Abstract
Background:
Three large randomized controlled trials of fluoxetine for stroke recovery have been performed. We performed an individual patient data meta-analysis (IPDM) on the combined data.
Methods:
Fixed effects meta-analyses were performed on the combined data set, for the primary outcome (modified Rankin scale (mRS) at 6 months), and secondary outcomes common to the individual trials. As a sensitivity analysis, summary statistics from each trial were created and combined.
Findings:
The three trials recruited a combined total of 5907 people (mean age 69.5 years (SD 12.3), 2256 (38%) females, 2–15 days post-stroke) from Australia, New Zealand, United Kingdom, Sweden, and Vietnam; and randomized them to fluoxetine 20 mg daily or matching placebo for 6 months. Data on 5833 (98.75%) were available at 6 months. The adjusted ordinal comparison of mRS was similar in the two groups (common OR 0.96, 95% CI 0.87 to 1.05, p = 0.37). There were no statistically significant interactions between the minimization variables (baseline probability of being alive and independent at 6 months, time to treatment, motor deficit, or aphasia) and pre-specified subgroups (including age, pathological type, inability to assess mood, proxy or patient consent, baseline depression, country). Fluoxetine increased seizure risk (2.64% vs 1.8%, p = 0.03), falls with injury (6.26% vs 4.51%, p = 0.03), fractures (3.15% vs 1.39%, p < 0.0001) and hyponatremia (1.22% vs 0.61%, p = 0.01) but reduced new depression (10.05% vs 13.42%, p < 0.0001). At 12 months, there was no difference in adjusted mRS (n = 5760; common OR 0.98, 95% CI 0.89 to 1.07). Sensitivity analyses gave the same results.
Interpretation:
Fluoxetine 20 mg daily for 6 months did not improve functional recovery. It increased seizures, falls with injury, and bone fractures but reduced depression frequency at 6 months.
Background
Stroke is a leading cause of adult disability globally, and new treatments are needed to reduce stroke-related disability.1,2 Following the encouraging results of the FLAME trial published in 2011, 3 we collaboratively designed three large trials to test the hypothesis that fluoxetine 20 mg given daily after stroke for 6 months would reduce dependency at 6 months. 4 Each trial was neutral with respect to the primary outcome.5–7
When the collaboration was initiated, we agreed to perform an individual patient data meta-analysis (IPDM) after all three trials had reported their primary results, in order to confirm or refute a smaller benefit of fluoxetine on our primary outcome than the individual trials had been powered to do, either overall or in particular subgroups, and to provide more precise estimates of any harms.8,9 The primary objective of the IPDM was to determine whether patients with a clinical stroke diagnosis (2–15 days after onset) who are prescribed a 6-month course of fluoxetine 20 mg daily have improved functional outcome, as defined by the modified Rankin Scale (mRS) score at 6 months, compared with placebo.8,9
Pre-specified secondary objectives for our IPDM were:
(a) Does fluoxetine influence the secondary outcomes at 6 and 12 months?
(b) If fluoxetine improves mRS score at 6 months, does any improvement persist after treatment stops?
(c) Does fluoxetine increase the risk of serious adverse events?
(d) Is fluoxetine associated with longer-term survival?
(e) Is the effect of fluoxetine vs placebo on the primary outcome modified by minimization variables and pre-specified subgroups?
(f) In patients with motor deficits at randomization, does fluoxetine improve motor function?
(g) In patients with aphasia at randomization, does fluoxetine improve communication?
(h) Is there a relationship between functional status at 6 months and mood, and is this relationship affected by fluoxetine?
(i) How does non-adherence to the study protocol influence outcome?
(j) Does the effect of fluoxetine vs placebo vary by country of randomization?
(k) Does the effect of fluoxetine vary by ethnicity?
(l) Does the effect of fluoxetine vary by trial? 9
In this article, we present results for all of these aims, except for (h), which will be reported separately as it requires separate statistical analyses.
Methods
We transferred data securely from the AFFINITY and EFFECTS teams to the FOCUS statistician, who combined the data sets.
Where data items were not identical, the chief investigators of each trial decided if/how to combine data. The combined data set included all variables, even if only collected in one or two of the trials. SAS v9.4 (SAS Institute, Inc., Cary, NC, USA) was used for the statistical analysis. We performed descriptive exploratory analyses to display differences in baseline characteristics of patients in the three trials (Supplemental Tables S1 and S2).
We reproduced the tables from the published articles.5–7 We re-analyzed each trial’s data to ensure that the main results could be reproduced.8,9 We had intended to describe the duration and type of hospital stays between randomization and discharge home, discharge to a residential or nursing home or death, but the three trials reported these data were reported in different ways, so this was not possible.
We performed a one-stage IPDM by combining all data into one model to produce estimates of treatment effect.8,9 The minimization algorithms of the three trials had already ensured that patients were allocated to fluoxetine or control in a way that minimized the treatment imbalance with a predefined probability, to ensure allocation was random. We used ordinal logistic regression adjusted for minimization factors but also reported in an unadjusted manner. We conducted an ordinal analysis of mRS by treatment allocation (fluoxetine vs placebo), under the assumption of proportional odds in the model, and tested this assumption using the score test for proportional odds assumption. Then, as a sensitivity analysis, we performed a two-stage IPDM, which involved the creation of summary statistics from each trial and then combining the summary statistics using fixed-effects meta-analysis and Forest plots. Had the one-stage and two-stage IPDD produced different answers, we would have explored why.
