Abstract
Background:
Although long-term stroke management is critically important, poor patient adherence to follow-up appointments threatens the validity of clinical trials. This cross-sectional survey aimed to identify contributing factors and potential consequences of lost to follow-up (LTFU) in long-term stroke management trials.
Methods:
We searched Medline, Embase, Web of Science, Cochrane library, and Scopus from inception to 20 August 2024 for randomized controlled trials of multimodal post-stroke care initiated within 1 year of stroke. Data on general trial and methodological characteristics were extracted. Univariable random-effects meta-regression analyses were performed to identify LTFU predictors. Furthermore, we assessed how assumptions about LTFU affected effect estimates for significant binary primary outcomes.
Results:
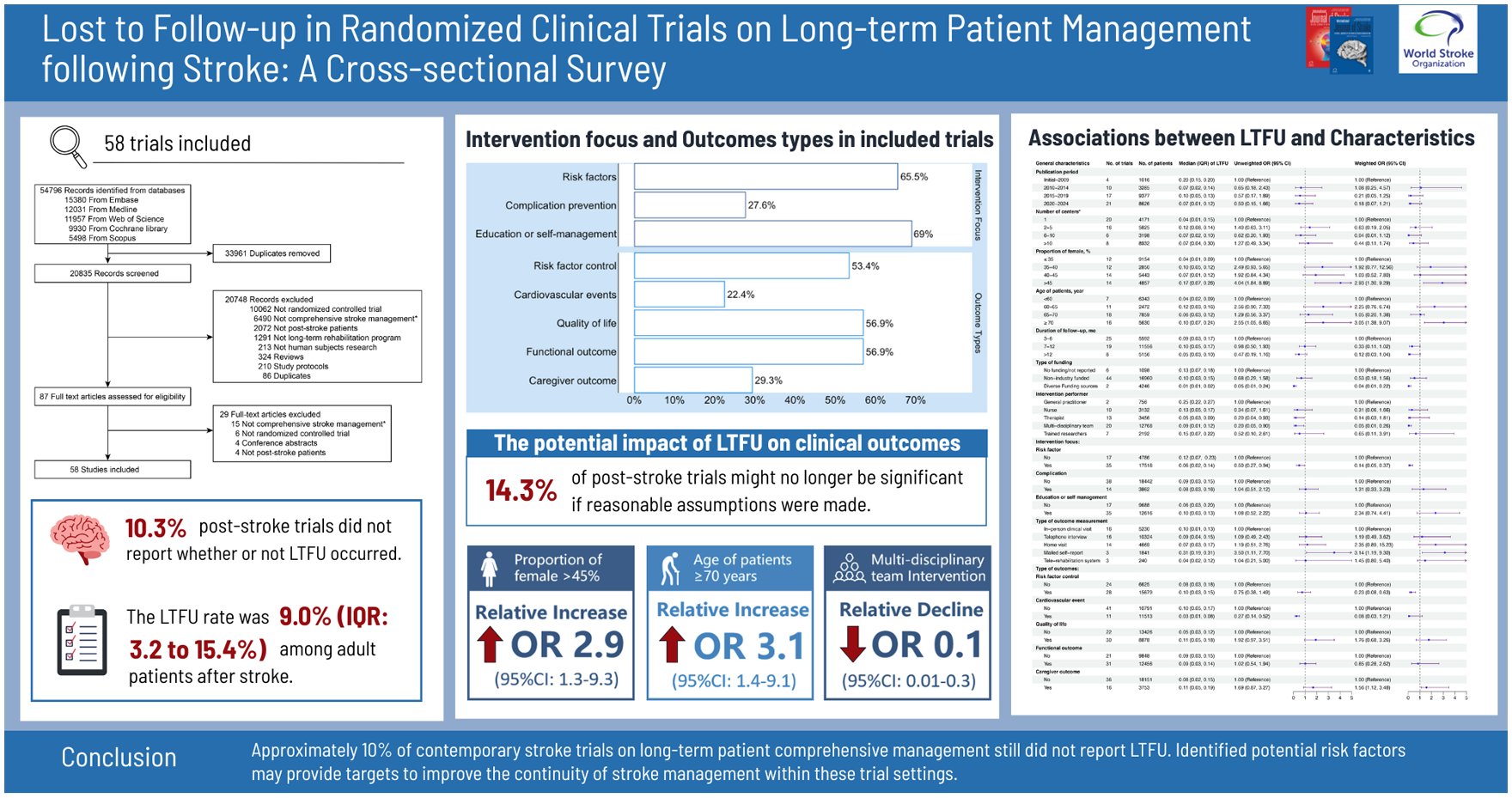
Among 58 eligible reports (27,575 patients and 3349 caregivers), six trials (10.3%) did not specify patient LTFU, while 8 of 17 caregiver-inclusive trials (47.1%) omitted LTFU reporting of caregivers. The median follow-up was 12 months (interquartile range (IQR): 6–12), with LTFU rates of 9.0% (IQR: 3.2–15.4%) for patients and 14.0% (IQR: 6.8–20.7%) for caregivers. Higher LTFU odds correlated with a higher proportion of females (odds ratio (OR): 2.93, 95% confidence interval (CI): 1.30–9.29) and older age (OR: 3.05, 95% CI: 1.38–9.07). Trials involving multidisciplinary rehabilitation teams showed lower LTFU (OR: 0.05, 95% CI: 0.01–0.26). When assuming different event rates for LTFU patients, 0–14.3% of significant results were no longer significant.
Conclusion:
Overall, approximately 10% of stroke trials on long-term patient management still did not report LTFU. Identified potential risk factors may provide targets to improve the continuity of stroke management within these trial settings. Attention to patient management is critical for ensuring valid trial conclusions.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
