Abstract
1-Ethyl-3-methylimidazolium trifluoromethanesulfonate ([emim][OTf]) is an efficient co-solvent and co-promoter for one-pot sequential glycosylation with the combined use of thioglycosides and trichloroacetimidates (or N-phenyltrifluoroacetimidates) donors at room temperature. One-pot glycosylation is efficient for the synthesis of triterpenoid saponins with the combined use of N-phenyltrifluoroacetimidate donors and 2-methyl-5-tert-butylphenyl (Mbp) thioglycoside donors in [emim][OTf].
Introduction
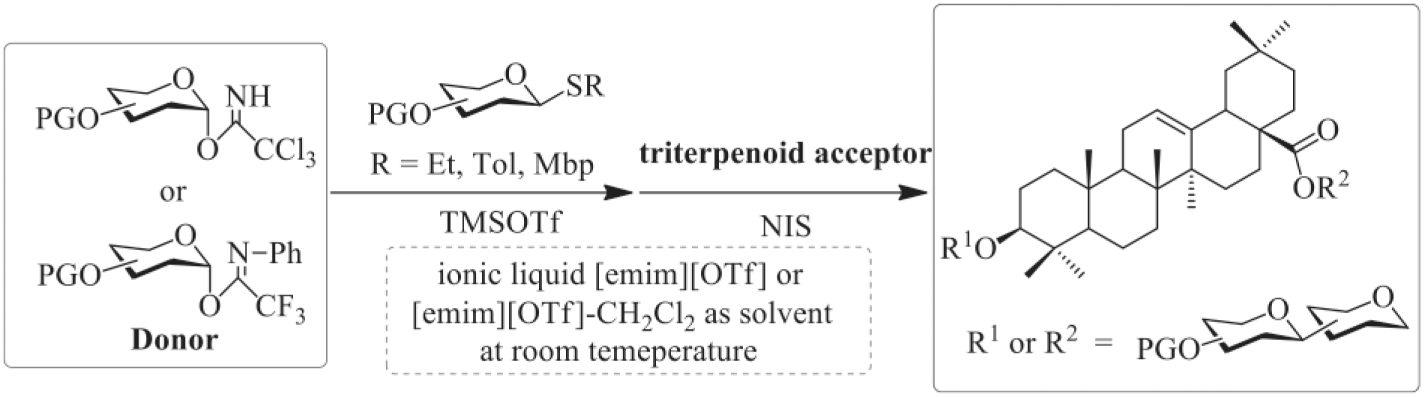
Triterpenoid saponins are glycosylated secondary metabolites that exist widely in terrestrial plants and some marine organisms and present a broad spectrum of well-defined biological and pharmacological activities.1–5 Attracted by these interesting biological activities, several research groups have reported on the synthesis of many oleanane-type triterpenoid saponins.6–8 Notably, the development of a ‘one-pot sequential glycosylation’ strategy has greatly facilitated the synthesis of this kind of glycoconjugate with complicated sugar moieties.9,10 Recently, we completed the synthesis of several natural and non-natural triterpenoid saponins by adopting the ‘one-pot sequential glycosylation’ strategy with the combined use of 2-methyl-5-tert-butylphenyl (Mbp) thioglycosides and trichloroacetimidates as donors (equation (1) in Scheme 1).11–13 Although this strategy is efficient for the assembly of saccharides onto aglycones, there are some drawbacks, such as low reaction temperatures (−78 oC), the use of an environmentally toxic and volatile organic solvent (CH2Cl2) and the need for molecular sieves as dehydrating agents (4 Å MS).