For binary outcomes, we compared treatment groups using a binary logistic regression and adjusted for factors used in the minimization algorithm. For continuous outcomes, we present descriptive statistics categorized by allocated treatment. We used a simple unadjusted analysis comparing the two treatment groups using a Mann–Whitney U-test (i.e., not adjusted for variables in the minimization algorithm) due to the distribution of the outcomes.
Missing data in IPDD
The mRS, our primary outcome, includes death; therefore, the number of patients with missing mRS at follow-up was small. Those with a missing mRS were not included in any analysis requiring mRS (complete-case analysis). For secondary outcomes for which missing data were expected because data were not available for patients who did not survive, we presented results for those who were alive at follow-up, and any discrepancies in death rates between groups were taken into account in the interpretation. Missing data for single questions within scores were handled in detail by each scoring method. Where responses to all questions within a scale or subscale were missing, that patient was not included in that particular analysis.
For secondary outcomes and subgroup analyses, we used binomial test for the comparison of proportions, Wilcoxon or Cox proportional-hazard model as appropriate, and compared treatment arms.
Subgroup analysis
We performed subgroup analyses by observing the change in log-likelihood when the interaction between the treatment and the subgroup was added to a logistic regression model.
Sensitivity analysis
We sequentially excluded patients from the trials to explore the influence of non-eligible patients being recruited, and compliance with the trial medication as already described in detail in the statistical analysis plan for the individual trials.
Risk of bias across studies
We rated the certainty of the evidence of the IPDM using Cochrane Grades of Recommendation, Assessment, development, and Evaluation (GRADE), for risk of bias, inconsistency, indirectness, imprecision, and publication bias, using the criteria high, moderate, low and very low; for our primary outcome.
We did not need to produce a PRISMA IPDM diagram as described in our protocol because data from all three trials were available and analyzed.5–7
Results
Primary results
We randomized 5907 patients (mean age 69.6, SD 12.3, 2256 women, 38.19%) (Table 1). Other baseline data are shown in Supplemental Table S1; there were expected differences in baseline data for age, NIHSS predicted outcome, and time to randomization between trials; these differences were due to the differences in inclusion criteria (Supplemental Table S2). There were no differences in the proportion of men and women between trials.
Baseline data comparing the characteristics of AFFINITY, FOCUS, and EFFECTS trial participants by randomized group.
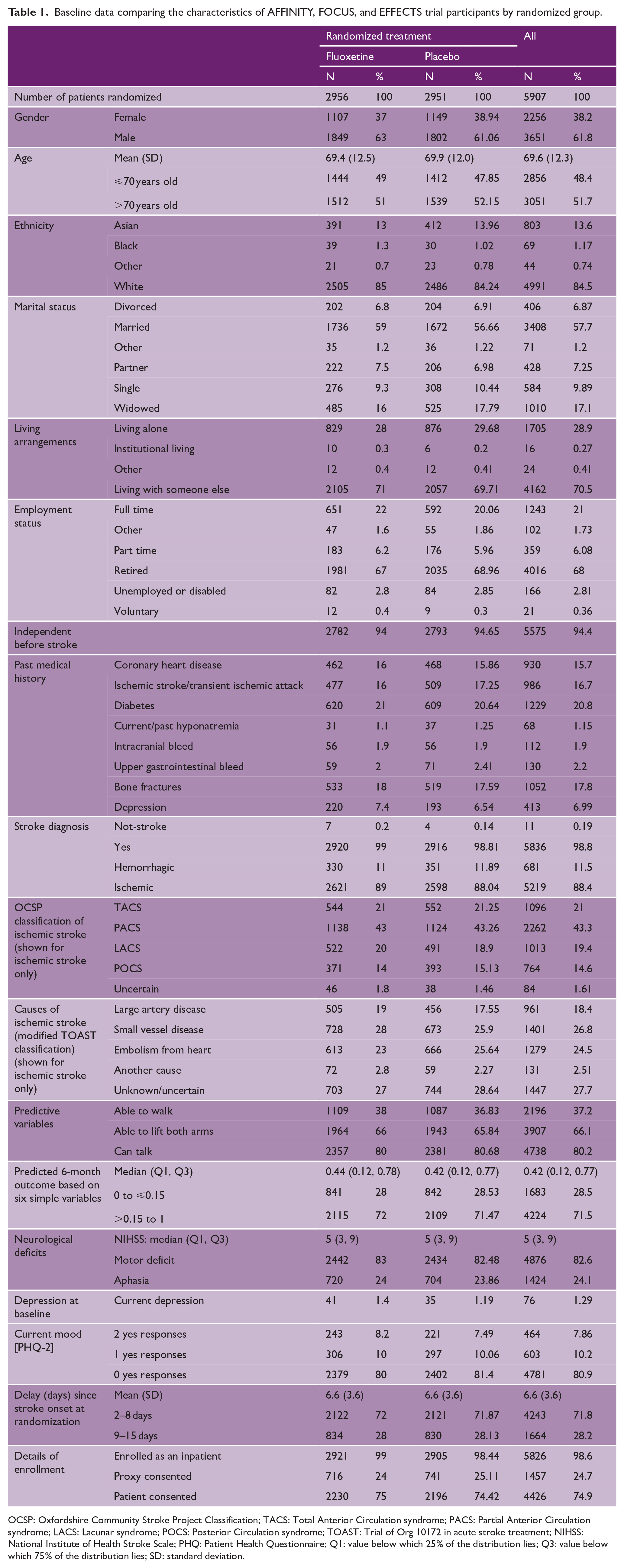
OCSP: Oxfordshire Community Stroke Project Classification; TACS: Total Anterior Circulation syndrome; PACS: Partial Anterior Circulation syndrome; LACS: Lacunar syndrome; POCS: Posterior Circulation syndrome; TOAST: Trial of Org 10172 in acute stroke treatment; NIHSS: National Institute of Health Stroke Scale; PHQ: Patient Health Questionnaire; Q1: value below which 25% of the distribution lies; Q3: value below which 75% of the distribution lies; SD: standard deviation.
