Abstract
A series of novel C-glycosides of heterocyclic derivatives containing a pyrimidin-2-amine or a 1H-pyrazolo[3,4-b]pyridine moiety were synthesized using condensation reactions of the substituted puerarin with guanidine or 3-amino-5-hydroxypyrazole in methyl alcohol. Their chemical structures were characterized by Fourier-transform infrared spectroscopy, nuclear magnetic resonance, and high-resolution mass spectrometry. In addition, their biological activity has been demonstrated by in vitro evaluation against the human leukemia cells K562 and human prostate cancer cells PC-3 by MTT-based assays, using the commercially available standard drug of cis-platin as a positive control. The results also demonstrated that most of the compounds showed considerable cytotoxicity to these two cell lines of K562 and PC-3, and indicated that novel C-glycosides of heterocyclic derivatives may be potential leads for further biological screenings and may generate drug-like molecules.

Introduction
Glycosides have received increasing interest in medicinal chemistry as carbohydrate biomimetics.1–3 As compared to O-glycosidic linkage in glycosides the C-glycosidic linkage offers stability to glycosidic bonds toward acidic or enzymatic hydrolysis and therefore these compounds are comparatively more stable. C-glycosides of synthetic and natural origins have been reported to possess a vast array of biological activities, such as antitumor, 4 antibiotic, 5 or SGLT2 inhibitor activity. 6 Among these compounds, C-glycosides of heterocycles are essential and reliable platforms for the development of many anticancer and antitumor drugs 7 and have been regarded as good glycosyl donors, in addition to their biological activities such as inhibition of enzyme activity. 8
In Chinese herbal medicine, flavonoids constitute the largest and most important group of polyphenolic compounds, which possess various beneficial biological properties, including anti-inflammatory,
9
antiviral,
10
and antitumor activities.
11
Puerarin, in which a β-D-glucopyranose is directly linked through a carbon–carbon bond to the A-ring of daidzein[7-hydroxy-3-(4-hydroxyphenyl)chromone], was isolated from Pueraria lobota,
12
one of the most popular Chinese herbal medicines. It has beneficial effects on cardiovascular, neurological, and hyperglycemic disorders.13,14 It could suppress the phosphorylation and degradation of NF-kB
15
and inhibit mRNA and protein expression of C-reactive protein in lipopolysaccharide (LPS)-stimulated peripheral blood mononuclear cells, suggesting anti-inflammatory activity.
16
In addition, previous research has found that the puerarin nanosuspensions suppressed the HT-29 proliferation, increased the early apoptosis, and enhanced the in vivo anticancer effectiveness toward human colon cancer HT-29 cells.
17
Subsequently, a finding suggested that a puerarin nanosuspension effectively inhibited the growth of HepG2 cells in vitro.
18
Recently, puerarin has been reported to significantly inhibit LPS-induced MCF-7 and MDA-MB-231 cell migration, invasion, and adhesion,
19
and suppress the enzymatic activity of cyclooxygenase-2 (COX-2) in A549 cells and its expression in tumor tissues with pulmonary metastasis.
20
In the reported experiments, the efforts were centered mostly on the substitutions on the aromatic rings (either phenolic hydroxyl or glucosyl group) of puerarin.21–24 It was reported that the chromone fragment can generate a 1,3-diketone equivalent in the presence of alkali, which readily reacts with amidines25,26 and hydrazine
27
to form the corresponding pyrimidines and pyrazoles. We report herein a facile construction of novel C-glycosides of heterocycles containing the pyrimidin-2-amine or the 1H-pyrazolo[3,4-b]pyridine
Results and discussion
Initially, the reaction of puerarin (
Screening of reaction conditions a .

