Abstract
Background:
Distal medium vessel occlusions (MeVOs) account for an estimated 25% to 40% of all acute ischemic strokes. Emerging evidence from non-randomized trials suggest that endovascular thrombectomy (EVT) can achieve high rates of successful reperfusion in MeVO strokes, with a safety profile comparable to EVT for proximal arterial occlusions. These findings underscore the need for a prospective randomized clinical trial to evaluate the safety and efficacy of EVT for MeVO stroke.
Objective:
This trial aims to evaluate the safety and efficacy of EVT for MeVO stroke.
Methods and design:
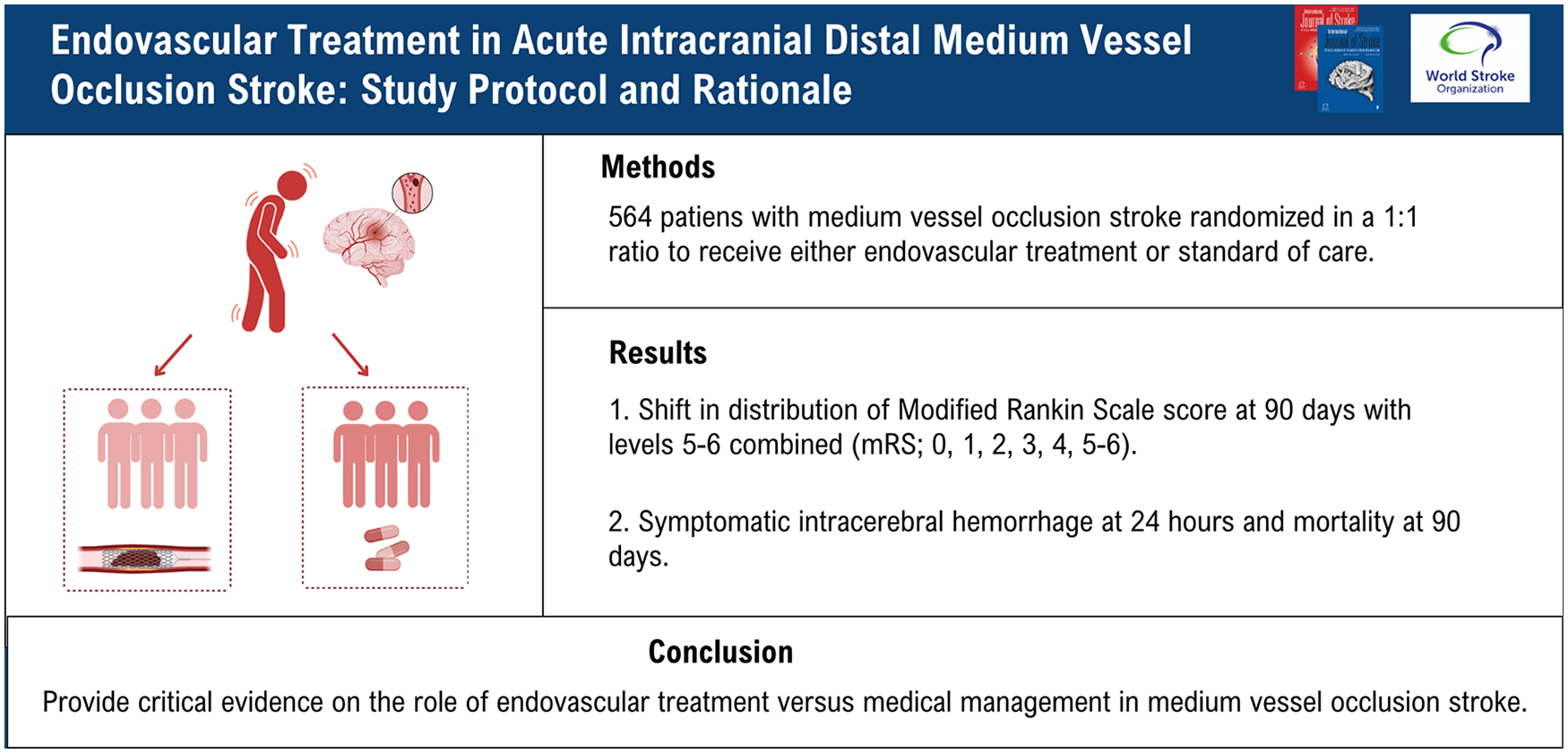
Endovascular treatment in acute intracranial distal medium vessel occlusion stroke (ORIENTAL-MeVO) is an investigator-initiated, multicenter, prospective, randomized clinical trial with open-label treatment and blinded endpoint assessment (PROBE). Up to 564 eligible patients will be consecutively randomized in a 1:1 ratio to receive either EVT or standard of care over a period of 2 years in over 50 comprehensive stroke centers in China.
Outcomes:
The primary outcome is a shift in the distribution of the modified Rankin Scale (mRS) at day 90s with levels 5–6 combined (mRS = 0, 1, 2, 3, 4, 5–6). Primary safety endpoints include symptomatic intracerebral hemorrhage at 24 h and mortality at 90 days.
Trial registration:
ClinicalTrials.gov NCT06146790.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
