Abstract
Background:
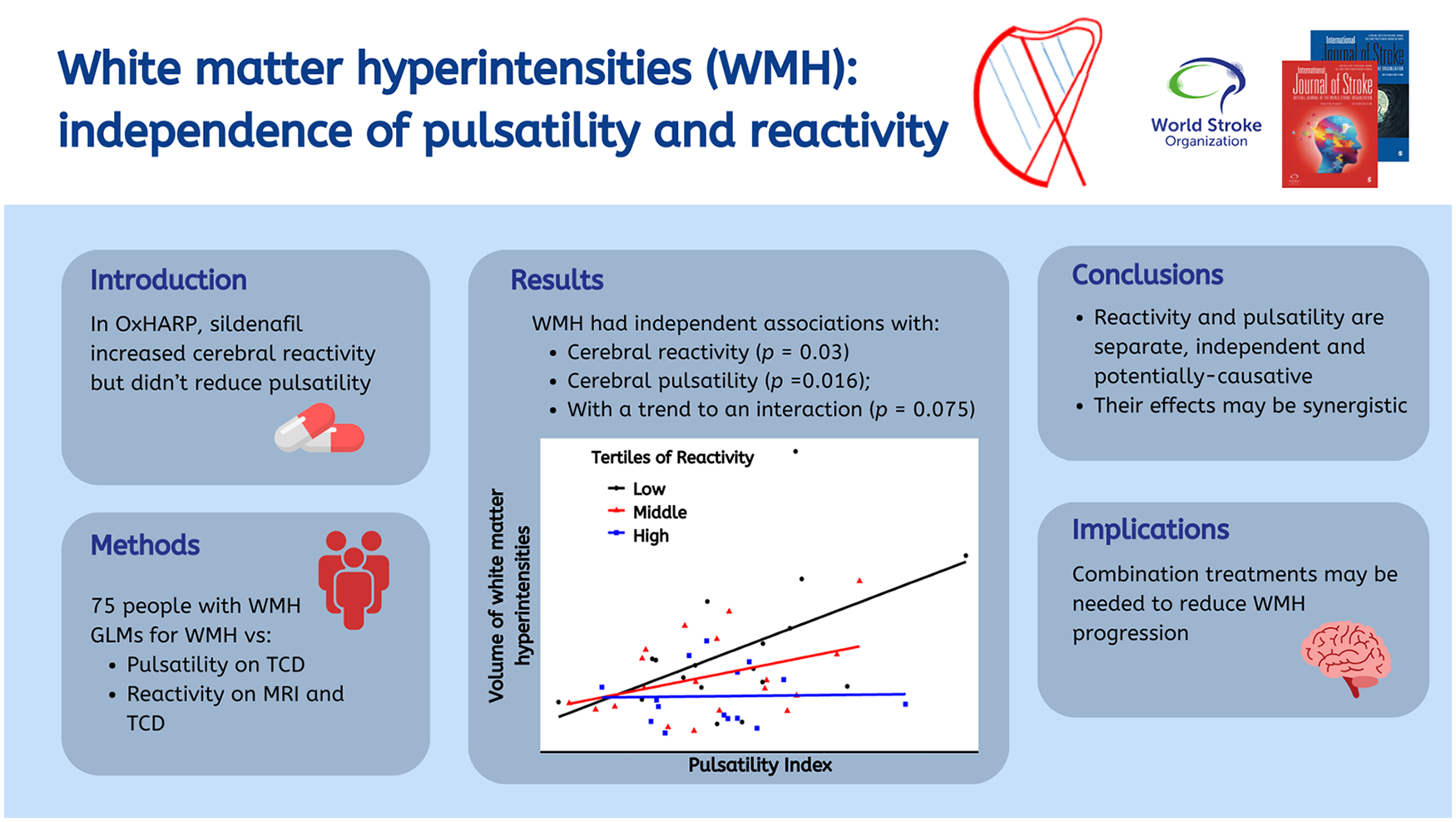
In the Oxford Haemodynamic Adaptation to Reduce Pulsatility trial (OxHARP), sildenafil increased cerebrovascular reactivity but did not reduce cerebral pulsatility, a marker of vascular aging. This analysis of OxHARP tested whether these potentially causative mechanisms were independently associated with the severity of white matter hyperintensities (WMHs).
Aims:
The aims were to determine independence of the relationship between severity of WMHs with both cerebral pulsatility and cerebrovascular reactivity in the same population.
Methods:
OxHARP was a double-blind, randomized, placebo-controlled, crossover trial of phosphodiesterase inhibitors in patients with mild-to-moderate WMH and previous minor cerebrovascular events. It determined effects on cerebrovascular pulsatility and reactivity on transcranial ultrasound and reactivity on magnetic resonance imaging (MRI). Associations were determined between baseline ultrasound measures, and averaged MRI measures across follow-up, with the severity of WMH on clinical imaging (Fazekas or modified Blennow scores) and WMH volume in the MRI substudy, by ordinal and linear regression.
Results:
In 75/75 patients (median 70 years, 78% male), cerebral pulsatility was associated with age (p < 0.001) whereas reactivity on ultrasound was not (p = 0.29). Severity of WMH in all participants was independently associated with decreased cerebrovascular reactivity and increased cerebral pulsatility (pulsatility p = 0.016; reactivity p = 0.03), with a trend to a synergistic interaction (p = 0.075). Reactivity on ultrasound was still associated with WMH after further adjustment for age (p = 0.017), but pulsatility was not (p = 0.31). Volume of WMH in the MRI substudy was also independently associated with both markers on ultrasound (pulsatility p = 0.005; reactivity p = 0.029) and was associated with reduced cerebrovascular reactivity within WMH on MRI (p < 0.0001).
Conclusion:
WMHs are independently associated with cerebral pulsatility and reactivity, representing complementary potential disease mechanisms and treatment targets.
Trial Registration:
clinicaltrials.org: https://classic.clinicaltrials.gov/ct2/show/NCT03855332.