Primary outcome data (mRS at 6 months) were available for 5833 (98.75%) of the 5907 patients randomized. An ordinal comparison of individual mRS categories at 6 months, adjusted for variables in the Minimization algorithm, was similar in the two groups (common OR (COR) 0.96, 95% CI 0.87, 1.05, p = 0.37) where a common OR in favor of placebo is < 1.0 (Figure 1), and also similar according to pre-specified subgroups. Our two-stage meta-analysis gave the same result (COR 0.96, 95% CI 0.87, 1.05, I2 0%, p = 0.98) (Figure 2).
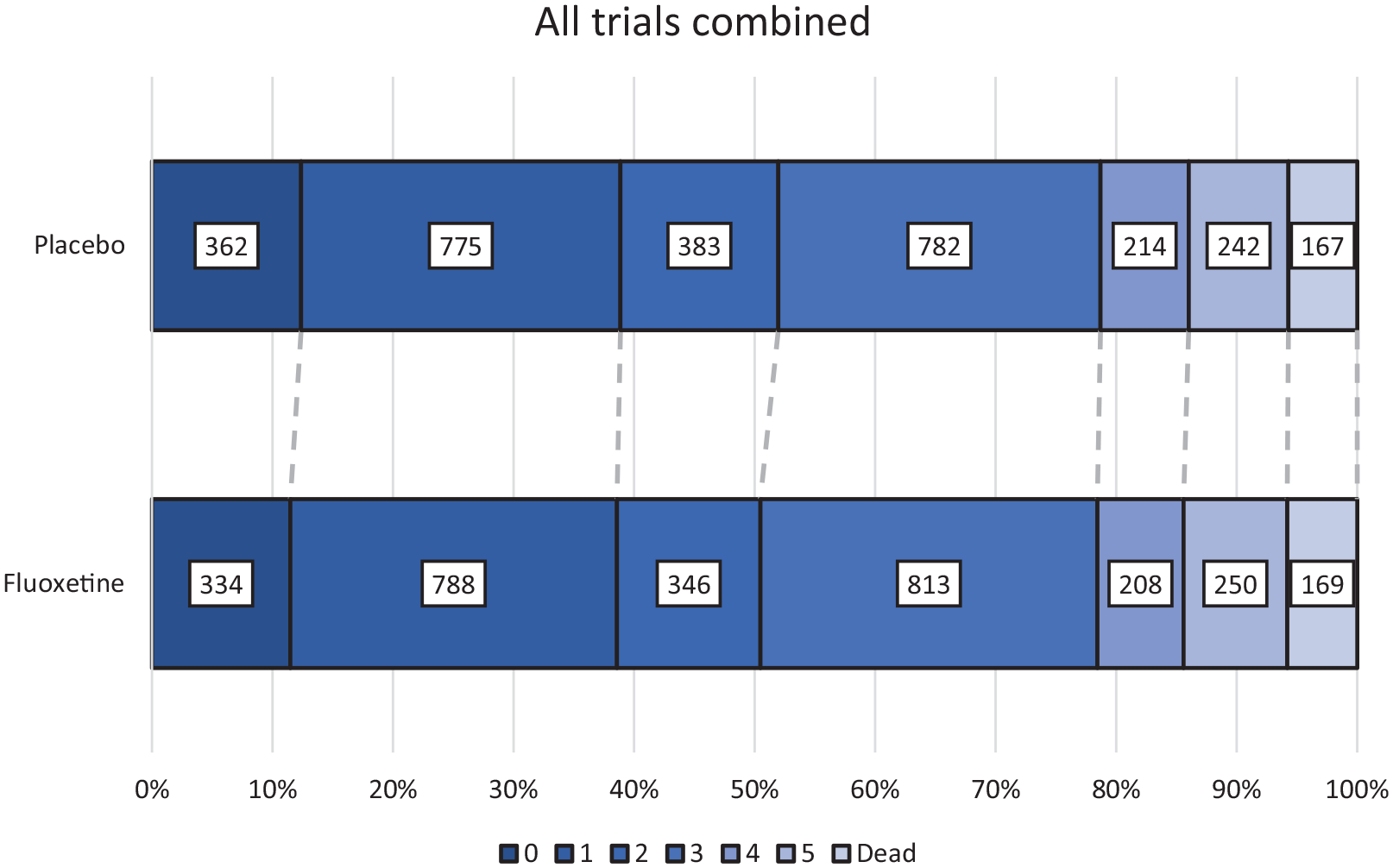
Combined individual participants’ mRS scores at 6 months in AFFINITY, FOCUS, and EFFECTS.
