Abstract
Background and aim:
Previous studies have suggested cardiovascular risk factors increase the risk of not only common sporadic stroke but also of stroke in patients with monogenic stroke disorders including CADASIL. We investigated the effects of the NOTCH3 Arg544Cys (R544C) variant and associated vascular risk factors on stroke in the Taiwanese population.
Methods:
This study was conducted using data from the Taiwan Biobank, consisting of at least 130,000 Han Chinese participants. The genotype was derived from customized genome-wide arrays for 650,000 to 750,000 single-nucleotide polymorphisms (SNPs). Individuals with NOTCH3 R544C were subsequently matched with noncarriers based on the propensity score at a 1:10 ratio by demographic and cardiovascular risk factors. The odds ratio (OR) for stroke or other phenotypes in NOTCH3 R544C carriers and matched noncarriers was then calculated. Univariate and multivariate regression analyses were performed on cardiovascular risk factors in NOTCH3 R544C carriers with and without stroke. The polygenic risk score (PRS) model, adopted from the UK Biobank, was then applied to evaluate the role of NOTCH3 R544C in stroke.
Results:
From the 114,282 participants with both genotype and questionnaire results, 1080 (0.95%) harbored the pathogenic NOTCH3 R544C variant. When compared to the matched controls (n = 10,800), the carriers presented with a history of stroke (OR: 2.52, 95% confidence interval (CI) (1.45, 4.37)), dementia (OR: 30.1, 95% CI (3.13, 289.43)), and sibling history of stroke (OR: 2.48, 95% CI (1.85, 3.34)) phenotypes. The risk of stroke increased with every 10-year increase in age (p = 0.006, Cochran–Mantel–Haenszel test). Among NOTCH3 R544C carriers, 16 (1.3%) of the 1080 carriers with a stroke history were older, male, and more likely to have hypertension, diabetes, dyslipidemia, and a family history of stroke. In the stepwise multivariate analysis, hypertension (OR: 11.28, 95% CI (3.54, 43.3)) and diabetes mellitus (OR: 4.10, 95% CI (1.31, 12.4)) were independently associated with stroke. Harboring the NOTCH3 R544C variant in the Taiwan Biobank is comparable with a 6.74 standard deviations increase in individual’s polygenic risk score for stroke.
Conclusion:
While the NOTCH3 R544C variant alone increased the risk of stroke, modifiable vascular risk factors also played a role in the occurrence of stroke in Taiwanese community-dwelling individuals carrying the NOTCH3 variant.
Introduction
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) due to cysteine-altering NOTCH3 variants is the most common hereditary stroke syndrome. 1 The patients with CADASIL exhibit broad-spectrum phenotypes, including stroke, cognitive deficit, gait disturbance, migraine with aura, and psychiatric disorders. The clinical symptom severity and leukoencephalopathy burden depend on the location of the variant in the epidermal growth factor-like repeat (EGFr) domains in NOTCH3. 2 As compared with cysteine-altering NOTCH3 EGFr 7-34 variants in population databases, variants of the NOTCH3 1-6 EGFr domain has a low frequency (1.98 per 100,000 adults). 3 Recent large-scale studies have reported a relatively low penetrance of the cysteine-altering NOTCH3 EGFr 7-34 variants, which leads to milder CADASIL phenotypes. 4
Importantly, vascular risk factors seemed to have a key influence on the risk of stroke and vascular dementia in CADASIL. From the 450,000-person UK Biobank investigation, the Framingham risk score contributed to stroke among 973 NOTCH3 variant carriers. 5 In the DiViNAS cohort study, sex and hypertension were found to be the major variables contributing to stroke and post-stroke prognosis risks. 6 Uncertainty surrounded the applicability of vascular risk factor control in Asian descendants harboring the NOTCH3 variants. From the Exome Aggregation Consortium (ExAC) database, NOTCH3 Arg544Cys (R544C, EGFr 13/14) was the most prevalent variant among individuals of East Asian descent. 3 In Taiwan, the prevalence of the NOTCH3 R544C variant was approximately 3% in patients with acute stroke and ~0.9% of the general population.7–9 Finding an optimal strategy for managing and preventing stroke in this population is crucial.
Aim and hypothesis
To address the issue at a population level, we aim to investigate the NOTCH3 R544C carriers in the Taiwan Biobank. We aim to characterize the phenotypes that were commonly observed in CADASIL, including stroke, cognitive deficit, gait disturbance, migraine, and psychiatric disorders, in the NOTCH3 R544C carriers. 10 We also aim to analyze the demographic and vascular risk factors difference in the NOTCH3 R544C variant with and without stroke. We hypothesized that the penetrance of stroke is related to acquired, modifiable vascular risk factors.
Methods
Study population
The Taiwan Biobank is a longitudinal, prospective cohort with self-reported questionnaires, physical examinations, blood tests, and genetic analysis information.7,11 The cohort population consists of at least 130,000 participants of Hans Chinese ancestry without a cancer history. All participants received routine health examinations at the Taiwan Biobank enrollment stations; 30 mL serum samples were obtained from them and sent for the standard laboratory examination. Participants underwent genotyping through a genome-wide association study (GWAS) with two generations of custom arrays: 653 million and 750 million single-nucleotide polymorphisms (SNPs) were tested on the C2-42 Axiom Genome-Wide TWB Array (TWBv1) and Axiom Genome-Wide TWB 2.0 Array Plate (TWBv2), respectively. The participants also reported health-related questionnaires to Taiwan Biobank enrollment stations.
