Abstract
Background:
Embolic stroke of undetermined source (ESUS) accounts for up to one quarter of ischemic strokes, with occult atrial fibrillation (AF) as a key underlying cause. Implantable cardiac monitors (ICMs) markedly improve AF detection but are limited by cost and resource demands. Existing AF-prediction models show modest accuracy and lack longitudinal validation. We developed and validated the CATCH-AF score to provide a simple, clinically applicable tool for stratifying early and long-term AF risk after ESUS.
Methods:
We analyzed 543 consecutive ESUS patients systematically monitored with ICMs. Variable selection used LASSO-penalized Cox regression. Model performance was assessed with time-dependent ROC curves, restricted mean survival time (RMST) analysis, and 10-fold cross-validation. To evaluate geographic generalizability, internal–external cross-validation was performed across seven participating centers, estimating discrimination and calibration for each held-out cohort. Based on the final multivariable model, a point-based score was derived including age, coronary artery disease, heart failure, and prior transient ischemic attack or ischemic stroke.
Results:
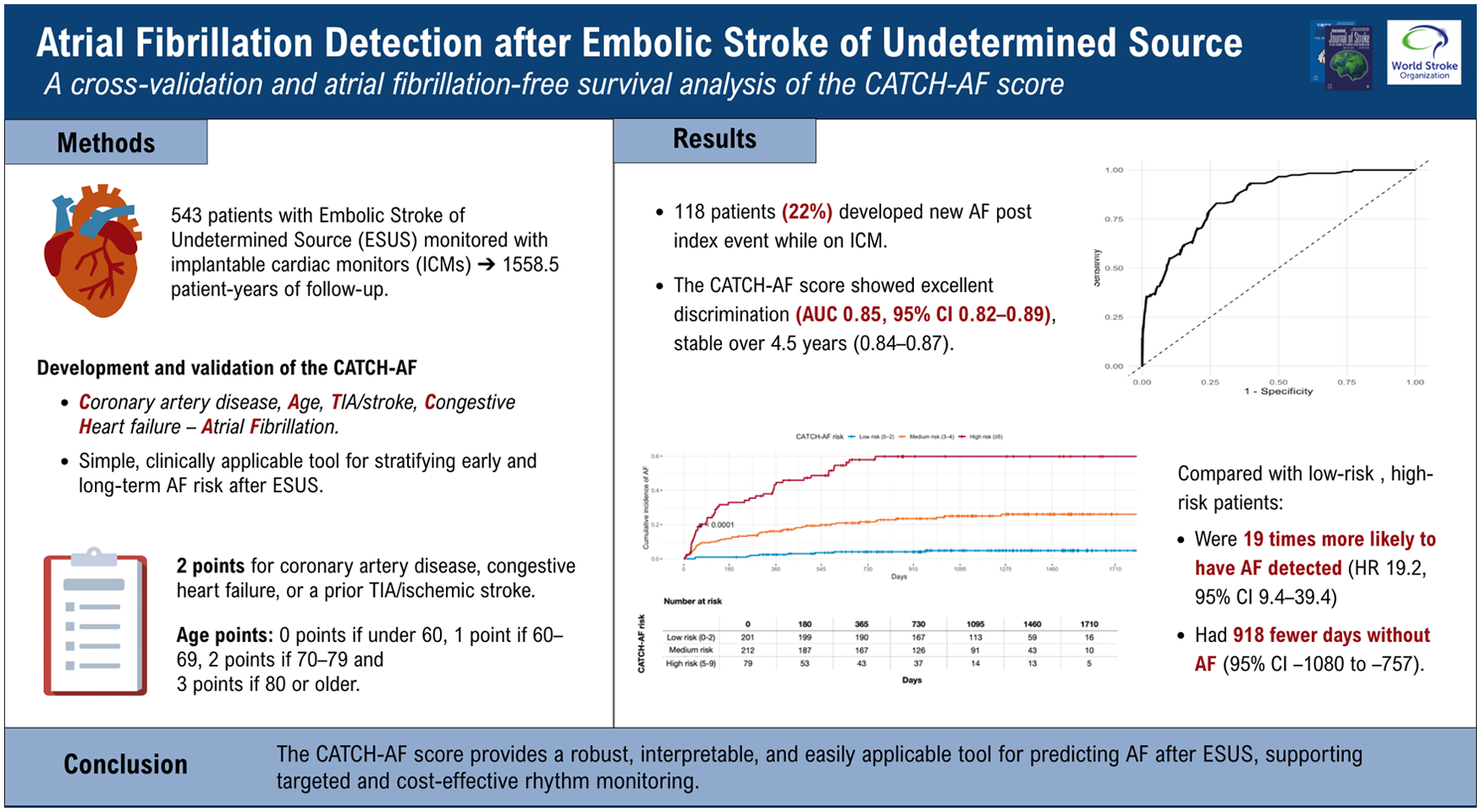
During 1558.5 patient-years of follow-up, 118 patients (22%) developed new AF. The CATCH-AF score showed excellent discrimination (AUC 0.85, 95% CI 0.82–0.89), stable over 4.5 years (0.84–0.87). Compared with low-risk patients (0–2 points), those at high risk (⩾5 points) had a 19-fold higher hazard of AF detection (HR 19.2, 95% CI 9.4–39.4; p < 0.001) and 918 fewer AF-free days (95% CI −1080 to −757).
Conclusions:
The CATCH-AF score provides a robust, interpretable, and easily applicable tool for predicting AF after ESUS, supporting targeted and cost-effective rhythm monitoring.
Keywords
Introduction
Embolic stroke of undetermined source (ESUS) accounts for up to one quarter of all ischemic strokes and represents a major clinical challenge due to its heterogeneous mechanisms and high risk of recurrence.1–3 Occult atrial fibrillation (AF) is a leading cause of ESUS, yet its diagnosis is often delayed or missed without prolonged cardiac monitoring. 2 Implantable cardiac monitors (ICMs) have markedly enhanced AF detection, but only a small proportion of ESUS patients undergo implantation, primarily due to limited technical and human resources and the considerable associated costs.4–7 This underscores the need for accurate risk stratification tools to identify those most likely to benefit from intensive monitoring.
