Abstract
Background:
Little is known about early major neurological improvement (EMNI) after intracerebral hemorrhage (ICH).
Aims:
We performed a post hoc analysis of the Intracerebral Hemorrhage-Deferoxamine trial (i-DEF; NCT02175225) to comprehensively evaluate EMNI and assess whether deferoxamine treatment affects it.
Methods:
Comparing repeated assessments of National Institutes of Health Stroke Scale (NIHSS) on days 2, 3, 4, and 7 (or discharge, if it was earlier) versus NIHSS score at presentation, and defining EMNI as an NIHSS score decrement of an absolute ⩾4 points from presentation, we determined its presence or absence on day 2, day 3, day 4, and day 7(/discharge). Using adjusted generalized linear mixed-effects or logistic models as appropriate, we examined the association of deferoxamine with EMNI as repeated measure, as well as EMNI’s overall frequency, time course, determinants, and association with favorable long-term outcome (modified Rankin Scale 0–2).
Results:
Among 291 i-DEF participants in the modified intention-to-treat population (median age 61 years, 38.5% female, median NIHSS score 13, 144 randomized to deferoxamine and 147 to placebo), the proportion of participants with EMNI continuously increased from 20% on day 2 to 36% on day 7(/discharge). Deferoxamine was associated with an average twofold higher odds of EMNI (odds ratio (OR): 2.30, 95% confidence interval (CI): 1.07 to 4.95, p = 0.033 after adjustment for the prespecified trial covariates onset-to-treatment time, baseline ICH volume, and presenting NIHSS score), without clear evidence for treatment-by-time interaction (pinteraction = 0.092). Secondary and sensitivity analyses using alternative EMNI definitions (as relative ⩾20% or ⩾30% NIHSS score decrement) and additional covariate adjustment yielded consistent findings. Race, ICH volume and location were also associated with EMNI. EMNI was independently associated with twofold to sixfold higher odds of favorable 90-day and 180-day outcome, regardless of assessment timepoint.
Conclusion:
In a post hoc analysis of the i-DEF trial, the likelihood of EMNI over the first week following ICH was higher with deferoxamine. EMNI showed a continuous upward trajectory and strong association with favorable long-term functional outcome.
Introduction
Intracerebral hemorrhage (ICH) is a devastating stroke form with sparse specific treatment options. 1 The early – acute and subacute– phase following ICH is known to be highly dynamic, but research on early clinical changes after ICH has almost exclusively focused on neurological deterioration.2–4 However, many patients with ICH do not worsen and may even improve early on.5,6 Early major neurological improvement (EMNI) has been extensively studied in ischemic stroke, particularly as an outcome predictor and treatment outcome per se.7,8 Little is known about EMNI after ICH. 9 Hardly any data exist about the time course and determinants of EMNI within the first week of ICH, its association with long-term outcomes, and how EMNI might be affected by treatments targeting secondary injury. 10
Iron toxicity has been implicated in secondary brain injury after ICH. 11 The Intracerebral Hemorrhage-Deferoxamine (i-DEF) randomized controlled trial showed signals for a positive effect of an early 3-day course of deferoxamine mesylate, an iron chelator, on functional outcomes and the trajectory of long-term recovery.12,13 In i-DEF, a substantially higher proportion of deferoxamine-treated participants achieved a modified Rankin Scale (mRS) score of 0–2, compared to placebo counterparts, within the first 7 days after ICH, indicating that deferoxamine might facilitate EMNI. 13
Aims
In this post hoc analysis of serial neurological assessments obtained using the National Institutes of Health Stroke Scale (NIHSS) over the first week of standardized follow-up within the i-DEF trial, we examined (1) the association of deferoxamine treatment with EMNI and (2) EMNI’s overall frequency, time course, determinants, and association with long-term outcome up to 180 days.
Methods
Study design and participants
Post hoc analysis of the i-DEF placebo-controlled, randomized, phase 2 clinical trial (NCT02175225) conducted across 40 centers in the United States and Canada. The trial methodology, data collection methods, and main results are described elsewhere.12,14 In short, i-DEF randomized participants aged 18–80 years with primary supratentorial ICH and without indication that withdrawal of care would be implemented within 72 h to receive daily intravenous infusions of deferoxamine (32 mg/kg/day) or placebo (saline) for three consecutive days, starting within 24 h of ICH onset. The randomization procedure included adjustment for key prognostic factors including the presenting NIHSS score. An NIHSS score ⩾6 was required for participation, and NIHSS was re-assessed serially on days 2, 3, and 4 (i.e. after each infusion at 24, 48, and 72 h following study drug initiation), as well as on day 7 (or discharge, whichever was earlier) by certified masked investigators. The exact time at which the actual assessments were carried out for each timepoint was also captured. Here, we included all participants comprising the modified-intention-to-treat population of the trial 12 who had at least one available NIHSS reassessment after presentation.
Further baseline assessments in i-DEF included standardized clinical (demographics/comorbidities) and neuroimaging evaluation. This included central assessment of CT scans by blinded trained raters at the core imaging laboratory to determine hematoma location, intraventricular hemorrhage (IVH) extension, and the volume of hematoma and perihematomal edema. Volumetry was done with an imaging analysis software (Analyze 11.0; AnalyzeDirect, Overland Park, KS, USA) using a validated Hounsfield-unit-threshold-based, semi-automated segmentation approach with manual correction, as described previously. 15 Follow-up evaluations included functional status on the mRS at 90 and 180 days by certified masked assessors.
