Abstract
Background:
Whether thrombolysis improves outcomes in non-arteritic central retinal artery occlusion (naCRAO) is uncertain. We aimed to evaluate the rate of visual recovery after intra-venous thrombolysis (IVT) or intra-arterial thrombolysis (IAT) administration of tissue plasminogen activator (tPA) or urokinase among patients with naCRAO and explore the parameters affecting the final visual acuity (VA).
Aim:
We systematically searched six databases. Logarithm of the minimum angle of resolution (logMAR) and VA of ⩾20/100 were used to quantify visual recovery. To explore the role of other factors on visual recovery, we defined two models for studies with aggregated data (designs 1 and 2) and 16 models for individual participant data (IPD, models 1–16).
Summary of Review:
We included data from 771 patients out of 72 publications in nine languages. Visual improvement for ⩾0.3 logMAR was reported in 74.3% of patients who received IVT-tPA within 4.5 h (CI: 60.9–86.0%; unadjusted rate: 73.2%) and 60.0% of those who received IAT-tPA within 24 h (CI: 49.1–70.5%; unadjusted rate: 59.6%). VA of ⩾20/100 was observed among 39.0% of patients after IVT-tPA within 4.5 h and 21.9% of those with IAT-tPA within 24 h. IPD models highlighted the association between improved visual outcomes and VA at presentation, at least 2 weeks follow-up before reporting the final VA, antiplatelet therapy, and shorter symptom onset to thrombolysis window.
Conclusion:
Early thrombolytic therapy with tPA is associated with enhanced visual recovery in naCRAO. Future studies should refine the optimum time window for thrombolysis in naCRAO.
Keywords
Introduction
Central retinal artery occlusion (CRAO) acutely compromises blood flow to the inner layers of the retina, leading to sudden painless and usually disabling monocular visual loss. Due to the poor visual recovery rates in the disease natural history1,2 and the absence of effective conservative therapies available to date,3 –5 the management of CRAO remained an important unanswered question. 6 As thrombolytic therapies are effective and constitute standard care in patients with cerebral ischemic strokes, 7 and retinal ischemia is a sub-type of ischemic stroke, 5 intra-venous thrombolysis (IVT) or intra-arterial thrombolysis (IAT) administration of various thrombolytic agents may also be effective in acute CRAO. 8 However, the lack of large-scale randomized clinical trials and heterogeneity among studies makes the effectiveness of thrombolytic therapies uncertain.9,10
Only six randomized controlled trials including 223 acute non-arteritic CRAO (naCRAO) patients were reported so far to compare various proposed interventions, and meta-analysis could not be conducted due to study heterogeneity. 11 Previous systematic reviews and meta-analyses appraising the visual recovery rates after thrombolysis in CRAO included observational studies and case series to try to derive a conclusion.12 –15 They compared the visual recovery rate among CRAO patients treated with thrombolysis versus non-thrombolytic therapies. However, the conservative therapies varied significantly among studies and many patients in the thrombolytic cohort concomitantly received them as well.14 –17 In addition, the eligibility window for thrombolysis, the severity of the visual loss, demographics, and various vascular comorbidities among different cohorts confounded the results and limited their conclusions.12,15
In 2020, a patient-level meta-analysis 15 visual recovery was conducted among CRAO patients. Although the authors intended to include publications presenting patients who received any IV thrombolytic agents within 48 h of symptom onset, meta-analysis was limited to English reports of patients who received IVT-tPA within 6 h. In 2021, an American Heart Association/American Stroke Association (AHA/ASA) scientific statement called for early recognition of patients with CRAO and proposed a treatment protocol with tissue plasminogen activator (tPA), either IVT or IAT, in select naCRAO patients that present in early time windows. 6
To further guide current clinical practice, we systematically reviewed all the available reports on the efficacy of thrombolysis in naCRAO at both individual participant data (IPD) level and aggregated data (AD) level. In addition, we explored the impact of different factors, including time, route and agent of thrombolysis, and additional therapeutic measures, such as heparin and antiplatelet administration, on the visual recovery of patients with naCRAO who received thrombolysis.
