Abstract
Control comparator selection is a critical trial design issue. Preclinical and clinical investigators who are doing trials of stroke recovery and rehabilitation interventions must carefully consider the appropriateness and relevance of their chosen control comparator as the benefit of an experimental intervention is established relative to a comparator. Establishing a strong rationale for a selected comparator improves the integrity of the trial and validity of its findings. This Stroke Recovery and Rehabilitation Roundtable (SRRR) taskforce used a graph theory voting system to rank the importance and ease of addressing challenges during control comparator design. “Identifying appropriate type of control” was ranked easy to address and very important, “variability in usual care” was ranked hard to address and of low importance, and “understanding the content of the control and how it differs from the experimental intervention” was ranked very important but not easy to address. The CONtrol DeSIGN (CONSIGN) decision support tool was developed to address the identified challenges and enhance comparator selection, description, and reporting. CONSIGN is a web-based tool inclusive of seven steps that guide the user through control comparator design. The tool was refined through multiple rounds of pilot testing that included more than 130 people working in neurorehabilitation research. Four hypothetical exemplar trials, which span preclinical, mood, aphasia, and motor recovery, demonstrate how the tool can be applied in practice. Six consensus recommendations are defined that span research domains, professional disciplines, and international borders.
Introduction
Appropriate control comparator design is critical for rigorously testing a trial hypothesis. The benefit of an experimental intervention is established relative to a pre-specified comparator. The ability to demonstrate a difference between an experimental and comparator intervention can be reduced by poor comparator choice. For example, the control comparator may include active ingredient(s) resulting in near identical interventions that will bias to the null hypothesis, or the control comparator may be unacceptable to participants resulting in a higher proportion of dropouts.1,2 Poor comparator design can negatively impact internal (i.e. can the outcome be causally attributed to the intervention?) and external (i.e. are the findings generalizable?) validity and the clinical utility of trial findings. Past reviews have demonstrated that little to no rationale is provided for comparator choice, and fewer words and references are used to describe a comparator as compared to the experimental intervention,2,3 which was reflected in scores obtained using the Template for Intervention Description and Replication (TIDieR). 4 Consequently, the rationale for and details of the comparator control intervention in trials are often unknown. This reduces our ability to advance upon and replicate results.
Stroke recovery and rehabilitation interventions can be complex and involve multiple interacting components or ingredients.5,6 Careful consideration of active and inactive ingredients within a trial is required.7,8 How to relate controls between preclinical and/or clinical trials is also a major barrier to translation. 9 Even a usual care comparator can pose considerable challenges, with vast variation at the micro (i.e. within and between trial sites) and macro (i.e. between countries) level. 10 While the challenge of appropriate control comparator design has been discussed in neurorehabilitation literature,11,12 there is no tool available to guide decision-making. This may explain why, for example, most upper limb rehabilitation trials almost always adopt a comparator that is usual care or dose-matched usual care2,13 rather than a control that seeks to have all but the active ingredients of the experimental intervention present. Such a one-size-fits-all approach overlooks other comparator options that may suit the research question, trial phase, 14 stage of stroke recovery, 15 experimental intervention (considering active/inactive ingredients), geography (e.g. health care system and its funding), and feasibility (e.g. funding, regulatory) considerations, as well as the perspectives of people with lived experience. Establishing guidance to support appropriate selection, description, and reporting of control comparator interventions is needed.
The control taskforce of the third Stroke Recovery and Rehabilitation Roundtable (SRRRIII) aimed to understand the challenges for control comparator design; produce a tool to guide control comparator selection, description, and reporting; and provide recommendations, which if adopted, would advance the science of preclinical and clinical trials in stroke recovery and rehabilitation. Definitions used throughout are provided in Box 1. We present four hypothetical exemplars (Supplemental 1) that apply the developed CONtrol comparator deSIGN (CONSIGN) decision support tool to diverse stroke domains, intervention approaches, trial phases, recovery stages, settings, and populations. Our broad and multidisciplinary focus highlights how control comparator considerations affect all domains of stroke recovery and rehabilitation, and the broader research community involved in the conduct of preclinical and clinical trials.
Definitions.

Methods
Results
The aggregated ranking of five identified challenges to control comparator design, according to ease (i.e. easy to hard) and importance (i.e. low to high), are presented in Figure 1. The challenge of “identifying appropriate type of control” was ranked easy to address and very important. This confirmed the need to develop a decision support tool. Interestingly, the challenge of “variability in usual care” was ranked hard to address and of low importance. This likely reflects that it is near impossible to address usual care globally from a clinical (e.g. different health care systems and funding structures worldwide) and preclinical (e.g. different veterinary guidelines and institutional husbandry support) perspective. “Understanding the content of the control and how it differs from the experimental intervention” was ranked as very important but not easy to address despite the availability of reporting checklists, for example, TIDieR. As “feasibility of delivering, monitoring, and recording control intervention content” was ranked easy to address, it provided a possible solution to include in the trial protocol that would ensure that variability in usual care is captured.