Outcomes
Primary outcome was EMNIa, defined as an absolute NIHSS score decrease by ⩾4 points compared to presentation. The NIHSS is a measure of neurological deficits ranging from 0 to 42, with higher scores indicating more severe impairment. We chose NIHSS instead of Glasgow Coma Scale (GCS), another widely used score of clinical severity that focuses on impairment of consciousness, because the former captures neurological impairment in a broader and more detailed manner, and was shown to be a more meaningful assessment tool and better outcome predictor in ICH.16,17 We chose a 4-point cutoff, because this definition has long been thought to reflect a clinically important change and is established in ischemic stroke,18–20 and has been considered by others for use in ICH. 9 Since a presenting NIHSS score ⩾6 was required for trial participation, the 4-point-decrement definition was universally applicable in trial participants. Using the NIHSS scores obtained at the prespecified assessment timepoints, we determined the presence or absence of EMNIa as dichotomous outcome on days 2, 3, 4, and 7 by comparing the respective NIHSS scores on days 2, 3, 4, and 7 versus the NIHSS score at presentation.
Secondary outcomes were alternative definitions of EMNI as relative NIHSS score decrement (1) ⩾20% (EMNIr20) or (2) ⩾30% (EMNIr30) compared to presentation, in accordance with prior research in ischemic stroke showing that relative rather than absolute NIHSS score changes by these thresholds may be more informative.21,22 EMNIr20 and EMNIr30 were again considered as repeated measures on days 2, 3, 4, and 7, as above.
Statistical analysis
We categorized participants according to a modified-intention-to-treat strategy to deferoxamine versus placebo, in keeping with the main trial. 12 We present baseline characteristics and outcomes using descriptive statistics, that is, frequencies and percentages for categorical data, and median and interquartile range (IQR) for continuous data. We compared categorical and continuous variables using χ2 or Fisher’s exact tests and Mann–Whitney U or t-tests as appropriate, respectively. We performed a longitudinal analysis of EMNI as repeated measure.
Primary analysis: association of deferoxamine with EMNIa
We used generalized linear mixed-effects models with logit link function and robust standard errors clustered at the participant level. Repeatedly assessed EMNIa during the first 7 days was included as the outcome, participant ID as random intercept, and NIHSS assessment time as continuous variable (exact time for each participant and timepoint, expressed in hours after presentation) as fixed effect. Random slopes were not included due to convergence issues. This approach accounts for the intra-individual correlation of repeated measures and leverages all available data for each participant. It assumes a linear change of logit-EMNIa with time, which was informed by initial exploratory descriptive analysis. To investigate the effect of deferoxamine on EMNIa, we fitted the model twice: (1) as a main-effects model, assuming the effect of deferoxamine is constant over time, and (2) as an interaction model to evaluate whether the effect of deferoxamine differs over time. In keeping with the main trial and previous i-DEF subanalyses,12,23 we adjusted all models for the prespecified categorical covariates onset-to-treatment time (⩽12 vs >12 h), baseline ICH volume (⩽10 vs >10 mL), and presenting NIHSS score (⩽10 vs >10).
Secondary analyses: association of deferoxamine with alternative EMNI definitions
Following the same approach, we used EMNIr20 and EMNIr30 instead of EMNIa as the outcome in the adjusted linear mixed models described above.
Sensitivity analyses
We repeated the primary analysis of EMNIa and the secondary analyses of EMNIr20 and EMNIr30 (1) alternatively adjusting for presenting NIHSS as continuous variable (instead of the prespecified categories) and (2) maximally adjusting for onset-to-treatment time, baseline ICH volume, and presenting NIHSS, all as continuous variables (after ensuring no gross violation of the linearity assumption), along with IVH presence and ICH location (lobar/thalamic/deep non-thalamic) as categorical ones. This was to account for imbalances of these established outcome predictors between the groups, as in previous i-DEF subanalyses. 13
Additional analyses: EMNI’s determinants and association with long-term outcome
We explored EMNIa’s determinants in the entire study population using the same approach as above to investigate the main effects of the following baseline characteristics in a single multivariable generalized linear mixed-effects model adjusted for randomized treatment: presenting NIHSS, age, ICH volume, edema extension distance (EED) 24 as continuous variables, and sex, race (White/Black/Asian/other), hypertension, diabetes, ICH location (lobar/thalamic/deep non-thalamic), and IVH presence as categorical ones. We repeated these analyses using EMNIr20 and EMNIr30, instead of EMNIa.
We investigated the association of EMNIa at each assessment timepoint with favorable functional outcome at 90 days, defined as mRS score 0–2, in keeping with the main trial.12,13 For this, we fitted four separate logistic models for EMNIa on day 2, day 3, day 4, and day 7/(discharge). All models were adjusted for randomized treatment and known outcome predictors including age, presenting NIHSS, ICH volume as continuous variables, and ICH location and IVH presence as categorical ones, as in previous research.4,6,25 We repeated these analyses using functional outcome at 180 days. Finally, we repeated all analyses of 90-day and 180-day outcome using EMNIr20 and EMNIr30 as predictors, instead of EMNIa.
For all analyses, we report model-based estimates as odds ratios (ORs) along with 95% confidence intervals (CI) and two-sided p-values. We applied no multiplicity correction given the nature of this post hoc analysis. Analyses were done using STATA v.18 (StataCorp LLC, College Station, TX, USA). We report this study in accordance with the STROBE statement.
Handling of missing values
Participants who died and were thus missing NIHSS assessment at any given timepoint were assigned absence of EMNI (using the sample median as assessment time for the purpose of analysis). No other imputations were done in case of missing NIHSS. Baseline data included as model covariates were complete. Given the low missingness in mRS, analysis of EMNI’s association with functional outcome was done on a complete-case basis.
Results
The entire i-DEF modified-intention-to-treat population comprising 291 participants was available for analysis (median age 61 years, 38.5% female, 147 allocated to placebo, and 144 to deferoxamine; Table 1). Information on presenting NIHSS was complete, with a median score of 13 (IQR 8–18, range 6–33); scores were well balanced between the treatment arms. Besides thalamic ICH and IVH (more common in the placebo arm), the remaining baseline data were reasonably balanced. A detailed comparison of baseline characteristics by treatment allocation has been published previously. 12
Baseline characteristics.
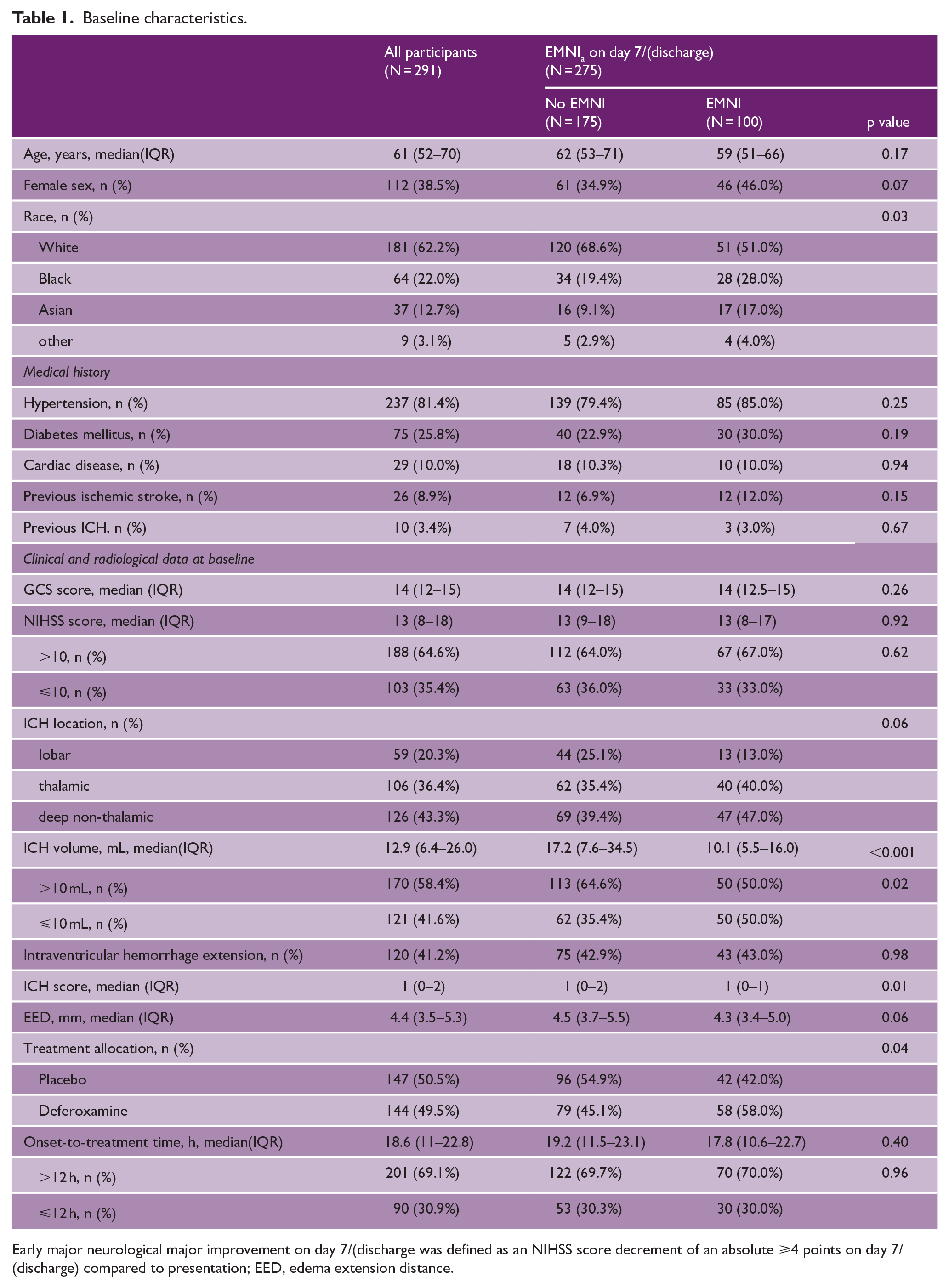
Early major neurological major improvement on day 7/(discharge was defined as an NIHSS score decrement of an absolute ⩾4 points on day 7/(discharge) compared to presentation; EED, edema extension distance.
Following presentation, all participants had at least one NIHSS reassessment. The median (IQR) time of NIHSS assessments was 26 (24–28), 50 (48–52), 74 (72–77), and 161 (139–169) h after presentation. The median (IQR) NIHSS score was 11 (7–17) on day 2 (11 [7–16] deferoxamine vs 12 [8–18] placebo), and decreased to 10 (6–16) on day 7 (9 [5–15] deferoxamine vs 11 [7–17.5] placebo]. Supplementary Figure 1 shows the detailed trajectory of NIHSS scores. A total of six participants died within 7 days and were therefore classified as “no EMNI” at a total of 7 assessment timepoints following their death. After this imputation, 31 participants with at least one missing NIHSS assessment remained (25 missing one, 6 missing two, and none missing three or more assessments). Among those, NIHSS was missing in 3 participants on day 2, 13 on day 3, 5 on day 4, and 16 on day 7(/discharge).