Methods
Search strategy
This systematic review and meta-analysis was conducted and reported following the Enhancing the Quality and Transparency of Health Research guidelines—Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement (Supplementary eDocument 1), 18 AHRQ Series on Complex Intervention Systematic Reviews (PRISMA-CI), 19 the PRISMA-IPD statement for inclusion of individual-level data, 20 and Meta-Analysis of Observational Studies in Epidemiology (Supplementary eDocument 2). 21 We limited this study to thrombolysis by tPA (the thrombolytic of choice by the AHA/ASA guideline) 7 or urokinase (UK, widely practiced in Eastern Countries12,22–24). The search protocol was designed as [retinal artery occlusion] AND [(Intraarterial OR intravenous) (thrombolytic therapy OR urokinase OR tissue plasminogen activator)], with the use of thesauri—MeSH search tags (mh) and (majr)—truncation, wild cards, and different Boolean operators. We searched potentially eligible reports through PubMed, Cochrane Library, ScienceDirect via Elsevier, International Clinical Trials Registry Platform, ClinicalTrials.gov, and Trip Medical Database until 15 October 2022, without any filter on study design, document type, or language. PubMed alerts were used to capture the latest published articles until 28 February 2023. The authors searched keywords in French, German, Arabic, and Persian to include published studies with titles and abstracts in languages other than English that were afterward translated into English for study selection and data abstraction. In addition, we attempted cross-referencing and citation tracking of retrieved reports in multiple languages via Google Scholar. We augmented the search results by manually searching the investigators who have registered trials in ClinicalTrials.gov and communicating with selected authors.7,12,22 –24
Eligibility criteria
We included patients with acute occlusion of the central retinal artery or branch retinal artery who received IVT or IAT with either tPA or UK. We excluded the following: (1) acute retinal arterial occlusion due to arteritis, genetic pathologies, and chronic inflammatory disorders, (2) combined arterial and venous occlusion, (3) unclear or multiple interventions, (4) use of thrombolytics other than tPA and UK, (5) continuous infusion of the thrombolytic agent for few days, and (6) without data on baseline (before thrombolysis) and follow-up (after interventions) visual acuity (VA). In the systematic review, patients were included irrespective of the interval between symptom onset to intervention and the severity of vision loss on presentation. However, sensitivity analyses were limited to the patients with CRAO and VA of 20/200 or worse on presentation who received thrombolysis within 24 h of vision loss.
Outcome measures
We recorded all available formats and time frames of baseline and follow-up VA. For statistical analysis, all values were converted to the logarithm of the minimum angle of resolution (logMAR) with the following equation: logMAR = −LOG10 (Fraction of Snellen Scale). LogMAR equivalencies for profound low vision were approximated based on the work of Lange et al. 25 We considered the closest logMAR value to the event as the baseline and the latest available value as a follow-up VA measure. The effect of thrombolytic therapy on visual recovery (primary outcome measure) was quantified threefold: (1) VA improvement of ⩾0.1 logMAR (one Snellen line), (2) VA improvement of ⩾0.3logMAR (three Snellen lines), and (3) mean change (MC) in logMAR VA between baseline and follow-up. We also dichotomized visual recovery as a final VA of 20/100 or better (secondary outcome measure). We defined adverse effects of thrombolytic therapy as incident intra-ocular hemorrhage, symptomatic intracranial hemorrhage, ischemic stroke, transient ischemic attack, and death.
Data abstraction and quality assessment
Details of abstract screening and data collection and abstraction are available in Supplementary sDocument 5. Two independent reviewers (S.S. and R.B.S.) conducted the quality assessment and critical appraisal of cohorts by Risk of Bias in Non-randomized Studies of Interventions tool26,27 and case reports using CARE guidelines for case reports. 28 Publication bias was visualized with funnel plots and the degree of bias was measured by the Egger bias test. 29 We adjusted the effect of significant publication bias on outcomes by using a trim and fill algorithm 30 and the inclusion of quality scores into analyses. 31 Potential outliers were excluded from further analyses after a full review of each study methodology.
