Abstract
Background:
Stroke is increasingly recognized as an important cause of morbidity and mortality in people living with HIV (PLWH). Although advances in antiretroviral therapy (ART) have transformed HIV into a chronic condition, cerebrovascular complications remain common and often underrecognized.
Aims:
This review summarized current understanding of the pathogenesis, clinical features, investigations, and management of stroke in PLWH, highlighting knowledge gaps and priorities for future research.
Summary of evidence:
HIV-associated stroke is multifactorial. Mechanisms include HIV-associated vasculopathy, opportunistic infections, immune reconstitution inflammatory syndrome (IRIS), and traditional vascular risk factors accelerated by HIV and ART. Clinical presentations may resemble those in HIV-negative individuals but occur at a younger age, with distinct subtype distributions and a higher frequency of concomitant infection. Investigation requires a tiered approach: standard stroke imaging and cardiac work-up, supplemented by cerebrospinal fluid analysis and advanced vessel wall imaging in patients with suspected inflammatory or infectious etiology. Reperfusion therapies appear feasible in stable HIV disease, but evidence remains limited. Outcomes are shaped by immune status, access to care, and comorbidities, with higher recurrence and cognitive decline reported in several cohorts. Real-world challenges include stigma, clinician awareness gaps, and inequities in diagnostics and treatment, particularly in low-resource settings.
Conclusions:
Stroke in PLWH is an emerging global health challenge. Clinicians should maintain a high index of suspicion in younger patients and those with advanced disease or recent ART initiation. Multidisciplinary care pathways and equitable access to diagnostics and secondary prevention are essential. Future research must address unresolved questions around vasculopathy, IRIS, small vessel disease, and long-term cognitive outcomes to guide evidence-based management.
Keywords
Introduction: why does this matter?
As the global population of people living with HIV (PLWH) ages, currently estimated at 40 million people, clinicians are increasingly facing the evolving challenge of a heightened risk of cerebrovascular disease in this population. Once considered a disease of acute opportunistic infections and immunodeficiency, HIV has become, in the era of effective antiretroviral therapy (ART), a chronic condition. This success has shifted the disease burden toward noncommunicable complications, including cerebrovascular disease (here used to encompass ischemic and hemorrhagic stroke, as well as small vessel disease, unless otherwise specified).
Stroke has emerged as a particularly important contributor to morbidity in PLWH. Meta-analysis of global epidemiological data suggests that HIV confers a 40% higher risk of stroke, with the population attributable fraction and disability-adjusted life years of stroke in PLWH nearly tripling between 1990 and 2019. 1 This rise is largely explained by improved survival and population aging. Although the rate of increase has slowed in the modern ART era, the burden remains substantial, particularly in sub-Saharan Africa. The prevalence of stroke in PLWH is estimated at 1.3%, rising to almost 4% in those aged ⩾ 50 years, and an incidence of 17.9 per 10,000 person-years. 2 Taken together, these meta-analyses confirm that stroke is a growing global problem for PLWH, raising urgent questions as to why cerebrovascular events continue to rise despite expanding ART coverage and improving viral suppression.
The consequence is a patient population that is often younger than typical stroke cohorts, presenting with cerebrovascular events that may be under-recognized or misattributed. For frontline clinicians, particularly in sub-Saharan Africa where the HIV burden is highest, awareness of this evolving challenge is crucial. Timely recognition is not only essential for acute management but also provides opportunities for earlier intervention, targeted prevention, and improved long-term outcomes.
This review aimed to provide a clinically grounded synthesis of current knowledge on HIV and cerebrovascular disease. By highlighting the burden, mechanisms, clinical features, diagnostic challenges, management strategies, and global health considerations, we seek to equip clinicians with the understanding needed to recognize, treat, and ultimately prevent stroke in PLWH.
Pathophysiology: what’s the link?
The pathogenesis of stroke in PLWH is multifactorial, reflecting a spectrum of vascular injury that varies across the stages of HIV disease. While not all individuals experience advanced immunosuppression, evidence linking low CD4 counts and high viral load to cerebrovascular events supports the concept that greater immune dysfunction and increased viral burden predispose to vascular injury and worse outcomes.3,4 The mechanisms span a continuum from infection-driven inflammation to premature vascular aging (Figure 1), reflecting overlapping contributions from viral, immune, coinfectious, metabolic, and traditional vascular factors.
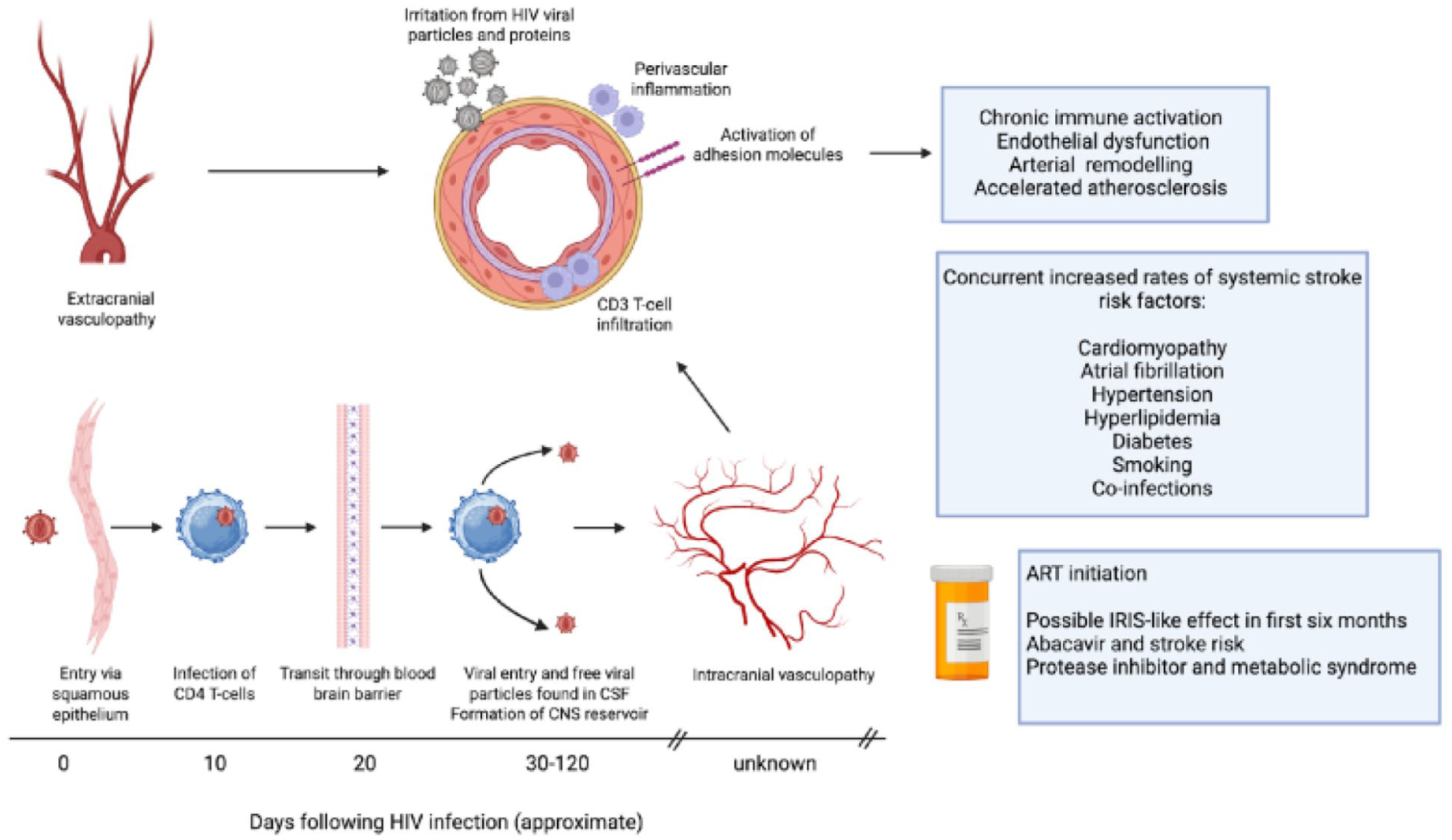
Potential pathophysiological mechanisms for stroke in PLWH.