One-pot sequential glycosylation with the combined use of thioglycosides and trichloroacetimidates (or N-phenyltrifluoroacetimidates) donors in [emim][OTf] at room temperature.
Ionic liquids (ILs) are a class of solvents which are quite different from traditional organic solvents due to their high polarity with poorly coordinating ion pairs, which usually results in improved accelerating effects in chemical reactions. 14 ILs are often fluid at room temperature and have a low vapour pressure. They have been widely applied as green replacements for organic solvents. 15 In particular, ILs have had many applications in oligosaccharide synthesis as green solvents and catalysts.16–19 Recently, Galan et al. 20 reported the successful application of [bmim][OTf] as a mild promoter in ambient three-component reactivity-based one-pot glycosylation reactions. In the light of these research findings, and in conjunction with our interest in searching for a general, efficient and green glycosylation strategy, we report in this article the use of [emim][OTf] 18 as co-solvent and co-promoter for one-pot sequential glycosylation with the combined use of thioglycosides and trichloroacetimidates (or N-phenyltrifluoroacetimidates) donors at room temperature (equation (2) in Scheme 1).
Experimental
Commercial reagents were all analytically or chemically pure and used without further purification unless specified. All anhydrous solvents were dried and redistilled prior to use in the usual way. Thin-layer chromatography (TLC) was performed on precoated E. Merck Silica Gel 60 F254 plates. Flash column chromatography was performed on silica gel (200–300 mesh, Qingdao, China). Optical rotations were determined with a Perkin–Elmer Model 241 MC polarimeter. 1H NMR and 13C NMR spectra were taken on a JEOL JNM-ECP 600 spectrometer with tetramethylsilane as the internal standard, and chemical shifts are recorded in δ values. Mass spectra were recorded on a quadrupole timeof- flight (Q-TOF) global mass spectrometer.
Results and discussion
In this research using the ionic liquid

Ionic liquid
One-pot glycosylation of different glycosyl trichloroacetimidates

All reactions were performed at room temperature.
The ratio of co-solvent
Yield refers to the isolated product by silica gel column chromatography.

To test the scope and limitations of the reaction, a wide range of trichloroacetimidate donor
One-pot glycosylation of different glycosyl trichloroacetimidates

All reactions were performed at room temperature.
The ratio of co-solvent
Yield refers to the isolated product by silica gel column chromatography.