Data synthesis and analyses
Aggregated data
In addition to the available reported AD, we calculated and included the summary of cohorts of five or more patients with IPD who met the eligibility criteria for each meta-analysis.13,14,16 We defined two study designs by including: (1) the route and agent of thrombolytic therapy (AD-Design 1: IVT-tPA vs IVT-UK vs IAT-tPA vs IAT-UK) and (2) the route of thrombolysis alone, regardless of the agent (AD-Design 2: IVT vs IAT). The heterogeneity among the studies was evaluated with the Cochran Q test (χ2 test for heterogeneity). The percentage of total heterogeneity to total variability was demonstrated by the I2 statistic and its 95% CI. An I2 statistic greater than 60% with a Q test with p < 0.10 was considered a significant statistical heterogeneity. To further assess the possible impact of other moderators on outcomes in each AD-Design, we performed a meta-regression including the following parameters: age (⩽55 vs >56 years), 32 gender, time from visual loss to thrombolysis, heparin administration (after the thrombolysis), antiplatelet therapy, the language of publication (English vs non-English), and having 2 or more weeks of follow-up before final VA. We used two-step random-effects models with double arcsine transformations and DerSimonian-Laird estimator for estimating ⩾0.1 logMAR, ⩾0.3 logMAR, and ⩾20/100 VA improvement. We used raw MC and standardized MC using change score standardization (SMCC) to assess the MC in logMAR after the intervention.33,34 Omnibus test was utilized to compare each meta-regression model versus null hypotheses.
Individual participant data
