Abstract
Background:
Depression is common after stroke and is associated with increased mortality. However, there are few data on the prevalence after transient ischemic attack (TIA), and it is unclear whether TIA can trigger depression and, if so, what factors might suggest susceptibility in an individual.
Methods:
We completed a systematic review (Medline/PsycINFO/EMBASE searched to 20 March 2025) of published cross-sectional or cohort studies that reported the prevalence of depression at any time point after a TIA and validated the findings in a population-based cohort (Oxford Vascular Study; OXVASC). Pooled prevalence rates were calculated, risk factors reported and regression analyses were used to determine the proportion of between-study heterogeneity that could be accounted for by study methodology.
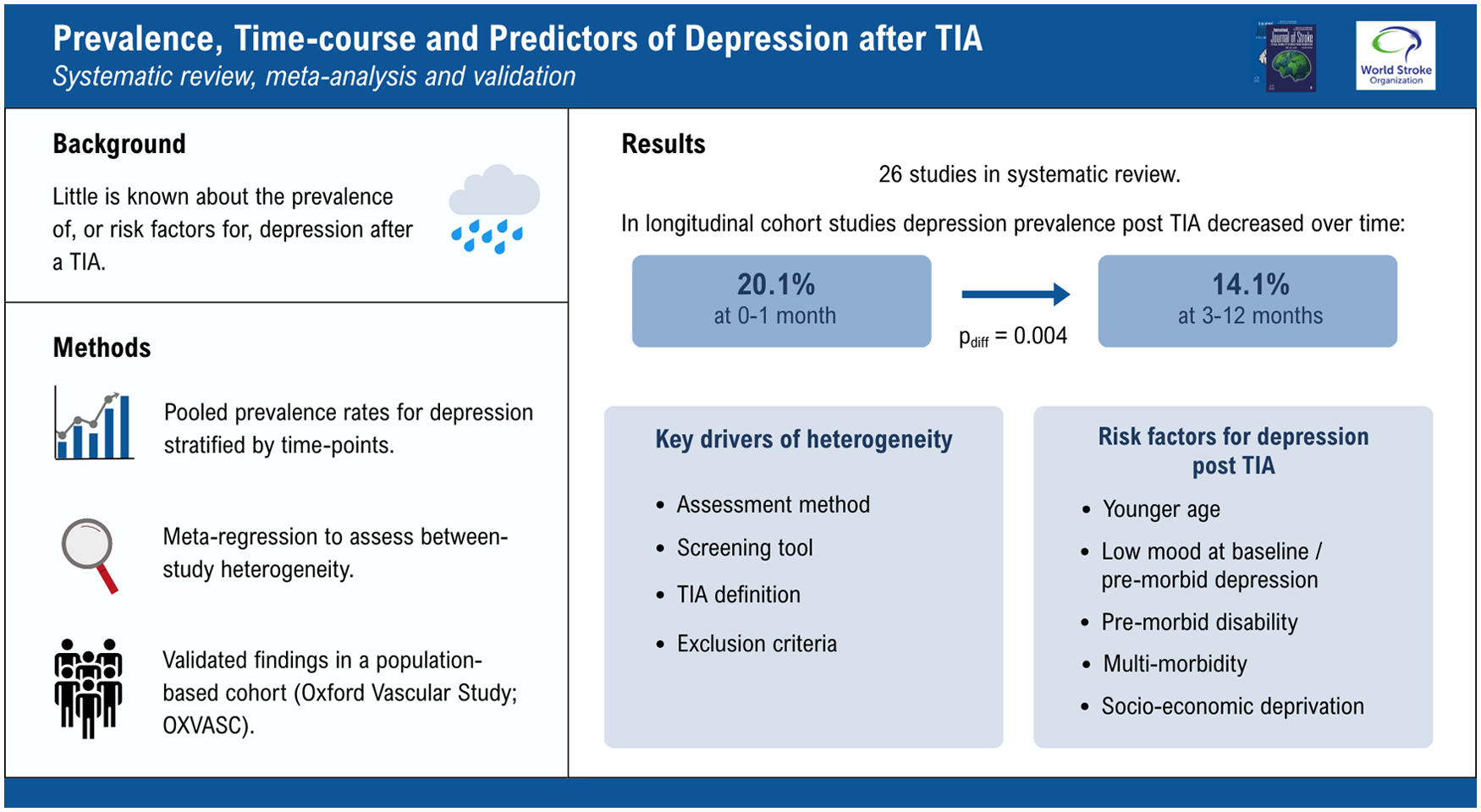
Results:
The search identified 26 studies; 23 reported data at a uniform time point after the TIA but the prevalence rates of depression were highly heterogeneous at each time point studied (phet < 0.001 at <1, 1–6 and 12 months). However, among studies with serial assessments (196 patients from four published longitudinal studies and 478 from OXVASC) there was less heterogeneity and the pooled prevalence of depression fell from 20.1% (16.9%–23.4%; phet = 0.35) at 0–1 month to 14.1% (11.5%–16.7%; phet = 0.40) at 3–12 months (pdiff = 0.004). In the few studies that reported risk factors for depression after a TIA, younger age was the only consistent predictor, but this was similarly validated in the OXVASC population. On regression analysis for heterogeneity, 83% of the variance in prevalence rates between studies was explained by assessment method (postal questionnaire: 5.6%, 95% CI 3.1–8.1 vs face-to-face interview 17.7%, 13.7–21.7; p < 0.001), the screening tool used, TIA definition and exclusion criteria. A model, based on these study methods in published studies, predicted a prevalence rate of 22.1% in OXVASC, which was close to the observed rate of 20.7% at 1 month validating the findings.