Effect of deferoxamine on primary outcome (EMNIa)
On day 2, 58/288 participants (20.1%) experienced EMNIa (22% deferoxamine vs 18.4% placebo). This increased to 100/275 (36.4%) on day 7(/discharge) (42.3% deferoxamine vs 30.4% placebo). Figure 1 shows the percentage of participants with EMNIa at consecutive assessment timepoints according to treatment allocation. In the main-effects model of the primary outcome including adjustment for the main trial’s prespecified categorical covariates, deferoxamine showed an average twofold higher odds of EMNIa compared to placebo (OR: 2.30, 95% CI: 1.07–4.95, p = 0.033). The interaction model yielded a weak signal for treatment-by-time interaction, such that the likelihood of EMNIa over time seemed to increase more rapidly with deferoxamine (pinteraction = 0.092).
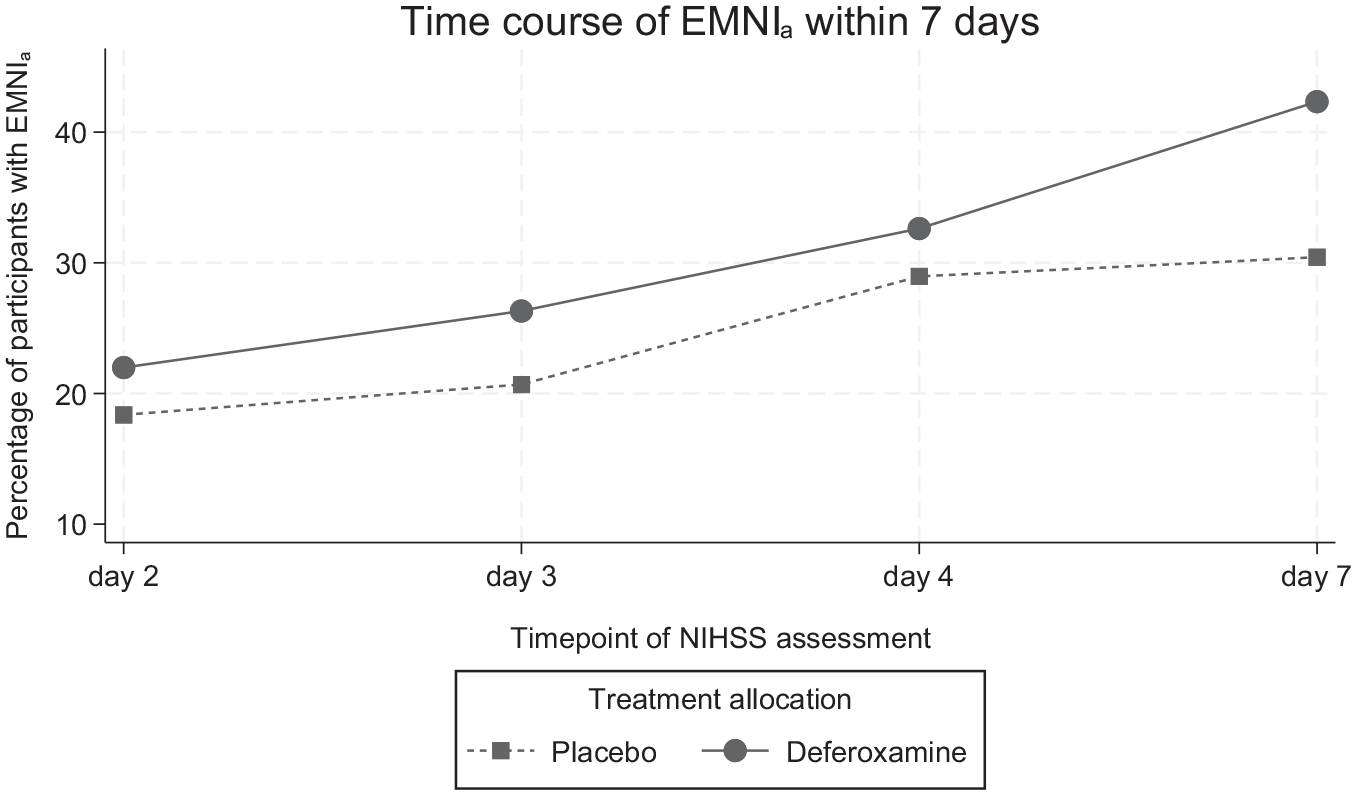
Percentage of participants with early major neurological improvement (EMNIa) at the prespecified NIHSS assessment timepoints according to treatment allocation. EMNIa was defined as an NIHSS score decrement of an absolute ⩾4 points between the respective timepoint and presentation.
Sensitivity analyses alternatively adjusting the models for continuous presenting NIHSS yielded comparable results (OR: 2.70, 95% CI: 1.24–5.86, p = 0.012 for deferoxamine in the main-effects model, pinteraction = 0.089 in the interaction model). Maximally adjusting (for presenting NIHSS, onset-to-treatment time, baseline ICH volume (all continuous), IVH presence, and ICH location) also showed consistent findings (OR 2.11, 95% CI: 0.94–4.74, p = 0.071 for deferoxamine in the main-effects model, pinteraction = 0.084 in the interaction model).
Effect of deferoxamine on secondary outcomes (EMNIr20 and EMNIr30)
The percentage of participants with EMNIr20 and EMNIr30 at consecutive assessment timepoints according to treatment allocation is shown in Figure 2. In the main-effects model, adjusted for the trial’s prespecified covariates, deferoxamine showed threefold higher odds of EMNIr20 compared to placebo (OR: 2.92, 95% CI: 1.29–6.62, p = 0.010). The interaction model showed no evidence of effect modification by time (pinteraction = 0.894). Results were consistent for EMNIr30, with the adjusted main-effects model yielding an OR of 4.01 (95% CI: 1.66–9.69, p = 0.002) for deferoxamine’s effect, and the interaction model again showing no evidence of treatment-by-time interaction (pinteraction = 0.948). Sensitivity analyses of both secondary outcomes in alternatively and maximally adjusted models showed consistent findings (Supplementary Table 1).