At one end of the spectrum lies vasculitis, historically termed HIV vasculitis. This involves lymphocytic infiltration of medium- and large-sized arteries, but there is little evidence of direct viral invasion from pathological studies. 5 Endothelial cells lack CD4 and CXCR5 receptors, and HIV RNA or proteins are rarely found within vessels, 6 suggesting this is largely immune-mediated rather than angioinvasive.
During periods of high viraemia, cytokine-driven endothelial activation upregulates adhesion molecules (ICAM, VCAM), promoting endothelial dysfunction. 7 Markers of macrophage activation (e.g. soluble CD163) remain elevated despite ART initiation and correlate with carotid plaque burden and intima–media thickness. 8 Beyond active replication, HIV-derived proteins such as Tat and circulating inflammatory cytokines may also injure the endothelium and sustain chronic inflammation, contributing to both extracranial and intracranial vessel remodeling. 9
In some patients, vascular inflammation may reflect immune reconstitution inflammatory syndrome (IRIS), typically within six months of ART initiation or re-initiation, but occasionally up to two years later.10,11 IRIS refers to a rebound effect whereby the recovering immune system mounts an exaggerated, pathogenic inflammatory response to an antigen. Classically, it is an antigen of a coinfective pathogen, and may present as unmasking IRIS (latent coinfection revealed) or paradoxical IRIS (worsening of known infection, Figure 2). However, the target antigen may also be an HIV protein, or even non-pathogenic self-antigens. 10 The arterial vessel wall can be a target of this process, 12 and early ART-associated strokes are more common in individuals with a low CD4 nadir and concurrent infections. 11
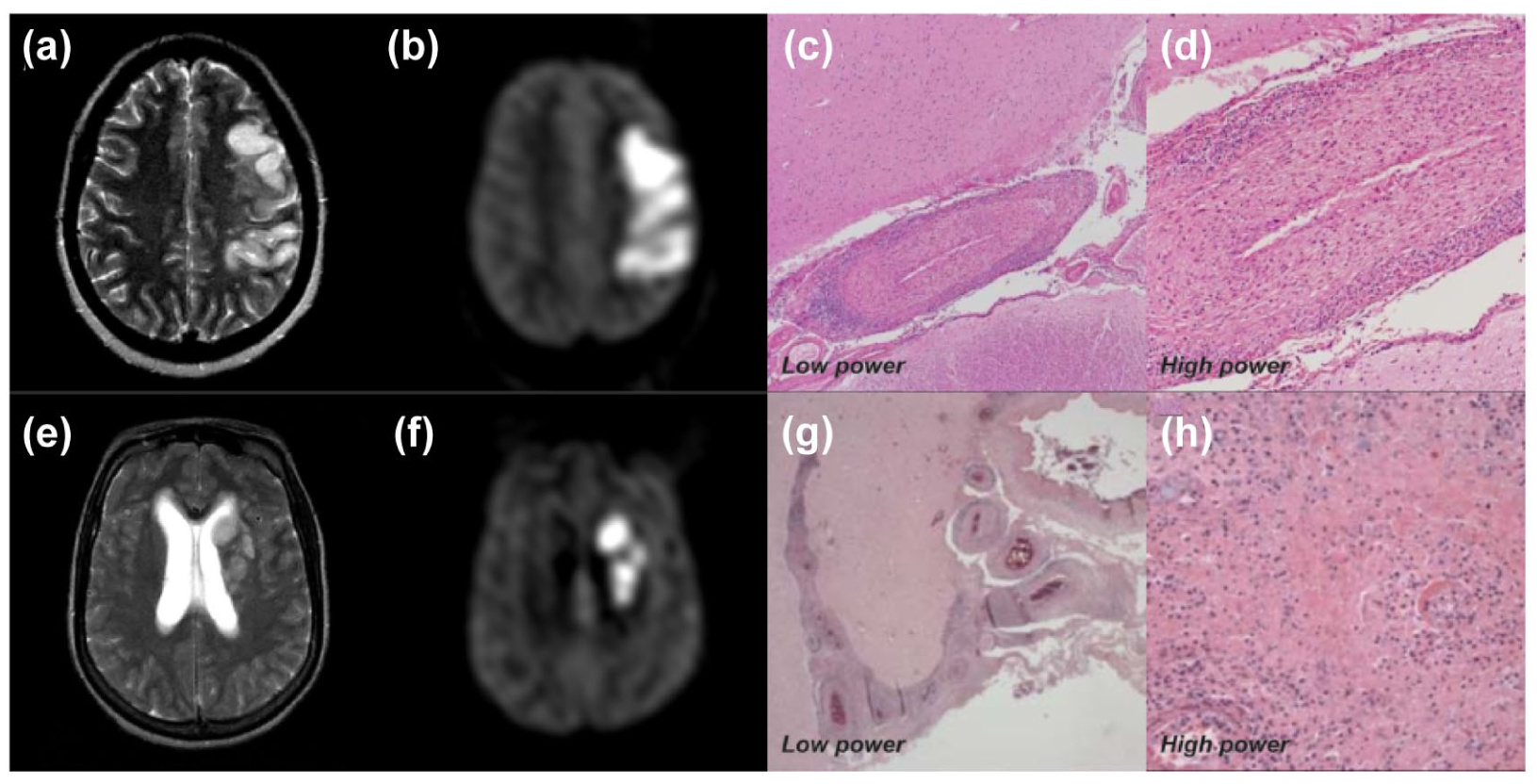
Illustration of paradoxical and unmasking intracranial IRIS-vasculopathy. © The Authors Benjamin et al 2017. Published by Oxford University Press for the Infectious Diseases Society of America. 13 Also note that this is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted reuse, distribution, and reproduction in any medium, provided the original work is properly cited
Opportunistic or coinfective vasculitides remain major causes of stroke in advanced HIV. Mycobacterium tuberculosis, Varicella Zoster virus (VZV), Treponema pallidum, Cytomegalovirus, Cryptococcus neoformans, and Hepatitis C virus have all been associated with vasculopathy, hypercoagulability, or atherosclerosis (Figure 3). VZV reactivation is notable and may occur without rash or CSF PCR positivity, highlighting the importance of intrathecal VZV IgG testing. 13 Coinfection with hepatitis C is linked to increased risk of both ischemic and hemorrhagic stroke, 4 and HIV-COVID-19 coinfection may compound cerebrovascular risk through endothelial injury and hypercoagulability, though data are limited. 14
An illustration of syphilis co-infection and Stroke in PLWH: A 45-year-old patient with a 7-year history of HIV who was compliant with antiretrovirals and a CD4 count of 560. The patient presented with unsteadiness and bilateral weakness. MRI showed diffusion restriction within the internal capsules (a), representing bilateral acute infarction within the territories of the anterior choroidal arteries. A time-of-flight angiogram (b) did not show any stenoses. Vessel wall imaging showed concentric enhancement of the terminal internal carotid arteries, corresponding to the origin of the anterior choroidal arteries (c, arrows). With a cerebrospinal fluid Treponema pallidum particle agglutination assay of > 1:1280 and rapid plasma regain of 1:4, a diagnosis of a neurosyphilis-related vasculopathy was made. The vessel wall enhancement improved following treatment with benzylpenicillin (d).