To better explore the predictors of primary outcome measures, we used IPD to define 16 models (Table 1 and Supplementary sDocument 6). IPD models included patients with VA > 20/200 at presentation, CRAO, and branch retinal occlusion (BRAO). All models were adjusted for patients’ age (⩽55 vs >56 years). 32
Summary of individual participants data models.
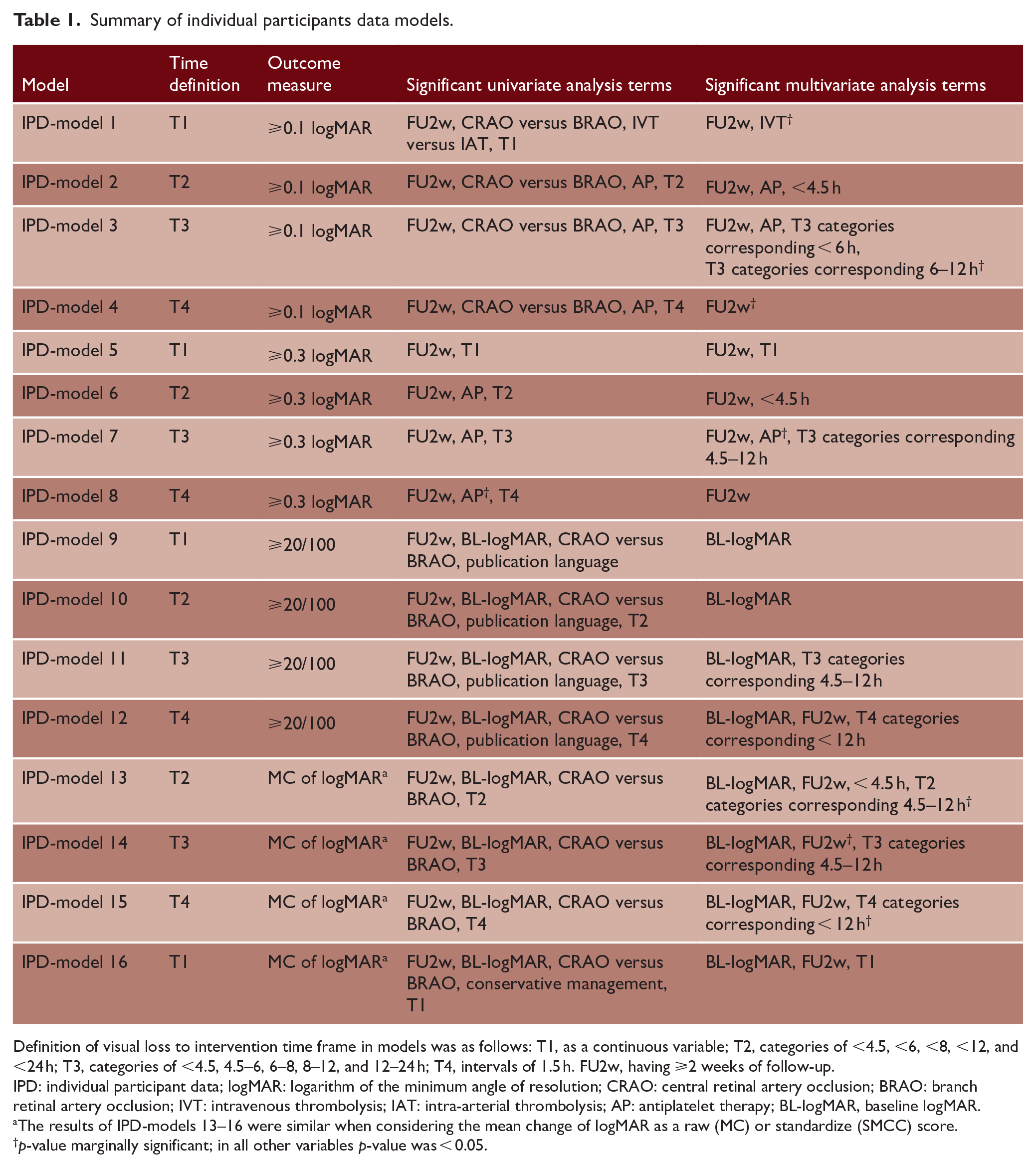
Definition of visual loss to intervention time frame in models was as follows: T1, as a continuous variable; T2, categories of <4.5, <6, <8, <12, and <24 h; T3, categories of <4.5, 4.5–6, 6–8, 8–12, and 12–24 h; T4, intervals of 1.5 h. FU2w, having ⩾2 weeks of follow-up.
IPD: individual participant data; logMAR: logarithm of the minimum angle of resolution; CRAO: central retinal artery occlusion; BRAO: branch retinal artery occlusion; IVT: intravenous thrombolysis; IAT: intra-arterial thrombolysis; AP: antiplatelet therapy; BL-logMAR, baseline logMAR.
The results of IPD-models 13–16 were similar when considering the mean change of logMAR as a raw (MC) or standardize (SMCC) score.
p-value marginally significant; in all other variables p-value was < 0.05.
For the I2 statistic, p < 0.1, univariate regression models p < 0.15, and in other analyses two-sided p < 0.05 were considered statistically significant. Data were synthesized by the “metafor” package, R program, version 4.2.2. Data manipulation was conducted via Python version 3.9.12.
Results
Literature review and study selection
Among 1153 identified studies through six databases search, 72 reports were included in the systematic review and 63 in the AD and IPD meta-analyses (Figure 1). Supplementary sTable 1 presents the study selection details for the systematic review and meta-analysis.
