Abstract
Ten novel ureido-substituted 1,3-benzoxazines were synthesized in moderate yields by La(OTf)3-catalyzed reaction of some aldehydes with four ureido-substituted 2-aminomethylphenols which were prepared by a one-pot method. Evaluation of their fungicidal activity revealed that most compounds showed moderate to good activity and one of the ureido-substituted 2-aminomethylphenols showed activity against Rhizoctonia solani and Sclerotonia sclerotiorum comparable to the control compound, chlorothalonil.

Introduction
Urea derivatives play an important role as pesticides and as drugs. Many of them have good biological activity as herbicides, 1 insecticides,2–4 fungicides, 4 and plant growth regulators, 5 and as antiviral,4,6 antitumor, 7 anticancer, 8 and antituberculosis agents. 9 They are also stearyl-CoA desaturase inhibitors, 10 and some are neuroprotection agents against Aβ-induced cytotoxicity. 11 The ureido group has high cytokinin activity and selectivity, and a series of bioactive compounds can be formed by integration of a ureido group with 1,5-diarylpyrazoles 12 and with other groups. 13 On the other hand, heterocyclic nitrogen compounds such as 1,3-benzoxazine derivatives have received more and more attention due to their wide range of biological activities including anti-inflammatory, 14 anti-mycobacterial, 15 anti-cancer, 16 anti-fungal17,18 and anti-plasmodial activity, 19 and as CCR2 and CCR5 dual antagonists, 20 and DNA-PK inhibitors. 21 Molecular hybridization, which involves the combination of two or more distinct bioactive units, is one of the successful strategies for the development of new drug classes.22,23 Hence, in a continuation of our programs aimed at synthesizing new fungicidal compounds with high fungicidal potency,17,18 we integrated the ureido group with the 1,3-benzoxazine ring to form novel ureido-substituted 1,3-benzoxazine hybrids. Here we wish to report the preparation and fungicidal activity of the target compounds.