Introduction
Cerebral small vessel disease causes 30% of ischemic stroke, 80% of hemorrhagic stroke, and 40% of vascular dementia. However, the pathophysiology of cerebral small vessel disease (cSVD) remains uncertain and thus has no specific treatments. 1 cSVD is most strongly associated with age, midlife hypertension, and subsequent vascular aging.2,3 This increases aortic stiffness 4 which causes greater transmission of aortic pulse pressure to the brain. This increases cerebral arterial pulsatility,4–6 the difference between the peak and trough cerebral blood flow velocity with each heart beat, divided by the mean flow. Small vessel disease (SVD) is also associated with cerebrovascular endothelial dysfunction7,8 commonly demonstrated by a smaller increase in cerebral blood flow induced by inhalation of carbon dioxide (CVR, cerebrovascular reactivity). Cerebral blood flow can either be assessed as flow velocity in large intracranial vessels with transcranial Doppler ultrasound (TCD), or through blood flow sensitive magnetic resonance imaging (MRI) sequences (BOLD, pcASL). However, previous cohorts have not assessed the association between cSVD, cerebral pulsatility, and cerebrovascular endothelial function in the same cohort.
Targeting cerebrovascular endothelial dysfunction with vasodilatory drugs is currently the most promising intervention. In the LACI-2 feasibility trial, isosorbide mononitrate (ISMN) 9 was associated with reduced cognitive decline, 10 but cerebral pulsatility and CVR were not measured. The recent OxHARP 11 trial targeted the same pathway, testing the effects of phosphodiesterase (PDE) inhibitors with sildenafil (a PDE 5 inhibitor) or cilostazol (a PDE 3 inhibitor). OxHARP demonstrated no reduction in cerebral pulsatility with either drug, but sildenafil resulted in a significant increase in cerebral perfusion and CVR, and a reduction in cerebrovascular resistance. This discordant effect on cerebral pulsatility compared to endothelium-dependent cerebrovascular function could imply either that these are independent mechanisms; that cSVD results from an interaction between these mechanisms; or that sildenafil acts downstream of common underlying processes. The relationship between these mechanisms with the severity of cSVD within the same cohort may discriminate their potential role in driving cSVD and guide the development of treatment strategies.
Aims
The aims were to determine independence of the relationship between severity of white matter hyperintensities (WMHs) with both cerebral pulsatility and CVR in the OxHARP trial.
Methods
Study design
OxHARP was a double-blind, randomized, placebo-controlled, crossover phase 2 trial with physiological endpoints, 12 that ran from 11 July 2019 to 6 December 2022, paused due to the COVID-19 pandemic from March to September 2020. Participants received placebo or sildenafil 25 mg thrice daily or cilostazol 50 mg twice daily, titrated to 50 or 100 mg, respectively, after 1 week, for 3 weeks of treatment in total. At screening and on the final day of treatment, participants underwent a physiological assessment to determine the primary outcome of middle cerebral artery pulsatility (MCA-PI) and the secondary outcome of CVR to carbon dioxide (CO2) on TCD. 12 At trial inception, 30 patients were intended to be recruited to undergo an MRI assessment of CVR to 6% CO2, while on sildenafil and placebo. This was amended during the trial to include up to 30 further patients to undergo MRI on all three treatments.
Study population
Patients were eligible if they had a history of a stroke or probable transient ischemic attack (TIA) more than 1 month previously, of cryptogenic or lacunar etiology, with mild-to-moderate WMH evident on their most recent clinical brain imaging (by Fazekas score on MRI or modified Blennow score on computed tomography (CT)). The full inclusion criteria have been reported previously. 12
Physiological assessment
At the baseline screening visit and on the final day of each trial medication, a physiological assessment was performed in a temperature-controlled laboratory (21-23 C) after 15 min of supine rest and 30 min after administration of the trial medication in the clinic. Middle cerebral artery flow velocity was assessed with 2 MHz probes (DWL), as the highest velocity waveform between 50 and 55 mm depth, including peak systolic velocity (PSV), mean flow velocity (MFV), and end-diastolic velocity (EDV). The primary outcome (MCA-PI) was Gosling’s Pulsatility Index ((PSV − EDV)/MFV). To assess CVR-TCD, bilateral monitoring of the middle cerebral artery was established where feasible, with two probes held by a DiaMon headset, acquired over 10 min with concurrent electrocardiogram (ECG), non-invasive blood pressure monitoring calibrated to an oscillometric brachial reading (FMS, Finometer Midi) and end-tidal carbon dioxide monitoring (etCO2, AD Instruments Gas Analyser ML206). CVR-TCD was determined during 2-min alternating periods of inhalation of medical air, 4% and then 6% CO2, delivered via a respiratory circuit with a well-sealed, non-invasive ventilation mask. Aortic blood pressure was determined by radial artery tonometry and aortic stiffness as carotid-femoral pulse wave velocity (PWV, Sphygmocor, At-Cor Medical, Sydney, Australia).
For each assessment, two TCD recordings were reviewed by 2 blinded reviewers (A.J.S.W, J.T.) to estimate PSV, EDV, and MFV from the average of three heart beats from each recording to derive MCA-PI. Where both sides were of a similar mean velocity (within 10%) and quality upon blinded review, an average of the sides was taken, otherwise the higher quality, higher velocity recording was used. Disagreements between reviewers were resolved by panel discussion. For CVR-TCD, the primary analysis side was selected in the same manner (A.J.S.W., O.L.). CVR-TCD was defined as the beta-coefficient from a linear model between MFV and etCO2 during inhalation of medical air, 4% and 6% CO2, after phase delay correction by cross-correlation and piecewise cubic Hermite interpolation.
MRI imaging
MRI was performed on a 3-Tesla Siemens Prisma scanner (Erlangen, Germany, at the Oxford Wellcome Centre for Integrative Neuroimaging), as reported previously. 11 The principal CVR-MRI outcome was determined as the percentage change in BOLD signal (multiband 6, repetition time (TR) 800 ms, time to echo (TE) 30 ms, 2.4 mm voxels) per mm Hg end-tidal CO2 during inhalation of two cycles of 2 min alternating periods of air and 6% CO2. Throughout imaging, participants had continuous non-invasive monitoring of etCO2 (AD Instruments Gas Analyzer ML206), respiratory motion, oxygen saturations, and blood pressure. 13 Derivation of CVR in WMH and normal-appearing white matter (NAWM) from BOLD-CVR MRI were determined by general linear models after correction for phase delay, as previously reported. 11 For comparison with baseline variables, the average CVR across all MRI scans for each individual was used.