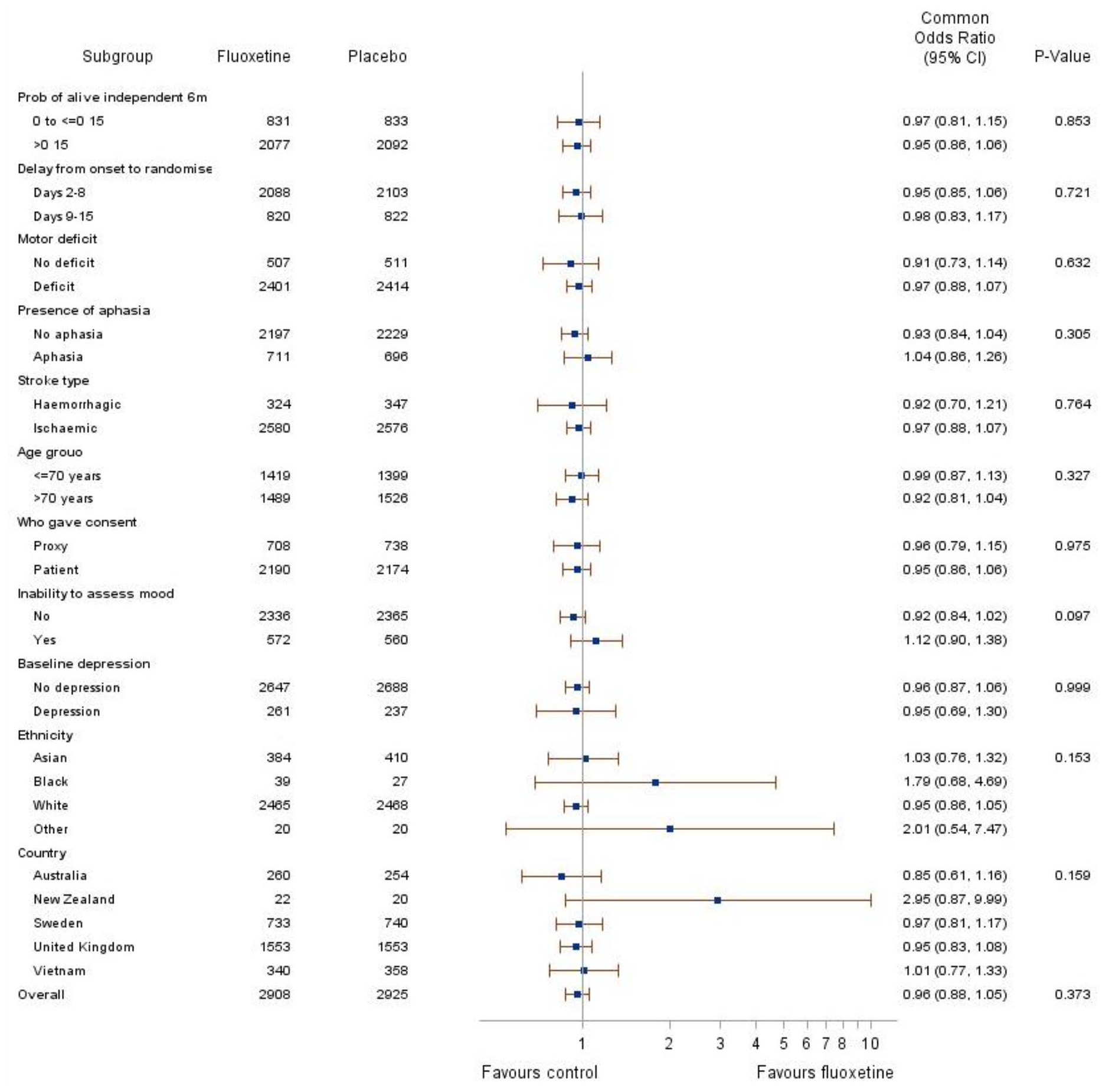
Forest plot showing AFFINITY, FOCUS, and EFFECTS participants’ probability of being alive and independent (mRS score 0–2) in pre-specified subgroups.
Secondary results
There was no difference in the secondary outcomes at 6 and 12 months, except for better SIS emotion score in all trials (Table 2), a reduced risk of new depression (10.05% vs 13.42%, p < 0.0001) and fewer new antidepressants (11.71% versus 15.28%, p < 0.001) at 6 months (Table 3). At 6 months, fluoxetine was associated with an increased the risk of seizures (2.64% vs 1.8%, p = 0.03), falls with injury (6.26% vs 4.51%, p = 0.03), fractures (3.15% vs 1.39%, p < 0.0001) and hyponatremia (1.22% vs 0.61%, p = 0.01) (Table 3).
Secondary outcomes for participants in AFFINITY, FOCUS, and EFFECTS at 6 months.