Conclusions:
Depression affects about 1-in-5 patients early after a TIA but prevalence falls with time, suggesting that it may be triggered by the event in some cases, but further work is required to identify risk factors. In addition, when planning future research, investigators must remain cognizant of the significant influence that study design has on the observed prevalence rates.
Keywords
Introduction
Major stroke can lead to a depressive disorder with a prevalence rate of around 30% in the first year, but data on depression after a transient ischemic attack (TIA) are sparse.1,2 A few small studies have reported a prevalence of 10% to 20% during the first few months but with limited data beyond that early time point. 3 While a TIA does not leave any permanent neurological impairment, lifestyle and medication changes are usually required and may have a negative impact upon an individual’s psychological wellbeing. A history of pre-morbid depression, severity of stroke, stroke lesion location, lack of social support and education level are all associated with an increased risk of poststroke depression. However, there is limited data on the risk factors in TIA patients. 4 A better understanding of the prevalence, risk factors and etiology of depression after a TIA might allow clinicians to identify vulnerable individuals through targeted screening. This is important as depression is known to be associated with numerous adverse outcomes such as poor compliance with medical therapy, increased risk of dementia and increased all-cause mortality.5–9 Reliable data on the time-course of depression after TIA might also help to determine the extent to which depression is triggered by the TIA or associated treatment or lifestyle changes
We therefore did a systematic review and meta-analysis of studies of published cross-sectional or cohort studies that reported the prevalence of depression at any time point after a TIA to identify the pooled prevalence rates and time-course of depression in the first year after a TIA along with any associated risk factors. We also assessed the associations between study methodology and reported prevalence to better understand the heterogeneity and then validated all the findings in a population-based cohort (Oxford Vascular Study; OXVASC).
Methods
To identify studies on post-TIA depression, we searched Ovid Medline (1946 onward), EMBASE (1974 onward) and PsycINFO (1806 onward) up until 20 March 2025 for studies of any form of psychological distress after a TIA. The search strategy is outlined in Figure S1, and the study protocol was registered on PROSPERO (CRD420251017152) and completed according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.10,11 Two reviewers (AM and PMR) independently screened the studies identified and determined eligibility. Reference lists of all eligible articles and relevant reviews were also hand searched.
Studies were included if they reported the prevalence of depression at any time point after a TIA in a series of consecutively recruited eligible patients. Both tissue-based and time-based definitions of TIA were eligible for inclusion. Studies were required to use a validated screening tool in order to quantify frequencies of depression, though to maximize yield, the tool did not have to have been validated specifically in a stroke or TIA population. Studies on only subsets of a TIA population (e.g. post carotid endarterectomy) were included in the review but the prevalence data were not included in the meta-analysis. Studies were excluded if the data relevant to the TIA population could not be isolated from the data of a mixed population of stroke and TIA, if the full text of the paper was unavailable, or if a translation of a non-English study was unavailable. Where multiple studies used a subset or the whole of the same data set, the duplicate studies were only included if frequencies at different time points were reported, otherwise the study of the largest patient series was included.
Risk of bias was assessed by AM for each study using principles from Cochrane and a systematic review of tools for assessing quality and susceptibility to bias in observational studies in epidemiology.12,13 From the included studies information was extracted on the study characteristics (type, follow-up duration, recruitment method, proportion of eligible assessed, assessment method, tools used and their cut-off values) and patient characteristics (mean age, gender, proportion with psychological symptoms and associated factors). Where provided, data on control (non-TIA/stroke) patients were extracted.
Original source data were also included from the Oxford Vascular Study (OXVASC). Details of the recruitment, follow-up and analysis of this cohort are included in the Supplementary Material (Text S1). 14 Briefly, OXVASC is a population-based cohort of all patients in a subpopulation (approx. 100,000) of Oxfordshire, UK, who sought medical attention with an acute vascular event. In patients presenting with TIA from 2014 to 2020, depression was assessed at 1 and 12 months after the event and related to risk factors (including infarction on brain imaging) with adjustment for covariates.