WMH volume on MRI was quantified on OxHARP trial scans from T1 and fluid-attenuated inversion recovery (FLAIR) images (BIANCA, FSL), and from both OxHARP and clinical MRI scans by the Fazekas score, by two independent blinded reviewers (C.S., A.J.S.W.). Severity of WMH on CT scans was determined by the modified Blennow score. 14 A blinded grading of WMH severity for all participants was performed using the best available imaging (1—OxHARP FLAIR MRI; 2—most recent clinical MRI FLAIR; 3—clinical T2; 4—most recent CT brain), prior to any statistical analysis, into mild (equivalent to Fazekas 1–2), moderate (equivalent to Fazekas 3), and moderately-severe (equivalent to Fazekas 4–6) groups. Where there was discordance between reviewers, a panel decision determined the final group allocation on the extent of WMH with the final responsibility with the chief investigator (A.J.S.W.).
Statistical analysis
Differences in frequency or mean and standard deviation (SD) of demographic characteristics were determined between trial subgroups by chi-square and analysis of variance (ANOVA) tests. Relationships between demographic measures and baseline measures of MCA-PI and CVR-TCD were determined by general linear models, with and without adjustment for age, sex, and cardiovascular risk factors (history of diabetes, hypertension, smoking, or dyslipidemia). Associations with concurrent physiological indices (e.g. aortic stiffness, blood pressure) were determined by mixed-effects linear models across all MRI scans at all visits, including subject as a random effects variable and allowing for repeated measures. Associations with the severity of WMH classified as mild, moderate, or moderately-severe were determined by ordinal regression, and with volume of WMH by general linear models, with and without interaction terms between MCA-PI and CVR-TCD, CVR-WMH, or CVR-NAWM. WMH volume or CVR were divided into tertiles to illustrate any interactions.
All participants gave informed written consent. OxHARP is sponsored by the University of Oxford, approved by the UK Health Research Authority and South Central—Oxford C Research Ethics Committee (19/SC/0022), and is registered with ClinicalTrials.org (NCT03855332).
Analyses were performed in FSL, RStudio (version 2024.09.0, glm, lme4, lrm) and MATLAB r2018.
Results
In total, 75/75 patients had valid baseline TCD measurements (59 men, 78%). There were no significant differences between patients recruited to each arm of the study, and the distributions of age, hypertension, diabetes, and smoking were consistent with previous physiological studies of TIA and minor stroke (Table 1).15–17 Approximately half of patients had mild WMH on CT or MRI, and the remaining patients had moderate or moderately-severe WMH. A total of 15 patients had three MRIs, 32 patients had two MRIs, and nine patients had one MRI.
Population in the OxHARP trial.
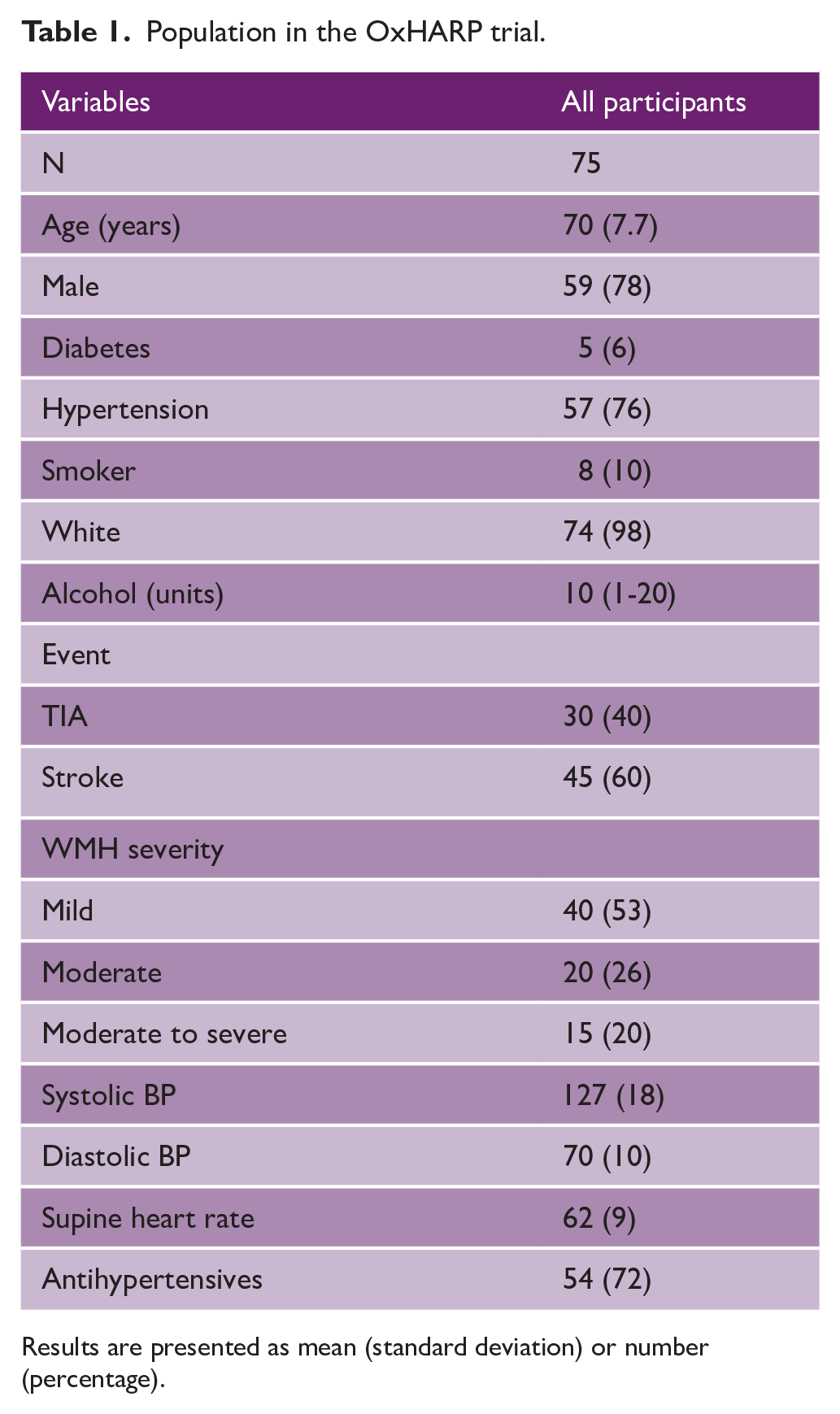
Results are presented as mean (standard deviation) or number (percentage).
