Abstract
Rationale:
Mechanical thrombectomy (MT), the standard of care for acute ischemic stroke (AIS) secondary to large vessel occlusion (LVO), is generally not offered to patients with large baseline infarct (core). Recent studies demonstrated MT benefit in patients with anterior circulation stroke and large core (i.e. Alberta Stroke Program Early Computed Tomography Score, ASPECTS 3–5). However, its benefit in patients with the largest core (ASPECTS 0–2) remains unproven.
Aim:
To compare the efficacy and safety of MT plus best medical treatment (BMT) and of BMT alone in patients with ASPECTS 0–5 (baseline computed tomography (CT) or magnetic resonance imaging (MRI)) and anterior circulation LVO within 7 h of last-seen-well.
Sample size estimate:
To detect with a two-sided test at 5% significance level (80% power) a common odds ratio of 1.65 for 1-point reduction in the 90-day modified Rankin Scale (mRS) score in the MT + BMT arm versus BMT arm and to anticipate 10% of patients with missing primary endpoint, 450 patients are planned to be included by 36 centers in France, Spain, and the United States.
Methods and design:
LArge Stroke Therapy Evaluation (LASTE) is an international, multicenter, Prospectively Randomized into two parallel (1:1) arms, Open-label, with Blinded Endpoint (PROBE design) trial. Eligibility criteria are diagnosis of AIS within 6.5 h of last-seen-well (or negative fluid-attenuated inversion recovery (FLAIR) if unknown stroke onset time), ASPECTS 0–5 (ASPECTS 4–5 for ⩾80-year-old patients), and LVO in the anterior circulation (intracranial internal carotid artery (ICA) and M1 or M1–M2 segment of the middle cerebral artery (MCA)).
Study outcomes:
The primary endpoint is the day-90 mRS score distribution (shift analysis) with mRS categories 5 and 6 coalesced into one category. Secondary endpoints include day-180 mRS score, rates of 90-day and 180-day mRS score = 0–2 and 0–3, rate of decompressive craniectomy, the National Institutes of Health Stroke Scale (NIHSS) score change, revascularization and infarct volume growth at 24 h, and quality of life at day 90 and 180. Safety outcomes (90-day all-cause mortality, procedural complications, symptomatic intracerebral hemorrhage, and early NIHSS score worsening) are recorded. A dynamic balanced randomization (1:1) is used to distribute eligible patients into the experimental arm and control arm, by incorporating the center and these pre-specified factors: baseline ASPECTS (0–3 vs 4–5), age (⩽70 vs >70 years), baseline NIHSS (<20 vs ⩾20), intravenous thrombolysis (no vs yes), admission mode (Drip-and-Ship vs Mothership), occlusion site (intracranial ICA vs MCA—M1 or M1–M2), intravenous fibrinolysis (no vs yes), and last-seen-well to randomization time (0–4.5 vs >4.5–6.5 h).
Discussion:
The LASTE trial will determine MT efficacy and safety in patients with ASPECTS 0–5 and LVO in the anterior circulation.
Trial registration:
LASTE Trial NCT03811769.
Keywords
Introduction and rationale
Mechanical thrombectomy (MT) associated with best medical treatment (BMT) for acute ischemic stroke (AIS) with large vessel occlusion (LVO) is considered the standard of care, based on the results of several randomized controlled trials (RCTs).1–3 However, a large initial infarct core is an independent factor of poor outcome, and the Alberta Stroke Program Early Computed Tomography Score (ASPECTS) ⩽6 was an exclusion criterion in most of these trials. Consequently, until recently, unequivocal evidence of MT benefit in this population lacked, and it was believed that the overall treatment effect was mostly driven by patients with ASPECTS ⩾7. Therefore, current guidelines recommend MT only for patients with ASPECTS ⩾6.4,5 Recently, three RCTs demonstrated MT superiority compared to BMT in patients with LVO and large baseline infarct, based on volumetric infarct analysis on magnetic resonance imaging (MRI) or computed tomography (CT) perfusion or on ASPECTS = 3–5 on non-contrast CT or MRI.6–8 However, when this study was designed, no level 1 evidence existed on MT benefits in patients with ASPECTS 0–5. In the pre-specified secondary analysis of the Highly Effective Reperfusion evaluated in Multiple Endovascular Stroke Trials (HERMES) meta-analysis, MT effect was favorable in all baseline categories (all infarct sizes), even in patients with baseline ASPECTS 3–5 (a minority of randomized patients). Conversely, no conclusion could be drawn for patients with ASPECTS 0–2 due to their extremely small number. 9 Meta-analyses, before the publication of the three large core RCTs, suggested that MT was associated with higher odds of favorable outcome at 3 months in patients with large core (i.e. ASPECTS 0–5).10,11 Three randomized trials recently confirmed MT benefit in patients with large core, but none of them included patients with baseline ASPECTS 0–2.6-8 Therefore, MT effects in this patient population remain unknown. The aim of the LArge Stroke Therapy Evaluation (LASTE) trial is to demonstrate the superiority of MT plus BMT over BMT alone in patients with AIS due to anterior circulation LVO and ASPECTS ⩽5.