Statistical analysis
Pooled prevalence rates for depression were calculated with stratification according to the time to assessment after a TIA. Cross-sectional studies in which an assessment was done at a specified time point were stratified as <1 month; 1–6 months; 12 months. Longitudinal studies that reported prevalence rates at different time points in the same cohort were stratified as <1 versus 3–12 months. The prevalence rates for each study were pooled with an inverse variance–weighted random-effects meta-analysis to generate a pooled prevalence rate for the prespecified time-points along with 95% confidence intervals. These 95% confidence intervals were calculated allowing for extra-binomial variation because standard methods of calculating confidence intervals produce artificially narrow intervals if there is heterogeneity of risk between different studies. 15 Reported p-values were two-sided, with significance set at less than 0.05.
Analyses of heterogeneity of prevalence across studies were done with χ2 tests. To determine the sources of any heterogeneity, studies were stratified according to the following methodological criteria: publication year, TIA definition (time vs tissue), follow-up time, face-to-face vs telephone vs postal assessment, screening tool, exclusion criteria (dementia, pre-event disability, pre-event depression) and mean patient age. We used prespecified subgroup analyses to explore the impact of these methodological factors with the pooled prevalence rates for each subgroup calculated by inverse variance–weighted random-effects meta-analysis and compared with χ2 tests. To further evaluate how much between-study heterogeneity was explained by these methodological factors, we performed an inverse-variance weighted meta-regression of the log risk against study type. We then applied this model to the OXVASC population to predict the expected prevalence, adjusting for the specific design characteristics of the OXVASC study. Statistical analyses were performed using SPSS Statistics 25.
Results
Our search (Figure 1, Figure S1) identified 26 published studies (23 cohorts; 3 cross-sectional) that reported at least some data on depression after TIA in addition to the OXVASC study (Table S1).14,16–41 Five studies focused on specific subpopulations (younger adults,24,33 younger adults with evidence of ischemia, 32 carotid artery stenosis, 29 middle cerebral artery events 27 ) and one included transient neurological attacks in addition to TIAs. 35
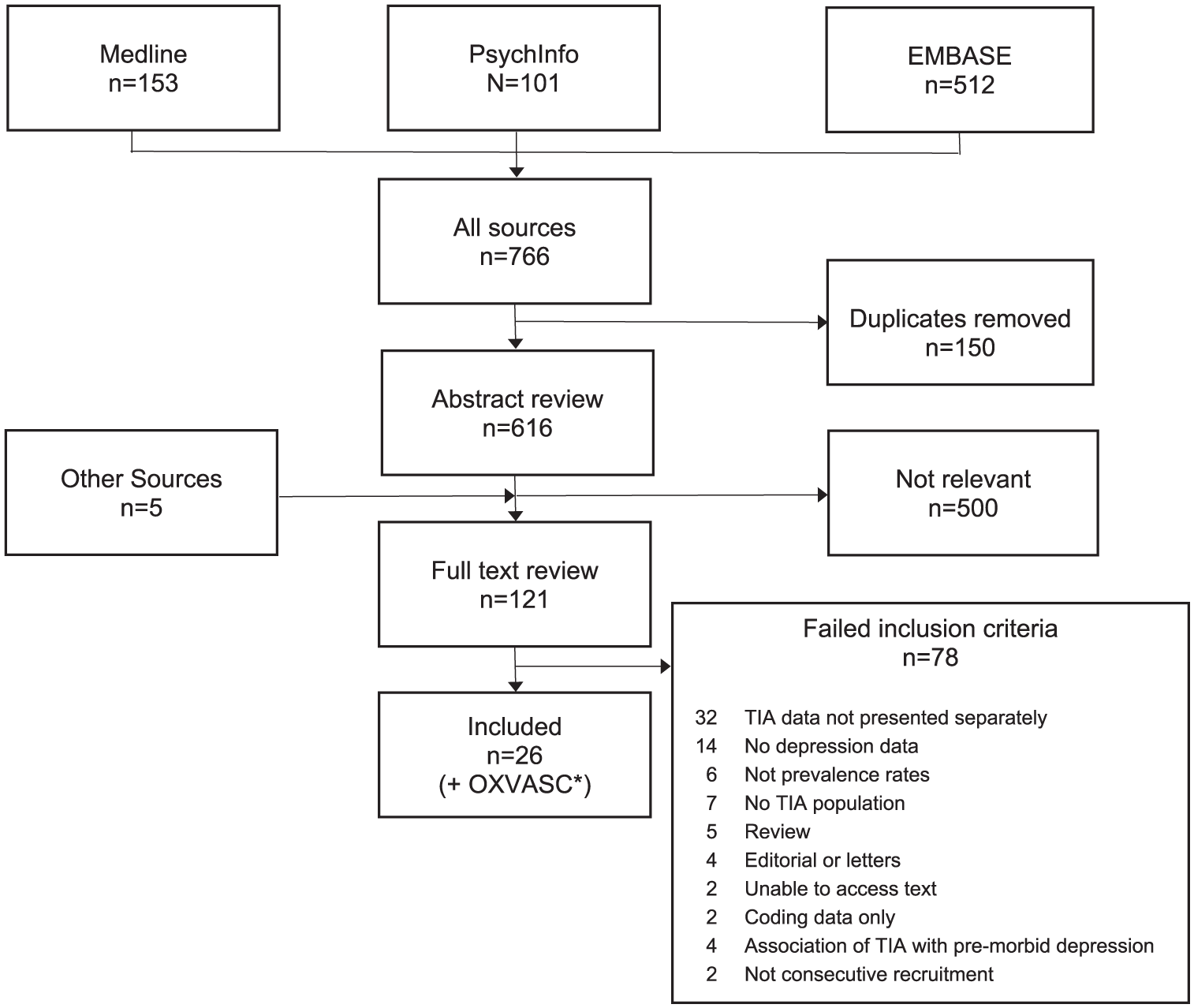
Literature search summary. *The OXVASC cohort has been analyzed separately and combined with the 26 other studies subsequently.
Assessments of depression were face-to-face in 18 studies, via postal questionnaires in three,26,30,34 telephone assessments in one, 18 a combination of face-to-face and telephone in two,39,41 a combination of postal and telephone in one, 28 a combination of email and telephone in one, 20 and not reported in one. 29 Eleven different screening tools were used with the Depression subscale of the Hospital Anxiety and Depression Scale (HADS-D) being the most common (10 of the 27 studies).16,20,24–27,30,32,34,35 Of the different screening tools used only 7 have been previously validated in stroke patients (Table S2).
The prevalence rates of depression were highly heterogeneous between studies at each time point reported (phet < 0.001 at <1, 1–6 and 12 months; Figure 2). For example, among the 7 studies that assessed TIA patients at the time of initial presentation, the reported prevalence of depression ranged from 4% to 30% with a pooled rate of 11.3% (3.9–18.7%). However, among five longitudinal studies (including OXVASC) that reported rates at more than one time-point, the pooled prevalence was 20.1% (16.9–23.4) at 0–1 months, decreasing (p = 0.004) to 14.1% (11.5–16.7) at 3–12 months, with little heterogeneity (phet = 0.35 and 0.40 respectively; Figure 3).
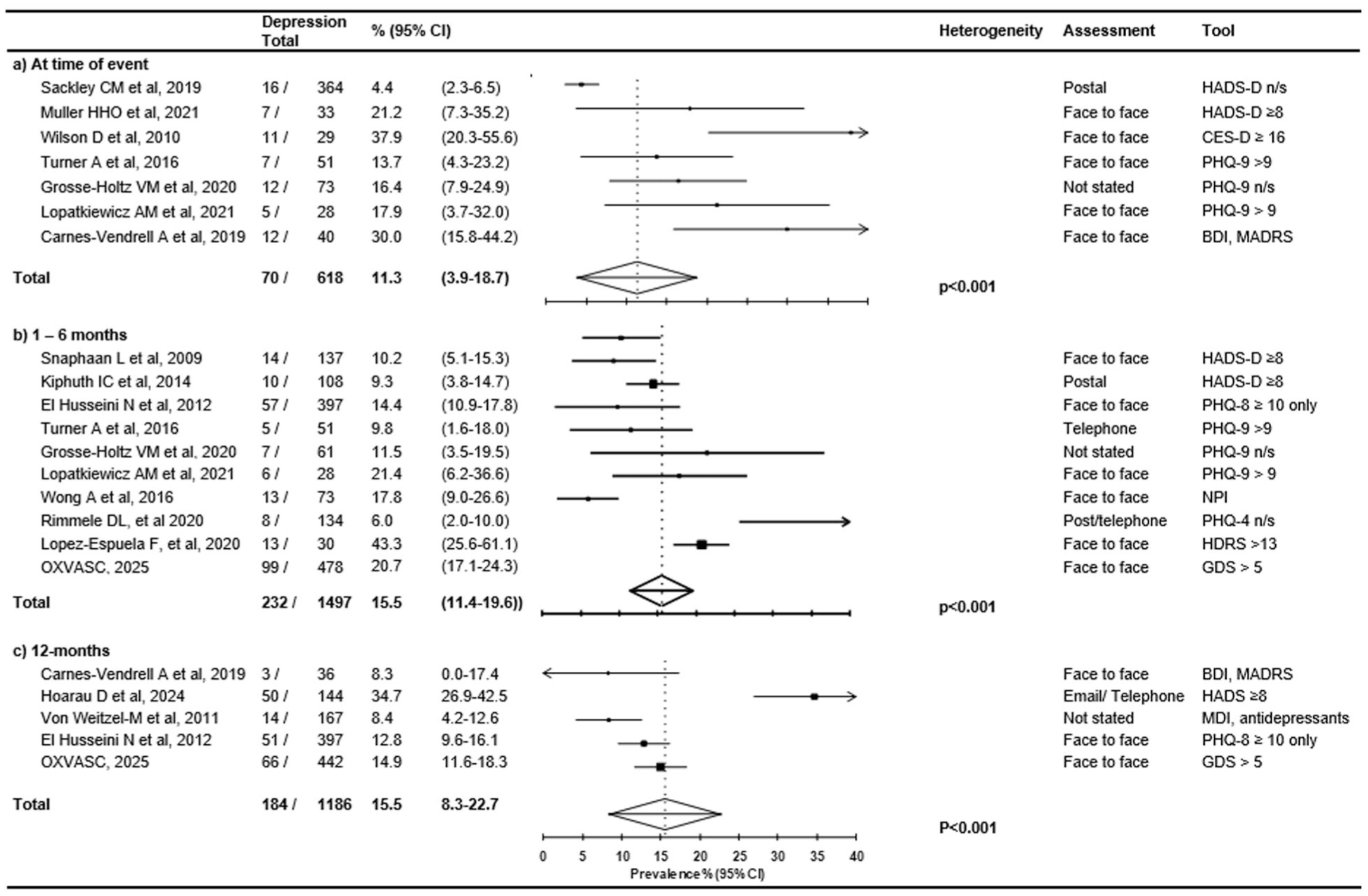
Pooled prevalence of depression after a TIA stratified by time in cross-sectional studies. Studies in which an assessment was at one of more specified time-points after a TIA were included and stratified as <1 month; 1–6 months; 12 months. The between-study heterogeneity in each subgroup is also reported. CI: confidence interval; n/s: not specified; BDI: Beck Depression Inventory; MADRS: Montgomery Asberg Depression Rating Scale; HADS-D: Hospital Anxiety and Depression Scale—Depression; GDS: Geriatric Depression Scale; PHQ-9: Patient Health Questionnaire version 9; CES-D: Center for Epidemiologic Studies Depression Scale; PHQ-8: Patient Health Questionnaire version 8; PHQ-4: Patient Health Questionnaire version 4; NPI: Neuropsychiatric Inventory; HDRS: Hamilton Depression Rating Scale; MDI: Major Depression Inventory.
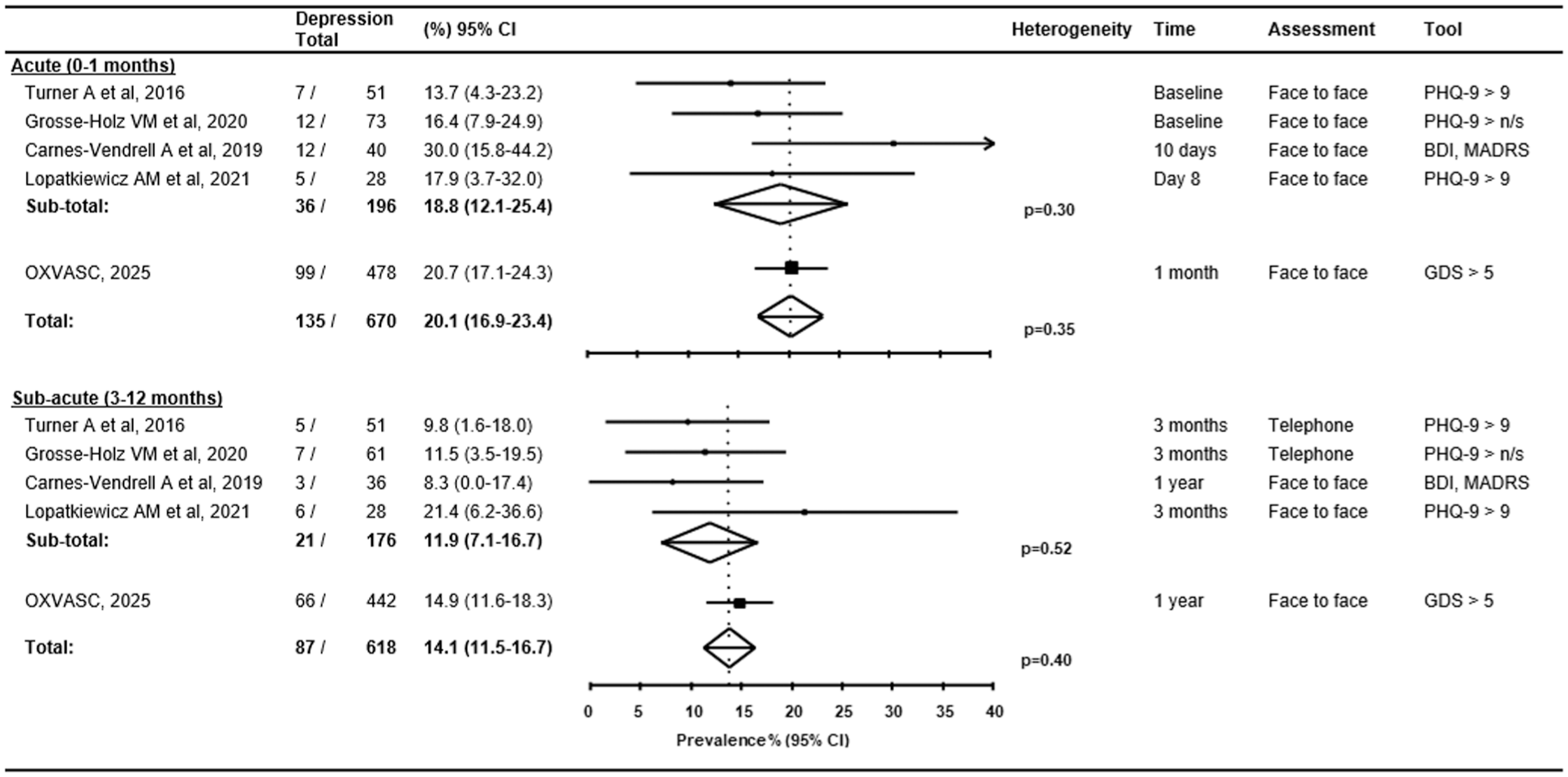
Pooled prevalence of depression after a TIA stratified by time in longitudinal cohort studies. Studies reporting prevalence rates at different time-points in the same cohort were included and stratified as acute (< 1 month) versus subacute (3–12 month). The between-study heterogeneity in each subgroup is also reported. CI: confidence interval; n/s: not specified; BDI: Beck Depression Inventory; MADRS: Montgomery-Asberg Depression Rating Scale; GDS: Geriatric Depression Scale; PHQ-9: Patient Health Questionnaire version 9. El Husseini reported prevalence rates in a longitudinal study but at 3 months and 12 months and therefore the data was not included.
Eight studies reported risk factors for depression after a TIA, with an additional 5 reporting risk factors only in a combined population of TIA and stroke. Among 8 studies, excluding OXVASC, (Table S1) that reported risk factors for depression after a TIA, younger age was associated in two of three studies,16,30,32 and female sex in one of two previous studies.32,33 Current smoking, chronic obstructive pulmonary disease, socio-economic deprivation, atrial fibrillation and a higher number of TIAs were each found to be associated in single studies.16,21,28 Only one study reported depression rates in relation to ischemic lesions on Diffusion Weighted MRI after TIA and found no association. 