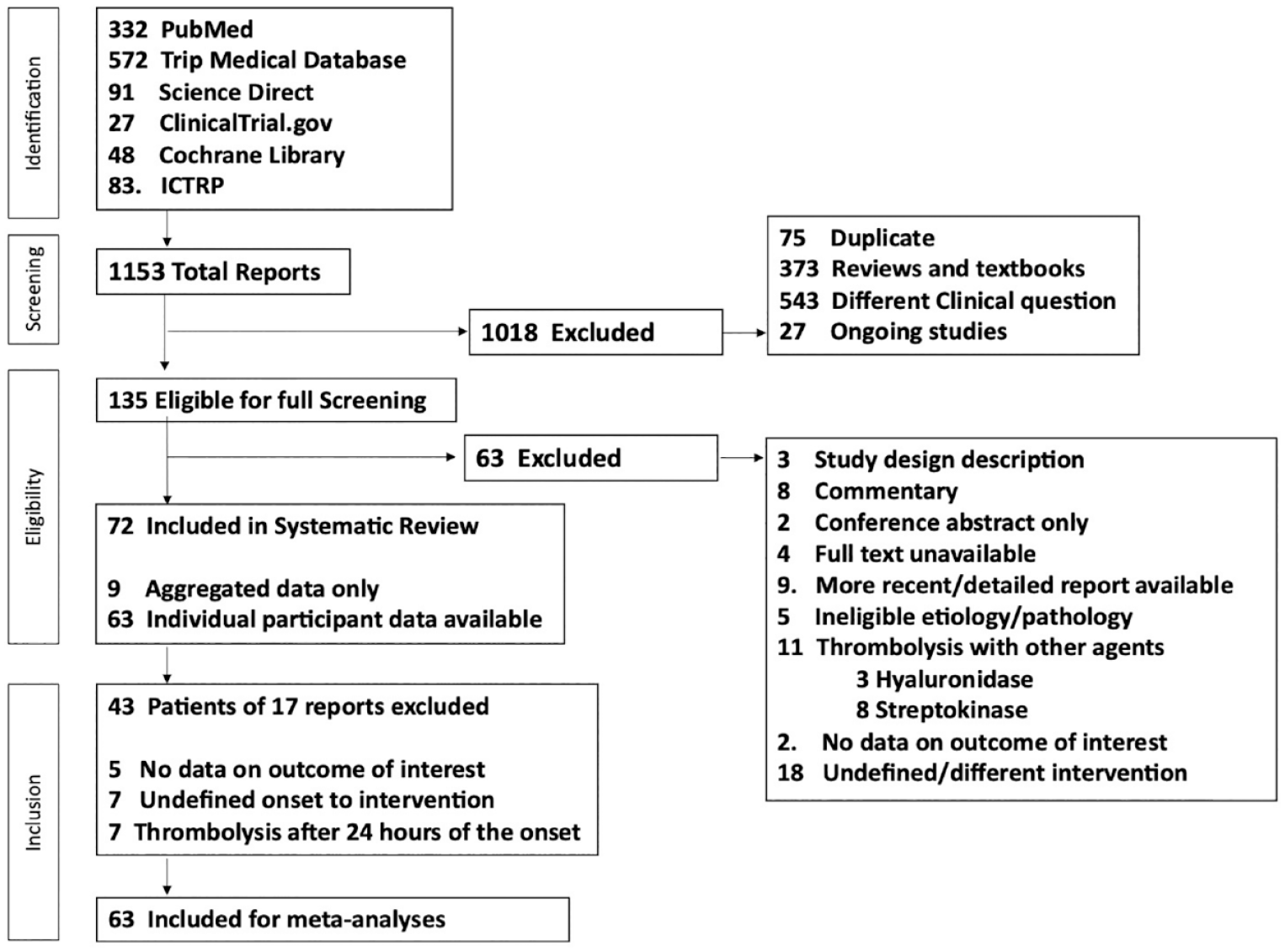
Flowchart indicating the process of database screening and study selection for the systematic review and meta-analyses. Please note that some studies were ineligible due to more than one criterion.
Patients’ characteristics
In the systematic review, we recorded data from 771 patients deriving from 72 publications, including 9 articles with AD (338 patients) and 63 articles with IPD (433 patients) reported in 9 languages (Supplementary sDocument 7). Age was available for 636 patients (82.5%, mean age 52.4 ± 15.03 years), and 56.8% (318 out of 560) were female. All CRAOs were monocular, and 55.4% (124 out of 224) affected the right eye. BRAO was observed in 19 patients (2.5%).
A total of 43 patients (17 reports) were excluded from further analyses due to receiving multiple interventions, 35 inadequate data on the outcome of interest,36 –40 unavailable onset to intervention time,36,40 –45 and visual loss onset to thrombolysis > 24 h 45 –51 (Supplementary sTable 1). Among the included patients in AD and IPD meta-analyses (N = 728, 63 articles; