Abstract
A simple, one-pot synthesis of six 3-aryl-2,4-diselenoxo-1,3,5-triazepane-6,7-diones has been achieved in moderate to good yields via a three-component condensation of variously substituted anilines, oxalyl chloride, and two molecules of potassium selenocyanate in acetone at room temperature.
Keywords

Multicomponent reactions (MCRs) have been frequently used by synthetic chemists as a facile means to generate molecular diversity from bifunctional substrates that react sequentially in an intramolecular fashion. 1 Molecules with triazepine skeletons have attracted much attention as a result of their interesting biological properties. 2 Triazepanediones represent a particular subset, of which a number of functional isomers have been synthesized.3–5 Applications of compounds with triazepanedione skeletons have been reported. 6 With regard to monocyclic triazepines, 7 1,2,4-triazepines have been prepared by the cycloaddition of 1-azirines to 1,2,4,5-tetrazines 8 or by a photochemical walk rearrangement of 3,4,7-triaza-2,4-norcaradienes. 9 Although the synthesis of seven-membered [1,2,4]-triazepine derivatives is well precedent in the literature,10–12 few reports are known for [1,2,5]-triazepine systems.13–17 The current interest in selenium-containing organic compounds stems from their remarkable synthetic and biological functions.18–22 The first synthesis of acyl isoselenocyanates, which were generated by the reaction of acyl chlorides and potassium selenocyanate, has been described by Douglas. 23
Recently, a synthesis of 3-aryl-2,4-dithioxo-1,3,5-triazepane-6,7-diones from oxalyl chloride, anilines, and two molecules of ammonium thiocyanate in acetone was reported. 24 Herein, we describe a synthesis of the seleno analog, namely 3-aryl-2,4-diselenoxo-1,3,5-triazepane-6,7-diones, from variously substituted anilines, oxalyl chloride, and two molecules of potassium selenocyanate in acetone at room temperature using a reported method. 24
Results and discussion
A recent report
24
described the synthesis of a series of 3-aryl-2,4-dithioxo-1,3,5-triazepane-6,7-diones by the reaction between oxalyl chloride, anilines, and two molecules of ammonium thiocyanate (NH4SCN) in acetone under ultrasound irradiation. We thought that a synthesis of some analogues of the 3-aryl-2,4-diselenoxo-1,3,5-triazepane-6,7-diones, 3-aryl-2,4-diselenoxo-1,3,5-triazepane-6,7-diones (

The reaction of substituted anilines, oxalyl chlorideand two molecules of potassium selenocyanate inacetone at room temperature.
So it proved, and a series of six such compounds were prepared in good yield using a variety of substituted anilines (
Yields of a series of 3-aryl-2,4-diselenoxo-1,3,5-triazepane-6,7-diones (
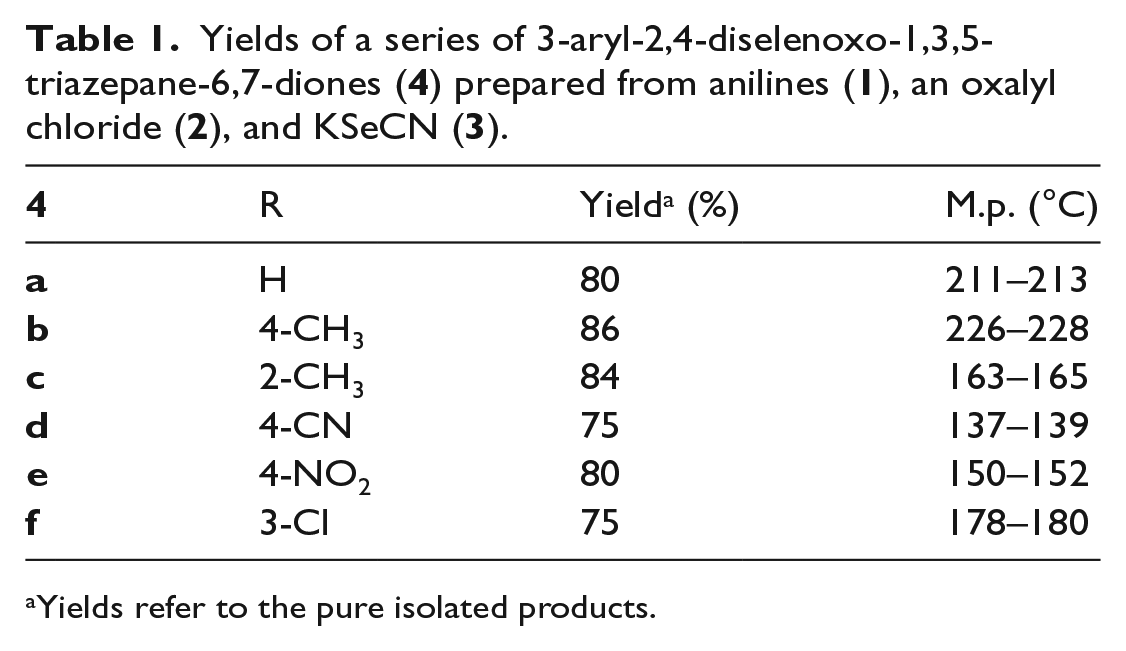
Yields refer to the pure isolated products.
The structures of compounds
A tentative mechanism for this transformation is proposed in Scheme 2. It is conceivable that oxalyl diisoselenocyanate

Suggested mechanism for formation of compound
Experimental
Melting points were determined with an Electrothermal 9100 apparatus and are uncorrected. Elemental analyses for C and H were performed using a Heraeus CHN-O-Rapid analyzer. Mass spectra were recorded on a FINNIGAN-MAT 8430 mass spectrometer operating at an ionization potential of 70 eV. IR spectra were recorded on a Shimadzu IR-470 spectrometer. NMR spectra were obtained on a Bruker DRX 500 Avance spectrometer (1H NMR at 500 Hz, 13C NMR at 125 Hz) in DMSO-d6 using TMS as an internal standard. Chemical shifts (δ) are given in parts per million (ppm). All of the chemicals used in this study were purchased from Merck and Fluka (Buchs, Switzerland) and were used without further purification.
Caution! All of the reactions involving selenium-containing compounds should be carried out in a well-ventilated hood.
General procedure
To a solution of potassium selenocyanate (2 mmol) in dry acetone (3 mL) was added oxalyl chloride (1 mmol) in dry acetone (3 mL). The reaction mixture was stirred at r.t. for 10 min. Anilines (1 mmol) in dry acetone (4 mL) was added to the mixture and stirring was continued at r.t. for 12 h. The progress of the reaction was monitored by TLC. After completion of the reaction, the resulting precipitate was collected by filtration on a Buchner funnel and washed with cold water (20 mL) and recrystallized from EtOH to afford the pure title compounds.
3-Phenyl-2,4-diselenoxo-1,3,5-triazepane-6,7-dione
3-(4-Methylphenyl)-2,4-diselenoxo-1,3,5-triazepane-6,7-dione
3-(2-Methylphenyl)-2,4-diselenoxo-1,3,5-triazepane-6,7-dione
4-(6,7-Dioxo-2,4-diselenoxo-1,3,5-triazepan-3-yl)benzonitrile
3-(4-Nitrophenyl)-2,4-diselenoxo-1,3,5-triazepane-6,7-dione
3-(3-Chlorophenyl)-2,4-diselenoxo-1,3,5-triazepane-6,7-dione
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