Cerebral pulsatility and CVR had different patterns of association with demographic factors. MCA-PI on TCD was strongly associated with increasing age, a history of stroke, and concurrent elevated systolic blood pressure (SBP; b = 0.013, p < 0.0001, Table 2), before and after adjustment for age, sex, and cardiovascular risk factors (Supplemental Table 1). In contrast, CVR-TCD was not associated with increasing age but was lower in men than women. CVR-MRI in NAWM was associated with increased alcohol intake and variability in blood pressure, but not with age, while CVR-WMH was associated with age (Supplemental Table 1).
Associations between clinical characteristics and middle cerebral artery pulsatility (PI) and cerebrovascular reactivity (CVR) on ultrasound (CVR-TCD) or in normal-appearing white matter (NAWM) or white matter hyperintensities (WMHs).
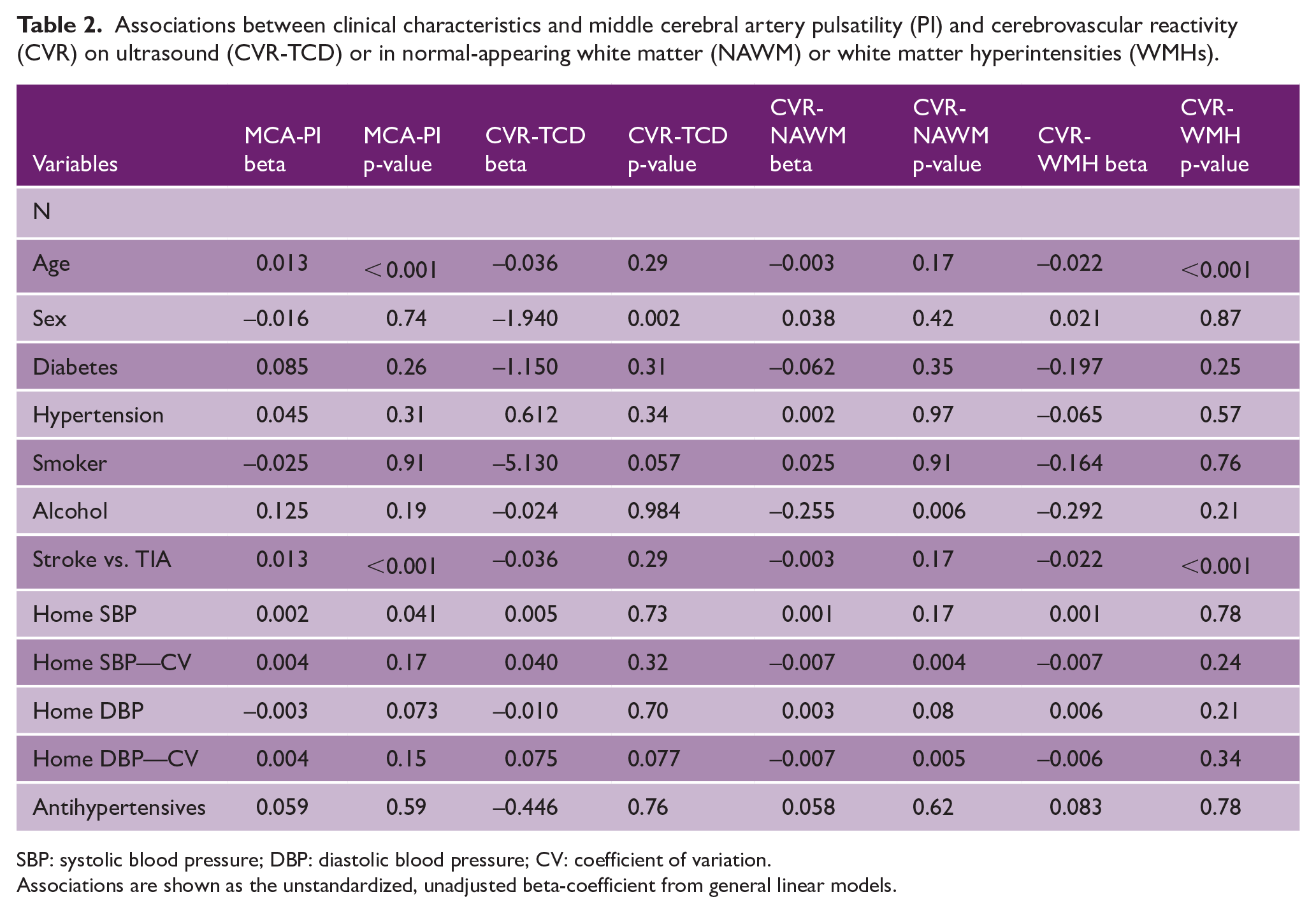
SBP: systolic blood pressure; DBP: diastolic blood pressure; CV: coefficient of variation.
Associations are shown as the unstandardized, unadjusted beta-coefficient from general linear models.
There were also discordant associations with systemic blood pressure. Increased SBP, reduced diastolic blood pressure (DBP), and increased PWV (Table 3, Supplemental Table 2) were associated with increased MCA-PI, while SBP was particularly associated with reduced PSV and EDV (Supplemental Table 3). However, blood pressure was not associated with any measure of CVR, although increased PWV was associated with a reduced CVR-TCD and CVR-MRI in both NAWM and WMH (Supplemental Table 2 and Table 3). CVR-MRI in deep gray matter was associated with a history of hypertension but no other clinical risk factors. In contrast, CVR-MRI in WMH was strongly associated with increasing volume of WMH, hypertension, increased MCA-PI, and age, whereas CVR-MRI in NAWM was only associated with MCA-PI (Supplemental Table 4).
Associations between systemic physiological measures and middle cerebral artery pulsatility (MCA-PI) and cerebrovascular reactivity (CVR) on ultrasound (TCD), in normal-appearing white matter (NAWM) or in white matter hyperintensities (WMHs).