Results and discussion
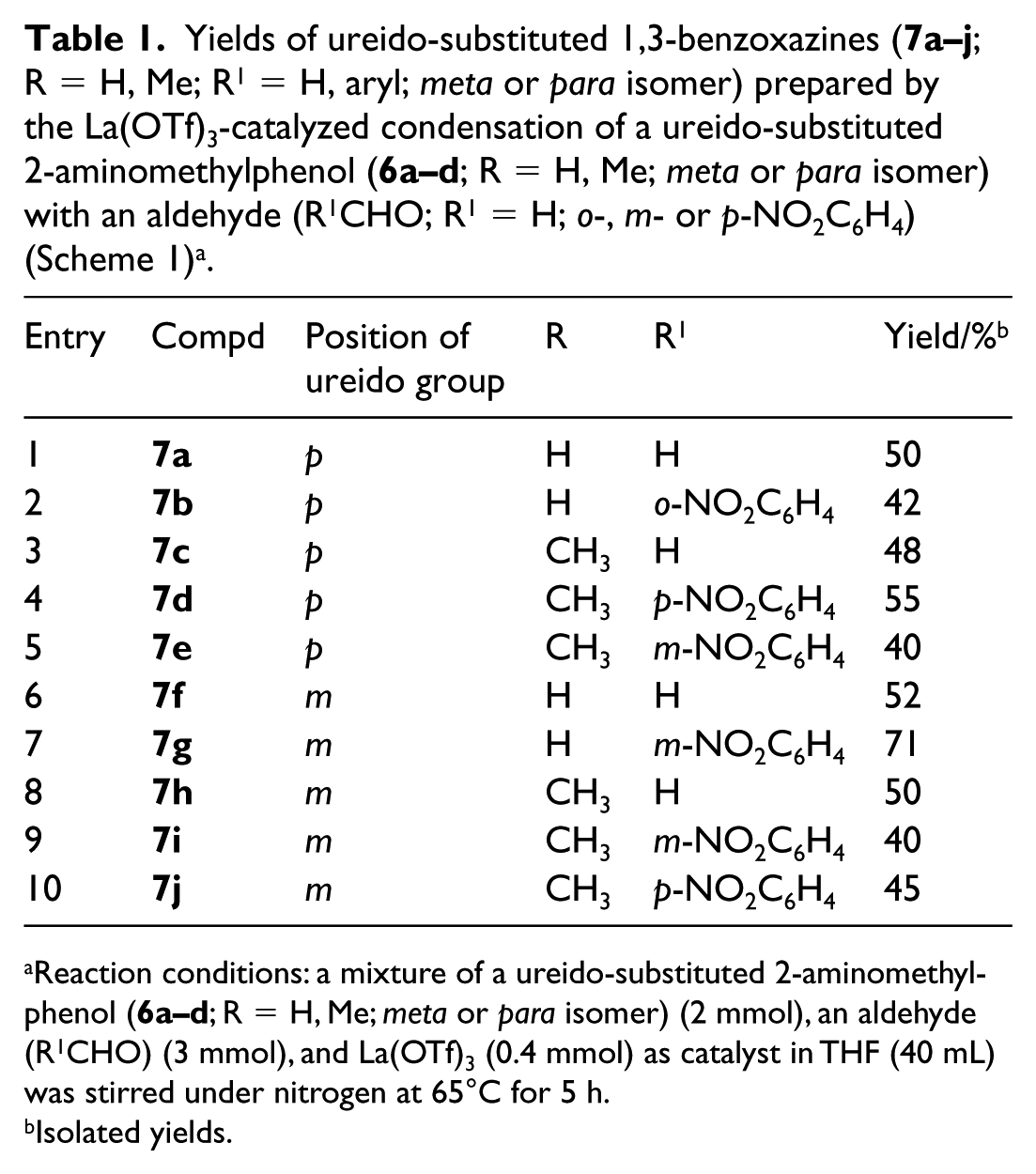
As shown in Scheme 1, the important intermediates (

Synthesis of compounds 7.
Reaction conditions: a mixture of a ureido-substituted 2-aminomethylphenol (
Isolated yields.
The in vitro fungicidal activities of the prepared compounds
Inhibition rate (%).
Chlorothalonil used as reference compound.
Conclusion
In summary, we have developed an efficient method to prepare ureido-substituted 1,3-benzoxazines by La(OTf)3-catalyzed reactions of ureido-substituted 2-aminomethylphenols with aldehydes under mild reaction conditions. The ureido-substituted 2-aminomethylphenols were synthesized by a one-pot reaction of phenylenediamine, N,N-diethylformyl chloride, substituted salicylaldehydes and NaBH4. The fungicidal activities of the prepared compounds against plant fungi were evaluated, and some compounds showed good activities. Compound
Experimental
NMR spectra were obtained on a Bruker AV-II 500 spectrometer (1H NMR at 500 MHz, 13C NMR at 125 MHz) in CDCl3 using TMS as internal standard. Chemical shifts (δ) are given in ppm and coupling constants (J) are given in hertz (Hz). Melting points were measured on a WRS-2A microcomputer melting point instrument and are uncorrected. HRMS were recorded on a Waters UPLC/XEVOQ-TOF LC/MS instrument. IR spectra were recorded on a Nicolet-6700 FTIR spectrometer. Solvents, reagents and catalysts were purchased from MERYER and Energy Chemical as analytical grades and used without further purification. Column chromatography was carried out using silica gel (200–300 mesh).