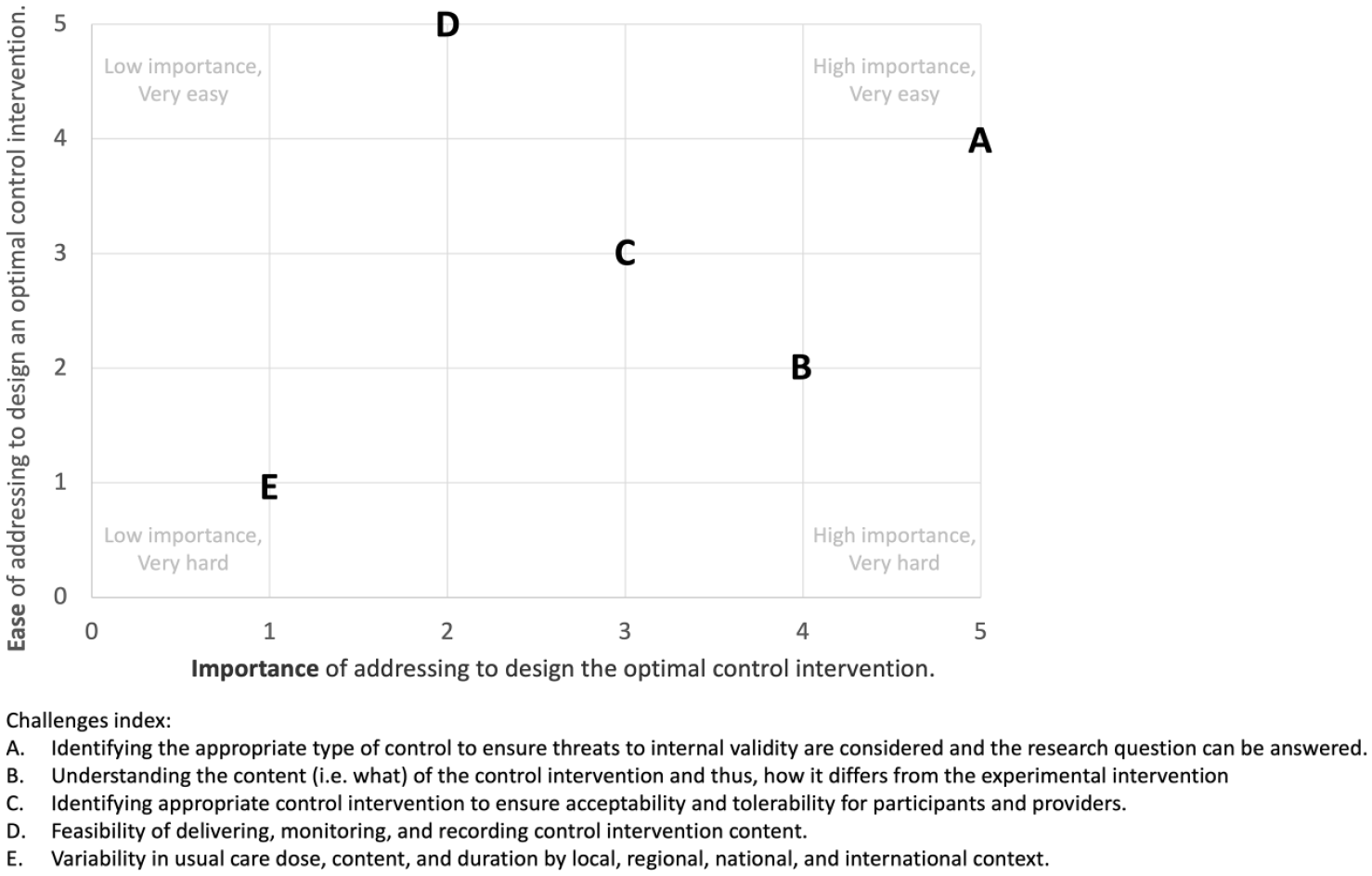
Scatter plot of aggregated rankings of identified control comparator challenges. 0 = low importance/very hard. 5 = low importance/very easy.