35 The analysis was based on only 28 patients with an acute ischemic lesion, but the finding is consistent with the result in OXVASC. In addition, in the OXVASC study in a multivariable analysis, younger age, reporting a low-mood at baseline and disability (modified Rankin score > 1) were all associated with depression at 1 and at 12 months after a TIA. A past history of depression and living alone were associated with depression at 1 month, and multi-morbidity and socio-economic deprivation were associated with depression at 12 months. Smoking status, atrial fibrillation and female sex were not found to be association with post-TIA depression in the OXVASC cohort (Table S3).
Apart from OXVASC, none of the studies reported data on any association between depression after TIA and risk of recurrent stroke, suicide, or all-cause mortality.
Of the potential sources of heterogeneity or bias (Figure S2), only 10 studies reported participation rate and only 11 studies reported rates of premorbid depression or whether patients with prior depression were excluded. Study methodology appeared to affect the reported prevalence of depression within 6 months of a TIA (Figure 4), including a lower rate in postal questionnaire studies than in face-to-face studies (5.6%, 3.1–8.1 vs 17.7%, 13.7–21.7; p < 0.001) and a lower rate in tissue-based definition of TIA compared with time-based (7.9%, 1.5–14.3% vs 17.7%, 12.8–22.5; p < 0.001). The screening tool used also appeared to affect the reported prevalence, though interpretation of this is limited by only study using the GDS. Exclusion of patients with pre-morbid depression or dementia did not appear to affect the prevalence in univariate modeling.