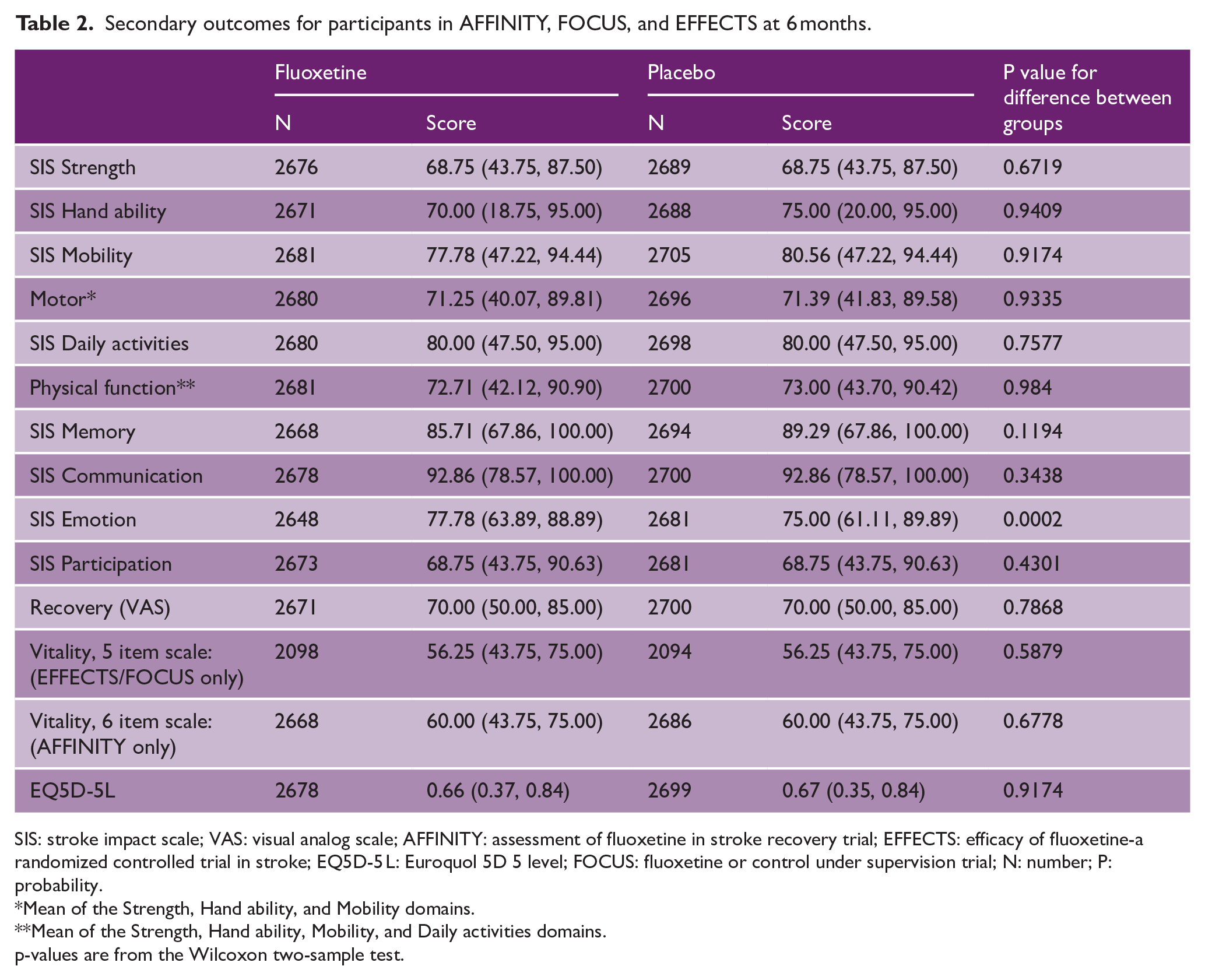
SIS: stroke impact scale; VAS: visual analog scale; AFFINITY: assessment of fluoxetine in stroke recovery trial; EFFECTS: efficacy of fluoxetine-a randomized controlled trial in stroke; EQ5D-5 L: Euroquol 5D 5 level; FOCUS: fluoxetine or control under supervision trial; N: number; P: probability.
Mean of the Strength, Hand ability, and Mobility domains.
Mean of the Strength, Hand ability, Mobility, and Daily activities domains.
p-values are from the Wilcoxon two-sample test.
Adverse events for participants in AFFINITY, FOCUS, and EFFECTS at 6 months.