Methods
Design
LASTE is an international, multicenter, Prospectively Randomized into two parallel (1:1) arms, Open-label, with Blinded Endpoint (PROBE design) trial to demonstrate the superiority of immediate MT plus BMT (experimental arm), compared with BMT (control arm), to improve the 90-day clinical outcome in patients with large stroke (ASPECTS ⩽5) and LVO in the anterior circulation.
Study population—inclusion and exclusion criteria
Table 1 lists all inclusion and exclusion criteria. The main inclusion criteria are as follows: ⩾18-year-old patients with AIS, pre-stroke modified Rankin Scale (mRS) score ⩽1, baseline ASPECTS 0–5 (ASPECTS 4–5 for ⩾80-year-old patients), assessed with non-contrast CT or diffusion-weighted MRI, within 6.5 h of last-seen-well (or negative fluid-attenuated inversion recovery (FLAIR) if unknown stroke onset time), and LVO in the anterior circulation (intracranial internal carotid artery (ICA) and M1 or M1–M2 segment of the middle cerebral artery (MCA)) on computed tomography angiography (CTA) or magnetic resonance angiography (MRA). For transferred patients, new imaging is performed if the initial imaging was done >3 h before arrival. All patients or their legally authorized representatives provide a written informed consent or meet the criteria for emergency consent according to the accepted country-specific requirements, before enrollment.
Inclusion and exclusion criteria.
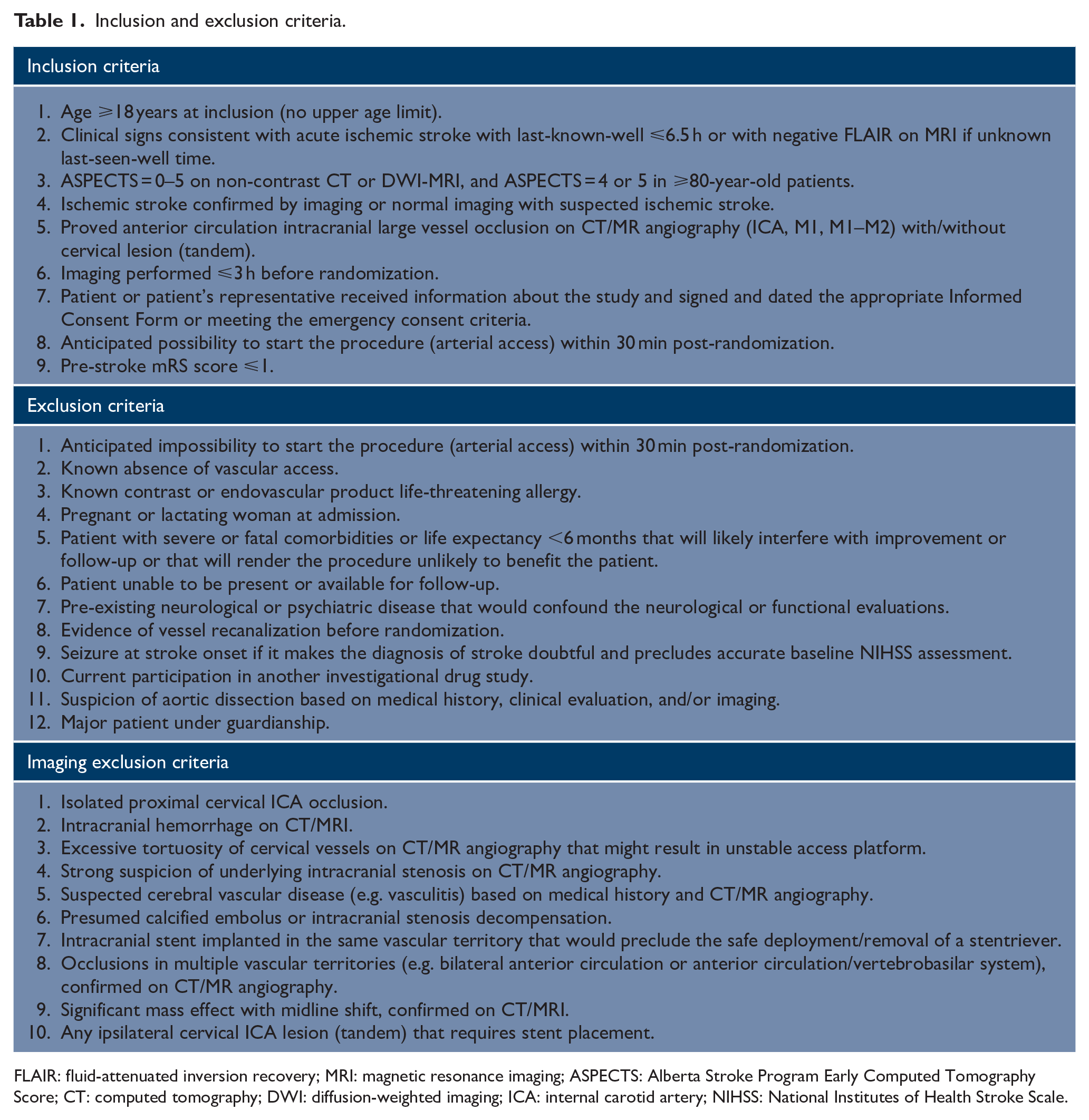
FLAIR: fluid-attenuated inversion recovery; MRI: magnetic resonance imaging; ASPECTS: Alberta Stroke Program Early Computed Tomography Score; CT: computed tomography; DWI: diffusion-weighted imaging; ICA: internal carotid artery; NIHSS: National Institutes of Health Stroke Scale.
Randomization
A dynamic balanced randomization (1:1) is used to assign eligible patients to the experimental arm or control arm, by incorporating the center and these pre-specified factors: age (⩽70 vs >70 years), occlusion site (intracranial ICA vs MCA—M1 or M1–M2), intravenous fibrinolysis (no vs yes), last-seen-well to randomization time (0–4.5 vs 4.5–6 h), ASPECTS (0–3 vs 4–5), the National Institutes of Health Stroke Scale (NIHSS) score (⩽20 vs >20), and admission mode (Drip-and-Ship vs Mothership).
Intervention
In the experimental arm, MT (started within 30 min post-randomization and limited to three attempts in a single vessel) is performed with any approved devices (stentriever or aspiration).
BMT
BMT is according to the current guideline–based best care. 4 Drugs (intravenous thrombolysis, anticoagulants, antiplatelet therapy) are administered at the treating physician’s discretion, but intra-arterial therapies may not be included.
Primary outcome
The primary outcome is the day-90 mRS score shift (mRS 5 and 6 are coalesced in the analysis) in the experimental group versus the control group.
Secondary outcomes
Secondary efficacy outcomes include mRS score shift (combining scores 5 and 6) at day 180, percentage of patients with day-90 and day-180 mRS scores ⩽2 and ⩽3, distribution of utility-weighted day-90 and day-180 mRS scores, NIHSS score changes, percentage of patients with secondary decompressive craniectomy, revascularization on MR/CT angiography, infarct volume growth at 24 h post-randomization, and quality of life at day 90 and day 180.
Safety outcomes include 90-day all-cause mortality, symptomatic intracerebral hemorrhage at 24 h (according to the Heidelberg classification), 12 rapid worsening (NIHSS score ⩾10), and serious procedure/device-related adverse events. The health economic evaluation endpoints (definition and analyses) will be presented in a separate document.