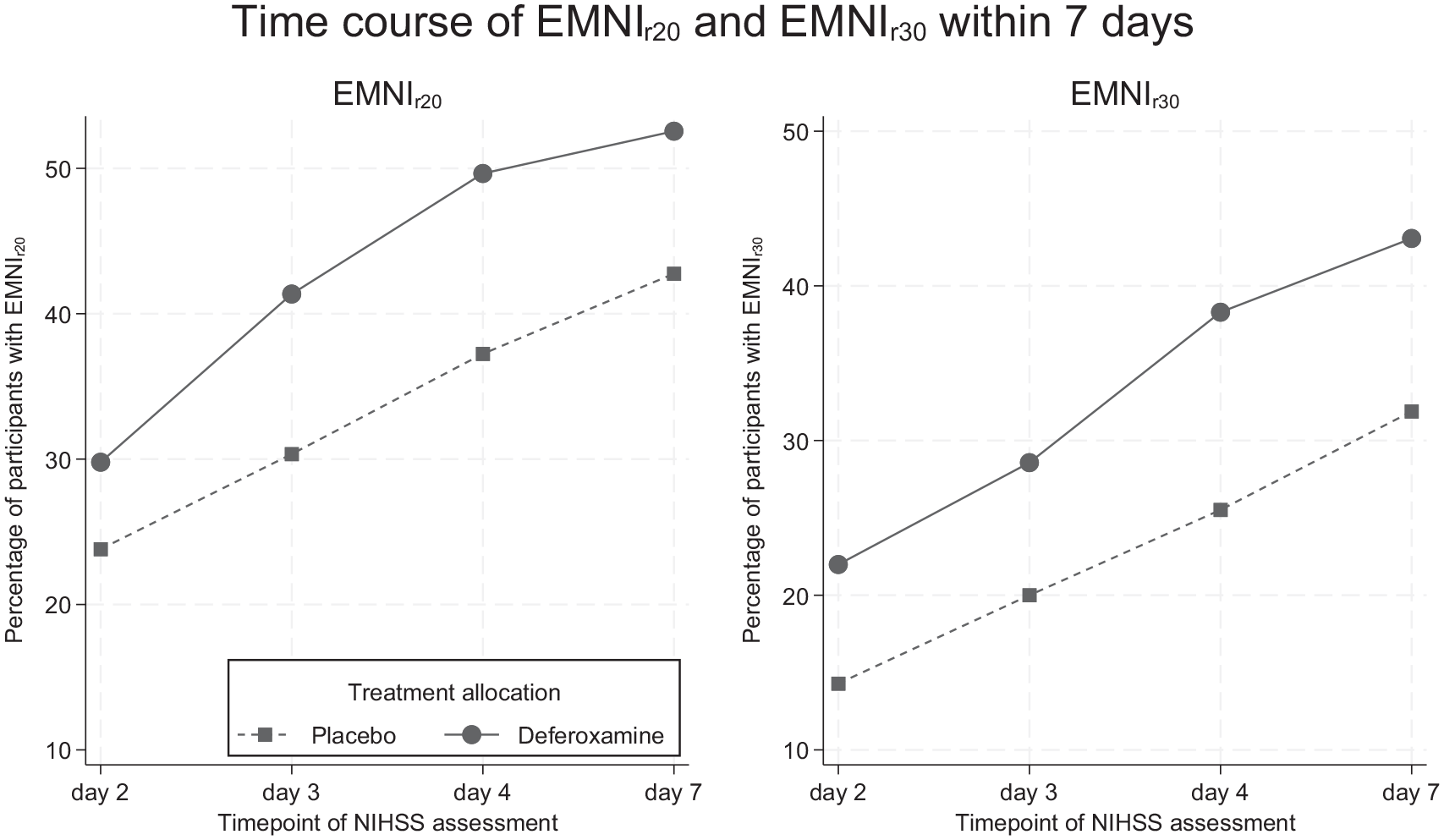
Percentage of participants with early major neurological improvement (left: EMNIr20; right: EMNIr30) at the prespecified NIHSS assessment timepoints according to treatment allocation. EMNIr20 and EMNIr30 were defined as an NIHSS score decrement of a relative ⩾20% or ⩾30%, respectively, between the respective timepoint and presentation.
Determinants of EMNI
Baseline characteristics according to EMNIa are given in Table 1. In the multivariable linear mixed model adjusted for randomized treatment, the odds of EMNIa across repeated assessments increased over time (OR: 1.45 per 24 h, 95% CI: 1.27–1.65) and with higher presenting NIHSS (OR: 3.32 per 5 points higher score, 95% CI: 2.09–5.29, both p < 0.001). Both Black (OR: 2.96, 95% CI: 1.13–7.71, p = 0.027) and Asian participants (OR: 6.02, 95% CI: 1.70–21.32, p = 0.005) had higher odds of EMNIa compared to their White counterparts. Larger hematoma volume was associated with lower odds of EMNIa (OR: 0.20 per 10 mL higher volume, 95% CI: 0.12–0.34, p < 0.001), while the odds were higher with both lobar (OR: 5.00, 95% CI: 1.03–24.40, p = 0.046) and deep non-thalamic hematomas (OR: 4.87, 95% CI: 1.70–13.98, p = 0.003) compared to thalamic ones. Figure 3 shows all model-based estimates. Repeated analyses using EMNIr20 and EMNIr30 yielded consistent results, except for showing no association of presenting NIHSS with EMNI (Supplementary Figure 2).