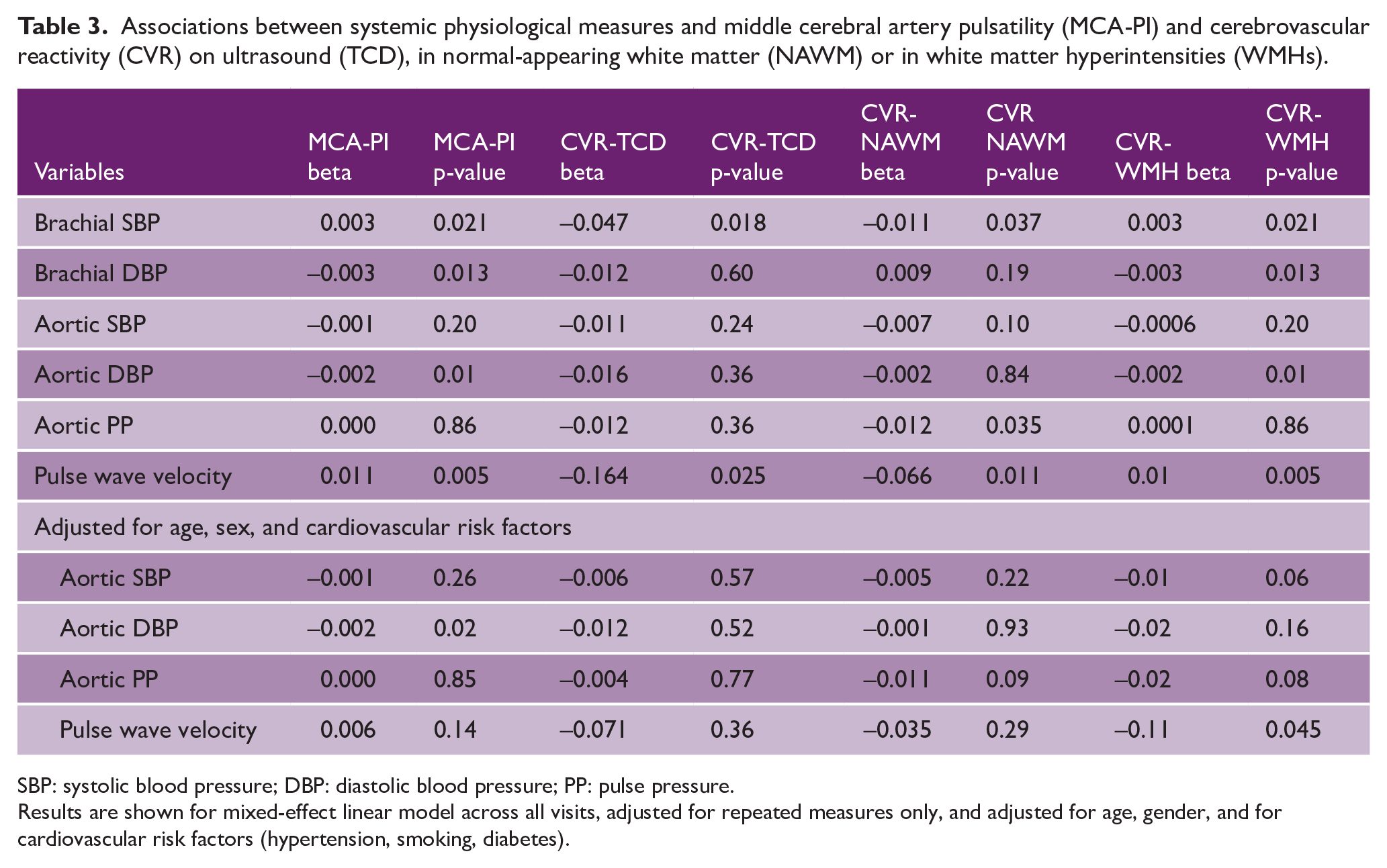
SBP: systolic blood pressure; DBP: diastolic blood pressure; PP: pulse pressure.
Results are shown for mixed-effect linear model across all visits, adjusted for repeated measures only, and adjusted for age, gender, and for cardiovascular risk factors (hypertension, smoking, diabetes).
In contrast, both markers were associated with WMH. Severity of WMH was independently associated with increased MCA-PI (odds ratio (OR) = 1.89, p = 0.016), decreased absolute blood flow velocities (Supplemental Figure 1), reduced CVR-TCD (OR = 0.57, p = 0.034), and CVR-MRI in WMH (OR = 0.24, p = 0.0004). There were similar associations with WMH volume in the MRI substudy, before and after adjustment for MCA-PI, CVR-TCD, age, and sex (Figure 1 and Table 4). Both MCA-PI and CVR-TCD were similarly associated with the severity of periventricular or deep WMH (Supplemental Figure 2).