THF: tetrahydrofuran; DMSO: dimethyl sulfoxide; DMF: dimethylformamide.
All reactions were carried out on a 1 mmol scale of
Reactions at reflux in EtOH, MeOH, THF, and MeCN at 90 °C in DMF and DMSO.
Isolated yield after chromatography.
No reaction.
The optimized conditions were applied to the cyclocondensation of the substituted puerarin (
Synthesis of C-glycosides of heterocycles
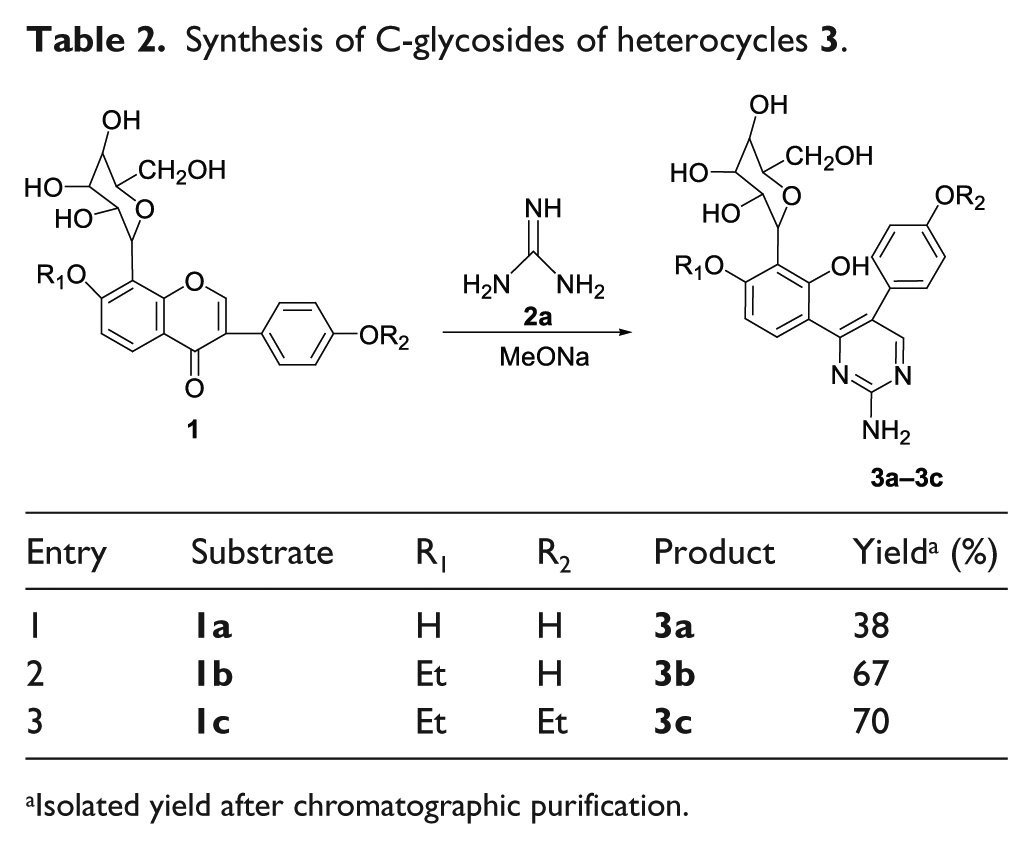
Isolated yield after chromatographic purification.