Taken together, our experimental results demonstrated that one-pot convergent approaches for glycosylation of triterpenoid saponins in the ionic liquid
Conclusion
In conclusion, we have shown the applicability of the ionic liquid
Synthesis of 1-ethyl-3-methylimidazolium trifluoromethanesulfonate ([emim][OTf])
Freshly distilled ethyl trifluoromethanesulfonate (8.95 g, 0.05 mol) was dropped slowly into magnetically stirred N-methylimidazole (4.00 g, 0.05 mol) at 0 °C over a period of 6 h. Stirring was continued for 12 h at 30 °C to complete the reaction. Finally the excess of ethyl trifluoromethanesulfonate (0.27 g, 0.0015 mol) was evaporated at 0.01 mbar and 25 °C. The product (12.7 g, 0.05 mol, quantitative yield) was obtained as a clear and colourless liquid.
General procedure for the preparation of triterpenoid saponins 2a–10a by one-pot sequential glycosylation
The glycosyl trichloroacetimidates
Compounds
3β-O-Acetyl oleanolate 28-O-2,3,4,6-tetra-O-acetyl-α-D-galacopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-β-D-glucopyranosyl-(1→6)-2,4-O-acetyl-3-benzyl-β-D-glucopyranoside (
1H NMR (CDCl3, 600 MHz): δ = 7.31–7.94 (m, 20H, Ph-H), 5.79 (t, J = 9.6 Hz, 1H, H-3″), 5.56 (t, J = 9.6 Hz, 1H, H-4″), 5.48 (dd, J = 9.6, 7.8 Hz, 1H, H-2″), 5.42 (dd, J = 9.2, 3.6 Hz, 1H, H-3″′), 5.40 (d, J = 7.7 Hz, 1H, H-1′), 5.35 (dd, J = 11.0, 3.2 Hz, 1H, H-2″′), 5.24 (t, J = 3.2 Hz, 1H, H-12), 5.08-5.14 (m, 3H, H-2′, H-3′, H-4′), 5.00 (d, J = 8.3 Hz, 1H, H-1″), 4.95 (d, J = 12.4 Hz, 1H, PhCHH), 4.92 (d, J = 3.6 Hz, 1H, H-1″′), 4.79 (d, J = 12.4 Hz, 1H, PhCHH), 4.47 (t-like, J = 8.3, 7.3 Hz, 1H, H-3), 4.25 (d, J = 11.5 Hz, 1H, H-6″′
13C NMR (CDCl3, 150 MHz): δ = 170.6 (C-28), 171.2, 170.6, 170.5, 170.4, 170.3, 169.9, 169.3, 165.9, 165.3, 165.1, 143.2 (C-13), 138.7, 133.8, 133.5, 130.0, 129.9, 128.8, 128.7, 128.6, 128.5, 127.8, 127.6, 126.5, 122.8 (C-12), 101.1 (C-1″′), 96.6 (C-1″), 91.7 (C-1′), 81.1 (C-3), 80.9, 74.6, 73.7, 73.2, 72.4, 71.2, 69.4, 68.4, 68.2, 67.7, 66.9, 66.6, 62.2, 62.1, 55.5, 47.6, 46.9, 45.9, 41.8, 41.1, 39.5, 38.3, 37.9, 37.1, 33.9, 33.2, 33.0, 31.9, 30.8, 29.9, 28.2, 27.9, 25.8, 23.7, 23.6, 23.5, 22.9, 21.5, 21.0, 20.9, 20.7, 18.4, 17.1, 16.9, 15.6.
HRMALDIMS: m/z calcd for C90H110O28Na [M + Na+]: 1661.7076; found: 1661.7097.
3,23-O-isopropylidene hederagenin 28-O-2,3,4,6-tetra-O-acetyl-α-D-galacopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-β-D-glucopyranosyl-(1→6)-2,4-O-acetyl-3-benzyl-β-D-glucopyranoside (10a )
[α]D22 + 15.6 (c 1.12, CHCl3); mp 156–158 oC. IR (KBr) νmax 3061, 2939, 1739, 1438, 1375, 1257, 1087 and 714 cm−1.
1H NMR (Acetone-d6, 600 MHz): δ = 6.98–8.24 (m, 20H, Ph-H), 6.35 (t, J = 9.6 Hz, 1H, H-3″), 5.96 (t, J = 9.6 Hz, 1H, H-4″), 5.89 (t, J = 9.7 Hz, 1H, H-4′), 5.71 (t, J = 9.6 Hz, 1H, H-3′), 5.63 (d, J = 8.2 Hz, 1H, H-1′), 5.59 (dd, J = 9.7, 8.3 Hz, 1H, H-2′), 5.44 (dd, J = 9.6, 3.7 Hz, 1H, H-3″′), 5.39 (d, J = 7.8 Hz, 1H, H-1″), 5.36 (t, J = 3.8 Hz, 1H, H-4″′), 5.34 (d, J = 3.2 Hz, 1H, H-1″′), 5.27 (d, J = 11.0 Hz, 1H, H-6″′-1), 5.10 (d, J = 11.9 Hz, 1H, PhCHH), 5.05 (d, J = 11.9 Hz, 1H, PhCHH), 4.73–4.76 (m, 1H, H-5′), 4.30–4.33 (m, 2H, H-2″′, H-6″′-2), 4.16 (dd, J = 12.1, 3.0 Hz, 1 H, H-6″-1), 4.06 (dd, J = 12.0, 5.4 Hz, 1H, H-6′-1), 3.98–4.02 (m, 3H, H-5″, H-5″′, H-6″-2), 3.91–3.95 (m, 2H, H-23), 3.84 (dd, J = 12.0, 2.3 Hz, 1H, H-6′-2), 3.80 (m, 1H, H-2″), 3.57 (dd, J = 12.0, 4.6 Hz, 1H, H-3), 2.84 (d, J = 13.1 Hz, 1H, H-18), 2.12, 2.11, 2.08, 1.97, 1.95, 1.82 (s each, 3H each, CH3CO × 6), 1.19, 1.14, 1.01, 0.96, 0.90, 0.69 (s each, 3H each, CH3 × 6).
13 C NMR (Acetone-d6, 150 MHz): δ = 176.0 (C-28), 170.8, 170.7, 170.6, 170.5, 170.4, 170.3, 166.2, 166.1, 165.9, 165.6, 165.2, 161.3, 144.1 (C-13), 134.9, 134.6, 134.5, 134.3, 130.9, 130.7, 130.5, 130.4, 130.3, 130.2, 130.1, 130.0, 129.9, 129.8, 129.6, 129.5, 129.4, 129.1, 128.7, 128.3, 123.4, (C-12), 101.2 (C-1′), 99.3 ((CH3)2
HRMALDIMS: m/z calcd for C91H112O28Na [M + Na+]: 1675.7232; found: 1675.7256.
Footnotes
Author contributions
YS and TG contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was financial supported by the financial support from the Foundation of Natural Science Basic Research and Social Development Plan of Shaanxi Province of China (2018JM2060 and 2016SF-240), Shaanxi Youth Science and Technology Star Project (2015KJXX-28), and the Foundation of Shaanxi Educational Committee (15JK1717 and 16JK1768).
