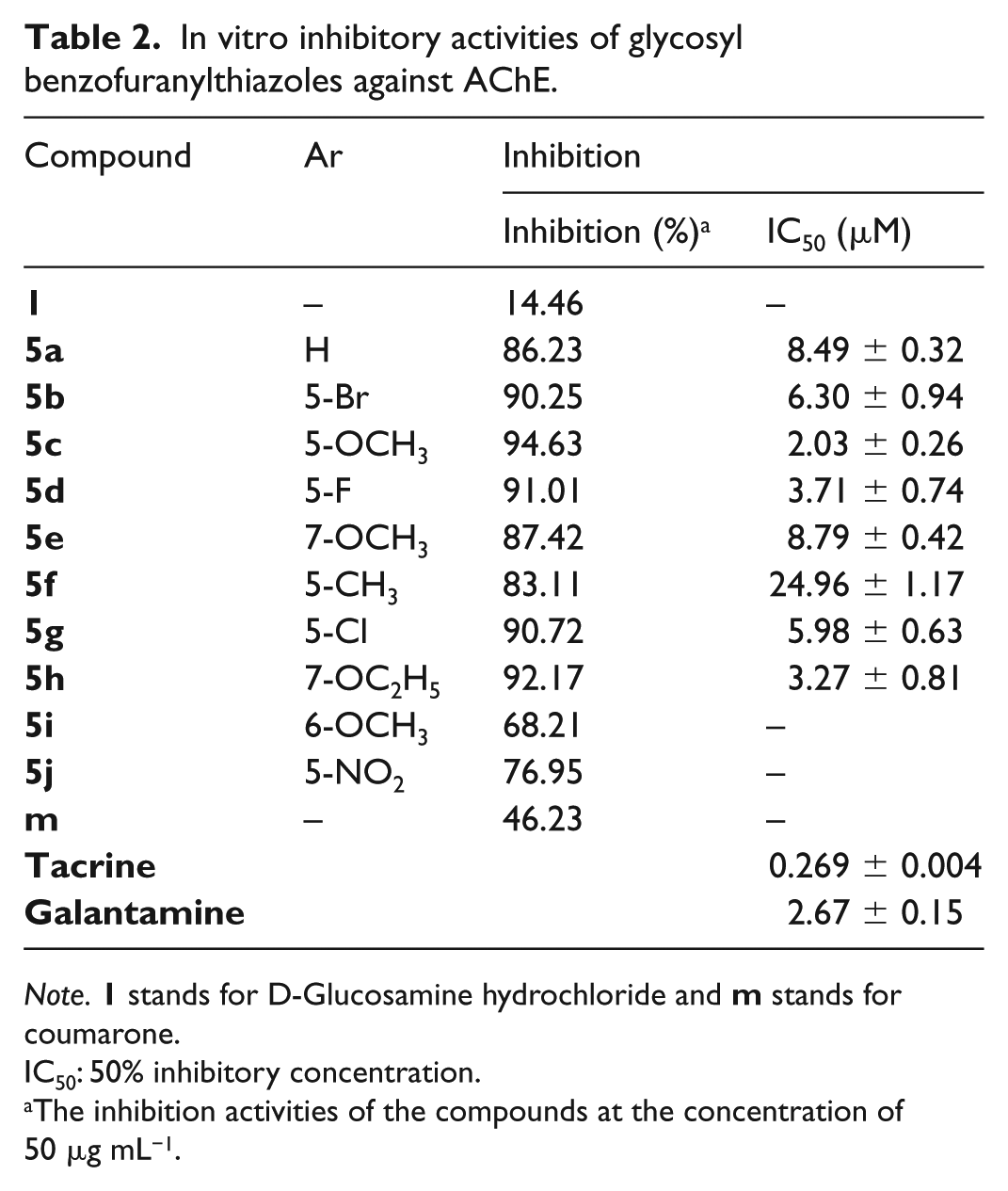
IC50: 50% inhibitory concentration.
The inhibition activities of the compounds at the concentration of 50 μg mL−1.
As shown in Table 2, it was found that 5 of the 10 desired compounds exhibited over 90.0% inhibition and these were subsequently evaluated for their IC50 values, while tacrine and galantamine were used as reference compounds. These results indicated that all the compounds gave higher inhibitory activities against AChE than the precursor compound coumarone (
Experimental
Chemistry
All chemicals were purchased from commercial sources and were used without further purification unless otherwise stated. Melting points (m.p.s) were determined on a Yanaco m.p. apparatus and were uncorrected. Infrared (IR) spectra were recorded on a Bruker Tensor 27 spectrometer with KBr pellets.
1
H NMR spectra were recorded on a Bruker Avance 500 MHz at ambient temperature using dimethyl sulfoxide (DMSO-
General procedure for the preparation of N
Using the aforementioned procedure the following compounds were similarly prepared.
In vitro cholinesterase activity assay
AChE, acetylthiocholine iodide (ATCI), 5,5-dithiobis-(2-nitrobenzoic acid) (DTNB), galantamine, and tacrine were purchased from Sigma-Aldrich. AChE activities were measured through Ellman’s colorimetric method with a slight modification,
20
using galantamine and tacrine as the reference compounds. An
Conclusion
A new series of C2-glycosyl benzofuranylthiazole derivatives were designed using methods involving mild conditions, convenient operations, and low toxicity. These novel compounds were characterised by nuclear magnetic resonance (NMR), IR, and HRMS. Most of the compounds are active against AChE. Compound
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project Funded by the Postgraduate Research & Practice Innovation Programme of Jiangsu Province (KYCX17-2074 and KYCX18-2580), Open-end Funds of Jiangsu Key Laboratory of Marine Biotechnology (HS2014007), Project 521 Funded by Lianyungang (LYG52105-2018023), and Public Science and Technology Research Funds Projects of Ocean (201505023).