In the modern ART era, the predominant vascular phenotype appears to be accelerated atherosclerosis with vessel wall remodeling rather than classic inflammatory vasculitis (Figure 4).13,15,16 Imaging studies reveal noncalcified, outwardly remodeled plaques and premature large-artery disease, consistent with accelerated vascular aging.15,17,18 This remodeling-heavy pattern parallels the progressive vascular injury seen after radiotherapy and likely represents early-stage atherosclerosis rather than a distinct process. Nevertheless, nonatherosclerotic vasculopathy, characterized by adventitial inflammation, media thinning resulting in inward remodeling, and dolichoectasia, remains recognized, particularly in intracranial arteries.16,19 Indeed, around two-thirds of strokes in PLWH involve the intracranial circulation, 20 potentially reflecting HIV’s neurotropism. HIV enters the CNS within days of infection and establishes a persistent viral reservoir within CNS macrophages and microglia which may sustain local immune activation and chronic endothelial injury. 21
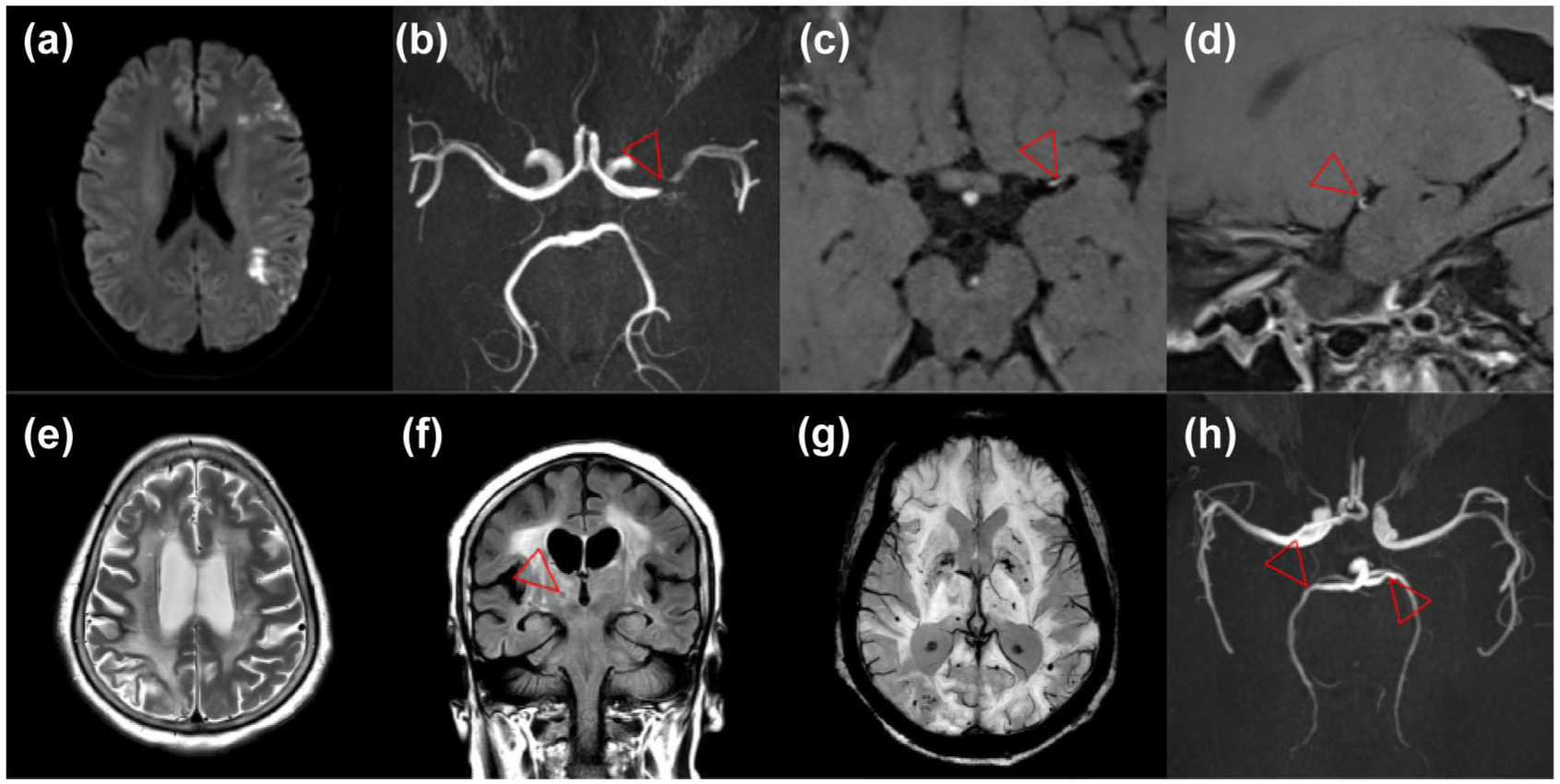
An illustration of stroke manifestations in stable HIV disease: A 45-year-old patient with HIV controlled on emtricitabine/tenofovir/bictegravir (CD4 count of 940, undetectable viral load) and no cardiovascular risk factors presented after developing right-sided weakness. MRI showed acute infarction in the left MCA borderzone territories (a). Time-of-flight angiography showed a severe left M1 MCA stenoses (b, arrow). With no evidence of infection on CSF analysis, the eccentric vessel wall enhancement (c and d, arrows) was typical for atherosclerosis. A 55-year-old patient with HIV controlled on emtricitabine/tenofovir, raltegravir, and darunavir (CD4 count 430, undetectable viral load) with multiple cardiovascular risk factors (type 2 diabetes mellitus, hypertension, and hypercholesterolemia) presented with cognitive impairment (mini-mental state examination of 13/30). MRI showed confluent white matter T2-weighted hyperintensity (e), multiple lacunar infarcts affecting the thalami (f, arrow), many deep and lobar microhemorrhages on susceptibility-weighted imaging (g), and multiple intracranial artery stenoses consistent with atherosclerosis (h, arrows). The extent of the cerebral hyperintensity that extended to the cortex and involvement of the middle cerebellar peduncles (not shown) suggested a combination of severe small vessel disease and HIV encephalopathy.
Depending on the regimen and cumulative exposure, ART may contribute to this spectrum of HIV-associated vasculopathy. Long-term exposure to protease inhibitors is associated with dyslipidemia, insulin resistance, and increased atherosclerotic risk. 22 Beyond arthrogenesis, epidemiological data suggest that abacavir is associated with increased stroke risk; potential mechanisms relate to platelet activation, endothelial dysfunction, or hypersensitivity-driven inflammation. 23 By contrast, integrase inhibitor-based regimens appear metabolically safer 24 and their increasing adoption may enable the contribution of ART to be disentangled from the role of the virus itself in the development of HIV-associated vasculopathy. A major limitation in understanding these mechanisms lies in the difficulty of directly characterizing vascular pathology and viral localization in the brain, particularly as biopsies and postmortem studies are rare, and spatial viral detection methods remain technically constrained. Consequently, most mechanistic models rely on peripheral measures (serum biomarkers, or extracranial imaging such as carotid intima-media thickness) and assume that these reflect intracranial pathology, which is unlikely given HIV’s neurotropism and the unique characteristics of the blood–brain barrier neurovascular unit.