CONSIGN tool
The CONSIGN tool was designed and implemented to provide decision support for design and development of a control comparator for a preclinical or clinical trial. We were inclusive of both participant groups to address the standard of control design across the discovery pipeline. 14 The tool is accessible at https://www.redcap.link/SRRR-CONSIGN and the International Stroke Recovery and Rehabilitation Alliance website https://www.strokerecoveryalliance.com/. Box 2 provides an overview of the tool. The tool addresses challenges identified including appropriate type of control selection (Step 3), threats to internal validity (Step 4), understanding the content of the control and how it differs from the experimental intervention (Step 2&6), and feasibility of the control intervention (Step 5). The tool also draws upon the established TIDieR checklist. 4 In developing the CONSIGN tool, the core group was driven to make the content and concepts accessible to all people involved in designing a trial that explicitly compares an experimental intervention(s) to a control comparator. While conceived for stroke recovery and rehabilitation trials, we intentionally developed the tool in a way that is agnostic to domain (e.g. aphasia and motor) or population (e.g. ischaemic or haemorrhagic stroke), increasing the potential for broad utility in trial design. In doing so, people who are experienced in trials (preclinical/clinical), as well as those learning to run trials may benefit equally from using this tool.
CONSIGN tool overview.
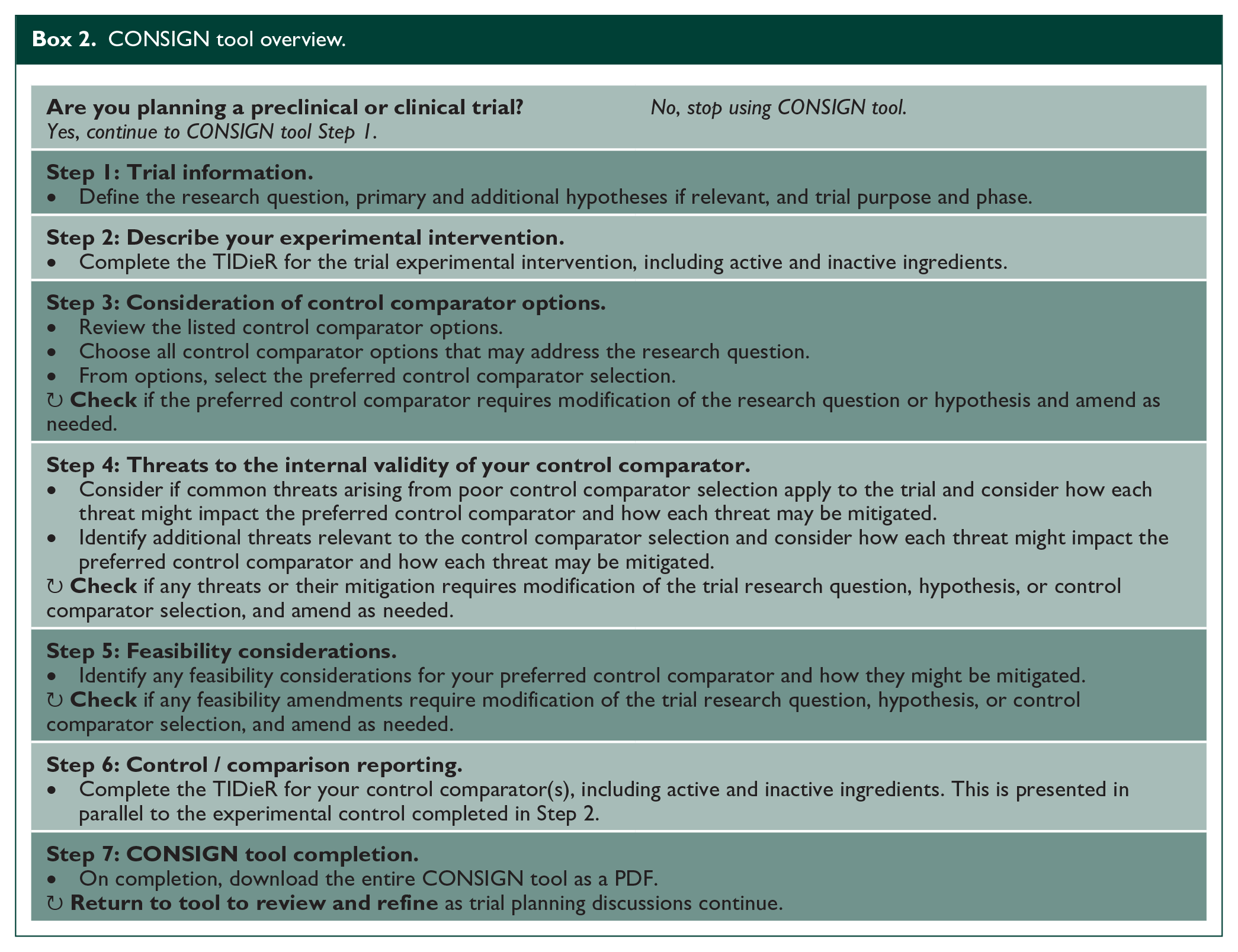
The following sections discuss the rationale and methodological considerations for key components of the CONSIGN tool. Supplemental 2 outlines key references with an associated summary of how the reference is relevant to control comparator design.