Isolated yield after chromatographic purification.
To explain the formation of

The proposed mechanism of cyclocondensation.
Subsequently, to further demonstrate the potential activities of these synthesized C-glycosides of heterocycles
In vitro antitumor activities of target compounds.
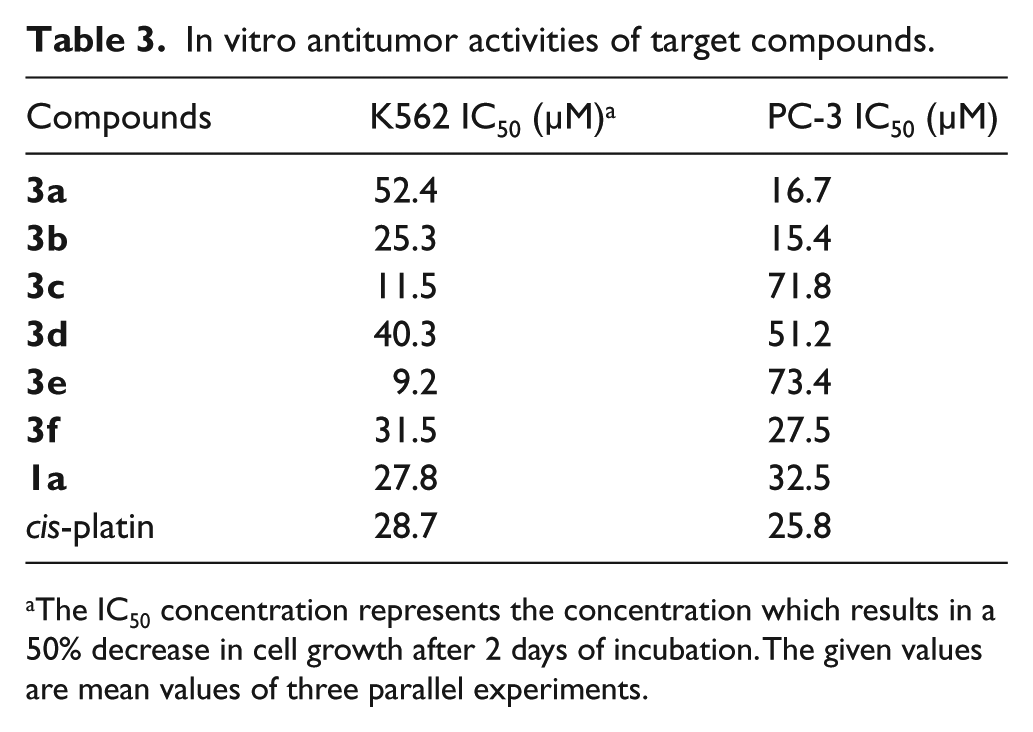
The IC50 concentration represents the concentration which results in a 50% decrease in cell growth after 2 days of incubation. The given values are mean values of three parallel experiments.
Experimental
The 1H and 13C NMR spectra were recorded on Bruker Avance DMX 300 MHz NMR spectrometers in dimethyl sulfoxide (DMSO)-d6 using tetramethylsilane (TMS) as the internal standard. Chemical shifts were reported as δ values (ppm). High-resolution mass spectrometry electrospray ionization (HRMS-ESI) spectra were obtained on a Micro™ Q-TOF mass spectrometer. The infrared (IR) spectra were recorded on a PerkinElmer One FTIR spectrograph using KBr pellets. Melting points (m.p.) were uncorrected and recorded on an Electrothermal IA9100 digital melting point apparatus.
Reagents were purchased from commercial sources and used as received unless mentioned otherwise. Reactions were monitored by thin-layer chromatography (TLC) using silica gel GF254 plates. Column chromatography was performed on silica gel (300–400 mesh).
Synthesis of 7-alkoxy-3-(4-alkoxyphenyl)-8-C-β-D-glucopyranosyl-4H-chromen-4-one (1b –c )
The mixture of puerarin
Synthesis of the ethylated products of puerarin (3a –c )
The mixture of puerarin or its derivatives (
4-(2,4-Dihydroxy-3-C-β-D-glucopyranosylphenyl)-5-(4-hydroxyphenyl)pyrimidin-2-amine (
4-(2-Hydroxy-3-C-β-D-glucopyranosyl-4-ethyoxyphenyl)-5-(4-hydroxyphenyl)pyrimidin-2-amine (
4-(2-Hydroxy-3-C-β-D-glucopyranosyl-4-ethyoxyphenyl)-5-(4-ethyoxyphenyl)pyrimidin-2-amine (
Synthesis of 3-hydroxy-5,6-diphenyl-1H-pyrazolo[3,4-b]pyridines (3d –f )
The mixture of puerarin or its derivatives (
3-Hydroxy-5-(4-hydroxyphenyl)-6-(2,4-dihydroxy-3-C-β-D-glucopyranosyl)-1H-pyrazolo[3,4-b]pyridine (
3-Hydroxy-5-(4-hydroxyphenyl)-6-(2-hydroxy-3-C-β-D-glucopyranosyl-4-ethyoxyphenyl)-1H-pyrazolo[3,4-b]pyridine (
4-Hydroxy-5-(4-ethyoxyphenyl)-6-(2-hydroxy-3-C-β-D-glucopyranosyl-4-ethyoxyphenyl)-1H-pyrazolo[3,4-b]pyridine (
Bioassay of the antitumor activities 29
Cell culture
The leukemic cells K562 and human prostate cancer cells PC-3 were incubated with an RPMI-1640 medium. 29 The number of tumor cells is 5 × 103 cells per hole in a 96-well plate and the tumor cells were incubated for 24 h at 37 °C, and the air in the incubator is humid air with 5% CO2.
Antitumor activity evaluation
DMSO solutions of compounds

The IC50 values were calculated by prohibiting regression using SPSS Statistics17.0 software.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Scientific Research Program Funded by Shaanxi Provincial Education Department (Nos 18JK0837 and 16JK1822), Natural Science Basic Research Plan funded by Shaanxi Province of China (Nos 2018JM2045 and 2016JM5024), Science and Technology Projects of Xianyang City (No. 2017k02–19), Scientific Research Project funded by Xianyang Normal University (No. XSYK18006), Youth Backbone Teachers Project funded by Xianyang Normal University (No. XSYGG201606), University Students Research and Innovation Training Program of Ministry of Education (201810722010), University Students Research and Innovation Training Program of Shaanxi Province (Nos 201828010 and 2490), University Students Research and Innovation Training Program of Xianyang Normal University (Nos 2018003, 2017060, and 201710722003), and Scientific Research Project funded by Ministry of Land Resources (No. SXDJ2017-3).