Antiphospholipid antibodies and reductions in proteins C and S have been reported in PLWH with stroke, but these findings are best regarded as bystander effects of immune activation not causal mechanisms. While they may contribute in select cases, current evidence does not support them as dominant drivers of stroke in PLWH. 6
Traditional vascular risk factors, including hypertension, diabetes, dyslipidemia, and smoking, are more prevalent and occur earlier in PLWH. 25 Cardioembolic mechanisms may also contribute. In a Californian cohort, the incidence of atrial fibrillation was over twice that of the general population. 26 In sub-Saharan African cohorts, attribution is complicated by higher background rates of cardiomyopathy, rheumatic valve disease, and endocarditis. 27 Social determinants such as stress, substance use, and healthcare access further amplify risk. 28
Overall, the interplay between viral persistence, immune dysregulation, coinfections, treatment effects, and traditional risks highlights that stroke in HIV is not a single entity but a dynamic and evolving spectrum of vascular injury, shaped by disease stage, immune status, and ART exposure.
Clinical features and etiology: what does stroke look like in PLWH?
The clinical presentation of stroke in PLWH is broadly similar to that in the general population, typically manifesting as a sudden onset of focal neurological deficit lasting more than 24 hours. However, in some cases, particularly in the context of multifocal vasculopathy, the presentation may be more stepwise or progressive, rather than abrupt. 6 Similarly, particularly in cases of coinfection, there may be a subacute history of additional symptoms including progressive headache, fever, neck stiffness, skin rashes or lesions, and constitutional features such as unintentional weight loss and night sweats.
Demographics
A consistent feature across cohorts is that stroke tends to occur at a younger age in PLWH. 6 In studies from high-income settings during the early ART era, the median age at presentation was 42.9 years in 1997 and 48.4 years in 2006; 29 another study reported a median of 42 years between 1997 and 2002. 30 In low- and middle-income countries (LMICs), the age at presentation was even younger: 33.4 years in Cape Town (2000-06) 31 and 39.8 years in Blantyre, Malawi (2008-09). 32 The influence of sex, however, remains uncertain. While male sex is a strong predictor of stroke in the general population, some HIV cohorts suggest a higher risk among women, particularly younger women living with HIV, pointing to potential sex-specific or hormonal interactions that warrant further investigation. 3
Stroke classification
Ischemic stroke predominates, comprising around three quarters of cases.4,10 Using our clinicopathological framework for describing the etiology of HIV-related ischemic stroke,6,13 in southern African cohorts the cause is opportunistic infections in 25–28%, HIV vasculopathy in 20–38%, coagulopathy in 9–19%, cardioembolic in 6–14%, and unknown/cryptogenic in 17–19%.13,31 Anatomically, ischemic strokes of the cortex and adjacent white matter tracts are the most common, but higher rates of basal ganglia infarct and multivessel involvement are observed.13,33 Hemorrhagic strokes are less common than ischemic strokes but still occur at rates higher than the general population. 4 Several cohorts have shown a signal for low CD4 count and intracerebral hemorrhage, even in the absence of opportunistic coinfection,4,34 supporting a role for HIV-associated vasculopathy. A candidate mechanism is the progression through inward remodeling, dolichoectasia, and fusiform aneurysm formation observed postmortem in the intracranial arteries of PLWH, although these histological changes have not been demonstrated in a hemorrhagic stroke-specific cohort. 20 Finally, given the proinflammatory milieu and potential endothelial dysfunction in HIV, one might expect a higher frequency of cerebral venous thrombosis. However, available data are limited to small case series; therefore, it remains uncertain whether venous strokes are truly more common in PLWH.
Subclinical cerebrovascular disease
There is increasing recognition of subclinical cerebrovascular disease in PLWH. While only 1–5% of PLWH are reported to have stroke in clinical series, autopsy studies have found evidence of cerebral ischemic lesions and vascular pathology in 4–34% of cases in the premodern ART era. 6 In the modern era, this remains frequent at autopsy. 16 Similarly, cerebrovascular disease in PLWH may manifest as cognitive decline rather than overt stroke and may be related to small vessel disease (SVD). 35 The link between progressive SVD, stroke, and HIV-associated neurocognitive disorder remains incompletely understood, but this overlap raises the possibility that what appears as cognitive decline may in some cases reflect vascular pathology. It remains uncertain whether the principal drivers of this are directly related to HIV itself, or whether these changes reflect overlapping processes associated with aging and traditional vascular risk factors (Figure 4).
Stroke across the HIV disease course
An important way to conceptualize stroke in PLWH is to consider it across the stages of HIV disease (Figure 5). In advanced untreated disease (Stage 1), the risk is largely mediated by opportunistic and co-infections such as tuberculosis, VZV, and syphilis, which can drive vasculitis or vascular occlusion. 13 Following ART initiation or re-initiation (Stage 2), stroke may arise not only from co-infections but also from IRIS vasculopathy. The timing is usually within the six months of ART initiation but can occur up to two years later. In stable treated HIV (Stage 3), the clinical spectrum resembles that of the aging general population, with contributions from atherosclerosis, arteriosclerosis, and cardioembolic disease, but occurs at an accelerated rate. Importantly, coinfections such as syphilis and VZV can occur at any disease stage (i.e. regardless of immunosuppression), and clinicians should maintain a high index of suspicion. Similarly, a detailed history should be sought to include any ART interruption or drug resistance to recognize stage 3 patients who may revert to stages 1 and 2.
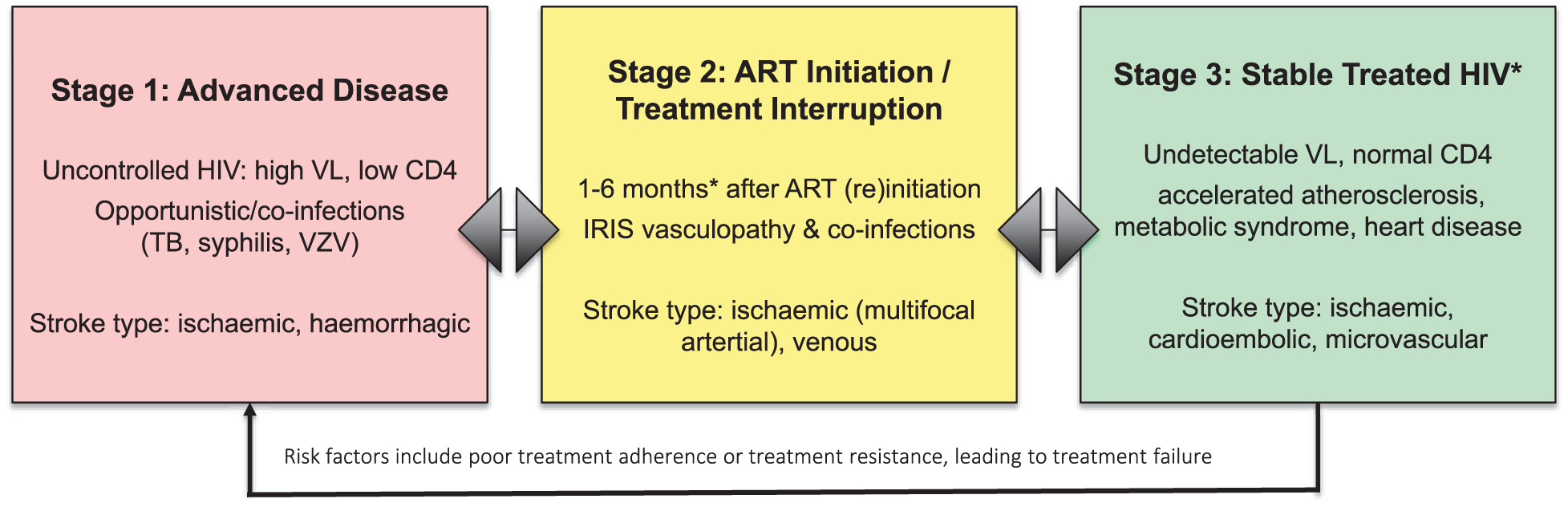
HIV and stroke risk across disease stages. In advanced untreated disease (Stage 1), the risk is largely mediated by opportunistic and coinfections such as tuberculosis, varicella zoster virus (VZV), and syphilis, which can drive vasculitis or vascular occlusion. In the period of ART initiation or treatment interruption (Stage 2), stroke may arise not only from opportunistic infections but also from an immune reconstitution inflammatory syndrome (IRIS) vasculopathy. *The timing is usually within the first six months of ART initiation but can occur up to two years later. In stable treated HIV (Stage 3), the clinical spectrum resembles that of the aging general population, with contributions from atherosclerosis, arteriosclerosis, and cardioembolic disease, but occur at an accelerated rate. Importantly, while some infections are only associated with immunocompromise (TB, cryptococcus), coinfections such as syphilis and VZV can occur at any disease stage. Interruptions to treatment and/or development of resistance may result in reversion to Stage 1 disease. For stroke type, these are the commonest at each stage, and are not exhaustive; background HIV vasculopathy (atherosclerosis, nonatherosclerosis, small vessel disease) causes ischemic and hemorrhagic strokes at a higher rate than the general population, across the stages of disease.
Stroke mimics
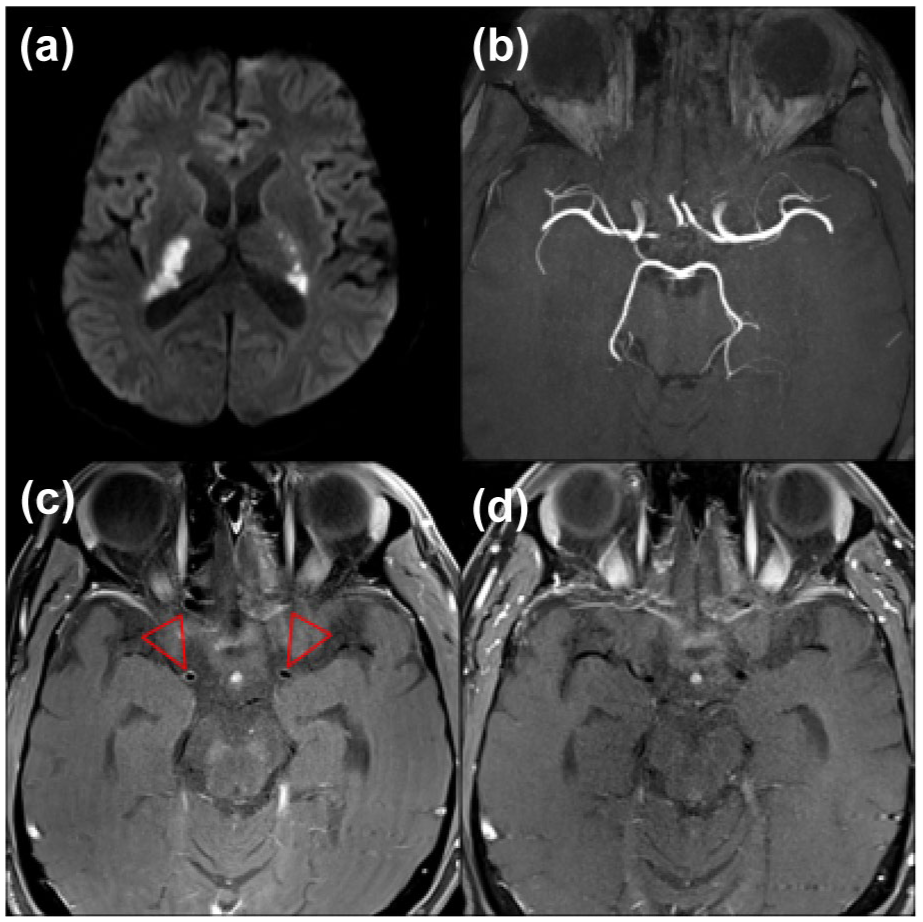
It is also important to recognize stroke mimics in PLWH. Common examples of HIV-related pathology which can present with focal neurology include CNS toxoplasmosis (Figure 6), tuberculomata, lymphoma, and progressive multifocal leukoencephalopathy. While these typically present subacutely, each may cause seizures; an associated Todd’s paresis may present as a sudden-onset deficit and masquerade as hyperacute stroke. Such conditions usually occur in stages 1 and 2 of HIV disease and should be considered particularly when imaging is unavailable, and the patient continues to deteriorate despite standard management. In stage 3 disease, conditions such as CNS escape (a term used to describe detectable virus in the CSF in the presence of lower or undetectable serum viral load) or CD8 encephalitis may arise, although these usually present with encephalopathy or cognitive decline rather than acute focal stroke-like syndromes. 36
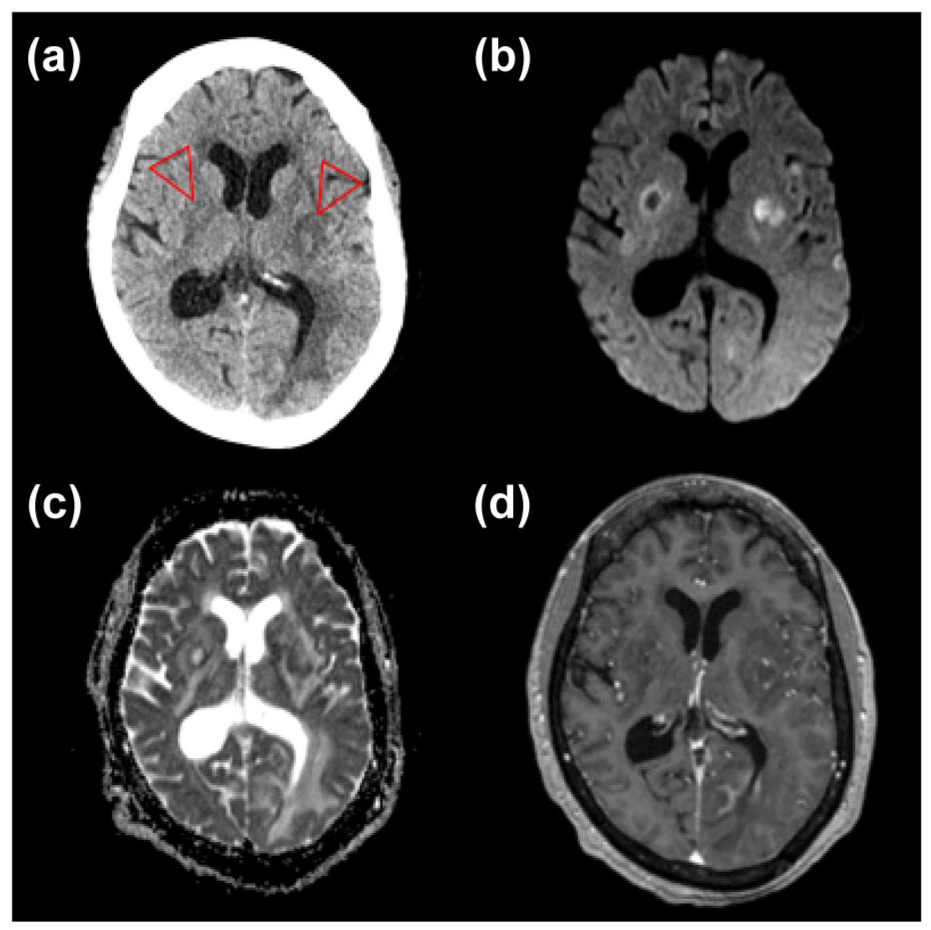
CNS toxoplasmosis. A 66-year-old patient, who presented with right-sided weakness and confusion, underwent a CT and CT angiogram due to clinical suspicion of a stroke. CT showed areas of low attenuation in the basal ganglia and the left occipital lobe (a, arrows). MRI, however, showed multiple cerebral lesions causing diffusion restriction (b and c) with peripheral enhancement as well as further smaller areas of subcortical enhancement. Toxoplasmosis was diagnosed based on a positive CSF PCR. HIV was also diagnosed on admission, with a CD4 nadir of 10 and viral load of 128,825, and she was commenced on emtricitabine/tenofovir and dolutegravir.
The staged approach (Figure 5) may be particularly useful in LMICs, where diagnostic resources are limited and reliance on careful history and disease staging may help guide assessment. Moreover, stroke can still represent the first presentation of an undiagnosed HIV infection. Recognizing associated clinical features, such as oral or esophageal candidiasis, seborrheic dermatitis, or Kaposi’s sarcoma skin lesions, can provide important diagnostic clues in such cases.
What should clinicians do?—Management
Investigations
Initial acute evaluation of a suspected stroke should include the National Institutes of Health Stroke Scale, noncontrast CT of the head, CT angiogram, and CT perfusion imaging, if available. We recommend that HIV screening is included in the standard stroke workup. In PLWH, a new stroke should prompt evaluation of disease status with CD4 count and HIV viral load, as well as a minimum evaluation for co-infections including syphilis (serology) and tuberculosis (chest X-ray and urine sample) (Figure 7). 37 Special attention should be paid to any atypical or accompanying clinical features, including features of infection. In these cases, further tests for coinfection, including lumbar puncture (LP), may be indicated. LP is an important adjunct, particularly when there is a high suspicion of infective or inflammatory vasculopathy. Standard cautions and contraindications to LP apply: severe systemic illness or raised intracranial pressure due to malignant cerebral edema or hemorrhage are notable. Indicators include systemic symptoms (e.g. fever, cough, constitutional symptoms, rash), laboratory evidence (raised inflammatory markers, CD4 count < 350), and/or neuroimaging features suggestive of a nonatherosclerotic process (white matter–predominant hyperintensities, contrast enhancement, or multifocal vasculopathy). LP may also be informative in PLWH who have recently initiated ART (within six months), had a low CD4 nadir, or show rapid immune reconstitution, where CSF pleocytosis or raised protein may point to underlying infection or IRIS. It is worth acknowledging that CSF pleocytosis or raised protein are suggestive but not specific for inflammatory etiology: up to 10% of patients with non-inflammatory/infectious ischemic stroke have CSF pleocytosis, albeit mild (below 50 cells/mm3). Finally, LP is necessary to identify uncontrolled HIV in the CNS compartment (send CSF HIV PCR). This is possible in any PLWH presenting with neurological symptoms, but is more likely in cases where treatment has not been started, was significantly delayed, or has been interrupted. 36 Identification of CNS escape and potential resistance is important because it may have implications for their ART regimen going forward .

Stroke workup in PLWH. Core investigations are in bold italic, alongside further investigations to be considered CT—computerized tomography.
Hyperacute management
Evidence on the safety of thrombolytics such as tissue plasminogen activator (tPA) or tenecteplase in PLWH is limited and largely derived from retrospective series. These data suggest no excess harm compared with the general population, although most reported cases are individuals with well-controlled HIV, i.e., presumed stage 3 disease. 38 Caution is therefore warranted in patients with suspected or confirmed infective or inflammatory vasculopathy, where vessel wall fragility may increase the risk of hemorrhage. Similarly, while there is no clear contraindication to endovascular therapy for large-vessel occlusions in PLWH, the role and safety of such interventions in co-infectious vasculopathies remains uncertain. These patients often present subacutely and are therefore typically ineligible for hyperacute reperfusion therapies.
ART and acute management
ART should be continued in patients established on treatment. For patients on ART who are not virally suppressed, a detailed social history should be obtained to identify barriers to ART access, tolerance, and adherence, and genotyping, when available, should be considered if there is concern for ART resistance. In people who report ART adherence on abacavir and protease inhibitor regimens, risk-benefit discussion should be held and alternative drug regimens considered, given their association with stroke. For patients not on ART, ART should be initiated regardless of viral load or CD4 count. However, in cases of stenotic or obliterative intracranial vasculopathy, particularly when an infectious or inflammatory cause is suspected, immediate ART initiation may precipitate IRIS. The optimal timing of ART initiation in this context remains uncertain due to the lack of high-quality data. 39 A pragmatic approach, supported by evidence from TB and cryptococcal meningitis, is to first treat the infection and control inflammation (e.g. prednisolone 1 mg/kg for 2 weeks) and defer ART. The U.S. National Institutes of Health guidelines recommend delaying ART for 4–6 weeks in cryptococcal and TB meningitis. 40 Likewise, in patients with stroke and concurrent CNS infection without frank vasculopathy, infection should be managed first, with ART introduction guided by the same principles. For patients already established on ART, therapy should not be interrupted.
Secondary prevention
Ongoing management after stroke is largely the same as in the general population. It is important to note that aspirin and other secondary preventive therapies are prescribed in only 50% of PLWH in whom these medications are indicated. 41 Thus, special attention should be paid to this to ensure treatment in accordance with guidelines. The prevalence of reported drug–drug interactions (DDIs) between antiplatelet medications and ART may be as high as 60% in PLWH. While aspirin is not known to interact with ART, P2Y12 inhibitor antiplatelet medications, including clopidogrel, ticagrelor, and prasugrel, are activated or metabolized by CYP enzymes that in turn are inhibited by some PIs, reverse transcriptase inhibitors, and ART regimen boosters, and thus may theoretically increase the risk of bleeding or thrombosis when used concurrently.42,43 However, the clinical significance of these interactions is not well established, and treatment with antiplatelet or dual antiplatelet therapy in PLWH is generally not precluded based on these interactions alone. In fact, one recent large study found no association with antiplatelet use and bleeding or adverse cardiac events in cases of ART DDIs. 44 For patients requiring full anticoagulation, use of Direct Oral Anticoagulants (DOACs) along with PI-containing regimens is generally contraindicated due to risk of CYP 450 enzyme inhibition, increased DOAC concentration, and bleeding. DDIs with warfarin and multiple classes of ART drugs are common and thus require close INR monitoring.43,45 There is no known adverse interaction between ART and heparin or low molecular weight heparin. 43 For statin therapy, generally pitavastatin, atorvastatin, pravastatin, and rosuvastatin are preferred, while fluvastatin and simvastatin should be avoided, especially with PIs. 43 A summary of DDIs and recommendations is found in Table 1; a further comprehensive resource is hosted by the University of Liverpool here: https://www.hiv-druginteractions.org.