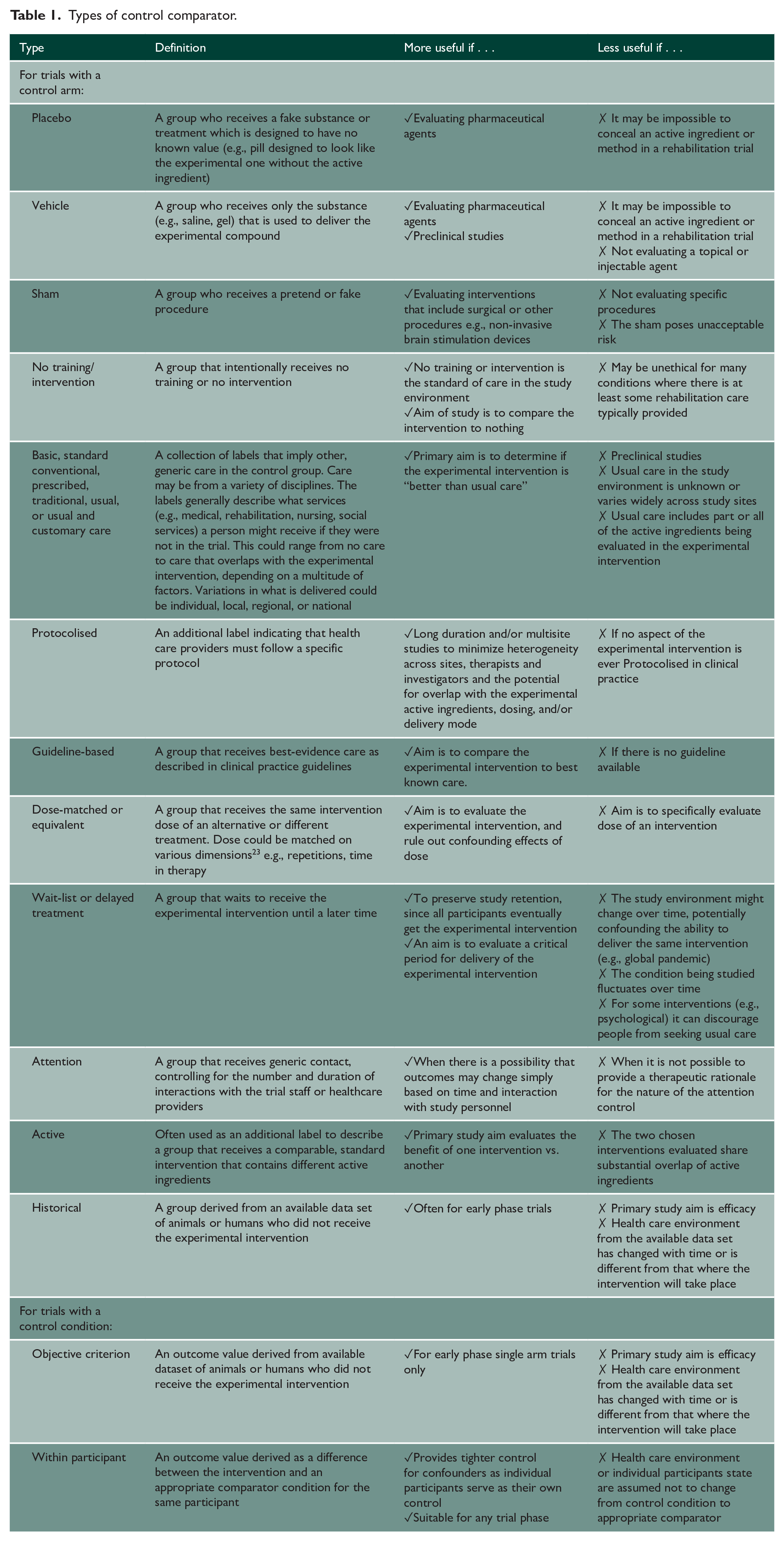
The starting point for control comparator selection is the research question (Step 1). The most appropriate comparator is one that allows a researcher to answer their specific research question. For example, if the research question asks if combination treatment “A + B” is better than “A” or “B” alone, then the comparator to “A + B” needs to be “A” alone and “B” alone. As another example, if the research question asks if the combination “A + B” is better than usual care, then the comparator ought to be usual care (Table 1).
Types of control comparator.