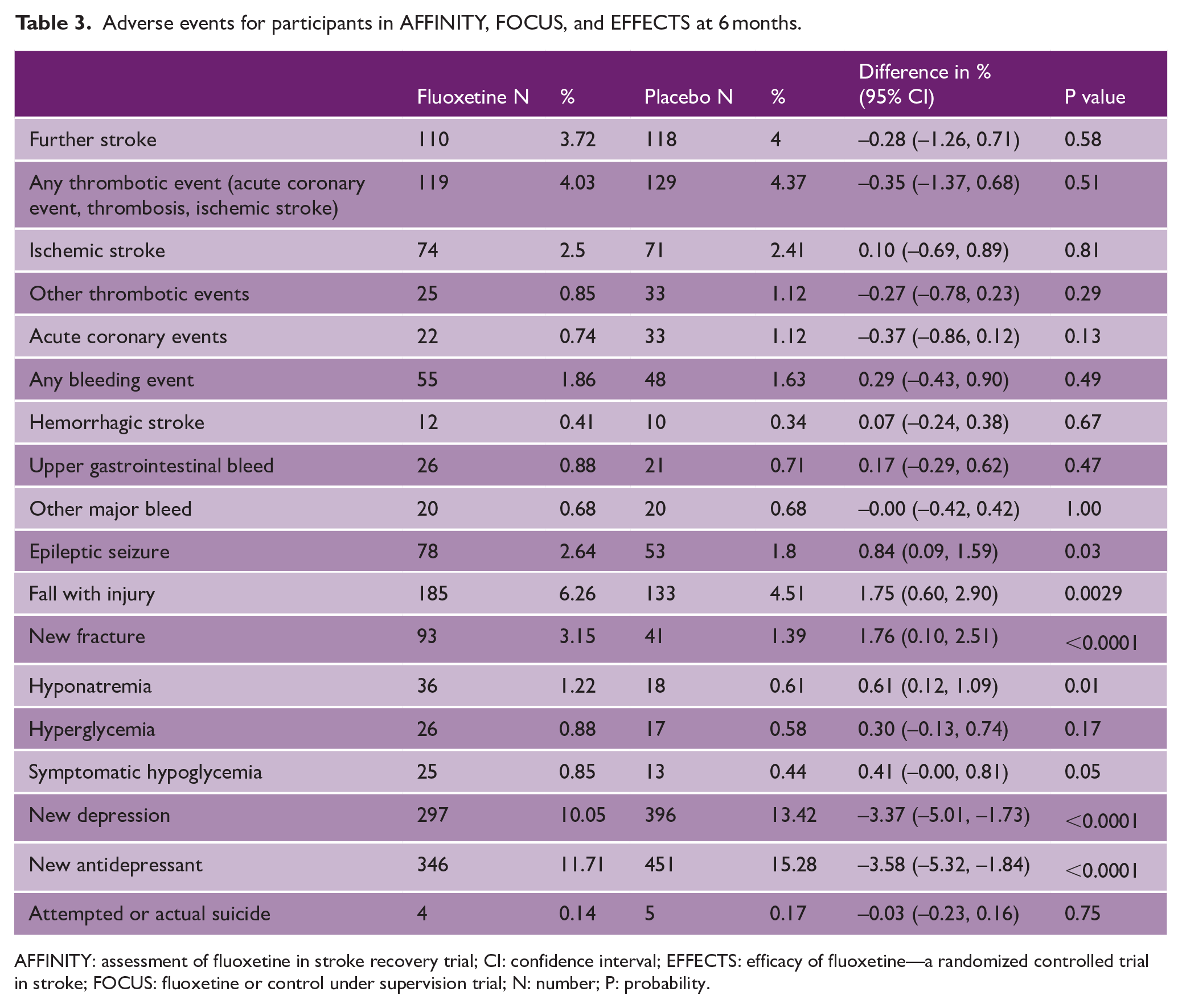
AFFINITY: assessment of fluoxetine in stroke recovery trial; CI: confidence interval; EFFECTS: efficacy of fluoxetine—a randomized controlled trial in stroke; FOCUS: fluoxetine or control under supervision trial; N: number; P: probability.
At 12 months, there was no difference between the fluoxetine and placebo groups for mRS (n = 5760; COR 0.98, 95% CI 0.89, 1.07) adjusted for minimization variables, or survival (hazard ratio 0.929, 95% CI 0.756, 1.141, p = 0.48) or our secondary outcomes. Twelve month adverse events are reported in the Appendix; the trial drug was stopped at 6 months and the trials collected adverse event data slightly differently so we cannot attribute any 12 month differences to treatment allocation.
The effect of fluoxetine on the primary outcome was not modified by baseline probability of being alive and independent at 6 months, time to treatment, motor deficit or aphasia or by pre-specified subgroups (including age, pathological type, inability to assess mood, proxy or patient consent, baseline depression and country) (Figure 2). The two stage IPD for subgroups also produced the same results for subgroups.
In patients with motor deficits at randomization, fluoxetine did not improve motor function (median (IQR): fluoxetine 64.81 (34.03, 86.16) n = 2199 and placebo 64.40 (34.95, 85.30) n = 2212; p = 0.76). In patients with aphasia at randomization (assessed by the NIHSS aphasia item), fluoxetine did not reduce communication problems (SIS domain) at 6 months (median (IQR): fluoxetine 75.00 (42.86, 92.86) n = 637; placebo 78.57 (50.00, 92.86) n = 613 p = 0.39). Non-adherence to the study protocol did not influence outcome at 6 months. The effect of fluoxetine vs placebo did not vary by country of randomization (United Kingdom, Australia, Vietnam, New Zealand, and Sweden) or ethnicity; or by trial (Figure 2). GRADE quality was high for our primary outcome (Table 4).
Grade assessment for the primary outcome in the combined AFFINITY, FOCUS, and EFFECTs trial data set.