Interactions of stroke medications and ART.
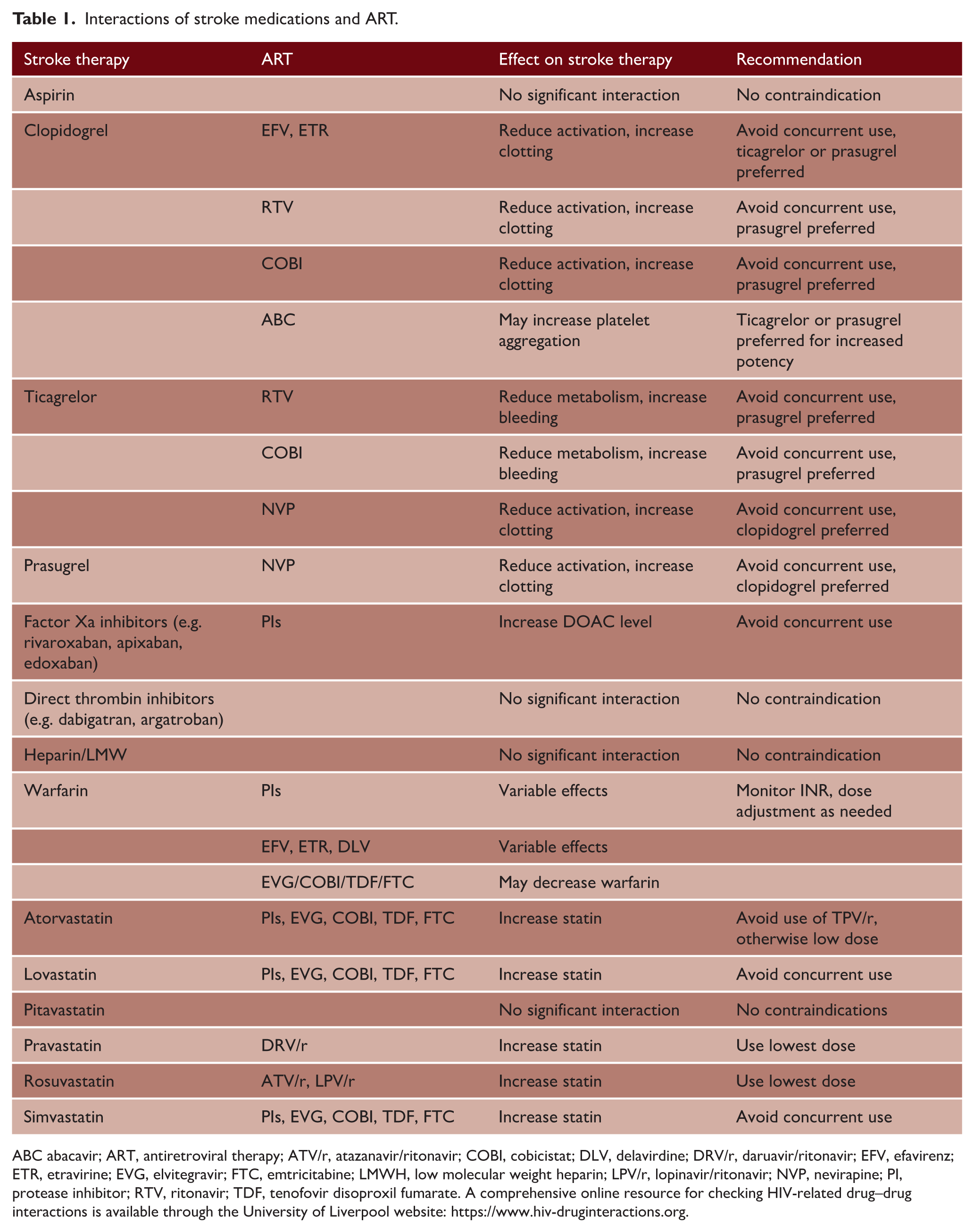
ABC abacavir; ART, antiretroviral therapy; ATV/r, atazanavir/ritonavir; COBI, cobicistat; DLV, delavirdine; DRV/r, daruavir/ritonavir; EFV, efavirenz; ETR, etravirine; EVG, elvitegravir; FTC, emtricitabine; LMWH, low molecular weight heparin; LPV/r, lopinavir/ritonavir; NVP, nevirapine; PI, protease inhibitor; RTV, ritonavir; TDF, tenofovir disoproxil fumarate. A comprehensive online resource for checking HIV-related drug–drug interactions is available through the University of Liverpool website: https://www.hiv-druginteractions.org.
Primary prevention
Clinical risk calculators used for primary prevention of stroke and other cardiovascular diseases, which were largely created using HIV-negative populations in high-income countries, appear to underestimate cardiovascular risk in PLWH. 46 Given that HIV acts as an independent risk factor for cerebrovascular disease, additional research into individualized risk calculators and prevention strategies is needed. Currently, there is no indication for antiplatelet or anticoagulation therapy for primary stroke prevention in PLWH. However, results from the REPRIEVE trial, which was halted early for efficacy, found that PLWH aged 40–75 with low-to-moderate cardiovascular risk who took pitavastatin had a 35% lower risk of major cardiovascular events vs placebo. 47 Accordingly, international HIV societies now recommend offering a medium-intensity statin to all PLWH above the age of 40, regardless of lipid profile or calculated cardiovascular risk. 48
Outcomes and prognosis: how do they do after a stroke?
Evidence on stroke outcomes in PLWH is mixed and varies by setting. In Malawi, a prospective cohort of 147 first-ever strokes found that 1-year mortality was 40% and poor functional outcome (mRS 4–6) nearly 48%, but importantly, HIV status itself was not associated with worse outcomes. 32 Similarly, in a South African tertiary hospital cohort, in-hospital outcomes (mortality, length of stay) did not differ between PLWH and HIV-negative matched controls. 33 In high-income settings, African American cohorts demonstrated younger stroke onset in PLWH, with worse outcomes at discharge associated with low CD4 and uncontrolled viraemia. 29 A Danish nationwide registry reported similar mortality after stroke compared with HIV-negative controls once comorbidities were adjusted for, but found a higher risk of recurrent stroke in PLWH. 49 Beyond mortality and recurrence, cognitive outcomes are also clinically relevant in that stroke may accelerate underlying HIV-associated neurocognitive disorder. Neuroimaging studies have demonstrated a higher burden of white matter hyperintensities (WMHs) in PLWH, consistent with small vessel disease, and in some cases, cerebral microbleeds.35,50 Collectively, these data suggest that while short-term functional outcomes can be comparable when access to care is equal, immune suppression, viraemia, and coinfections remain key modifiers of prognosis, with recurrence and cognition requiring particular attention.