Defining the content of the experimental intervention is key to knowing what needs to be controlled (Step 2). TIDieR 1 is an established tool to guide the reporting of intervention content and is a recognized extension to Item 5 of CONSORT. 9 We made one modification to TIDieR by expanding the “what” section. Our tool specifically requires users to define known or hypothesized active and inactive ingredients of the experimental intervention (Box 1). This modification addresses the established notion that it is commonplace in pharmacological trials to define active and inactive ingredients, but it is not standard in non-pharmacological trials, for example, a behavioral intervention. 7
Selecting the type of control is critical to enable researchers to demonstrate that the outcome observed is due to the experimental intervention as opposed to many other, alternative explanations (Step 3). This can be achieved by ensuring that the comparator intervention is as similar as possible to the experimental intervention in all aspects except the active ingredient(s). There are other considerations that may come into play during selection, such as the ability to conceal active ingredients, availability of evidence of effective treatments or clinical practice guidelines, acceptability of the control comparator for people with lived experience, as well as regulatory and ethical requirements. Table 1 provides an overview of possible control comparator options and contextualizes the considerations for when each may (or may not) be useful. While the list provides distinct options, they can and often are used in combination, for example, placebo vehicle, protocolised usual care, or active attention control.
Careful consideration of alternative explanations for the outcome of interest other than the experimental intervention must be considered during control comparator design (Step 4). Such alternative explanations can threaten trial internal validity; hence they are termed threats18,19 (Box 1). Alternate explanations may occur before the trial begins or during the conduct of the trial, for example, because of differences in the natural trajectory of recovery (e.g. the rate of spontaneous recovery may be different in the experimental and control groups due to timing of enrollment); participants exhibiting a learning effect as they become familiar with the outcome measure (e.g. a cognitive outcome was performed on more of the experimental group just before the study started as compared to the control); how the intervention is delivered (e.g. at the clinic for experimental while at home for the control); or the rate of dropout (e.g. the control intervention is considered a waste of time and many participants dropout). Considering all possible alternative explanations a priori means the control comparator can be designed to minimize potential threats using appropriate mitigation strategies. Figure 2 provides an overview of some common threats and options for mitigation.
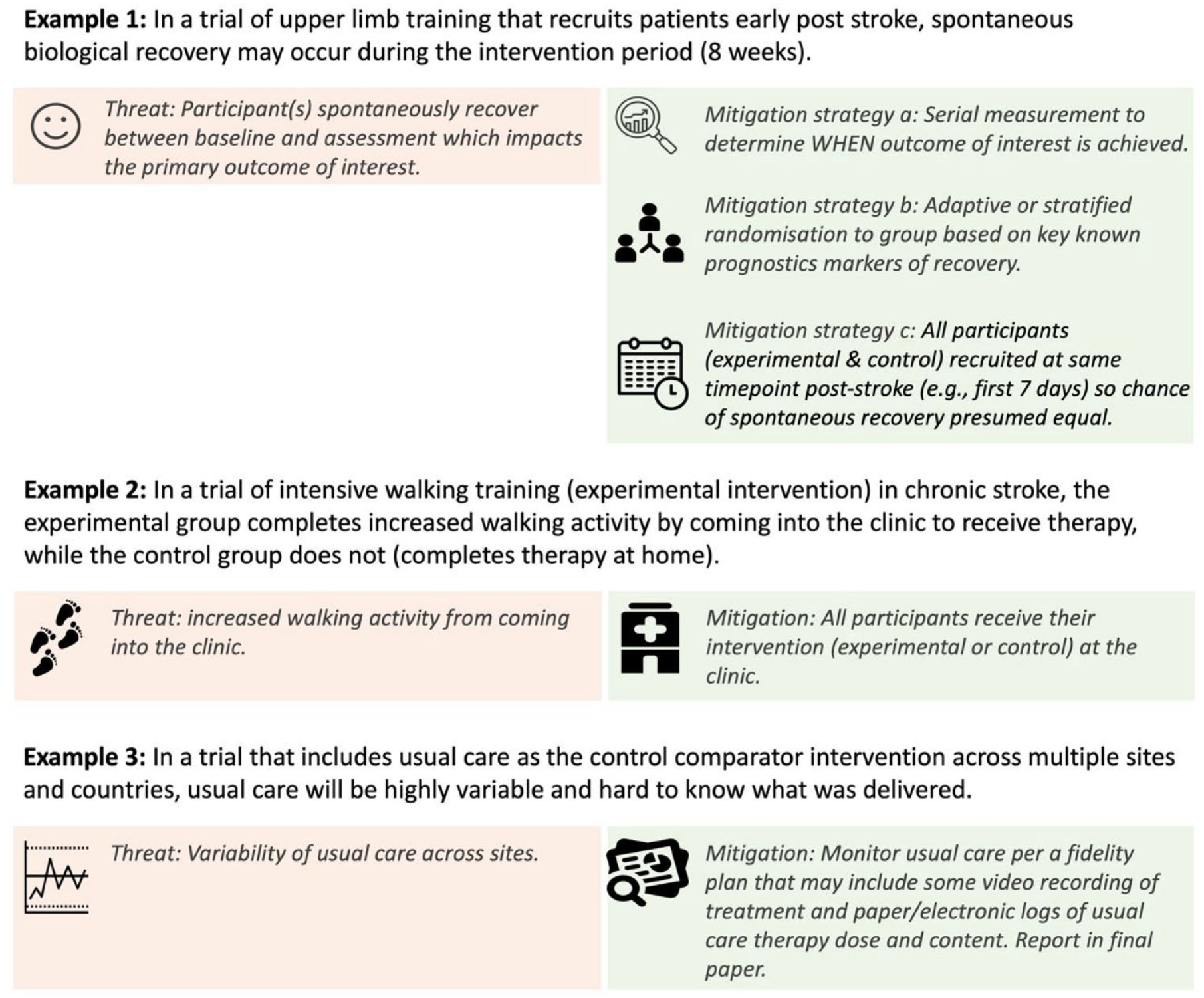
Demonstration of how threats may impact control comparator selection and potential mitigation strategies.