Selection criteria and genetic analysis
We analyzed data from the Taiwan Biobank between 2012 and 2021 using a genotype-first methodology. The NOTCH3 R544C genotype was tested on either or both customized GWAS plates, and variants were extracted using PLINK software (version 1.90). There were 251 and 838 NOTCH3 R544C heterozygote carriers detected on TWBv1 and TWBv2 GWAS chips, respectively. 250 NOTCH3 R544C heterozygotes participants had both chips tested. One homozygote NOTCH3 R544C mutant was detected on both arrays. The observed and expected heterozygosity of R544C was 0.913% and 0.916% (p = 0.4413) on the TWBv1 array, respectively, whereas it was 0.949% and 0.946% (p = 0.728) on the TWBv2 array, respectively. After confirming the Hardy–Weinberg equilibrium of the data from the two arrays, 1080 NOTCH3 R544C carriers were identified and extracted.
Propensity score matching
Demographic data, including age, sex, education, and cigarette and alcohol use, were extracted along with self-reported medical histories, including hypertension, diabetes, dyslipidemia, stroke, dementia, migraine, Parkinson’s disease, epilepsy, depression, schizophrenia, and bipolar disorder; and family histories of stroke and dementia were extracted from the self-reported health-related questionnaires administered from 2012 to 2021 at Taiwan Biobank enrollment stations. Then, we performed a 1:10 (NOTCH3 R544C carriers vs. noncarriers) propensity score matching by age, sex, hypertension, diabetes mellitus, dyslipidemia, education level, alcohol intake, and smoking status retrieved from self-reported health-related questionnaires. The matching process was performed using the nearest neighbor algorithm with generalized linear modeling using the MatchIt package (version 4.3.2) in R. 12 We also extracted body mass index (BMI), blood pressure measurement, lipid profile, glycated hemoglobin, fasting glucose level, serum creatinine, and uric acid results from the released biobank data and compared the variables on a case–control series basis.
Statistical analysis
We compared the demographic, vascular risk factors, and laboratory data between the NOTCH3 R544C carriers and noncarrier controls by using the Student t-test, chi-square test, or Fisher exact test with correction, as appropriate. A standard mean difference was calculated between the two groups. The OR for the association of the NOTCH3 R544C variant with self-reported neuropsychiatric diseases and relevant family histories was then calculated using the epitools package (version 0.5.10.1) in R. 13
We then divided the NOTCH3 R544C group into carriers with and without stroke histories. Chi-square and unpaired two-sided t-tests were performed to analyze categorical and continuous cardiovascular risk factor variables, respectively. Finally, a multivariable logistic regression model was constructed to assess the independent risk factors for stroke in the NOTCH3 R544C group, with the stepwise selection of categorical variables based on a p-value of <0.10 for entry and of <0.05 for stay, with age and sex forced into the model. All analyses were performed using R (version 4.1.2).
Polygenic risk score analysis
In order to assess the risk associated with NOTCH3 variants compared to common stroke variants, the data from the carriers and matched noncarriers were compared using the PRS model. 14 We utilized a PRS constructed by a meta-analytic strategy for ischemic stroke (metaGRS) that captures the totality of information from five stroke outcomes and 14 stroke-related phenotypes GWAS summary statistics, 14 and analyses this score in the Taiwan Biobank data. Taiwan Biobank samples were genotyped using the Axiom Genome-Wide TWB array and imputed using SHAPEIT4 (version 4.1.2) and IMPUTE2 (version 2.3.2) with a merged reference panel consisting of data from an East Asian population from 1000 Genomes Project phase 3 (n = 504) and Taiwan Biobank whole-genome sequencing (n = 1451). The weighted PRS was calculated using PLINK (version 1.9). All relevant SNPs were included in the calculation, regardless of their imputation quality. On a case–control basis, the predictivity of each PRS unit for stroke was calculated between stroke (defined as a binary trait) and standardized PRS via logistic regression. We compared the odds ratio (OR) for stroke in individuals carrying NOTCH3 variants versus noncarriers, as well as the OR for one standard deviation (SD) increase in the ischemic stroke PRS to assess the risk associated with NOTCH3 variants. The Z-score was then calculated to estimate the equivalent PRS standard deviations of carrying the NOTCH3 R544C variant. 15
Standard protocol approval, registration, and patient consent
This study was approved by the National Taiwan University Hospital Research Ethics Committee B (approval number 202103031RINB). Relevant data were acquired from the Taiwan Biobank (TWBR11004-02) after endorsement from an independent research ethics committee. Participant consent for data use was obtained by the Taiwan Biobank at enrollment.
Results
Characteristics of R544C carriers and matched controls
Of the 132,715 individuals who completed the questionnaire and 114,599 who underwent genotyping, 114,282 had both a complete questionnaire at enrollment and genotypic SNP results for NOTCH3 R544C. Of them, 1080 (0.95%) participants carried the NOTCH3 R544C variant. Through propensity score matching, 10,800 controls were matched from the 113,202 noncarriers. The flowchart of the current study is presented in Figure 1.
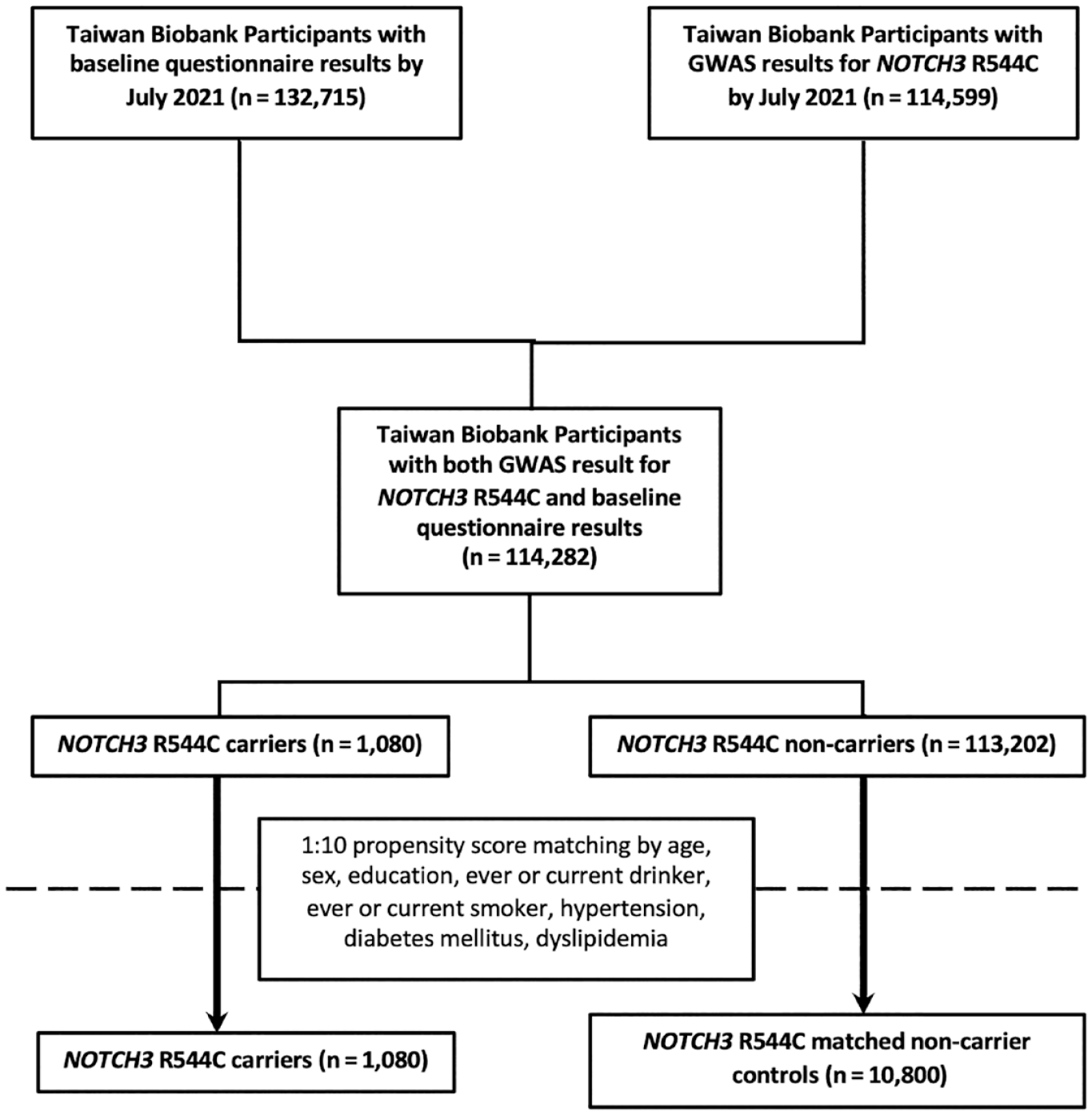
Participant recruitment and matching flowchart.
NOTCH3 R544C carriers (n = 1080, mean age 49.8 ± 10.9 years) were paired with noncarriers (n = 10,800, mean age 49.7 ± 10.9 years). The NOTCH3 R544C carriers and matched noncarriers exhibited similar age, sex, education levels, alcohol consumption, smoking status, medical history, and biochemical laboratory data, with an absolute standardized mean difference of mostly under 0.1 (Table 1). Systolic blood pressure was slightly higher in the matched control group; on the other hand, serum uric acid levels showed marginal elevations among the carriers.
Basic demographics and baseline laboratory values in NOTCH3 R544C carriers and matched controls.