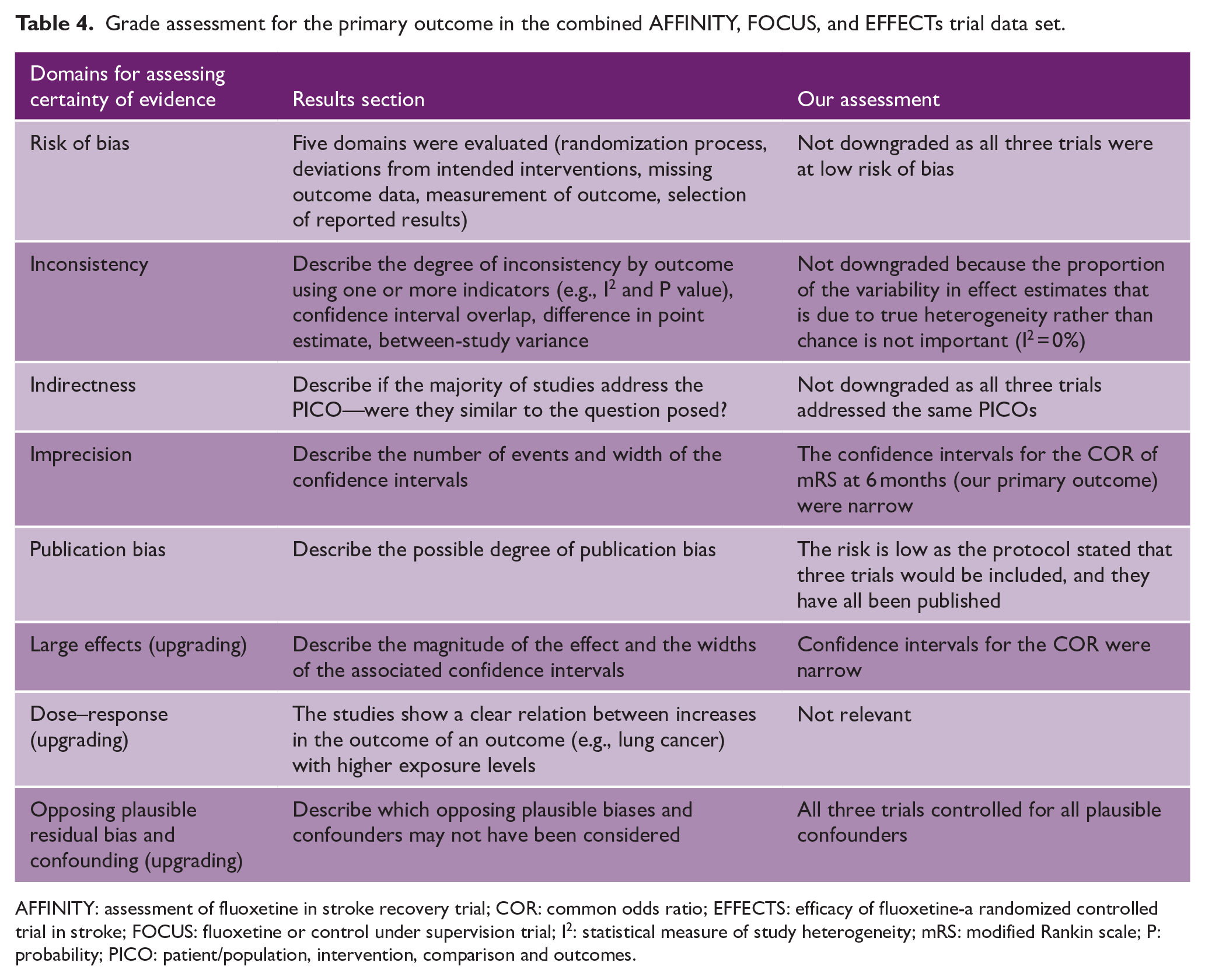
AFFINITY: assessment of fluoxetine in stroke recovery trial; COR: common odds ratio; EFFECTS: efficacy of fluoxetine-a randomized controlled trial in stroke; FOCUS: fluoxetine or control under supervision trial; I2: statistical measure of study heterogeneity; mRS: modified Rankin scale; P: probability; PICO: patient/population, intervention, comparison and outcomes.
Discussion
This IPDM in almost 6000 stroke patients demonstrated that fluoxetine 20 mg daily for 6 months did not improve functional outcome, reduced the risk of depression and new antidepressant prescriptions, and improved the 6-month emotion score in the stroke impact scale. However, fluoxetine increased the risk of seizures, falls with injury, bone fractures, and hyponatremia at 6 months—though the absolute risks of these events were very low. The evidence is of high certainty for our primary outcome. 10 We had previously calculated that with 6000 patients in an IPDM, there would be 90% power to detect a very small effect size equivalent to a COR of 1.16. 11 Thus, even if this IPDM has missed a beneficial effect, the effect size would have been extremely small and unlikely to be clinically relevant. Furthermore, the direction of benefit in this IPDM was in favor of placebo. We demonstrated no benefits in our pre-specified subgroups and our minimization variables. The external validity of this IPDM is high, with patients recruited from five countries. Like many stroke trials, more men than women were recruited, but there is no evidence of any difference in treatment effect (fluoxetine versus placebo) in men and women.
The strengths of this IPDM are that we published a protocol in advance and we included three large high-quality trials that had been designed to facilitate an IPDM. We were able to explore intervention by covariate interactions. 12
This IPDM adds to our Cochrane review by identifying a statistically significant effect on seizures and falls with injuries and by demonstrating no differences in treatment effects by subgroups, countries, ethnicity, or adherence to study medication. Also, we found no increased risk of bleeding, which had been a theoretical concern based on the pharmacology of fluoxetine. Access to the individual participant data enabled analyses of adjusted treatment effects overall and in pre-specified subgroups, which could not be done with tabulated data in the Cochrane review. The Cochrane review reports the relative and absolute risk of recovery as a group average for the overall trial population, whereas the IPDM used multivariate Cox proportional hazard regression to estimate the baseline risk of recovery from all measured, independent, significant participant prognostic factors and the relative and absolute risk change with fluoxetine versus placebo within each strata of baseline risk to identify which (if any) patient populations derived the greatest benefit/risk of adding fluoxetine to standard care.