Several clinical scores have been proposed to predict AF detection after ischemic stroke.8–15 Nevertheless, these models share important limitations. Most of them were derived from heterogeneous populations, often including patients with cryptogenic or mixed stroke subtypes rather than focusing exclusively on ESUS patients. Notably, none of the existing models has been systematically evaluated within a longitudinal framework as most available scores were assessed at single time points, without accounting for the evolving nature of AF risk after stroke. Consequently, since existing models have not been evaluated with time-dependent methods, their predictive performance over time remains unknown, and it is unclear whether they are better suited to identify early or delayed AF occurrence after ESUS. Moreover, the lack of time-to-event analyses means that these previous models do not provide insights into AF-free survival or cumulative incidence across risk categories, both of which are critical to inform clinical decisions about the duration and intensity of rhythm surveillance.
To address these limitations, we developed the CATCH-AF score in a large and homogeneous cohort of ESUS patients systematically monitored with ICMs, with the aim of deriving a simple, pragmatic, and robust clinical tool for predicting incident AF.
Methods
We conducted a retrospective, multicentre, observational cohort study of patients with ESUS who underwent ICM implantation (Reveal LINQ, Medtronic Inc, Minneapolis, MN, USA). Retrospective data were drawn from prospective, ongoing stroke registries at seven stroke centers (Table S1) included cases between 2021 and 2024 internationally. Anonymized data was pooled centrally. Where applicable, approval from local ethics committees was obtained. We adhered to the STROBE guidelines 16 to ensure accurate and transparent reporting of the study findings. The analysis plan was finalized and circulated before data collection and analysis. Individual patient data from this study cannot be shared due to data protection regulations; however, aggregated data can be provided upon request.
Definition and outcome
Consecutive patients captured in the prospective registries of the participating comprehensive stroke centers were screened for eligibility. Eligible patients were 18 years of age or older and had received a diagnosis of ESUS stroke or TIA, that was supported by consistency between symptoms and findings on brain magnetic resonance imaging or computed tomography. ESUS was defined according to the criteria proposed by the Cryptogenic Stroke/ESUS International Working Group, as follows: (1) stroke detected by magnetic resonance or computed tomography imaging that is not lacunar, (2) absence of extracranial stenosis causing ⩾50% luminal stenosis in arteries supplying the area of ischemia detected with a magnetic resonance angiography or computed tomography angiogram, (3) no major-risk cardioembolic source, and (4) no other specific cause of stroke identified (e.g. arteritis, dissection, vasospasm, drug misuse). 1 The vessel assessment was performed by any available imaging including ultrasound, CT-angiography, MR-angiography, and digital subtraction angiography. Patients on anti-arrhythmic drugs were not excluded from the analysis. For this analysis, we excluded patients with life expectancy less than 6 months, prosthetic mechanical valve, pacemaker, hepatic disease associated with coagulopathy (prothrombin time prolonged beyond the normal range) and clinically relevant bleeding risk including cirrhotic patients with Child Pugh B and C and estimated glomerular filtration rate (eGFR) < 15 mL/min/1.73 m2 (Study flow chart, S Figure 1). Twelve-lead electrocardiography, transthoracic or transoesophageal echocardiography, and cardiac monitoring for at least 24 hours were performed before determining the indication for ICM implantation. After written informed consent, the patients underwent ICM implantation under local anesthesia. The primary outcome was the occurrence of newly diagnosed AF during follow-up. AF was defined as an episode of irregular heart rhythm, without detectable P waves, lasting more than 30 seconds. Outcome assessment was carried out during scheduled outpatient visits and/or through contact with the patient, their relatives, or the patient’s primary physician. At each evaluation, all available clinical documentation was reviewed, including hospital discharge summaries, correspondence from primary care physicians, and reports of investigations performed in the outpatient setting. The timing of ICM implantation was determined at the discretion of the treating physician.
Definition of variables
All variables were prospectively collected at baseline according to standardized definitions (see supplemental material).
Statistical analysis
Baseline characteristics were summarized as medians (interquartile ranges) or counts (percentages) and compared using the Wilcoxon rank-sum and χ² or Fisher’s exact tests, as appropriate. The primary outcome was new-onset AF detected during ICM follow-up.
Time-to-event analyses were performed using Cox proportional hazards regression. To identify independent predictors, a penalized Cox regression with least absolute shrinkage and selection operator (LASSO) was applied, followed by a standard multivariable Cox model including all predictors with non-zero coefficients. Regression coefficients from the final model were scaled and converted into a point-based score (CATCH-AF). Model performance was assessed in terms of discrimination, calibration, and prognostic stratification. Receiver operating characteristic (ROC) curves and time-dependent AUCs were calculated to evaluate predictive accuracy over time, while AF-free survival was analyzed using Kaplan–Meier estimates and Cox regression across risk categories. Restricted mean survival time (RMST) analyses provided absolute differences in AF-free days between groups. Internal validity was assessed through 10-fold cross-validation, and model generalizability was tested using an internal–external cross-validation approach across the seven participating centers. All analyses were performed with R (R Foundation for Statistical Computing, Vienna, Austria). A two-sided p-value < 0.05 was considered statistically significant. Further methodological details, including LASSO penalization procedures, time-dependent ROC estimation, bootstrap comparisons, calibration plots, and internal–external cross-validation workflow, are provided in the Supplementary Methods.
Results
A total of 543 consecutive patients with ESUS undergoing ICM were included (62% male) with a median age of 63 years (interquartile range (IQR): 55–74) (Figure S1). All patients were discharged on antiplatelet treatment. All patients were free from AF events during the interval between the index event and ICM implantation. The baseline characteristics of the patients are summarized in Table 1. The overall follow-up period was 1558.5 patient-years, corresponding to a median follow-up of 1104 days (IQR: 695–1454.5). During follow-up, 118 (22%) developed new AF (ICMAF group) while 425 (78%) remained free from AF (ICMNO AF group). Patients who developed AF were significantly older compared with those without detection (p < 0.001). Sex distribution was similar across groups (63% vs 59% males, p = 0.500). All patients with AF detected during ICM monitoring were subsequently initiated on oral anticoagulation according to current secondary prevention guidelines.
Baseline characteristics of the study population overall and according to AF detection during ICM follow-up.
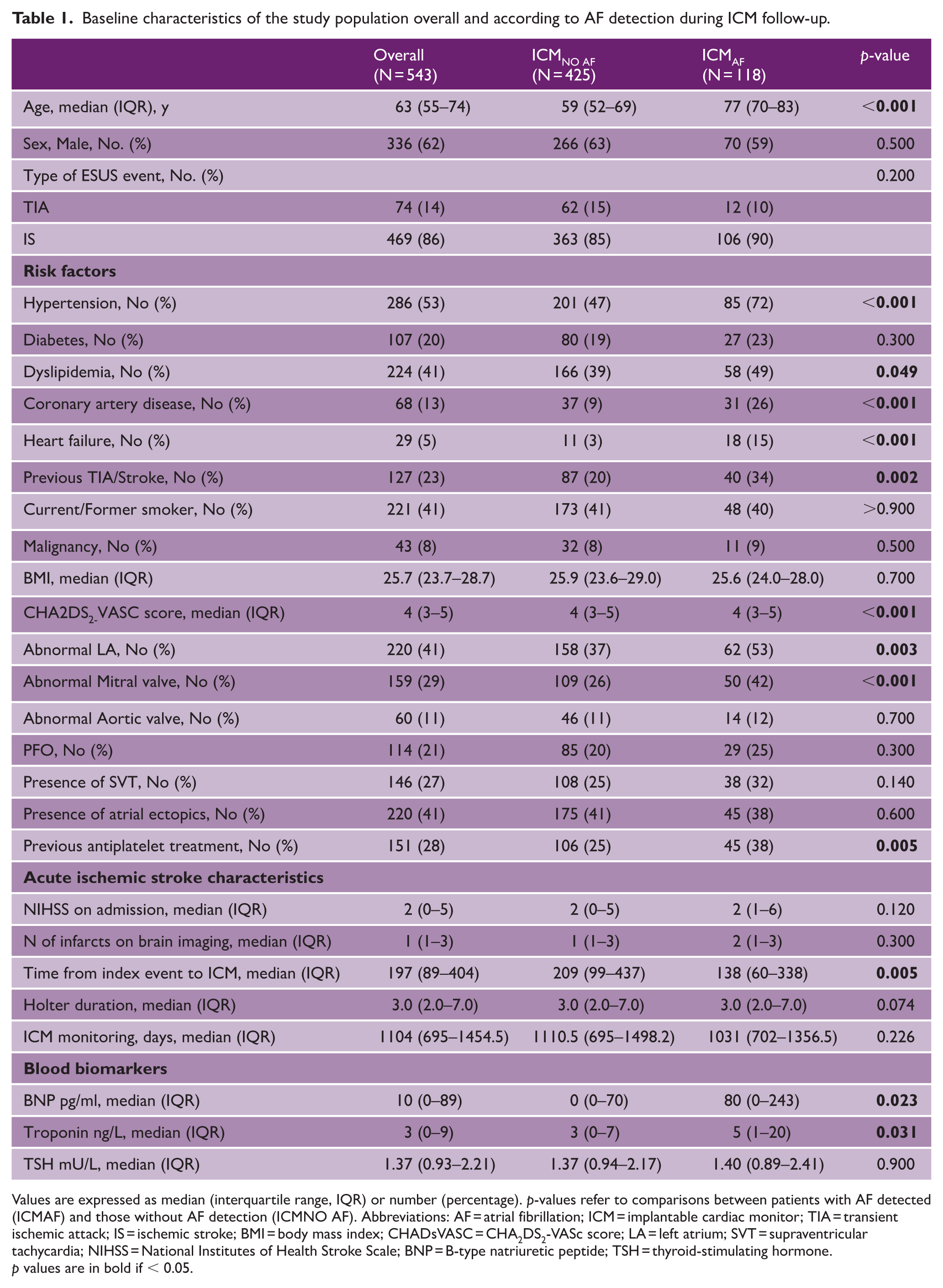
Values are expressed as median (interquartile range, IQR) or number (percentage). p-values refer to comparisons between patients with AF detected (ICMAF) and those without AF detection (ICMNO AF). Abbreviations: AF = atrial fibrillation; ICM = implantable cardiac monitor; TIA = transient ischemic attack; IS = ischemic stroke; BMI = body mass index; CHADsVASC = CHA2DS2-VASc score; LA = left atrium; SVT = supraventricular tachycardia; NIHSS = National Institutes of Health Stroke Scale; BNP = B-type natriuretic peptide; TSH = thyroid-stimulating hormone.
p values are in bold if < 0.05.
Lasso-based variable selection and multivariable modeling
Variable selection was performed using Cox proportional hazards regression penalized with the LASSO. The optimal penalty parameter was identified through 10-fold cross-validation (λ.min = 0.0814; Figure S2), corresponding to the model with the lowest partial likelihood deviance. At this level of penalization, several variables retained non-zero coefficients (Table S2). The strongest positive coefficients were observed for coronary artery disease (β = 0.3558), congestive heart failure (β = 0.3262), abnormal left atrium (β = 0.2798), and age (β = 0.0814 per year). Previous transient ischemic attack or ischemic stroke also contributed positively (β = 0.1603), together with body mass index (β = 0.0475). In contrast, abnormal mitral valve disease (β = −0.1053), atrial ectopics (β = −0.0813), and troponin (β = −0.000057) showed small negative coefficients. Hypertension, diabetes, and sex, as well as BNP and CHA2DS2-VASc score, were fully shrunk to zero and thus excluded from the penalized model. The coefficient trajectories (Figure S3) illustrate the stability of these predictors across varying levels of penalization. Age, previous TIA/stroke, coronary artery disease, and heart failure consistently retained non-zero coefficients up to the optimal λ, confirming their robustness as key predictors. At the optimal penalization parameter (λ.min), the LASSO Cox regression retained nine predictors with non-zero coefficients (Table S3). The Cox proportional hazards model including predictors selected by LASSO (at λ.min) showed that age was a continuous predictor, with each additional year conferring a 10% higher risk (HR 1.10, 95% CI 1.08–1.13; p < 0.001). Previous TIA/stroke was associated with nearly a twofold increase in hazard (HR 1.76, 95% CI 1.09–3.45; p = 0.050), while coronary artery disease (HR 2.03, 95% CI 1.22–3.38; p = 0.007) and congestive heart failure (HR 1.84, 95% CI 1.11–3.50; p = 0.044) were also significant predictors. Other variables retained by the penalized model, such as abnormal left atrium (p = 0.801), abnormal mitral valve (p = 0.284), abnormal aortic valve (p = 0.379), atrial ectopics (p = 0.279), and troponin (p = 0.501), did not achieve statistical significance. The overall model demonstrated excellent discriminatory ability, with a concordance index of 0.813 (SE 0.018).
Derivation of the CATCH-AF score
From the predictors selected by the LASSO procedure, a parsimonious multivariable Cox regression model was derived (Table 2). Four variables remained independently associated with incident AF detection during follow-up after ESUS. Increasing age was a strong continuous predictor, with each additional year conferring a 10% higher risk of AF (HR 1.10, 95% CI 1.08–1.13; p < 0.001). A history of previous TIA or ischemic stroke was also associated with a significantly increased hazard (HR 1.76, 95% CI 1.09–3.45; p = 0.050). Coronary artery disease (HR 2.03, 95% CI 1.22–3.38; p = 0.007) and congestive heart failure (HR 1.84, 95% CI 1.11–3.50; p = 0.044) were both strong independent predictors of AF detection. Based on the magnitude of regression coefficients from the final Cox model, a simplified point-based system was constructed (Table 3). Coronary artery disease, congestive heart failure and prior TIA or ischemic stroke were assigned 2 points. Age was categorized to optimize clinical applicability, with point values increasing across strata: 0 points for < 60 years, 1 point for 60–69 years, 2 points for 70–79 years, and 3 points for ⩾80 years. The resulting score, termed CATCH-AF (Coronary artery disease, Age, TIA/Stroke, Congestive Heart failure—Atrial Fibrillation), ranges from 0 to 9 points.
Multivariable Cox model derived from LASSO selection.

HR = hazard ratio; CI = confidence interval; TIA = transient ischemic attack.
Point assignment for the CATCH-AF score.
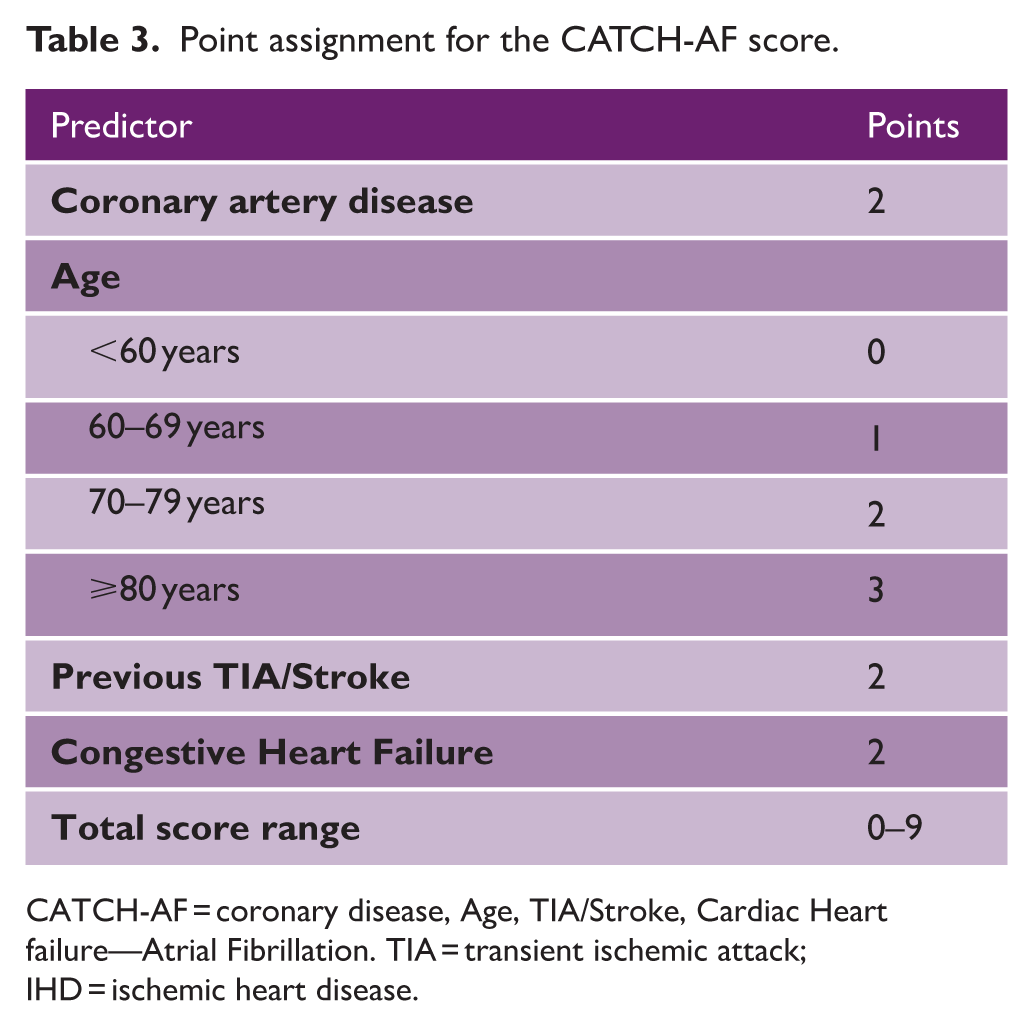
CATCH-AF = coronary disease, Age, TIA/Stroke, Cardiac Heart failure—Atrial Fibrillation. TIA = transient ischemic attack; IHD = ischemic heart disease.
Distribution of the CATCH-AF score and risk categories
The distribution of CATCH-AF scores in the study population is reported in Table S4. Among 543 patients, the majority clustered between 2 and 5 points, with only a minority at the extremes of the scale (0 points, n = 31 [5.7%]; 9 points, n = 8 [1.5%]). According to predefined thresholds, 226 patients (41.6%) were classified as low risk (0–2 points), 224 (41.3%) as intermediate risk (3–4 points), and 93 (17.1%) as high risk (⩾5 points). Patients with AF detection during follow-up were disproportionately represented in the intermediate- and high-risk categories, underscoring the discriminative capacity of the score.