Synthesis of ureido-substituted 2-aminomethylphenols (6a–d ; R = H, Me; meta or para isomer); general procedure
Under a nitrogen atmosphere, into a 100 mL three-necked round-bottom flask equipped with a condenser, p- or m-phenylenediamine (0.216 g, 2 mmol), N,N-diethylformyl chloride (0.49 g, 3.6 mmol), Et3N (0.202 g, 2 mmol) and CH2Cl2 (30 mL) were added. The reaction was heated to 40°C in an oil bath for 12 h. After removing the solvent under reduced pressure, salicylaldehyde (0.733 g, 6 mmol) or 5-methylsalicylaldehyde (0.817 g, 6 mmol) and ethanol (40 mL) were added. The reaction mixture was refluxed for 5 h and cooled to room temperature. Then, NaBH4 (0.11 g, 3 mmol) was added portion-wise at 0°C and the mixture was stirred for 1 h. After concentration, the mixture was diluted with ethyl acetate (40 mL) and the organic layer was washed successively with distilled water (20 mL × 3) and saturated NaCl solution (20 mL × 3). The organic solvent was dried over Na2SO4, evaporated under reduced pressure, and the residue purified by column chromatography (petroleum ether (boiling point is 60-90°C)/ethyl acetate = 2:1) affording product
N,N-Diethyl-N′-(4-{[(2-hydroxyphenyl)methyl]amino}phenyl)urea (
N,N-Diethyl-N’-(4-{[(2-hydroxy-5-methylphenyl)methyl]amino}phenyl)urea (
N,N-Diethyl-N’-(3-{[(2-hydroxyphenyl)methyl]amino}phenyl)urea (
N,N-Diethyl-N’-(3-{[(2-hydroxy-5-methylphenyl)methyl]amino}phenyl)urea (
Synthesis of ureido-substituted 1,3-benzoxazines (
Under a nitrogen atmosphere, into a 100 mL three-necked round-bottom flask equipped with a condenser, compound
N,N-Diethyl-N’-[4-(3,4-dihydro-2H-1,3-benzoxazin-3-yl)phenyl]urea (
N,N-Diethyl-N’-{4-[2-(2-nitrophenyl)-3,4-dihydro-2H-1,3-benzoxazin-3-yl]phenyl}urea (
N,N-Diethyl-N’-[4-(6-methyl-3,4-dihydro-2H-1,3-benzoxazin-3-yl)phenyl]urea (
N,N-Diethyl-N’-{4-[6-methyl-2-(4-nitrophenyl)-3,4-dihydro-2H-1,3-benzoxazin-3-yl]phenyl}urea (
N,N-Diethyl-N’-{4-[6-methyl-2-(3-nitrophenyl)-3,4-dihydro-2H-1,3-benzoxazin-3-yl]phenyl}urea (
N,N-Diethyl-N’-[3-(3,4-dihydro-2H-1,3-benzoxazin-3-yl)phenyl]urea (
Light yellow solid; m.p. 143.7–145.2°C; yield 52%; IR (KBr) ν: 3319, 2965, 1639, 1607, 1542, 1439, 1367, 1224, 1168, 754 cm–1; 1H NMR (500 MHz, CDCl3): δ 7.34 (t, J = 2.0 Hz, 1H), 7.12 (dt, J = 15.5, 8.0 Hz, 2H), 7.01 (d, J = 7.4 Hz, 1H), 6.89–6.79 (m, 3H), 6.76 (dd, J = 8.2, 2.2 Hz, 1H), 6.25 (s, 1H), 5.35 (s, 2H), 4.63 (s, 2H), 3.36 (q, J = 7.2 Hz, 4H), 1.22 (t, J = 7.2 Hz, 6H); 13C NMR (125 MHz, CDCl3): δ 154.4, 154.3, 148.9, 140.2, 129.4, 127.7, 126.8, 121.0, 120.7, 116.9, 112.5, 112.1, 109.3, 79.1, 50.3, 41.6 (2C), 13.9 (2C); HRMS (ESI) m/z calcd for C19H24N3O2: [M + H]+: 326.1869; found: 326.1858.
N,N-Diethyl-N’-{3-[2-(3-nitrophenyl)-3,4-dihydro-2H-1,3-benzoxazin-3-yl]phenyl}urea (
N,N-Diethyl-N’-[3-(6-methyl-3,4-dihydro-2H-1,3-benzoxazin-3-yl)phenyl]urea (
N,N-Diethyl-N’-{3-[6-methyl-2-(3-nitrophenyl)-3,4-dihydro-2H-1,3-benzoxazin-3-yl]phenyl}urea (
N,N-Diethyl-N’-{3-[6-methyl-2-(4-nitrophenyl)-3,4-dihydro-2H-1,3-benzoxazin-3-yl]phenyl}urea (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos. 21877034, 21372070), the Scientific Research Fund of Hunan Provincial Education Department (17A066), and the Open Foundation of Key Laboratory of Theoretical Organic Chemistry and Functional Molecule of the Ministry of Education (LKF1603).