A 2023 systematic review and meta-analysis of fluoxetine for stroke recovery, published after our Cochrane review, concluded that fluoxetine improved the Fugl Myer Motor score (a score of motor recovery), but this particular analysis was based on only four small trials recruiting 287 patients, some of which were at high risk of bias 13 . Our IPDM clearly shows that in patients with motor deficits, fluoxetine did not improve patient-reported motor recovery.
There are some weaknesses. Although our three trials were collaboratively designed, substantial work was needed to harmonize the datasets because of the slight differences in trial methodology to suit local contexts. Thus, it took longer than we had hoped to complete this IPDM. The GRADE assessments were performed by authors of the three trials and so may have overestimated the certainty of evidence. However, the GRADE assessment performed by the lead author of our Cochrane review of SSRIs for stroke recovery, who had not been involved in the three trials, was similar.
Based on our data, there is no justification for further research exploring the impact of fluoxetine (and by extrapolation-probably other SSRIs too) on functional recovery after stroke. This applies to all stroke types, all severity, and irrespective of baseline deficits. There are, however, unanswered questions about the utility of fluoxetine and other SSRIs for purposes other than functional recovery in patients with stroke for example what is their role in treating mood disorders, particularly in people with aphasia who are generally underrepresented in trials.
Although there was a very small reduction in the risk of depression at 6 months, the potential benefits of using fluoxetine prophylactically to reduce depression could be outweighed by the risks. An absolute reduction of 3.37% in a diagnosis of depression at 6 months was associated with an absolute increase in new fractures of 1.76%.
In summary, fluoxetine should not be given routinely to improve stroke functional recovery or prevent depression, unless the risk of developing depression is considered high and the patient is willing to accept a higher risk of adverse events.
Research in context
Evidence before this study
The three individual trials of fluoxetine showed that fluoxetine did not improve recovery at 6 months when given 2–15 days after stroke but did reduce depression. The 2021 Cochrane review of SSRIs for stroke recovery confirmed this and also demonstrated an increased risk of bone fractures with SSRIs. A subsequent 2023 meta-analysis suggested that fluoxetine improved motor recovery and recommended further large trials. 13 There have been no further large trials of fluoxetine for stroke recovery published since our three trials (PubMed search using the terms fluoxetine AND stroke on 2 October 2023).
Added value of this study
Fluoxetine increases seizure risk, risk of falls with injuries, fractures, and hyponatremia when given to people early after stroke but does not increase bleeding risk despite theoretical reasons why it could do.
There is no impact of fluoxetine on functional recovery in subgroups.
In patients with motor deficits and aphasia, there is no improvement in motor function and communication, respectively.
Compliance with treatment did not affect results.
Further large trials of fluoxetine for stroke recovery are not needed.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930241242628 – Supplemental material for Individual patient data meta-analysis of the effects of fluoxetine on functional outcomes after acute stroke
Supplemental material, sj-pdf-1-wso-10.1177_17474930241242628 for Individual patient data meta-analysis of the effects of fluoxetine on functional outcomes after acute stroke by Gillian Mead, Catriona Graham, Erik Lundström, Graeme J Hankey, Maree L Hackett, Laurent Billot, Per Näsman, John Forbes and Martin Dennis in International Journal of Stroke
Supplemental Material
sj-pdf-2-wso-10.1177_17474930241242628 – Supplemental material for Individual patient data meta-analysis of the effects of fluoxetine on functional outcomes after acute stroke
Supplemental material, sj-pdf-2-wso-10.1177_17474930241242628 for Individual patient data meta-analysis of the effects of fluoxetine on functional outcomes after acute stroke by Gillian Mead, Catriona Graham, Erik Lundström, Graeme J Hankey, Maree L Hackett, Laurent Billot, Per Näsman, John Forbes and Martin Dennis in International Journal of Stroke
Footnotes
Roles of authors
Gillian Mead: conceptualization, investigation, methodology, project administration, supervision, visualization, and writing the original draft.
Catriona Graham: data curation, formal analysis, validation, visualization, and editing draft.
Erik Lundström: conceptualization, data curation, methodology, reviewing, and editing draft.
Graeme J Hankey: conceptualization, data curation, methodology, reviewing, and editing draft.
Maree L Hackett: conceptualization, data curation, methodology, reviewing, and editing draft.
Laurent Billot: conceptualization, data analysis, data curation, methodology, reviewing, and editing draft.
Per Näsman: conceptualization, data analysis, data curation, methodology, reviewing, and editing draft.
John Forbes: conceptualization, data analysis, data curation, methodology, reviewing, and editing draft.
Martin Dennis: conceptualization, investigation, methodology, supervision, visualization, reviewing, and editing original draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Stroke Association, National Institute of Health Research, Australian Government National Health and Medical Research Council, Swedish Research Council, Swedish Heart-Lung Foundation, Swedish Brain Foundation, Swedish Society of Medicine, King Gustav V and Queen Victoria’s Foundation of Freemasons, and STROKE-Riksförbundet.
Data sharing statement
The statistical analysis plan for the IPDM and the main findings from the individual trials have already been published in open access journals. De-identified participant data and a data dictionary defining each field in the set, will be made available to others, after the planned analyses as described in our statistical analysis plan have been published. Data will shared only after investigator approval. Researchers requesting the data will need to write a protocol which will be approved by the investigators. Then a signed data access agreement will be produced. The mechanism for sharing the data will be determined by the information governance requirements of the relevant institutions and countries. Requests for data sharing should be made to Professor Gillian Mead in the first instance.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