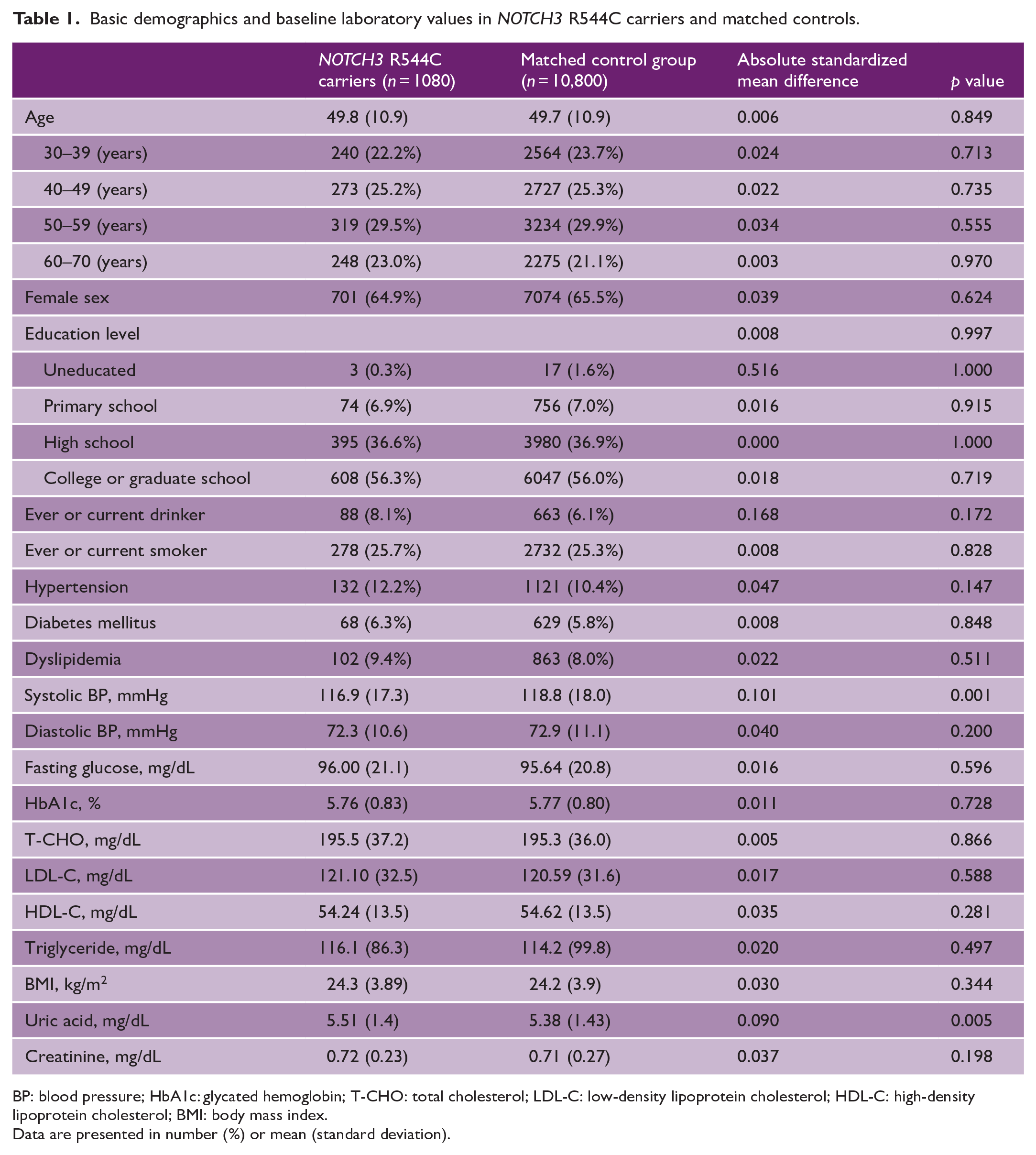
BP: blood pressure; HbA1c: glycated hemoglobin; T-CHO: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; BMI: body mass index.
Data are presented in number (%) or mean (standard deviation).
Phenotypic presentations of NOTCH3 R544C carriers
The proportion of participants with histories of stroke (1.48% vs. 0.59%) and dementia (0.28% vs. 0.009%) was higher in the carriers than in the noncarriers (Table 2). The propensity score-adjusted OR (95% confidence interval (CI), p value) for stroke in carriers compared to matched noncarriers was 2.52 (1.45, 4.37), p < 0.001), but that for dementia was 30.1 ((3.13, 289.43), p < 0.001). Between the two groups, Parkinson’s disease, migraine, schizophrenia, and other neuropsychiatric disorders did not differ significantly (p > 0.05).
Neuropsychiatric manifestations and family history of stroke and dementia in NOTCH3 R544C carriers and matched controls.
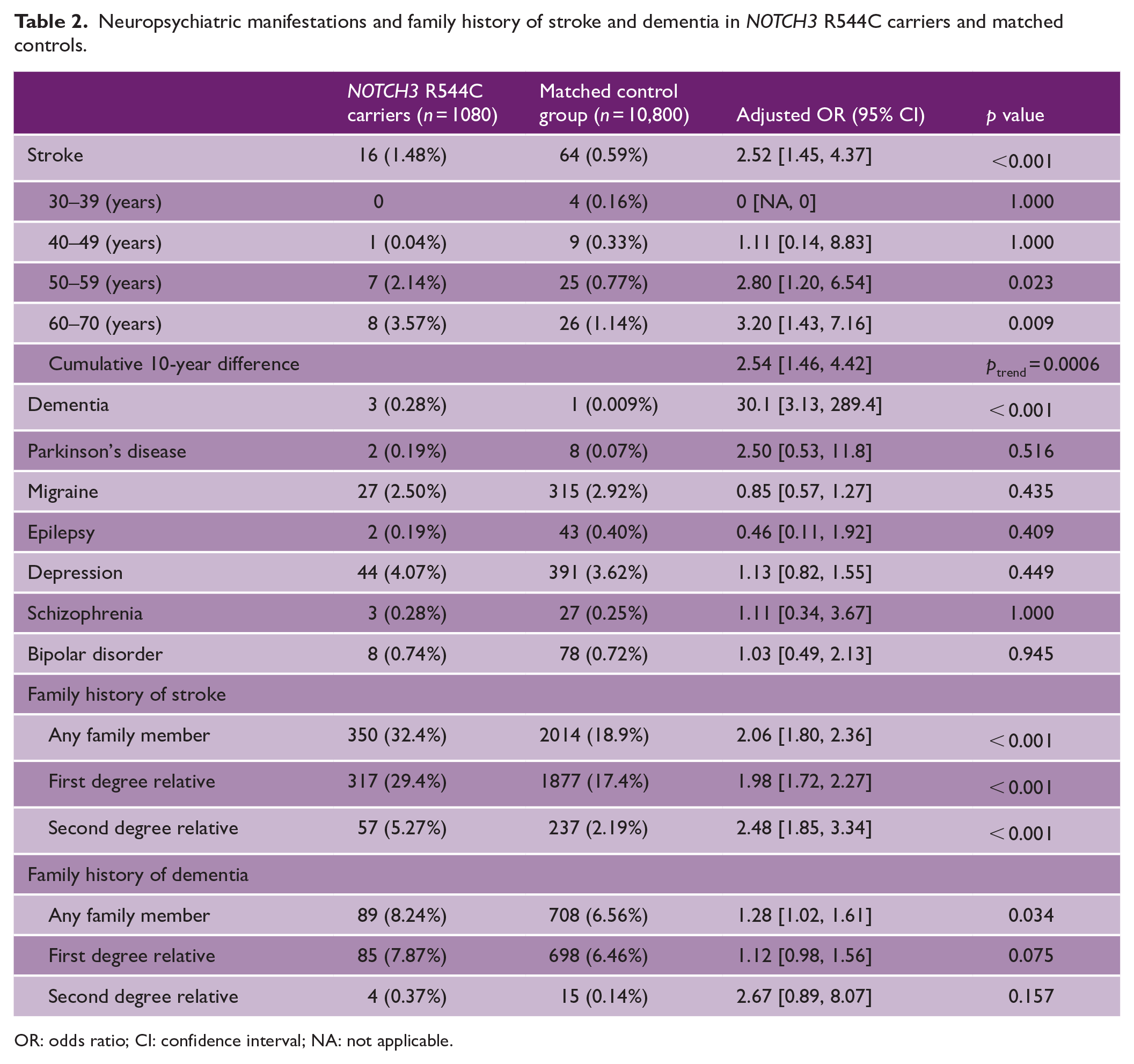
OR: odds ratio; CI: confidence interval; NA: not applicable.
After stratification by age at 10-year intervals, we noted a serial increase in adjusted OR (95% CI) for stroke in the R544C carriers; it was 1.11 ([0.14, 8.83), p = 0.99), 2.8 ((1.20, 6.54), p = .023), and 3.2 ((1.43, 7.16), p = 0.009) in the 40 to 49, 50 to 59, and 60 to 69 years age groups, respectively. Overall, the OR (95% CI) was 2.54-fold ((1.46, 4.42); ptrend = 0.0006, Cochran–Mantel–Haenszel test) for every 10-year increase in age.
NOTCH3 R544C variant carriers were more likely than their matched noncarrier counterparts to have a family history of stroke (adjusted OR: 2.06, 95% CI (1.80, 2.36), p < 0.001) and dementia (adjusted OR: 1.28, 95% CI (1.02, 1.61), p = 0.034) (Table 2). The OR (95% CI) for the family history of stroke was higher in second-degree relatives than in first-degree relatives (second-degree: 2.48 (1.85, 3.34); p < .001 vs. first-degree: 1.98 (1.72, 2.27); p < .001).
Factors associated with stroke development in R544C carriers
Of the 1080 NOTCH3 R544C carriers, 16 (1.3%) had a history of stroke (Table 3). Carriers with a history of stroke were more likely to be older, male, and to have hypertension, diabetes, dyslipidemia, a habit of smoking, and a family history of stroke. Moreover, the carriers with a history of stroke had higher blood pressure, BMI, fasting glucose, serum glycated hemoglobin, uric acid, and creatinine levels than did the carriers without stroke. Conversely, in the carriers without a stroke history, total cholesterol, low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C) levels were higher, and triglyceride levels were lower than those with a stroke history.
Comparison between NOTCH3 R544C carriers with and without stroke.