The CONSIGN tool was deliberately structured to design for rigor first and adjust for feasibility (Step 5) later, after selecting the preferred control comparator type that considers all possible threats. Addressing feasibility later in the tool enables the trial design team to understand the implications of altering their control comparator design due to such considerations. Feasibility considerations may relate to access to participants, funding, regulatory policies, and trial sponsors.
In the final step, the tool directs users to complete a control comparator TIDieR (Step 6). For this step, the completed experimental TIDieR (Step 3) is visually presented alongside the control TIDieR for comparison. Inclusion of an independent TIDieR for the control comparator(s) specifically addresses a major gap in current practice 3 and provides the trial design team with materials that can be used as supplemental data in any trial publication.
To demonstrate CONSIGN tool utility, we present four exemplar trials in Supplemental 1 and provide an overview of each in Table 2. The variability in exemplars demonstrates application to different trial phases, 14 domains, 24 phases of recovery, 15 control comparator considerations (Table 1), threats to internal validity (Figure 2), and feasibility constraints.
Overview of the four exemplar that apply the CONSIGN tool to control comparator design.
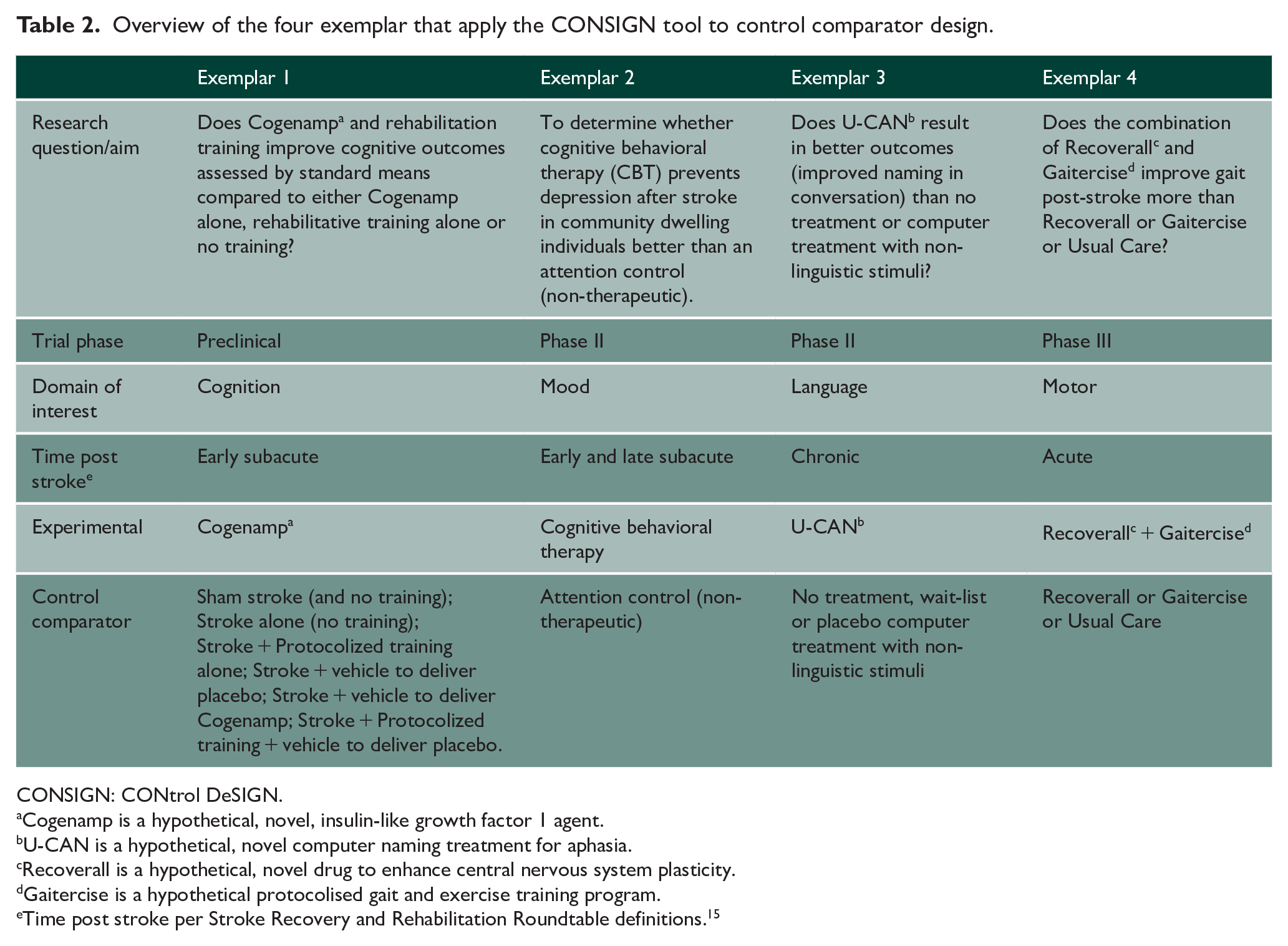
CONSIGN: CONtrol DeSIGN.
Cogenamp is a hypothetical, novel, insulin-like growth factor 1 agent.
U-CAN is a hypothetical, novel computer naming treatment for aphasia.
Recoverall is a hypothetical, novel drug to enhance central nervous system plasticity.
Gaitercise is a hypothetical protocolised gait and exercise training program.
Time post stroke per Stroke Recovery and Rehabilitation Roundtable definitions. 15
Recommendations
Box 3 outlines recommendations that address the identified challenges (Figure 1), support the use of the CONSIGN tool for control comparator design, promote control comparator monitoring, and uptake of the TIDieR 4 reporting guideline.
Control comparator design recommendations.

Discussion
Rigorously testing an experimental intervention and the associated primary hypothesis requires an appropriate control comparator design. The CONSIGN tool was developed to address the identified challenges (Figure 1) and provides decision support for design of a control comparator for preclinical and clinical trials. This tool steps the user through important considerations during the design process. It does not provide a single “perfect” control comparator design on completion because of the heterogenous nature of research questions and the potential for feasibility considerations. From the outset, we agreed to align preclinical and clinical control comparator design to avoid repeating past mistakes, as well as aid translation of intervention testing from preclinical to clinical populations. 