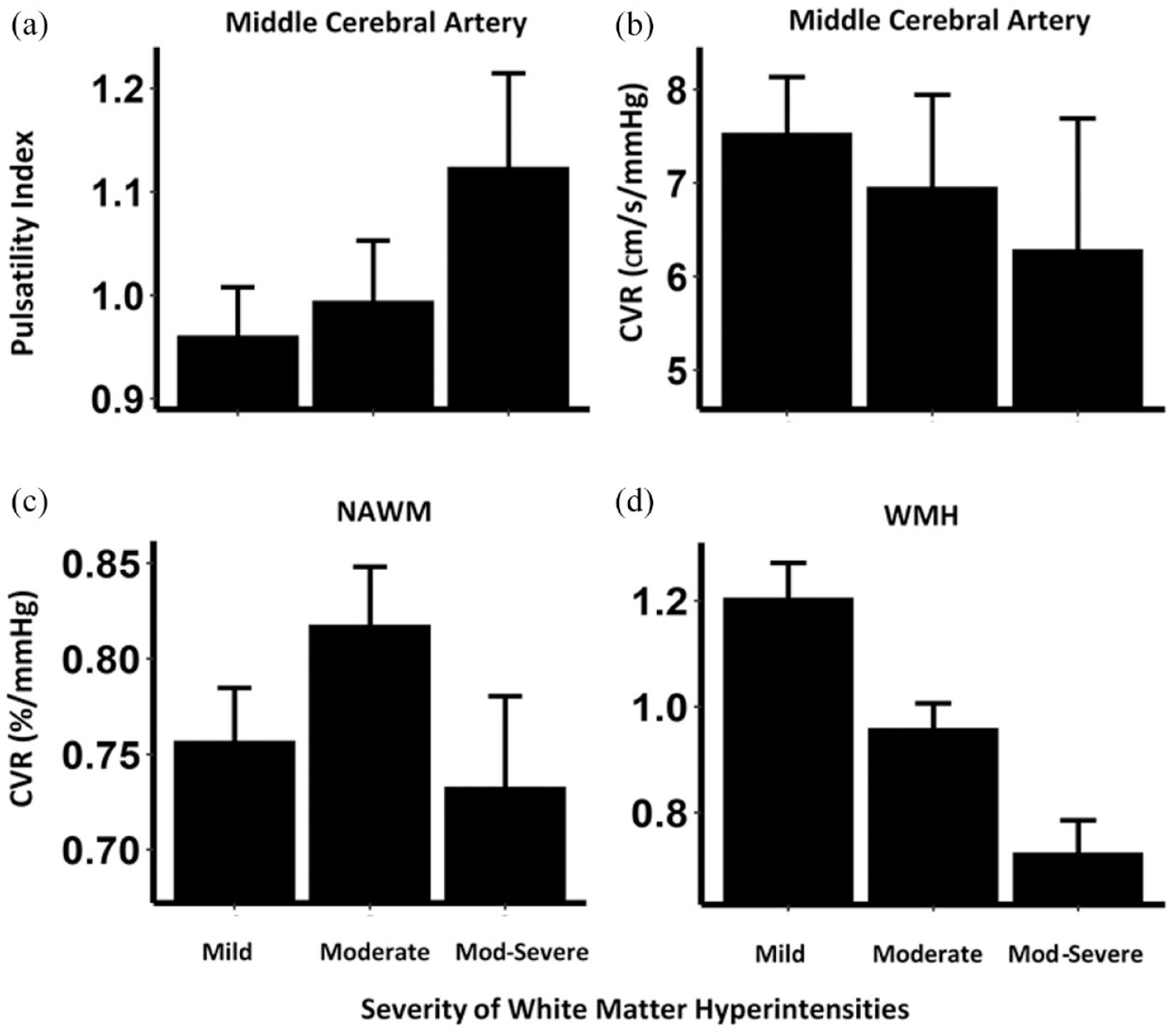
Relationship between pulsatility index (MCA-PI), white matter hyperintensities (WMHs), and cerebrovascular reactivity (CVR) on TCD and MRI. Mean values with 95% confidence intervals are shown across categories of WMH severity on CT or MRI for all patients for Gosling’s Pulsatility Index (a), cerebrovascular reactivity (CVR) on TCD (b), CVR-MRI in normal-appearing white matter (NAWM, c) and in WMHs (d).
Standardized associations between cerebral pulsatility (MCA-PI) or cerebrovascular reactivity (CVR) on transcranial Doppler ultrasound (TCD) or MRI, with the severity of small vessel disease.
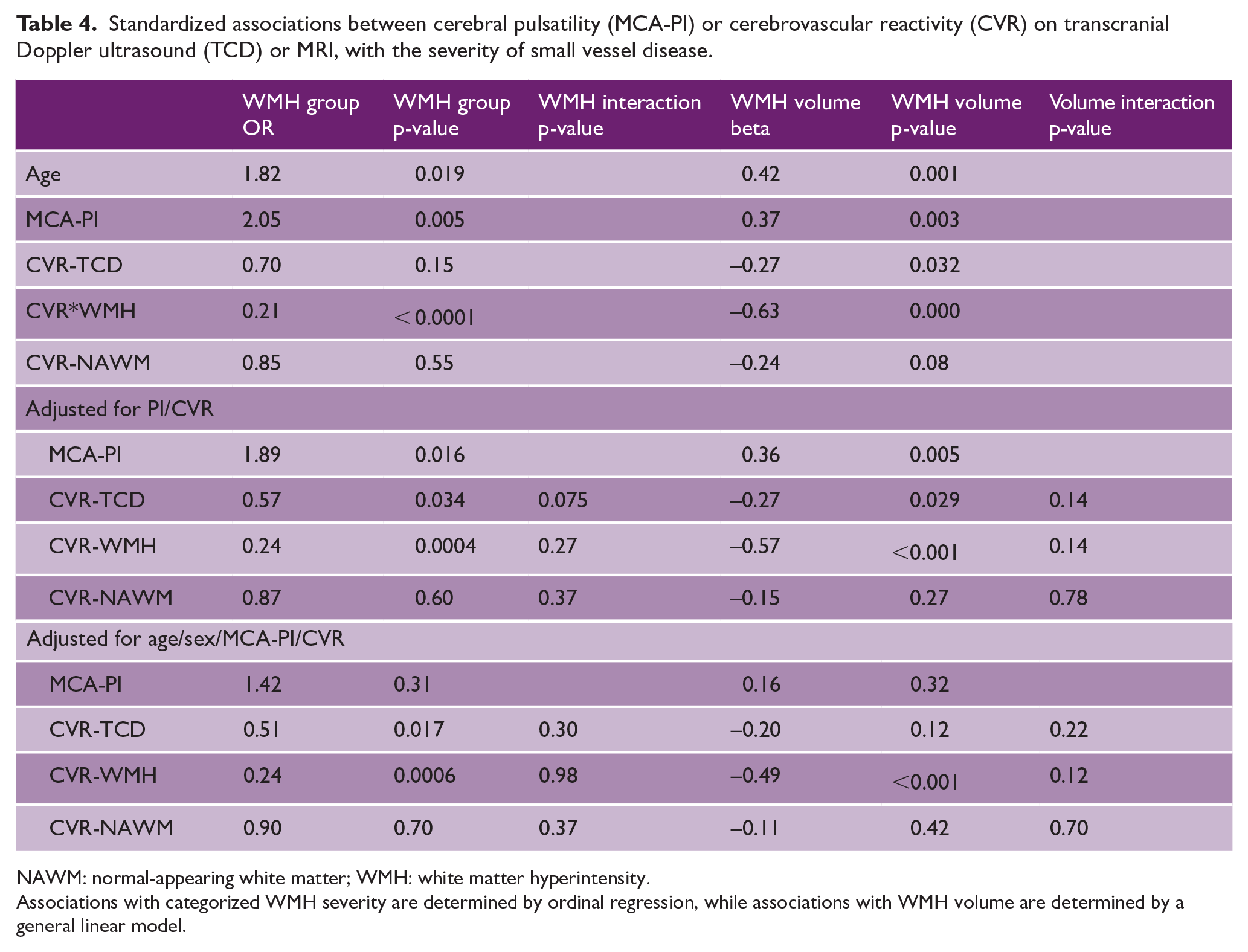
NAWM: normal-appearing white matter; WMH: white matter hyperintensity.