Real-world challenges of stroke in PLWH: stigma, access, and awareness
Addressing the challenges of stroke in PLWH extends beyond biological mechanisms to the realities of care delivery. PLWH often have many co-occurring social, educational, and economic factors, which lead to increased vulnerability and barriers to medication adherence and follow-up. These may include financial barriers such as inability to pay for medications or transportation to follow-up appointments, reduced educational level or cognitive impairment, and distrust of the healthcare system due to provider and systemic biases. Stigma, inequities in access to diagnostics, clinician awareness gaps, and systemic underrecognition in policy all contribute to poorer outcomes, particularly in LMICs. Practical steps to mitigate these barriers are summarized in the Practice Recommendations Box (Figure 8), highlighting the need for integrated, context-specific solutions.

Practice recommendations—addressing real-world challenges.
Unanswered questions and future research
Despite advances in understanding stroke in PLWH, key uncertainties remain (Table 2). At the mechanistic level, it is still unclear whether HIV-associated vasculopathy represents a distinct entity or an accelerated form of atherosclerosis, and distinguishing direct from indirect consequences of the virus remains challenging. Vessel wall imaging and postmortem data are valuable but limited, and better phenotyping is needed to clarify whether these changes are unique or overlap with traditional vascular disease. Progress in omics technologies may prove crucial in unraveling underlying mechanisms, but this will depend on well-characterized cohorts, cerebrospinal fluid repositories, and postmortem resources with high-quality nucleic acid preservation. 51
Key unanswered questions and future research priorities in HIV-associated stroke.
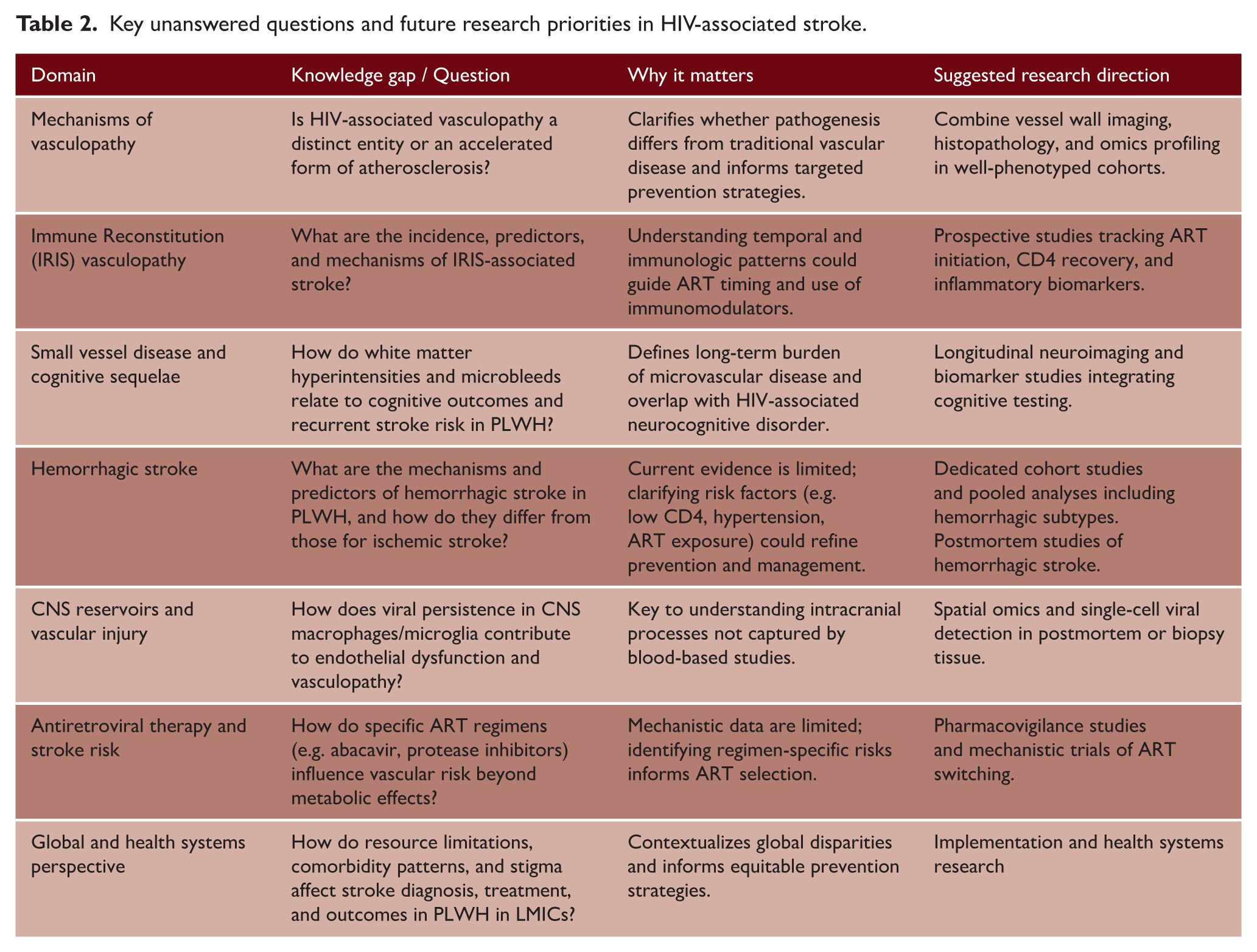
IRIS-associated vasculopathy also remains poorly defined. Case–control studies suggest a strong temporal relationship between ART initiation and stroke risk in immunosuppressed individuals, but prospective validation and interventional trials, including studies of ART timing and immunomodulatory therapies, are lacking.
The contribution of small vessel disease and microvascular pathology to long-term outcomes requires clarification. Cross-sectional imaging has shown a higher burden of WMH and microbleeds in PLWH, but longitudinal data linking these findings to cognitive decline, functional recovery, or recurrent stroke remain sparse. Susceptibility-weighted imaging and vessel wall imaging may help characterize this burden in vivo. An important caveat is that recent consensus on cognitive impairment in PLWH now separates HIV-associated brain injury from cerebrovascular disease. 52 While this reduces over-attribution of impairment to HIV, it risks relegating vascular contributions/mediators. Given the increasing burden of stroke and small vessel disease in this population, cerebrovascular injury must be integrated into future research on HIV-related cognitive outcomes. Prospective multimodal studies that combine vascular, viral, and immune biomarkers with cognitive phenotyping will be essential to disentangle these overlapping mechanisms.
Conclusion
Important gaps remain in understanding the mechanisms, natural history, and outcomes of stroke in PLWH. Future work must better define how HIV-related vascular injury interacts with traditional risk factors, immune reconstitution, and small vessel disease, while also addressing the impact of emerging challenges such as COVID-19. Integrating advanced imaging, biomarker studies, and longitudinal cohorts will be crucial; so too will pragmatic research in low-resource settings, where the burden is greatest. A coordinated, multidisciplinary approach is needed to ensure that cerebrovascular disease is not overlooked in the broader HIV research agenda and that findings translate into improved care worldwide.
Footnotes
Acknowledgements
We have no acknowledgements.
Ethical considerations
This article is a review of previously published literature. No new studies involving human participants or animals were conducted by the authors. The radiological images were obtained from an institutional teaching archive and are fully anonymized, with no identifiable patient information included. Ethical approval was therefore not required.
Consent to participate
The images used in this manuscript are fully anonymized and contain no identifiable patient information. Individual informed consent was therefore not required.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LB is supported by a Wellcome Trust fellowship (222102/Z/20/Z) and UCL/UCLH NIHR Biomedical Research Centre.
Data availability statement
There are no primary data in this review.