Data monitoring body
The Clinical Research and Innovation Department, Montpellier University Hospital (the study promoter) supervises the overall trial conduct monitoring. Data are collected in electronic case report forms by the site personnel.
All serious adverse events are analyzed by two members of the internal safety review committee. The executive committee and the promoter decide whether to continue, hold, or stop the study based on the recommendations by the safety monitoring board (one neurologist, one neuroradiologist, and one methodologist who do not participate in this RCT and are not affiliated to the sponsor).
Sample size estimates
The primary objective is to detect a shift in the day-90 mRS score distribution between the intervention and control arms. mRS scores of 5 and 6 are combined on the basis of the hypothesis that MT will improve the status of patients with mRS <5, but not with mRS 5 and 6. A sample size of 225 patients/group will have 80% power to detect, with a two-sided test at 5% significance level, a common odds ratio for 1-point improvement in the 90-day mRS score of 1.65 for the experimental arm versus the control arm by assuming the following 90-day mRS score distribution in the control group: 0 (1.9%), 1 (11.5%), 2 (13.5%), 3 (15.4%), 4 (19.4%), and 5 + 6 (36.5%). This sample size takes into account 10% of patients with missing primary endpoint (dropouts or lost to follow-up).
Statistical analysis
Statistical analyses will be independently performed with SAS 9.4 (SAS Institute Inc., Cary, NC, USA). The statistical analyses for the primary and secondary outcomes are listed in the statistical analysis plan (Supplemental Appendix).
The primary efficacy outcome analysis will be done using a mixed ordinal logistic regression model adjusted for factors considered in the minimization randomization algorithm: baseline ASPECTS (0–3 vs 4–5), last-seen-well to randomization time (0–4.5 vs >4.5–6.5 h), age (<70 vs ⩾70 years), baseline NIHSS score (<20 vs ⩾20), intravenous thrombolysis (no vs yes) and admission mode (Drip-and-Ship vs Mothership) as fixed effects, and center as random effect to account for the correlation between patients of the same center (cluster effect). Common proportional odds ratios for 1-point improvement in the mRS score will be derived from this model as treatment effect size (intervention vs control) with their 95% confidence interval. In case of deviations in the proportional odds assumption, the 90-day mRS score will be compared with the Mann–Whitney U and Wilcoxon–Mann–Whitney tests. Generalized odds ratios will be calculated as effect size. Missing data for the primary endpoint (day-90 mRS score) will be handled using a multiple imputation procedure and a regression switching approach (chained equation with n = 20 imputations), with the main baseline characteristics and treatment. 13
Study organization and funding
The study promoter monitors the overall trial conduct. The executive committee (four principal investigators from France and the United States) is responsible for the overall study design, interpretation, and supervision, including amendments and provides guidance during weekly videoconferences. The industry sponsors (Stryker Neurovascular, MicroVention, Medtronic, Cerenovus, and Balt Extrusion) are not involved in the study design, supervision, and publications.
Blinding
Treatment allocation is open; it is impractical to offer a sham endovascular procedure.
Each site will choose an evaluator blinded to randomization and not involved in patient care for the day-90 (primary endpoint) and day-180 mRS scoring. An independent core laboratory, blinded to randomization, will review the neuroimaging data (baseline and secondary endpoints).
Discussion
Among the RCT on MT for stroke, LASTE is unique because it examines MT safety and efficacy (vs BMT) in patients presenting with large infarcts (baseline ASPECTS 0–5), and particularly the largest infarcts (ASPECTS 0–2). The recently published large core RCTs are expected to change patient selection paradigms, leading to offer MT also to patients with large baseline infarcts who were not considered to be candidates for MT.6–8 By including patients with large core regardless of its size, LASTE aim is to determine whether in the early time window (0–7 h), knowledge of core size is relevant for patient selection. Although core size remains a strong prognostic factor, the demonstration of MT benefit in LASTE would imply that in the early time window, it does not constitute a treatment effect modifier. Moreover, LASTE will hopefully clarify the contentious issue of reperfusion potential harm in patients with the largest baseline infarcts.