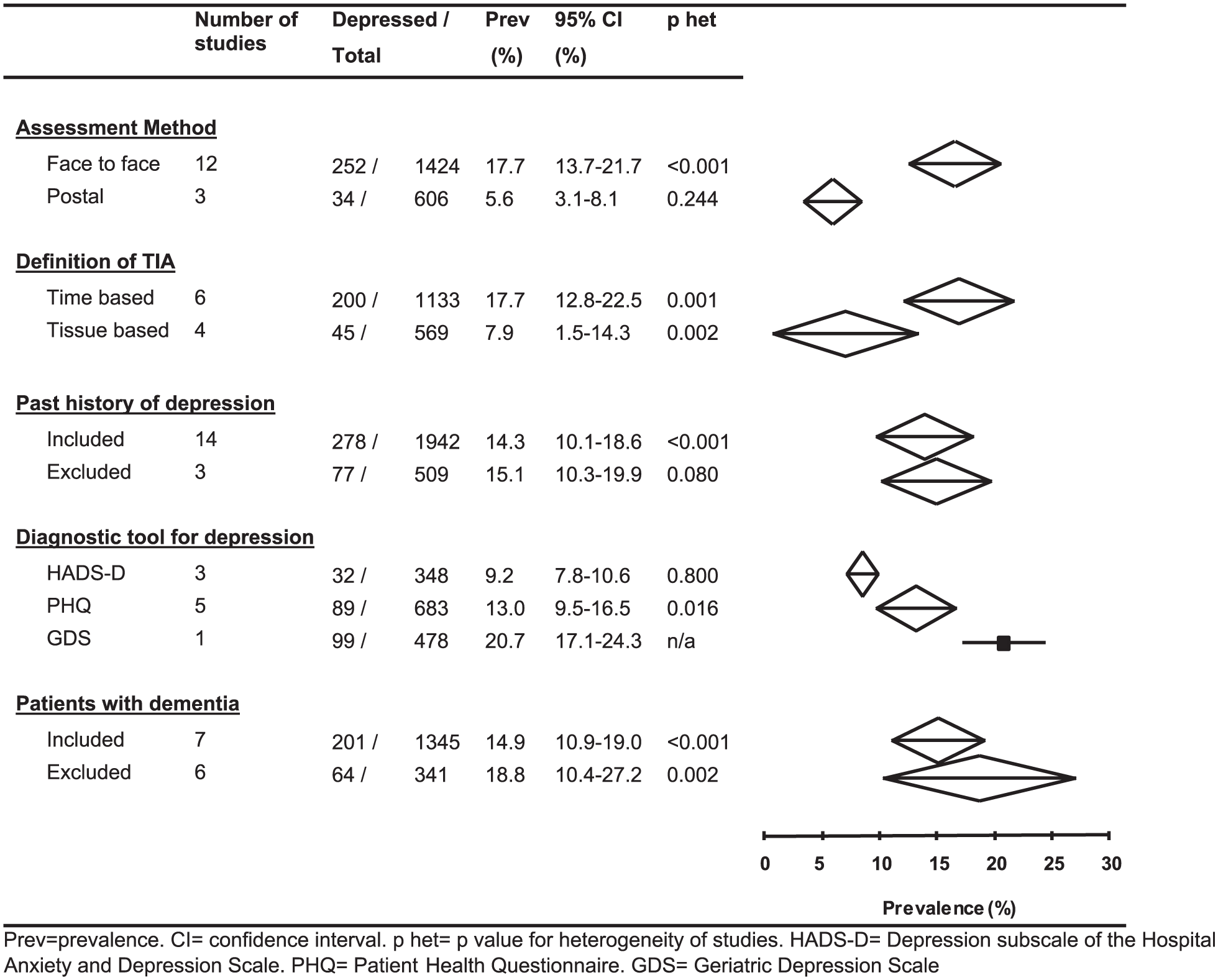
Impact of assessment method, definition of TIA and inclusion/exclusion criteria on the prevalence rate of post-TIA depression (within the first 6 months after the event). Where multiple prevalence rates were present within a study, the prevalence rate of depression was taken from the earliest assessment. Not all studies presented data on the different variables therefore different numbers included for each subset. Prev: prevalence; CI: confidence interval; phet: p value for heterogeneity of studies; HADS-D: Depression subscale of the Hospital Anxiety and Depression Scale; PHQ: Patient Health Questionnaire; GDS: Geriatric Depression Scale.
In an inverse-variance weighted meta-regression of the log risk against study type the screening tool used (HADS-D vs others), follow-up method (face vs telephone vs postal), TIA definition (time vs tissue) and exclusion criteria (dementia/high Rankin scores) together accounted for 84% of the observed between study heterogeneity. When this model was applied to the OXVASC population, the predicted prevalence—based on its specific study design—was 22.1%. This closely aligned with the OXVASC observed prevalence of 20.7% at 1-month follow-up, confirming the model’s predictive validity across different methodological frameworks.