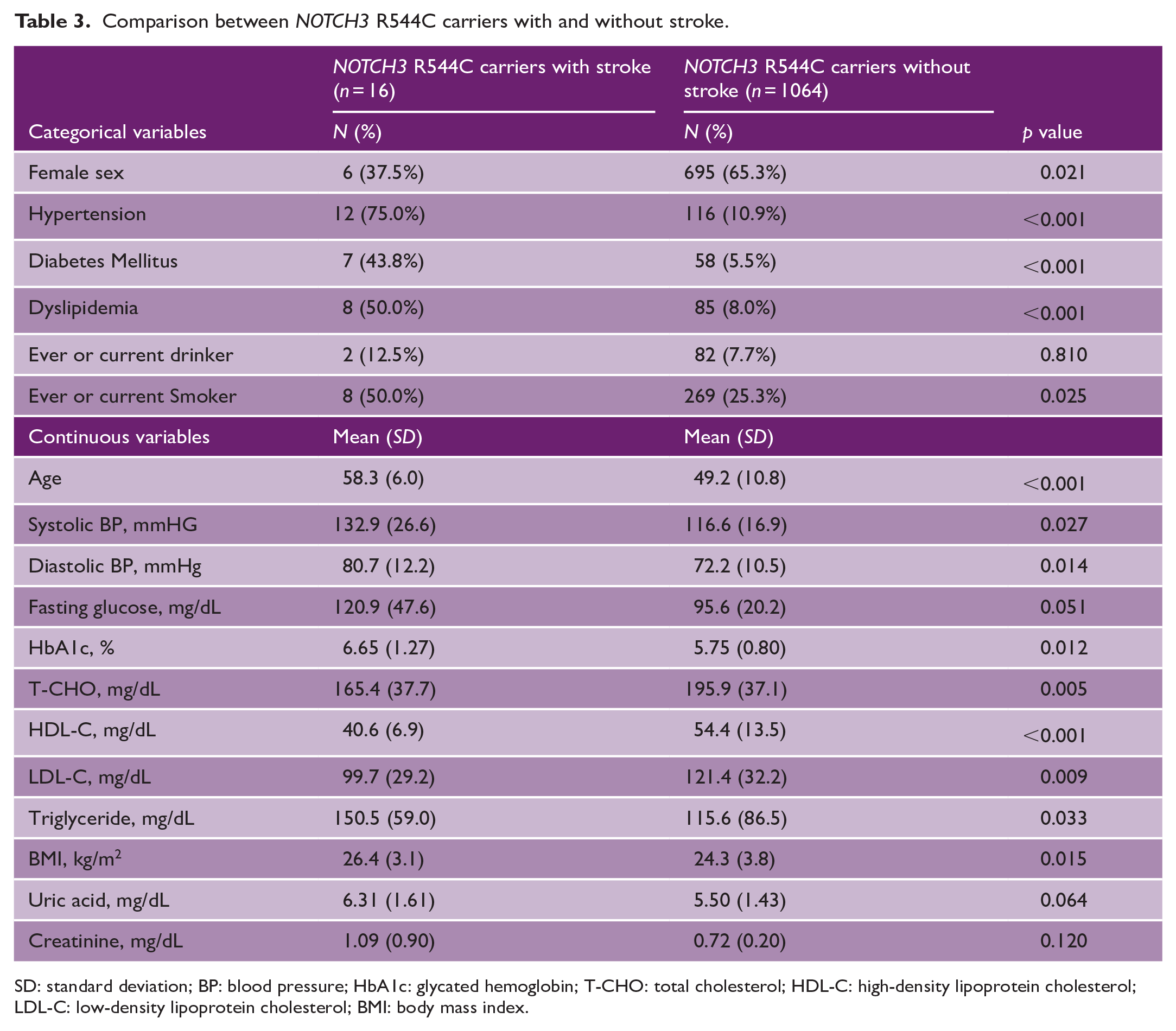
SD: standard deviation; BP: blood pressure; HbA1c: glycated hemoglobin; T-CHO: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; BMI: body mass index.
In our stepwise multivariate regression model including age, sex, hypertension, and diabetes as covariates, hypertension (adjusted OR: 11.28, 95% CI (3.54, 43.3), p < 0.001), and diabetes mellitus (adjusted OR: 4.10, 95% CI (1.31, 12.4), p = 0.012) remained considerably associated with stroke (Table 4).
Multivariable model of vascular risk factors for stroke in NOTCH3 R544C carriers.
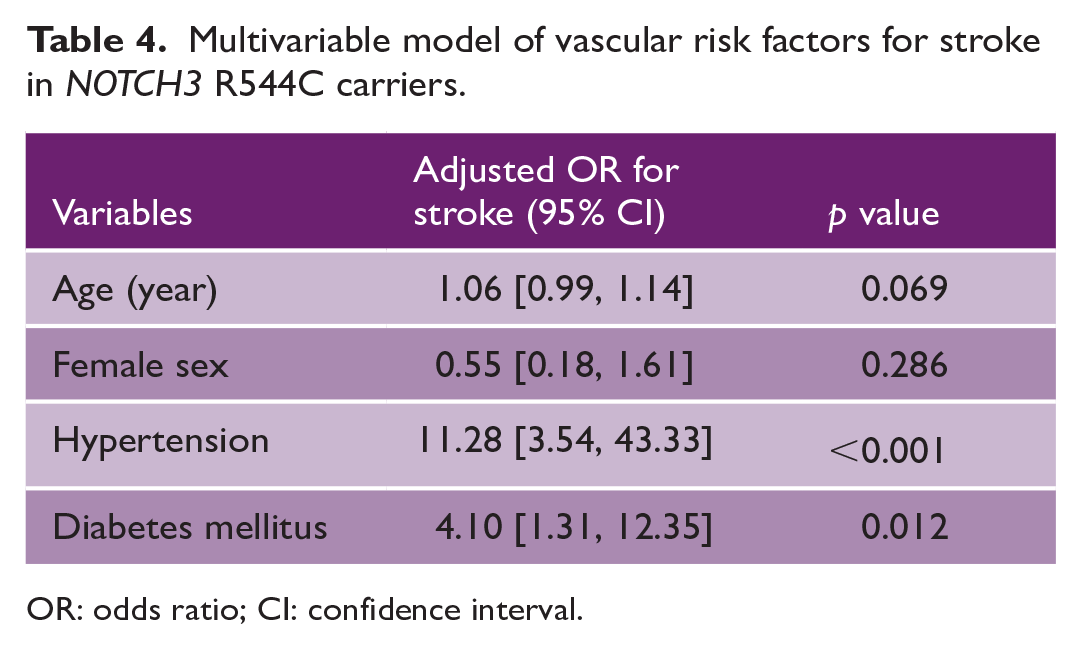
OR: odds ratio; CI: confidence interval.