Summary and conclusion
Evidence of MT benefit in patients with ASPECTS 0–5 and LVO is lacking. This large trial will provide information on whether MT plus BMT can improve their clinical outcome compared with BMT alone.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231191033 – Supplemental material for Evaluation of acute mechanical revascularization in large stroke (ASPECTS ⩽5) and large vessel occlusion within 7 h of last-seen-well: The LASTE multicenter, randomized, clinical trial protocol
Supplemental material, sj-docx-1-wso-10.1177_17474930231191033 for Evaluation of acute mechanical revascularization in large stroke (ASPECTS ⩽5) and large vessel occlusion within 7 h of last-seen-well: The LASTE multicenter, randomized, clinical trial protocol by Vincent Costalat, Bertrand Lapergue, JF Albucher, Julien Labreuche, Hilde Henon, Benjamin Gory, Igor Sibon, Grégoire Boulouis, Christophe Cognard, Nasreddine Nouri, Sebastien Richard, Gauthier Marnat, Federico Di Maria, Mariam Annan, Alain Duhamel, Federico Cagnazzo, Tudor Jovin and Caroline Arquizan in International Journal of Stroke
Supplemental Material
sj-docx-2-wso-10.1177_17474930231191033 – Supplemental material for Evaluation of acute mechanical revascularization in large stroke (ASPECTS ⩽5) and large vessel occlusion within 7 h of last-seen-well: The LASTE multicenter, randomized, clinical trial protocol
Supplemental material, sj-docx-2-wso-10.1177_17474930231191033 for Evaluation of acute mechanical revascularization in large stroke (ASPECTS ⩽5) and large vessel occlusion within 7 h of last-seen-well: The LASTE multicenter, randomized, clinical trial protocol by Vincent Costalat, Bertrand Lapergue, JF Albucher, Julien Labreuche, Hilde Henon, Benjamin Gory, Igor Sibon, Grégoire Boulouis, Christophe Cognard, Nasreddine Nouri, Sebastien Richard, Gauthier Marnat, Federico Di Maria, Mariam Annan, Alain Duhamel, Federico Cagnazzo, Tudor Jovin and Caroline Arquizan in International Journal of Stroke
Supplemental Material
sj-docx-3-wso-10.1177_17474930231191033 – Supplemental material for Evaluation of acute mechanical revascularization in large stroke (ASPECTS ⩽5) and large vessel occlusion within 7 h of last-seen-well: The LASTE multicenter, randomized, clinical trial protocol
Supplemental material, sj-docx-3-wso-10.1177_17474930231191033 for Evaluation of acute mechanical revascularization in large stroke (ASPECTS ⩽5) and large vessel occlusion within 7 h of last-seen-well: The LASTE multicenter, randomized, clinical trial protocol by Vincent Costalat, Bertrand Lapergue, JF Albucher, Julien Labreuche, Hilde Henon, Benjamin Gory, Igor Sibon, Grégoire Boulouis, Christophe Cognard, Nasreddine Nouri, Sebastien Richard, Gauthier Marnat, Federico Di Maria, Mariam Annan, Alain Duhamel, Federico Cagnazzo, Tudor Jovin and Caroline Arquizan in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.F.A., J.L., H.H., G.B., N.N., S.R., F.D.M, M.A., A.D., F.C., and C.A. declare no conflict of interest. V.C. received educational grant, consultation fees, research grant from Medtronic, Stryker, MicroVention, Cerenovus, and Balt; educational grant and consultation fees from phenox. B.L. received a research grant from MicroVention, Balt, and phenox. C.C. received consultation fees as consultant for Medtronic, MicroVention, Stryker, MIVI, and Cerenovus. B.G. received consultation fees from MIVI, Medtronic, MicroVention, and Penumbra. I.S. received consultation fees from Medtronic. G.M. received consultation fees from MicroVention, Stryker, and Balt; paid lectures from Medtronic, Johnson & Johnson, and Phenox. T.J. is advisor and investor for Anaconda, Route92, Viz.ai, FreeOx, Kandu, and Methinks. He received personal fees in his role on Cerenovus DSMB and steering committee and on Contego Medical screening committee. He received grant support from Medtronic and Stryker Neurovascular.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