25 Although developed for stroke recovery and rehabilitation trials, the utility of the tool extends to other trial populations and interventions, since the structure of the tool is not condition or content specific. It is important to note that CONSIGN complements existing guidelines for reporting standards for comparative trials including CONSORT 17 and TIDieRt4, as well as previous tools produced by the SRRRs including the Trial Development Framework that guides trialists to establish GO, NO-GO decision points important to overall trial design. 26
A key lesson learned during this consensus process was that there is no “one-size-fits-all” control comparator selection that can be applied to all stroke recovery and rehabilitation trials. Stroke recovery and rehabilitation interventions are often complex, and active ingredients may be hard to identify. We acknowledge that this makes designing a control comparator more difficult than selecting a comparator in a pharmacological trial. Many stroke recovery and rehabilitation trials have adopted “usual care” or “dose-matched usual care” as the control comparator.2,13 It is possible that this approach has not yielded breakthrough intervention(s) because the (a) active ingredients of the usual care comparator intervention(s) overlap with the experimental, and/or (b) heterogeneity in usual care produces heterogeneity in outcomes that in turn limit the ability to detect between-groups differences. There may also be funding or regulatory considerations that have driven the control comparator selection used in prior trials. While in many fields, it may be feasible to test an experimental intervention against a placebo (e.g. a pharmacological trial), where there is no active substance or treatment provided, this is often impractical in rehabilitation research, and may be unethical as it might require modification or withdrawal of standard care. Indeed, usual stroke care can have a large impact on functional outcomes. 3 In recognizing this, CONSIGN as a tool provides a breadth of available control comparator types for consideration (Table 1), which can be used individually or collectively. Extending beyond the initial trial design stage, we urge researchers, funding agencies, and publishers to carefully consider control comparator design during manuscript and grant review processes. We encourage researchers to include the CONSIGN tool in supplemental materials to demonstrate why a particular control comparator was selected.