Comparison of NOTCH3 R544C variant with standardized PRS for stroke
In the PRS applied to the Taiwan Biobank participants, the standardized PRS of stroke demonstrated an absolute risk increase of 1.15 (95% CI (0.92, 1.43), p = 0.22) per SD. The NOTCH3 R544C variant demonstrated a 6.74 SD equivalent increase in the PRS of stroke risk on average (log(ORstroke NOTCH3 R544C) − log (ORstroke PRS)). Conservative estimation (log(lower 95% CI ORstroke NOTCH3 R544C) − log (ORstroke PRS)) of 2.48 SD increase and the most conservative estimation (log(lower 95% CI ORstroke NOTCH3 R544C) − log (upper 95% CI ORstroke PRS)) of a 0.95 SD increase for stroke were noted (Figure 2).
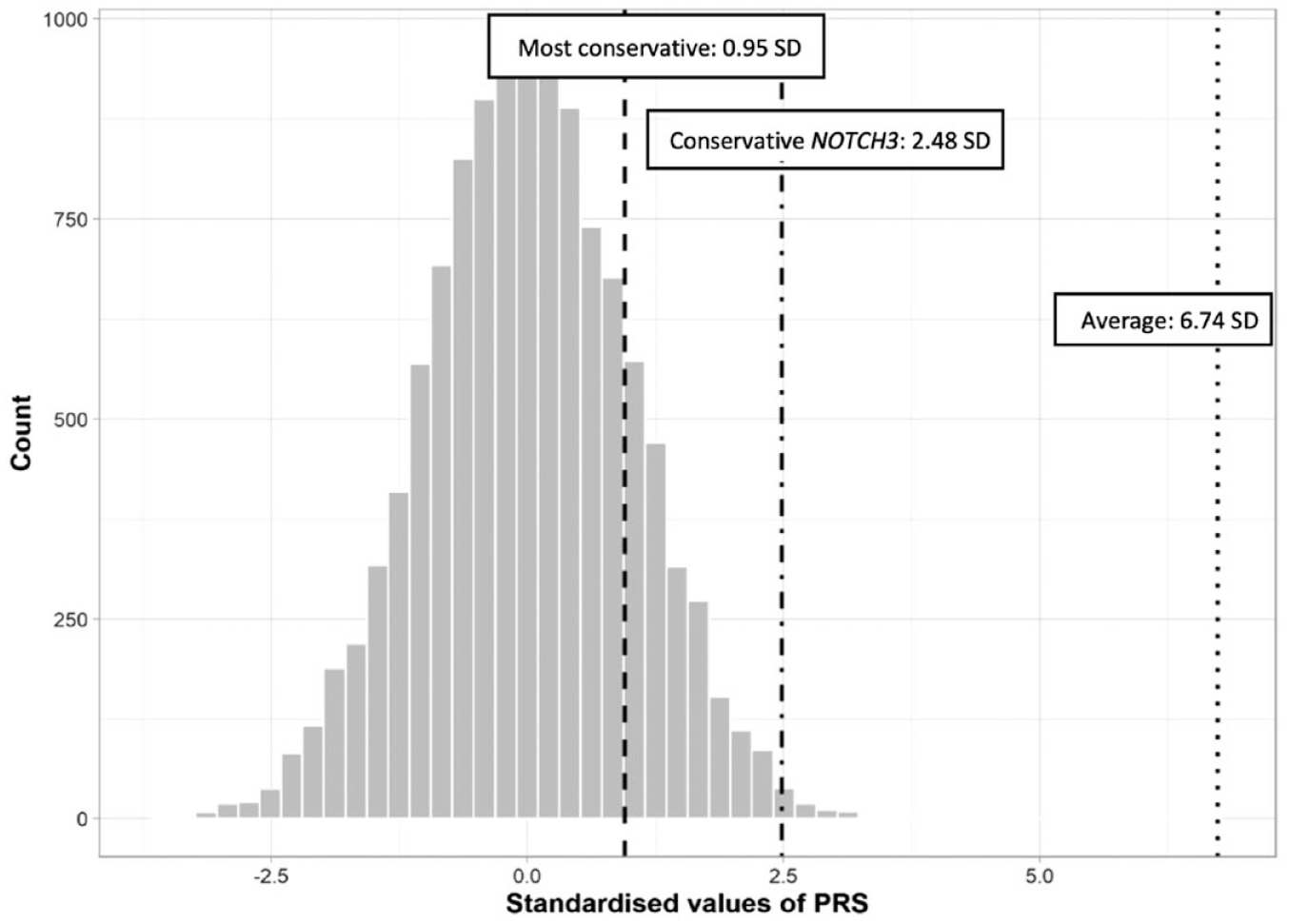
Contribution of the NOTCH3 R544C variant in polygenic risk score model for stroke. All 11,880 NOTCH3 R544C carriers and matched noncarriers (controls) were imputed into the metaGRS model from the UK Biobank. The distribution of individuals with standardized PRS units was presented in the bar chart. The dotted lines showed average (ORstroke NOTCH3 R544C vs. ORstroke PRS), conservative (lower limit 95% CI for ORstroke NOTCH3 R544C vs. ORstroke PRS), and most conservative (lower limit 95% CI for ORstroke NOTCH3 R544C vs. upper limit 95% CI for ORstroke PRS) estimates of PRS SD equivalents by carrying the NOTCH3 R544C variant. ORstroke: Odds ratio for stroke; SD: standard deviation; PRS: polygenic risk score.