Discussion
In the systematic review, we found that in the first month after a TIA the prevalence of depression is about 20%. The majority of studies only assessed for depression at a single time point but in the five longitudinal studies there was a statistically significant decrease in the rate of depression to around 14% by 3–12 months suggesting resolution in a proportion of patients.
Our findings have some clinical implications. That 20% of patients in the early stages after a TIA report symptoms of depression is significant as low mood is associated with non-compliance with medical treatment and non-acceptance of life-style adjustments, and at least, in the general population and in stroke patients, an increased mortality.5–9 In the systematic review, younger age and socio-economic deprivation were found to be associated with post-TIA depression but associations with other factors were inconsistent. Targeting patients with these specific risk factors may enable clinicians to use resources strategically to diagnose and support patients with post-TIA depression. However, given the suggestion of, at least a partial, resolution of depression symptoms after a TIA, it is less clear how clinicians should proceed in those identified with symptoms in the early, acute post-TIA period. Further studies to understand the risks and benefits of interventions for post-TIA depression, and to identify predictors of persistent depression, are required to guide clinicians in their practice.
The systematic review also found that there was significant heterogeneity in rates across all studies. This appeared to be largely explained by methodological differences, including varied participation rates and associated selection and attrition biases, different screening tools, and face-to-face versus postal assessments. There was much less heterogeneity in reported rates in the longitudinal studies, among which other aspects of study methodology were more similar. Low, or lack of reporting of, participation rate was found to be a potential source of bias in 17 of the studies. This is particularly an issue as patients with depression may be comparatively less likely to engage with a study or in the follow-up assessments. Similarly, patients with depression may be apathetic about completing and returning a postal assessment, compared with face-to-face assessment perhaps partly explaining the low rates of depression found in postal studies. Lack of reporting of pre-morbid depression was also a potential source of bias. Premorbid depression has been associated with an increased risk of depression after stroke,1,2 but was assessed for in less than half of the TIA studies. In the OXVASC study, pre-morbid depression was strongly associated with an increased risk of post-TIA depression. By excluding, or not identifying patients with pre-morbid depression, it is uncertain whether subsequent levels are related to the TIA or simply reflect pre-event status. However, our finding in the longitudinal studies that the risk is highest at one-month after a TIA and then declines significantly by one-year suggests some temporal association with the event. The method for diagnosis of depression also varied between studies with the most commonly used screening tool being HADS-D but with only one study assessing patients against a DSM criteria for a depressive disorder. 21
This systematic review has several limitations. First, a significant amount of heterogeneity was observed in the reported depression rates among cross-sectional studies, and although this appeared to be mainly explained by differences in methodology and participation rates we cannot be absolutely certain. Second, for the majority the studies employed screening tools for depression rather than more detailed neuropsychological assessments against DSM criteria. 42 Third, few studies looked specifically at susceptibility factors for depression in TIA patients therefore limiting understanding of the at risk population. Finally, although we found evidence of resolution of depression on early follow-up in the five longitudinal studies, data on use of antidepressant or other treatments was not routinely available.
In conclusion, the systematic review identified that depression affects about 1-in-5 patients early after a TIA, but prevalence falls with time, suggesting that it may be triggered by the event in some cases, but further work is required to identify risk factors and to determine the prognosis beyond one year. In addition, future studies must prioritize careful study design due to its significant influence on observed prevalence rates.
Supplemental Material
sj-docx-1-wso-10.1177_17474930261432616 – Supplemental material for Prevalence, time-course and predictors of depression after transient ischemic attack: A systematic review, meta-analysis, and validation
Supplemental material, sj-docx-1-wso-10.1177_17474930261432616 for Prevalence, time-course and predictors of depression after transient ischemic attack: A systematic review, meta-analysis, and validation by Aubretia J McColl, Sarah Pendlebury and Peter M Rothwell in International Journal of Stroke
Footnotes
Acknowledgements
The authors thank the staff in the general practices that collaborated in the Oxford Vascular Study.
Author contributions
AJM performed the search, study selection, data abstraction, quality assessment, statistical analysis, and wrote the first draft of the manuscript. SP revised the manuscript. PMR conceived and designed the study, provided supervision and funding, provided the independent study screen, interpreted the data, and revised the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PMR received compensation from Abbott Vascular for Consultant services. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Oxford Vascular Study is supported by grants to Professor P.M.R. from the National Institute for Health Research (NIHR) Oxford Biomedical Research Center (IS-BRC-1215-20008), Wolfson Foundation, the Wellcome Trust 104040/Z/14/Z and the Masonic Charitable Foundation.
Ethical approval
OXVASC was approved by the Oxfordshire Research Ethics Committee REC Ref: 05/Q1604/70.
Data availability statement
Requests for access to data should be submitted for consideration to the OXVASC Study Director (
Supplemental material
Supplemental material for this article is available online.