Associations with categorized WMH severity are determined by ordinal regression, while associations with WMH volume are determined by a general linear model.
Finally, there was no direct association between CVR-TCD and MCA-PI, for all patients or when stratified by severity of WMH (p = 0.41, Figure 2). Both MCA-PI and CVR-TCD were independently associated with WMH volume, but there was only a non-significant trend to an interaction between them (p = 0.075, Table 4), as the study was not powered to test for this. However, the direction of this trend was consistent for CVR-TCD and CVR-MRI in WMH, with MCA-PI associated with WMH volume predominantly in patients with reduced CVR-TCD (Figure 2 and Supplemental Figure 3).
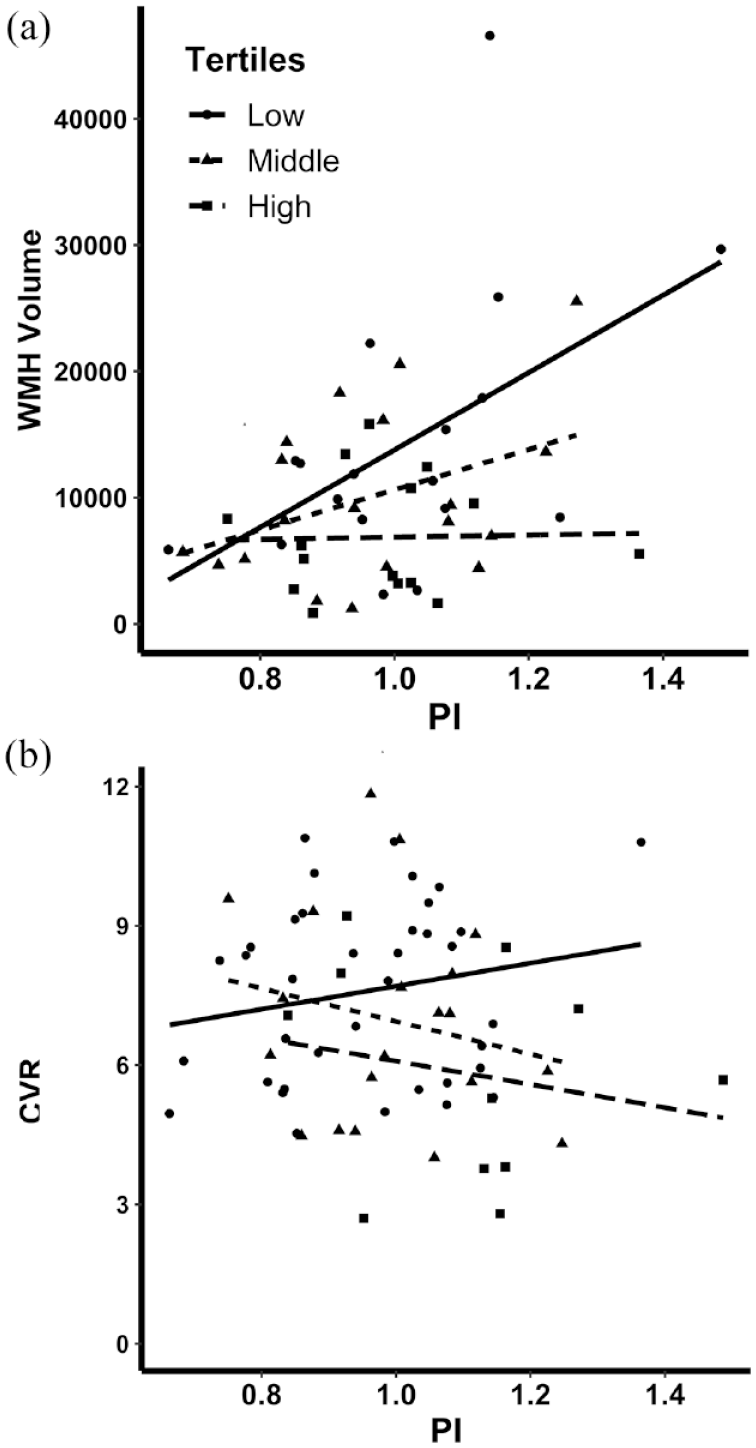
Interactions between pulsatility index, white matter hyperintensities (WMHs), and cerebrovascular reactivity (CVR) on TCD. In participants undergoing MRI during the trial, the linear relationship between cerebral pulsatility (PI) and WMH volume (a) is stratified by tertiles of cerebrovascular reactivity on TCD, from the lowest tertile (solid), to the middle (short dash), and to the highest CVR (long dash). The linear relationship between MCA-PI and CVR (b) is stratified by tertile of WMH.
Discussion
In 75 patients, mild to moderately-severe WMH were associated with increased MCA pulsatility and reduced CVR-TCD. However, there was no direct association between CVR-TCD and MCA-PI. They were independently associated with the severity of WMH, and there was a trend toward an interaction such that MCA-PI was only associated with WMH severity in patients with impaired CVR-TCD. Although not significant, if confirmed, this would suggest a synergistic relationship between these independent mechanisms, consistent with a mechanism whereby cSVD results from impaired ability of the brain to adapt to abnormal systemic hemodynamics. This is consistent with the differential effect of sildenafil on cerebrovascular hemodynamics in OxHARP.