Discussion
In this study, the majority of the Taiwanese NOTCH3 R544C carriers remained asymptomatic, with stroke (1.48%) and dementia (0.28%) being the most prominent phenotypes. In prior UK Biobank investigations, NOTCH3 EGFr 7-34 variant carriers had a low incidence of stroke (1.9%–5.4%) and dementia (0.9%).15,16 The phenotype spectrum of the NOTCH3 R544C variant was similar to previous reports,8,9,17 with migraine less characteristic in this genotype. 18 Aging, hypertension, and diabetes, which contribute to the vulnerability of the neurovascular unit, 19 led to a higher stroke penetrance in the current study.
Several epidemiological studies have supported the evidence of cardiovascular risk factors contributing to stroke in NOTCH3 variant carriers. In the Dutch registry study, male gender, hypertension, and diabetes influence incident age, stroke severity, and magnetic resonance imaging features. 6 In the UK Biobank, the NOTCH3 variant carriers with a high cardiovascular risk factor burden had a 7.73-fold risk of ischemic stroke than noncarriers with a low Framingham risk score. 5 Similarly, modifiable vascular risk factors such as hypertension and hypercholesterinemia were associated with stroke penetrance in a French CADASIL study. 20 To date, no randomized controlled trials have been conducted to examine whether reducing cardiovascular risk can effectively mitigate the risk of stroke in individuals with NOTCH3 variants.
The NOTCH3 variant landscape in the East Asian population was diverse, yet the NOTCH3 R544C variant (EGFr 13/14) and the NOTCH3 R607C variant (EGFr 15) top the most frequent variants. 21 In hospital-recruited studies of the NOTCH3 R544C carriers, sibling history of stroke and hypertension was related to intracerebral hemorrhage incidence.9,22 Given the unavailable information on the NOTCH3 R544C carriers with hemorrhagic stroke in Taiwan Biobank, it may be suggested that further studies of the cohort are needed with vascular risk contributions to specific stroke types. Among the symptomatic NOTCH3 R607C variant carriers, the prevalence of vascular risk factors was around one-third, but the relationship with stroke was not elaborated. 21 From a community-based perspective, our biobank study provides information on the cardiovascular risk factors impact on NOTCH3 variants in greater magnitude.
Contradictory lipid profiles were seen in this study, whereas HDL-C and LDL-C levels were both lower in those with stroke history. On the other hand, reported histories of dyslipidemia were higher in those with stroke history. This may be a result of previously medically treated patients reporting a medical history of having dyslipidemia. A previous study had shown impaired lipid metabolism to be a protective factor for cerebral microbleeds in CADASIL. 23 We assume it is farfetched to deduce the role of dyslipidemia in affecting stroke penetrance in the NOTCH3 variant carriers from the study. The current guideline was neither supportive nor against the treatment with commitment hyperlipidemia of these monogenic stroke carriers. 24
Carrying the NOTCH3 R544C variant in the Taiwan population remained crucial to developing stroke. We noted a stronger single SNP effect in the current standardized PRS model (average: 6.74 SD of stroke PRS unit) than in the UK Biobank (average: 4.85 SD of stroke PRS unit). 15 At the most conservative estimation, the risk of stroke associated with this variant was at least equivalent to one SD of the polygenic risk score. However, the risk scores obtained from the ischemic stroke PRS model should be interpreted with caution because the scores were originally derived for a Caucasian (UK) population and were applied to the Taiwan Biobank participants in the current study. 25
Strength and limitations
The current study has several strengths. First, we characterized the phenotype of 1080 NOTCH3 R544C carriers from a large-scale East Asian Biobank cohort. Second, we observed that cardiovascular risk factors significantly contributed to stroke risk in the NOTCH3 R544C carriers. Third, by applying a PRS model, we noted that the risk for stroke of the NOTCH3 R544C variant was comparable to the top polygenic risk scores in the Taiwan population.
Our study has several limitations. First, this is a cross-sectional analysis of the NOTCH3 R544C carriers from the Taiwan community. Therefore, selection bias in the severity of CADASIL phenotypes, propensity score matching variables, and NOTCH3 variants included in this study may exist. An association between cardiovascular risk factors and stroke may exist, but a causal inference cannot be drawn. Applicability across different NOTCH3 variants and East Asian ethnic groups may be limited. Second, there was limited access to neuroimaging, electronic health records for International Classification of Diseases (ICD) coding, and detailed data on cardiovascular risk-reducing medication from the current database. Third, few participants with dementia were seen in the matched cohort; thus, a large CI for dementia of the NOTCH3 R544C variant was observed. Furthermore, stroke cases in the Taiwan Biobank were insufficient for constructing our own PRS model. A detection bias may be present in the cross-ethnicity application when applying the PRS from the UK Biobank.
Conclusion
In conclusion, we investigated 1080 NOTCH3 R544C carriers from 110,000 participants enrolled in the Taiwan Biobank. These carriers had a high risk of stroke as well as a family history of stroke. Conventional vascular risk factors such as hypertension and diabetes were also noted to be associated with stroke risk in the carriers, and the need for adequate control of these risk factors should be emphasized.
Footnotes
Acknowledgements
The authors would like to thank the Taiwan Biobank for its assistance in providing and analyzing the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the Ministry of Science and Technology (Taiwan) (grant no. MOST110-2314-B002-155-MY3) and Academia Sinica (grant no. AS-GC-111-L04).
Data access statement
The anonymized material related to the publication may be shared upon request.