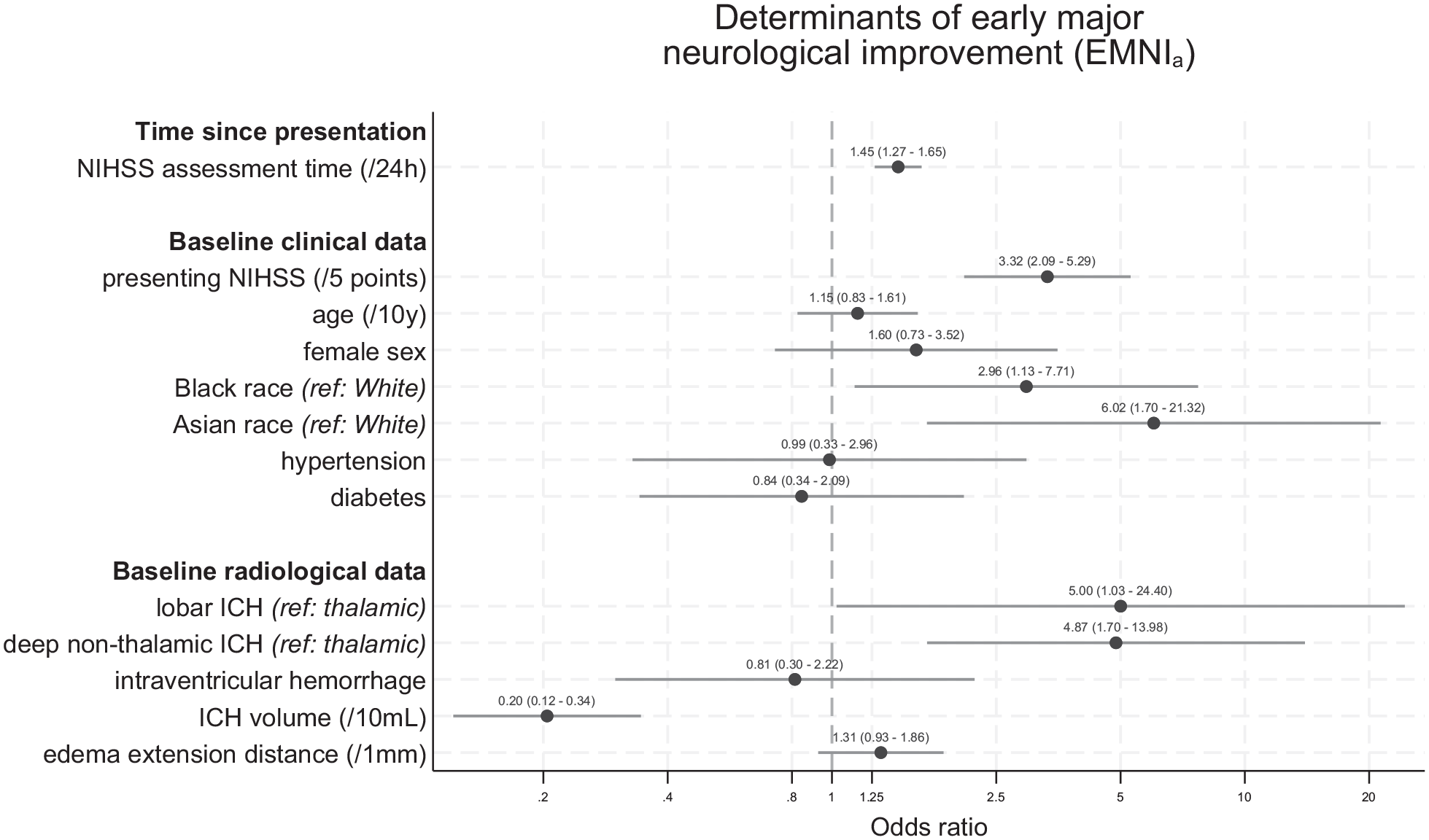
Odds ratio estimates for the association of time and baseline characteristics with EMNIa across all assessment timepoints during the first 7 days following ICH from the linear mixed model. EMNIa was defined at each timepoint as an NIHSS score decrement of an absolute ⩾4 points from presentation.
Association of EMNI with functional outcome
Data on 90-day mRS were available in 286 of 291 participants (98.3%). Regardless of whether it was assessed on day 2, day 3, day 4, or day 7(/discharge), EMNIa was independently associated with favorable 90-day outcome (mRS 0–2) after adjustment for age, presenting NIHSS, ICH volume, ICH location, IVH, and randomized treatment. The association was strongest for EMNIa on day 4 (OR: 5.96, 95% CI: 2.73–13.02, p < 0.001). We obtained similar results when using 180-day mRS (available in 275/291 participants (94.5%)). Here, the association with favorable 180-day outcome was strongest for EMNIa on day 3 (OR: 5.56, 95% CI: 2.42–12.77, p < 0.001); Figure 4). EMNIr20 and EMNIr30 showed similar associations with 90-day and 180-day outcome (Supplementary Figures 3 and 4).