The association between WMH and cerebral arterial pulsatility is consistent with the relationship between cSVD and vascular aging in previous cohorts.3,18–23 In contrast, only a limited number of studies have reported the association between WMH and CVR.24–26 OxHARP demonstrated that large vessel CVR-TCD in the MCA is related to impaired CVR-WMH more than CVR-NAWM. This may reflect either impaired endothelial function within both the MCA and WMH, with relative preservation within NAWM, or greater transmission of the altered endothelial response in large vessels to WMH, with reduced local compensation within WMH. Therefore, although impaired CVR within NAWM may be a preferential therapeutic target to prevent development of WMH, impaired CVR within WMH may still provide a sensitive marker of treatment effects on reactivity more widely within the cerebrovasculature.
The independent and possibly synergistic relationship between MCA-PI and CVR-WMH implies both systemic hemodynamic dysfunction, and impaired central compensatory mechanisms may be necessary for the presence of WMH. If so, effective prevention of WMH may be achieved by improving either systemic hemodynamics or compensatory mechanisms by targeting cerebrovascular endothelial dysfunction,1,10,13,27 but that synergistic benefits may be gained by targeting both mechanisms. To achieve reduced arterial pulsatility requires a greater reduction in systolic flow than diastolic flow. This may theoretically be achieved by vasodilators reducing peripheral wave reflection 28 or improving the Windkessel function of the aorta to reduce aortic pulsatility, but no such effect was seen in OxHARP. Alternatively, it has recently been demonstrated that pulsatility is strongly dependent upon heart rate, 17 such that drugs that increase heart rate may reduce pulsatility. 29
To improve CVR, the most promising agents are drugs that target the cerebrovascular endothelial NO-cGMP vasodilatory pathway. CVR on MRI was improved with cilostazol and isosorbide mononitrate in LACI-1, while (ISMN) was associated with potential improvements in cognition in LACI-2. 10 However, the mechanism of effect of these drugs on other mechanisms such as cerebral pulsatility or cerebral blood flow was not available in the clinically focused LACI-2 trial. The effects of sildenafil on both cerebral perfusion and cerebrovascular function in OxHARP are consistent with a predominant cerebral vasodilatory effect of phosphodiesterase type 5 (PDE5) inhibitions.
This study has limitations. First, it was powered to test the effects of the medications rather than to formally test for interactions between the predictors. Second, not all patients had an MRI scan as part of the study, as the primary and secondary outcomes were TCD based. This limited the power of potentially more sensitive MRI-based analyses. Furthermore, a significant proportion of patients had WMH estimated from previous clinical imaging. Third, the study only included patients with mild-to-moderate WMH and may not be applicable to patients with more severe disease. Fourth, the principal analysis used mixed-effects linear modeling across all visits, although the number of visits differed between patients. This method increases the sensitivity of the analysis but could have been biased by differential drop-out of some patients. However, the results were similar when only including baseline measurements. Finally, the participants were part of a clinical trial with greater ascertainment bias than in observational studies and a high proportion of men and very few participants of non-white ethnicities, that could limit external validity given that sex was associated with CVR-TCD. However, the study also has some unique strengths, including the concurrent measurement of systemic blood pressure and autonomic function, aortic blood pressure, large artery blood flow, and MRI-based measures of parenchymal blood flow.
These results, and the primary results of OxHARP, demonstrate the need for further research to determine the potential of PDE5 inhibition in cSVD. First, the mechanistic relationships and effects of interventions on further physiological outcomes need assessing in OxHARP, including autonomic function, cerebral autoregulation, and peripheral vascular reactivity. It is also necessary to validate these observational findings in larger cohorts with greater power to confirm the interaction between CVR and pulsatility and critically to identify the optimal physiological, short-term markers that both predict response to an intervention and predict disease progression and clinical outcomes. Furthermore, these cross-sectional associations, and the treatment effect of sildenafil in OxHARP, do not prove that either MCA-PI or CVR-TCD are causally associated with progression of cSVD, which needs testing in longer interventional trials. However, this analysis confirms that separate interventions are likely to be necessary to target MCA-PI and CVR, with sildenafil being a feasible intervention only for the latter. Finally, clinical trials are needed with clinical outcomes. Any such trials should also determine the utility of different physiological markers to predict both future clinical events and the response to treatment.
Conclusion
MCA pulsatility and CVR were independently associated with WMHs, the most prevalent manifestation of small vessel disease. However, pulsatility covaried with markers of vascular aging while CVR predominantly reflected disease severity, with a trend to a synergistic interaction, such that dysfunction in both systemic cardiovascular function and cerebrovascular compensation was necessary for an association with more severe WMH. This is consistent with the hypothesis that cSVD is driven by the interaction between systemic cardiovascular dysfunction and impaired compensatory mechanisms.
Supplemental Material
sj-pdf-2-wso-10.1177_17474930241306987 – Supplemental material for White matter hyperintensities are independently associated with systemic vascular aging and cerebrovascular dysfunction
Supplemental material, sj-pdf-2-wso-10.1177_17474930241306987 for White matter hyperintensities are independently associated with systemic vascular aging and cerebrovascular dysfunction by Alastair JS Webb, Karolina Feakins, Amy Lawson, Catriona Stewart, James Thomas and Osian Llwyd in International Journal of Stroke
Supplemental Material
sj-tif-1-wso-10.1177_17474930241306987 – Supplemental material for White matter hyperintensities are independently associated with systemic vascular aging and cerebrovascular dysfunction
Supplemental material, sj-tif-1-wso-10.1177_17474930241306987 for White matter hyperintensities are independently associated with systemic vascular aging and cerebrovascular dysfunction by Alastair JS Webb, Karolina Feakins, Amy Lawson, Catriona Stewart, James Thomas and Osian Llwyd in International Journal of Stroke
Footnotes
Acknowledgements
The authors acknowledge the contribution of the patients who participated in the trial and the support of the staff at the Wolfson Centre for Prevention of Stroke and Dementia.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.J.S.W. has received consulting fees from Woolsey Pharmaceuticals. K.F., A.L., O.L., C.S., and J.T. have no relevant conflicts of interests to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: K.F. and A.J.S.W. were funded by an Alzheimer’s Society grant (450-AS-PG-18-018). O.L. was funded by a Stroke Association research fellowship (SA PDF 21\100029). The OxHARP trial was funded by AJSW’s Wellcome Trust Clinical Research Development Fellowship (206589/Z/17/Z).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Proposals should be directed to
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