Longitudinal predictive performance and risk stratification of the CATCH-AF score
The standard ROC analysis of the CATCH-AF model yielded an AUC of 0.853 (95% CI 0.817–0.888) (Figure 1). Time-dependent ROC analyses further demonstrated robust and stable performance over longitudinal follow-up (Figure 2, Table S5), with AUC values of 0.849 (95% CI 0.803–0.892) at 6 months and 0.844 (95% CI 0.803–0.883) at 12 months, improving slightly at 24 months (0.867, 95% CI 0.831–0.902) and 36 months (0.872, 95% CI 0.833–0.907), and remaining consistently high thereafter, including at 1710 days (0.858, 95% CI 0.773–0.935). Supplemental Table S6 reports pairwise comparisons of time-dependent AUC values across follow-up intervals. No statistically significant differences were observed between any two time-points after correction for multiple testing (all BH-adjusted p > 0.80). The estimated changes in AUC (ΔAUC) were minimal, with 95% confidence intervals consistently spanning zero (e.g. ΔAUC between 6 and 36 months = 0.029, 95% CI −0.005 to 0.062; BH-adjusted p = 0.845). Kaplan–Meier estimates of AF–free survival demonstrated marked stratification across CATCH-AF risk categories (Figure 3). Patients classified as high risk (⩾5 points) showed a rapid and steep increase in cumulative incidence within the first months after implantation, with more than 40% experiencing AF detection within the first year, and approaching 60% at long-term follow-up. In contrast, medium-risk patients (3–4 points) exhibited a more gradual increase, with an incidence plateauing at approximately 20% after 3 years, while low-risk patients (0–2 points) maintained a very low cumulative incidence, remaining below 10% even at 1710 days. The log-rank test confirmed highly significant differences across the three categories (p < 0.0001). Risk stratification analyses confirmed a steep and clinically meaningful gradient across categories (Figure S4, Table S7). Compared with low-risk patients, those in the high-risk group had nearly a 20-fold higher hazard of AF detection (HR 19.2, 95% CI 9.4–39.4; p < 0.001). Medium-risk patients also showed significantly elevated hazards compared with the low-risk group (HR 6.1, 95% CI 3.0–12.4; p < 0.001), while high-risk patients had more than a threefold higher risk than those in the intermediate category (HR 3.1, 95% CI 2.1–4.7; p < 0.001).
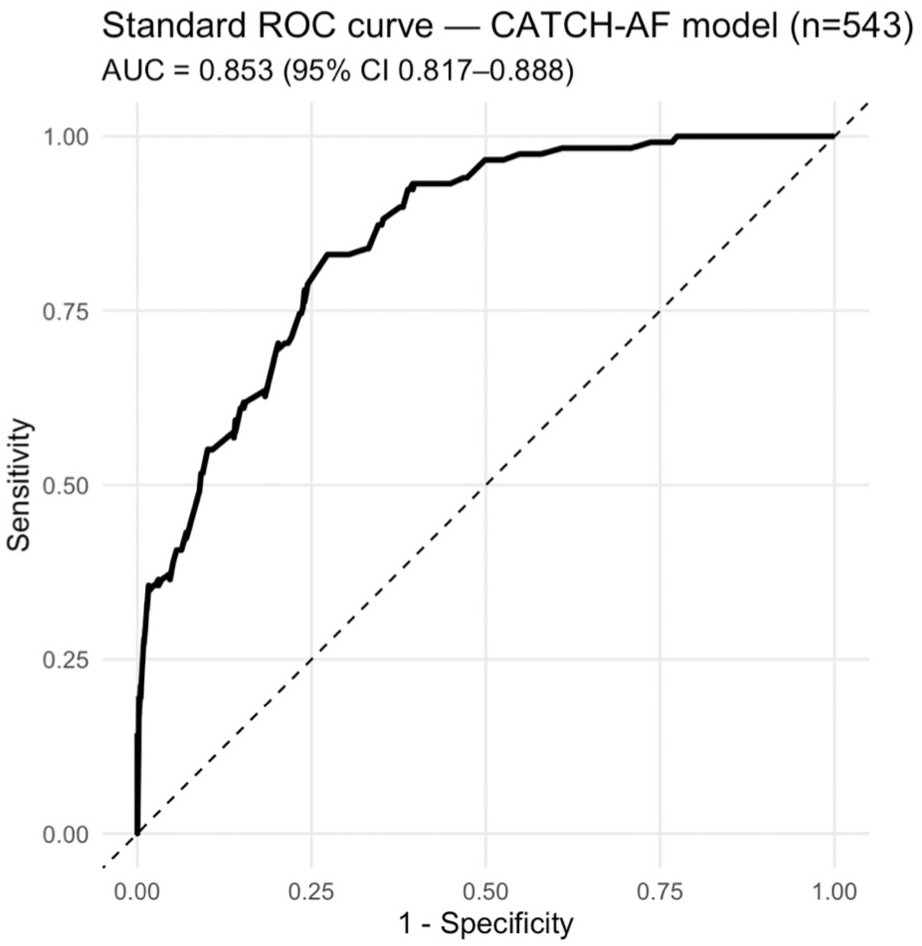
Standard ROC curve for the CATCH-AF model. Receiver operating characteristic (ROC) curve showing the discrimination performance of the CATCH-AF model in predicting atrial fibrillation (AF) detection after ESUS. The model achieved an area under the curve (AUC) of 0.853 (95% CI 0.817–0.888), indicating excellent discriminatory ability between patients who did and did not develop AF during follow-up.
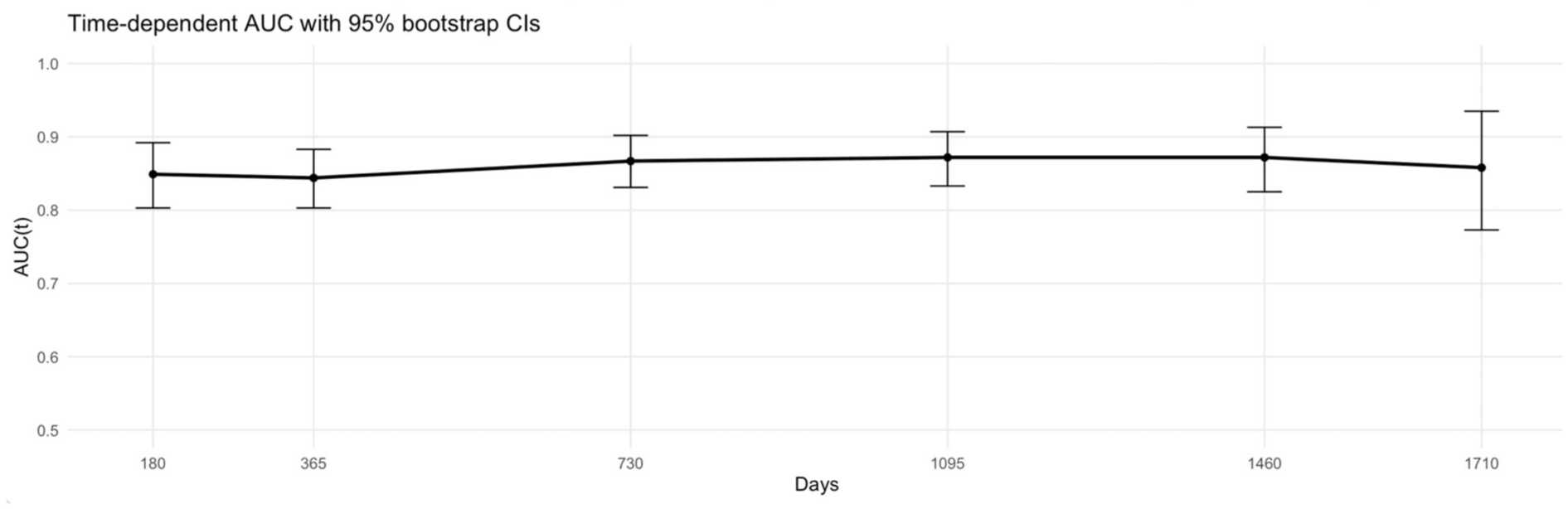
Time-dependent AUC of the CATCH-AF model. Time-dependent AUC curve with 95% bootstrap confidence intervals showing the longitudinal discriminative performance of the CATCH-AF score across follow-up time points (up to 1710 days). The predictive ability remained stable over time, with AUC(t) values consistently between 0.84 and 0.87, indicating that the model preserved accuracy for both early and delayed AF detection.
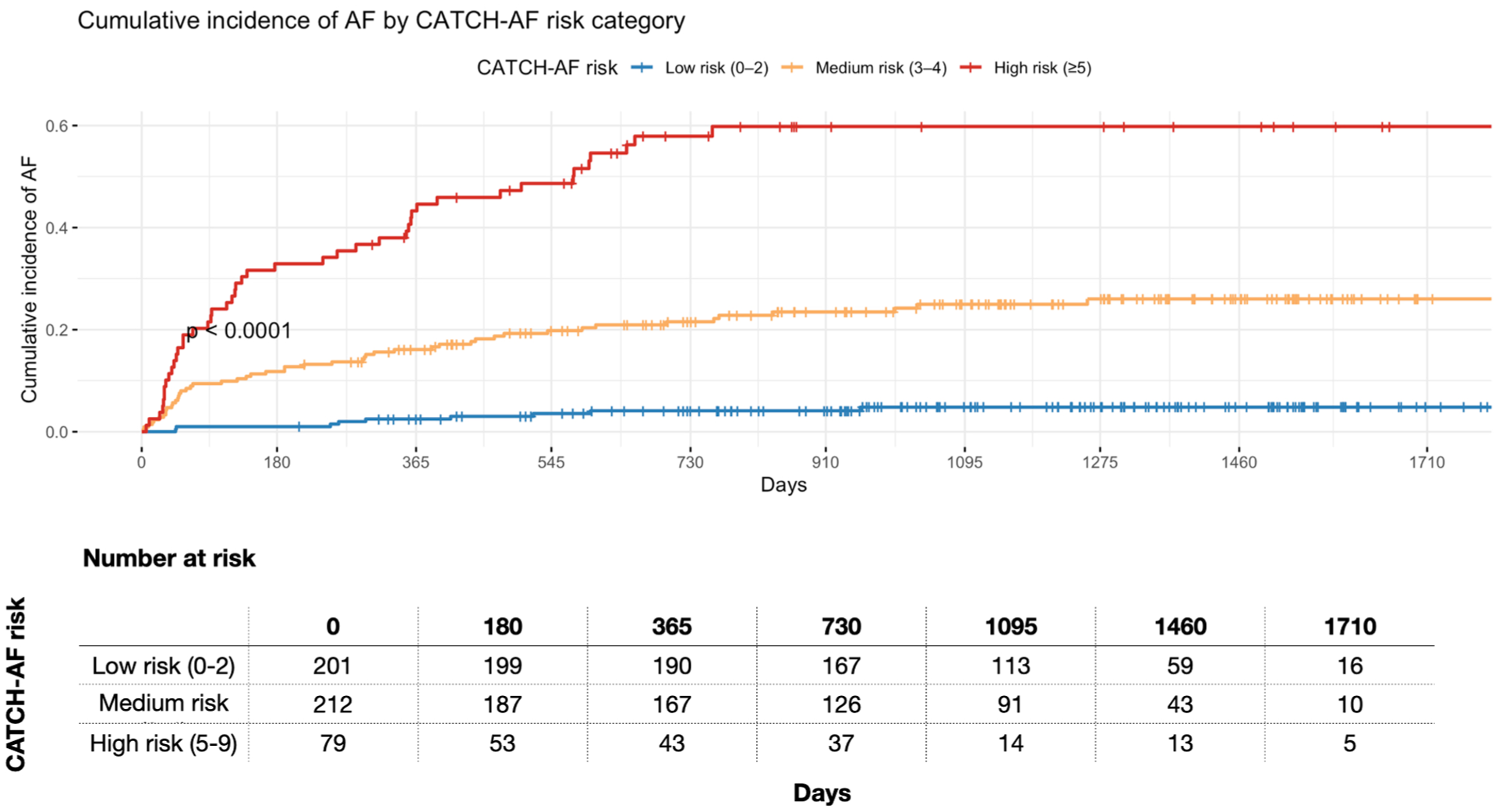
Cumulative incidence of AF according to CATCH-AF risk categories. Cumulative incidence curves for AF detection stratified by CATCH-AF risk categories: low (0–2 points), medium (3–4 points), and high (⩾5 points). High-risk patients exhibited the greatest cumulative incidence of AF (p < 0.0001, log-rank test). The number of patients at risk at each time point is displayed below the graph.
Restricted mean AF-free survival across CATCH-AF risk categories
RMST analyses provided an absolute measure of AF-free survival across CATCH-AF risk categories (Table S8, Figure S5). Patients classified as high risk (>4 points) experienced a profound reduction in AF-free survival compared with both low- and medium-risk categories. Specifically, high-risk patients survived on average 918.5 days fewer without AF than low-risk patients (95% CI −1080.2 to −756.8; p < 0.001) and 613.6 days fewer than medium-risk patients (95% CI −793.0 to −434.2; p < 0.001). Medium-risk patients also showed a significant reduction compared with the low-risk group, with an average loss of 305.0 AF-free days (95% CI −401.4 to −208.6; p < 0.001).
Internal and internal–external validation of the CATCH-AF score
Internal validation through 10-fold cross-validation and internal–external cross-validation across the seven participating centers confirmed the robustness of the CATCH-AF model, showing consistent discrimination and calibration. Detailed validation results are provided in the Supplementary Appendix (Figures S6 to S11, Tables S9 and S10).
Discussion
This study introduces the CATCH-AF score, a pragmatic tool to identify ESUS patients at increased risk of AF during systematic ICM follow-up. In our cohort of 543 patients, the score demonstrated excellent discrimination, with performance remaining stable over more than 4 years of observation. Patients classified as high risk (⩾5 points, 17.1% of the cohort) exhibited a nearly 20-fold higher hazard of AF detection compared with the low-risk group, while medium-risk patients also showed significantly elevated hazards. Beyond relative risk estimates, restricted mean survival time analysis revealed that high-risk patients lost an average of 918 AF-free days compared with low-risk patients, underscoring the clinical relevance of the stratification. Collectively, these results support the CATCH-AF score as a robust and clinically relevant instrument for stratifying long-term AF risk after ESUS and for informing the selection of patients most likely to benefit from intensive rhythm monitoring.
A key innovative element of our study lies in the longitudinal evaluation of predictive performance of the CATCH-AF score using time-dependent ROC analysis. Unlike previously published scores such as HAVOC, STAF, Brown ESUS-AF, AF-ESUS, and ACTEL, which were assessed primarily through static area under the receiver operating characteristic curve (AUROC) values at single endpoints, we systematically examined the discriminative capacity of the CATCH-AF score across the entire follow-up period. This approach acknowledges the inherently dynamic nature of AF risk after ESUS, where arrhythmia may be detected either early, within the first months, or several years after the index event. Our results demonstrate that the predictive ability of CATCH-AF was not only excellent at baseline (AUC 0.85) but also remained remarkably stable over time, with time-dependent AUC values consistently between 0.84 and 0.87 up to 1710 days. Our results suggest that the score maintains predictive accuracy for both early and delayed AF detection. This stability over time strengthens its potential clinical utility, allowing it to inform monitoring strategies beyond the short-term and across the extended course of follow-up.
Several AF prediction scores have been proposed in patients with cryptogenic stroke or ESUS, including HAVOC, 11 STAF, 14 ACTEL, 12 Brown ESUS-AF, 15 AF-ESUS, 8 and Decryptoring. 13 Across these studies, reported discriminative performance has generally ranged from moderate to good, with area under the receiver operating characteristic curve values typically between 0.70 and 0.85. However, direct comparison across scores is limited by substantial heterogeneity in study populations, rhythm monitoring strategies, follow-up duration, and outcome definitions. Importantly, many previously published scores were derived from heterogeneous cryptogenic stroke populations and were evaluated using short-term or intermittent rhythm monitoring, which may underestimate delayed AF detection. In contrast, the CATCH-AF score was developed and evaluated in a strictly defined ESUS cohort undergoing systematic ICM follow-up, with longitudinal assessment of predictive performance over more than 4 years. In contrast, the CATCH-AF score was developed and evaluated in a strictly defined ESUS cohort undergoing systematic ICM follow-up, with longitudinal assessment of predictive performance over more than 4 years. In this setting of intensive and prolonged rhythm monitoring, the score demonstrated excellent and stable discrimination over time, supporting its robustness and potential clinical applicability.
A further distinctive feature of this study is the application of restricted mean survival time analysis to quantify AF-free survival across CATCH-AF risk categories. The key clinical advantage of this approach lies in its ability to provide an absolute and interpretable measure of disease burden associated with increasing risk levels. In our cohort, patients classified as high risk (⩾5 points) experienced on average 918 fewer days free from AF compared with low-risk individuals, and 614 fewer days compared with those in the intermediate-risk category. Even patients in the medium-risk group demonstrated a meaningful reduction of approximately 305 AF-free days compared with the low-risk group. By reframing risk in terms of absolute AF-free survival time, this analysis offers direct clinical relevance—it quantifies how much earlier AF is likely to be detected in higher-risk patients, facilitates more effective communication with patients, and supports evidence-based prioritization of ICM use in those most likely to benefit. In contrast, previous prediction models have typically relied on relative measures such as hazard ratios or odds ratios, which, although statistically valid, are often less intuitive in clinical decision-making. 17 The use of RMST in our analysis therefore provides a novel and practical dimension to risk quantification, translating complex statistical differences into clinically meaningful time-based outcomes.
The clinical relevance of the CATCH-AF score extends beyond overall risk stratification to the temporal dynamics of AF detection. Using restricted mean survival time analysis, high-risk patients demonstrated a marked and early reduction in AF-free survival compared with low- and intermediate-risk groups, corresponding to a loss of more than 2 years of AF-free time. This finding indicates that high-risk patients are exposed not only to a higher probability of AF but also to substantially earlier AF occurrence. This temporal dimension is particularly relevant in real-world practice, especially within publicly funded healthcare systems where ICMs are effective but costly and not universally available. In this context, the CATCH-AF score may support prioritization of prolonged rhythm monitoring by identifying patients in whom early access to intensive monitoring is most likely to yield timely AF detection and earlier initiation of anticoagulation. Importantly, the score is not intended to preclude further investigation in patients classified as low risk, but rather to inform shared decision-making and support rational sequencing of monitoring strategies while preserving individualized clinical judgment.
This study has several notable strengths. First, it introduces the CATCH-AF score, a simple, pragmatic, and clinically intuitive tool derived from a large and homogeneous cohort of ESUS patients systematically monitored with ICMs. The score demonstrated excellent discriminative ability, with performance that remained stable over long-term follow-up exceeding 4 years. The inclusion of time-dependent ROC analysis represents a key methodological innovation, providing a dynamic evaluation of predictive accuracy across time.
Second, our study applies restricted mean survival time analysis to AF prediction after ESUS, translating relative risks into absolute, time-based outcomes. Third, the CATCH-AF score relies exclusively on readily available clinical variables—age, coronary artery disease, heart failure, and prior ischemic events—making it easily applicable across a wide range of healthcare settings. Several limitations should also be acknowledged. To strengthen the model’s external generalizability, future validation in independent ESUS populations from different healthcare systems is warranted. Nevertheless, the use of internal–external cross-validation across seven participating centers provided a rigorous assessment of the model’s transportability, demonstrating consistent discrimination and calibration across geographically distinct sites. Although model robustness and geographical transportability were assessed using internal cross-validation and an internal–external cross-validation framework across participating centers, the CATCH-AF score has not been validated in a fully independent external cohort. Internal–external cross-validation, whereby data from one center are sequentially held out for testing while the model is trained on the remaining centers, represents an intermediate validation strategy in multicentre datasets, but does not replace true external validation. External validation in independent ESUS populations, ideally across different healthcare systems and diagnostic pathways, is therefore required before widespread clinical implementation. Second, despite comprehensive adjustment for clinical covariates and uniform, continuous rhythm monitoring, residual confounding related to unmeasured factors cannot be entirely excluded. Third, while the time-dependent AUC and RMST analyses offer robust temporal insight into model performance, longer follow-up might further refine the score’s long-term predictive accuracy, especially beyond 5 years. Finally, the CATCH-AF score was developed and validated exclusively among ESUS patients undergoing systematic ICM. This homogeneous design ensured accurate AF detection and minimized ascertainment bias, but may limit the applicability of the model to broader stroke populations or to those monitored with less intensive or shorter-duration modalities. Future external validation studies in non-ICM settings are needed to confirm the generalizability of the score across diverse clinical contexts.
In conclusion, the CATCH-AF score represents a pragmatic and clinically accessible tool for predicting AF after ESUS. Derived from a large and homogeneous cohort systematically monitored with ICMs, it demonstrated excellent discrimination and sustained predictive accuracy throughout extended follow-up. The longitudinal evaluation confirmed stable time-dependent performance, supporting its capacity to predict both early and delayed AF detection. Furthermore, by incorporating restricted mean survival time analysis, the study provides an absolute, time-based quantification of AF-free survival, enhancing the interpretability and clinical relevance of risk stratification. Because it relies solely on simple and readily available clinical variables, the CATCH-AF score can be easily implemented in routine practice, facilitating timely identification of high-risk patients and optimizing resource allocation for prolonged monitoring. Future external validation across independent ESUS populations and different monitoring strategies will be essential to confirm its generalizability and integration into post-stroke care pathways.
Supplemental Material
sj-docx-1-wso-10.1177_17474930261428118 – Supplemental material for Atrial fibrillation detection after embolic stroke of undetermined source: Development and validation of the CATCH-AF score
Supplemental material, sj-docx-1-wso-10.1177_17474930261428118 for Atrial fibrillation detection after embolic stroke of undetermined source: Development and validation of the CATCH-AF score by Lucio D’Anna, Francesco Favruzzo, Claudio Baracchini, Alessandra Pes, Fionn Mag Uidhir, Diletta Rosin, Mariarosaria Valente, Gian Luigi Gigli, Liqun Zhang, Nathan Leung, Manav Sohal, Simona Sacco, Raffaele Ornello, Federico De Santis, Ubaldo Coppola, Gabriele Prandin, Michele Romoli, Valentina Tudisco, Federica Nicoletta Sepe, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Irtiza Syed, Soma Banerjee, Selina Edwards, Ceylan Safak, Roberto Avila, Joan Cruz, Ashley Laurie, Gaurav Desai, Maryam Haneef, Anne Idian, Arvind Chandratheva, Phang Boon Lim, Giovanni Merlino, Matteo Foschi and Robert Simister in International Journal of Stroke
Supplemental Material
sj-docx-2-wso-10.1177_17474930261428118 – Supplemental material for Atrial fibrillation detection after embolic stroke of undetermined source: Development and validation of the CATCH-AF score
Supplemental material, sj-docx-2-wso-10.1177_17474930261428118 for Atrial fibrillation detection after embolic stroke of undetermined source: Development and validation of the CATCH-AF score by Lucio D’Anna, Francesco Favruzzo, Claudio Baracchini, Alessandra Pes, Fionn Mag Uidhir, Diletta Rosin, Mariarosaria Valente, Gian Luigi Gigli, Liqun Zhang, Nathan Leung, Manav Sohal, Simona Sacco, Raffaele Ornello, Federico De Santis, Ubaldo Coppola, Gabriele Prandin, Michele Romoli, Valentina Tudisco, Federica Nicoletta Sepe, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Irtiza Syed, Soma Banerjee, Selina Edwards, Ceylan Safak, Roberto Avila, Joan Cruz, Ashley Laurie, Gaurav Desai, Maryam Haneef, Anne Idian, Arvind Chandratheva, Phang Boon Lim, Giovanni Merlino, Matteo Foschi and Robert Simister in International Journal of Stroke
Supplemental Material
sj-docx-3-wso-10.1177_17474930261428118 – Supplemental material for Atrial fibrillation detection after embolic stroke of undetermined source: Development and validation of the CATCH-AF score
Supplemental material, sj-docx-3-wso-10.1177_17474930261428118 for Atrial fibrillation detection after embolic stroke of undetermined source: Development and validation of the CATCH-AF score by Lucio D’Anna, Francesco Favruzzo, Claudio Baracchini, Alessandra Pes, Fionn Mag Uidhir, Diletta Rosin, Mariarosaria Valente, Gian Luigi Gigli, Liqun Zhang, Nathan Leung, Manav Sohal, Simona Sacco, Raffaele Ornello, Federico De Santis, Ubaldo Coppola, Gabriele Prandin, Michele Romoli, Valentina Tudisco, Federica Nicoletta Sepe, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Irtiza Syed, Soma Banerjee, Selina Edwards, Ceylan Safak, Roberto Avila, Joan Cruz, Ashley Laurie, Gaurav Desai, Maryam Haneef, Anne Idian, Arvind Chandratheva, Phang Boon Lim, Giovanni Merlino, Matteo Foschi and Robert Simister in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SB is a key opinion leader for RAPIDAI. RS is partially funded by the UCLH Biomedical Research Center. All other authors have no conflicts of interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Data availability
Data available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