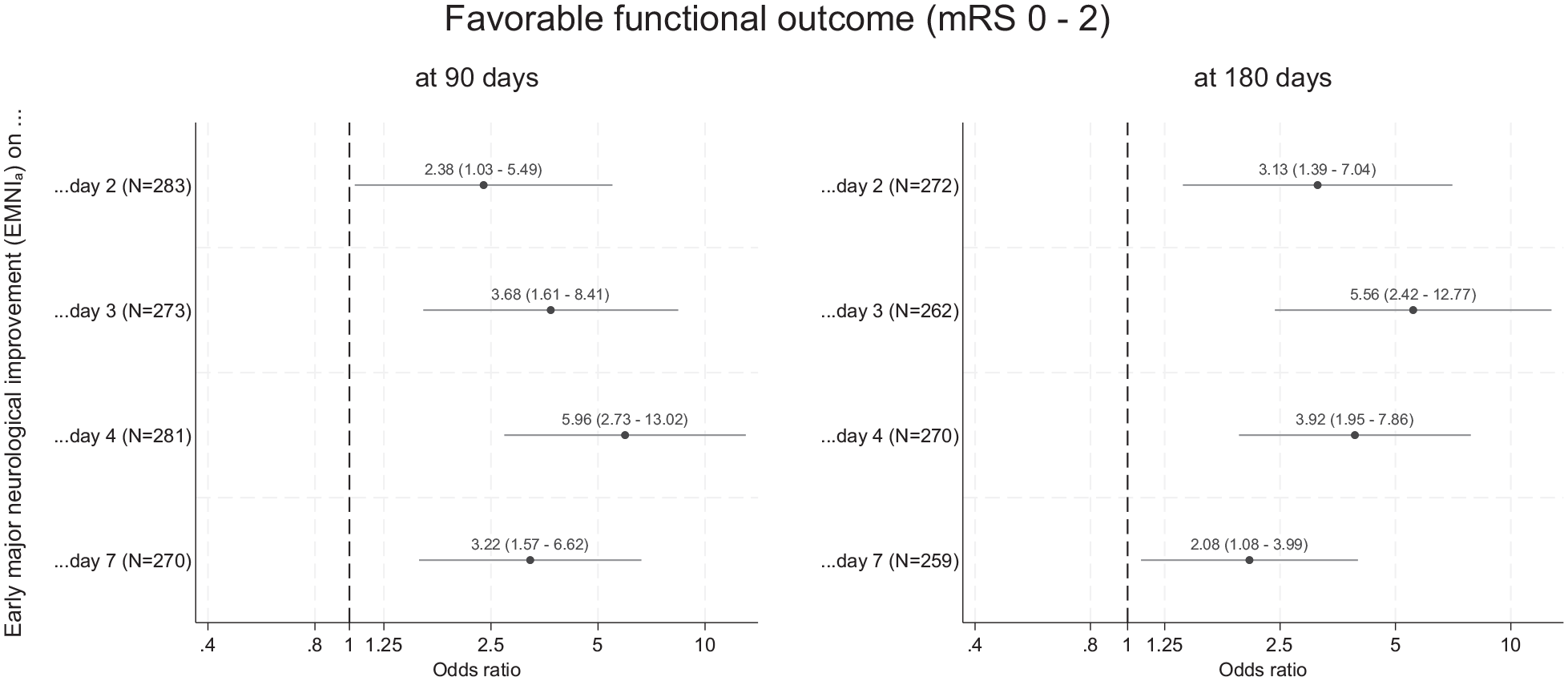
Association of EMNIa at repeated assessments with favorable functional outcome at 90 and 180 days. EMNIa was defined at each assessment timepoint (i.e. day 2, day 3, day 4, or day 7(/discharge)) as an NIHSS score decrement of an absolute ⩾4 points between the respective timepoint and presentation.
Discussion
This post hoc analysis of the i-DEF trial using serial neurological assessments over 7 days after ICH revealed the following key findings: (1) the likelihood of EMNI was higher with deferoxamine; (2) regardless of treatment, the frequency of EMNI was substantial, grew continuously over time, and was higher in non-White participants, those with higher presenting NIHSS score, smaller and non-thalamic hematomas; (3) EMNI was strongly and independently associated with favorable long-term functional outcome up to 180 days.
Adopting an established definition of EMNI as an absolute NIHSS decrement ⩾4 points, an analytic approach that leverages repeated measures, and prespecified methodological principles of the main trial, we found novel evidence supporting an early treatment effect of deferoxamine on neurological recovery within the first 7 days after ICH in the main-effects model of our primary analysis. Reassuringly, this persisted after additional adjustment for several potential confounders in sensitivity analyses, and was maintained when using alternative—arguably more clinically meaningful 21 —definitions of EMNI as relative (⩾20% or ⩾30%) NIHSS decrements, on which deferoxamine showed even larger effect sizes. Taken together, our findings expand on previous observations from i-DEF that the proportion of participants with early good functional outcome at 7 days was higher with deferoxamine 13 and bear biological plausibility. Preclinical data indicate that the beneficial effects of deferoxamine in reducing iron accumulation, oxidative damage, and neuroinflammation are evident within this timeframe in ICH animal models.26–28 These mechanisms might underlie our findings.