The application of the CONSIGN tool to four exemplars demonstrates the broad utility of this tool. The challenge of appropriate control comparator design exists across domains, including interventions that target cognition (Exemplar 1), mood (Exemplar 2), language (Exemplar 3), and motor (Exemplar 4) outcomes. We recognize there may be different considerations when selecting a type of control depending on the domain, for example, a waitlist control may be inappropriate as it may discourage people with mood disorders from seeking help, or that guideline care may not be possible for new and emerging scientific areas or geographical regions that have yet to establish clinical practice guidelines. Distinct opportunities may be available to preclinical trials, such as using a higher number of control comparators to systematically evaluate an intervention. To increase translatability, preclinical controls should explicitly report control conditions and attempt to mimic clinical controls. For example, preclinical investigations often allow larger differences between experimental and control and may even allow non-treated control arms. 9 To overcome this gap, preclinical trials should use some form of clinically inspired best practice. 25
The use of the CONSIGN tool does not negate the need for input of trial methodologists, biostatisticians, clinicians, and people with lived experience on the research team. As design of the experimental intervention evolves during trial preparation, it is highly likely that the needs of the control comparator design may also change. We view control comparator design as an iterative process where drafts of the CONSIGN tool output can be brought back to the research team (including people with lived experience) repeatedly until there is agreement on a rigorous and feasible control comparator condition(s) that appropriately addresses the research question of interest and statement of hypothesis, and controls for the active ingredients of the experimental intervention and possible threats to internal validity.
Improved planning and reporting of control comparators, including usual care, will ultimately facilitate the ability to synthesize findings across trials. The current heterogeneity of stroke trial control comparator types and lack of information about their content means that data syntheses often group different interventions with different active ingredients together. Sharing the content of the experimental and control comparator interventions using the TIDieR, 4 or an equivalent standard, will greatly enhance future research synthesis efforts.
Conclusion
This SRRR used expert consensus to identify the current challenges with control comparator design, and developed recommendations that span research domains, professional disciplines, and international borders to move the field forward. An important product emerging from this effort is the CONSIGN tool to guide control comparator selection, description, and reporting. This tool is designed to stimulate critical thinking about control comparator design but does not provide a single “perfect” control comparator design on completion. Adhering to the recommendations can improve control comparator design and ultimately establish a stronger rationale for a selected comparator. This has the potential to foster translation of trial outcomes that deliver benefits for people living with stroke. Our group looks forward to continuing to develop and improve the utility of the tool.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231199336 – Supplemental material for Control intervention design for preclinical and clinical trials: Consensus-based core recommendations from the third Stroke Recovery and Rehabilitation Roundtable
Supplemental material, sj-docx-1-wso-10.1177_17474930231199336 for Control intervention design for preclinical and clinical trials: Consensus-based core recommendations from the third Stroke Recovery and Rehabilitation Roundtable by Kathryn S Hayward, Emily J Dalton, Jessica Barth, Marian Brady, Leora R Cherney, Leonid Churilov, Andrew N Clarkson, Jesse Dawson, Sean P Dukelow, Peter Feys, Maree Hackett, Steve R Zeiler and Catherine E Lang in International Journal of Stroke
Supplemental Material
sj-pdf-2-wso-10.1177_17474930231199336 – Supplemental material for Control intervention design for preclinical and clinical trials: Consensus-based core recommendations from the third Stroke Recovery and Rehabilitation Roundtable
Supplemental material, sj-pdf-2-wso-10.1177_17474930231199336 for Control intervention design for preclinical and clinical trials: Consensus-based core recommendations from the third Stroke Recovery and Rehabilitation Roundtable by Kathryn S Hayward, Emily J Dalton, Jessica Barth, Marian Brady, Leora R Cherney, Leonid Churilov, Andrew N Clarkson, Jesse Dawson, Sean P Dukelow, Peter Feys, Maree Hackett, Steve R Zeiler and Catherine E Lang in International Journal of Stroke
Footnotes
Acknowledgements
The authors acknowledge Drs Hayward and Kwakkel for co-chairing and convening the SRRRIII and Dalton for managing SRRRIII conduct and in-person meeting organization. The third international Stroke Recovery and Rehabilitation Roundtables are an initiative of the International Stroke Recovery and Rehabilitation Alliance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Receipt of financial support from the Canadian Partnership for Stroke Recovery, NHMRC Center of Research Excellence to Accelerate Stroke Trial Innovation and Translation (grant no. GNT2015705) and unrestricted educational grants provided by Ipsen Pharma and Moleac.
Statement of endorsement
The World Stroke Organization (WSO) endorses the goals of the third international Stroke Recovery and Rehabilitation Roundtable (SRRRIII) research activity which are consistent with the mission of the WSO.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