We found no clear evidence for treatment-by-time interaction overall. Some of our interaction analyses yielded weak signals for potential effect modification by time, such that deferoxamine might appear to further accelerate the time-dependent increase in the likelihood of EMNI over the first 7 days following ICH. While power to detect a treatment-by-time interaction may have been low, these findings do not contradict our main results, but rather leave open the possibility that deferoxamine’s effects might become even more pronounced with time. This would be compatible with previous observations from i-DEF that deferoxamine seemed to accelerate the trajectory of longer-term recovery as measured on the mRS. 13 Finally, these weak signals for interaction might be related to dose-dependent effects, considering that deferoxamine was applied as an early treatment course over the first 3 days after ICH in i-DEF. On the whole, our study offers new data that strengthen the case for deferoxamine as a promising treatment option in ICH and warrant further study. Our findings also highlight the potential of applying longitudinal analyses of repeated outcome measures to clinical trial datasets, as these may improve power to detect treatment effects.29,30
We provide a novel comprehensive appraisal of the early course of neurological recovery after ICH, which has been largely unexplored. So far, research focused on neurological worsening, which may occur earlier or later within this timeframe, leads to poor outcome, and may represent a treatment target.2–4 However, absence of worsening is not equivalent to improvement, which is worth exploring in its own right. Using a definition of neurological improvement similar to ours but restricted solely between the 24-h and 7-day timepoints, only a single study has recently shown that some degree of subacute improvement is common after ICH. 9 Expanding on these observations with a granular account of the trajectory of early improvement, we show that a substantial proportion of patients may improve as early as the first day after ICH, and that this proportion will continue to increase up to day 7. This, being in the context of a randomized trial of maximally treated patients and regardless of trial-specific treatments, has several implications: (1) it strengthens the case against early prognostication in ICH, 1 and (2) it identifies early improvers as a potentially distinct patient subgroup. Future research should determine whether early improvers might be more responsive to treatments with modest therapeutic effects, which may be masked or negated in non-improving, more severely affected patients. In line with this hypothesis, a previous i-DEF subanalysis suggested that deferoxamine’s effects might be confined to patients with medium-sized hematomas, while those with large hematomas did not benefit from treatment. 31
In that regard, identifying EMNI’s determinants is relevant. We found both smaller hematomas and non-thalamic ones—be it lobar or deep—to carry higher odds of EMNI, which expands on previous data about the adverse prognosis of large hematoma volume 25 and thalamic location. 32 We also found that higher presenting NIHSS score was associated with higher odds of EMNIa. This is unsurprising, considering that the margin for improvement by that definition (using an absolute score decrement) is larger the higher the baseline score is. Notably, this “reverse ceiling effect” disproportionately favoring EMNI in patients with higher baseline stroke severity was not present when exploring alternative definitions of EMNI as relative NIHSS decrements (EMNIr20 and EMNIr30), strengthening the case in favor of relative definitions of EMNI.21,22 Black and Asian participants showed higher odds of EMNI than their White counterparts in our study. This was after adjustment for established outcome predictors, and might therefore reflect true racial differences in ICH, expanding on previous observations.25,33 However, the limited size of these subgroups and substantial uncertainty around the estimates, as reflected in the wide 95% CI, warrant caution, as this may also reflect a chance finding.
Our finding that EMNI—regardless of whether assessed on day 2, 3, 4, or 7 after ICH—is independently associated with favorable long-term functional outcome up to 180 days validates the clinical relevance of EMNI as a phenomenon worthy of study in ICH. Notably, the association with long-term outcomes seemed strongest for EMNI assessed on days 3 and 4 and weaker when assessed on days 2 and 7. This suggests that EMNI may be less clinically significant when assessed too early (i.e. when patients still face a risk of later deterioration) or too late (i.e. when delayed neurological improvement may no longer be sufficient to translate into better functional outcomes). While the reasons for this remain speculative, the timing of clinical improvement has been previously shown to impact long-term outcome after ischemic stroke, 22 and our data indicate that 48–72 h after presentation might be the optimal time to assess EMNI in ICH. Regardless, these data stress the lasting importance of EMNI and advances recent findings from the INTERACT2 trial, where subacute neurological improvement on day 7 was also associated with functional outcome, albeit up to 90 days. 9 Of note, NIHSS-based assessment within 1 week was recently proposed as a surrogate endpoint for clinical trial use to reduce trial duration and costs in the field of ischemic stroke. 34 The benefits of EMNI and shorter time to achieve independence to ICH survivors and their caregivers and healthcare expenses are similarly evident and require further scrutiny.
Our study has several strengths: (1) Although not a prespecified analysis, we applied predetermined methodological aspects of the main trial to explore deferoxamine’s association with outcomes defined using NIHSS scores obtained by certified blinded assessors, thus limiting the risk of spurious findings; (2) several lines of statistical inquiry, different EMNI definitions, and extensive covariate adjustment did not change our main results, underscoring their robustness. We acknowledge the following limitations: (1) the post hoc nature of this analysis and multiple testing limit causal inference and position our findings as hypothesis-generating rather than definitive. (2) The analysis of categorical data as determinants of EMNI is limited by small subgroup sizes, which may reduce statistical power and introduce imprecision and instability into our model-based estimates, warranting caution in their interpretation. (3) Selected using specific eligibility criteria in a phase 2 trial setting, our cohort may be less reflective of the broader ICH patient population and is limited in size. The generalizability/external validity of our findings needs to be assessed in other, larger ICH cohorts.
In conclusion, major neurological improvement starts early and continuously progresses within the first week after ICH in a large proportion of patients and has a strong and lasting association with favorable long-term functional outcome. In this post hoc analysis of the i-DEF trial, treatment with deferoxamine following ICH seemed to enhance EMNI.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930251348088 – Supplemental material for Early neurological improvement with deferoxamine after intracerebral hemorrhage: A post hoc analysis of the i-DEF trial
Supplemental material, sj-pdf-1-wso-10.1177_17474930251348088 for Early neurological improvement with deferoxamine after intracerebral hemorrhage: A post hoc analysis of the i-DEF trial by Alexandros A Polymeris, Vasileios-Arsenios Lioutas, Lydia D Foster, Diego Incontri, Elizabeth C Heistand, Juliette Marchal, Alexa Lazar, Urs Fischer, Stefan T Engelter, David J Seiffge, Sharon D Yeatts and Magdy H Selim in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: i-DEF was funded by NIH/NINDS (U01NS074425). Dr Polymeris was funded by Swiss National Science Foundation (SNSF; P500PM_221974) and Freiwillige-Akademische-Gesellschaft-Basel.
Informed consent
All i-DEF participants (or a legally authorized representative) provided written informed consent.
Ethical approval
The study was approved by the Local Ethics Committee and Institutional Review Board at each participating center.
Data availability
The data that support these findings may be provided to qualified investigators upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
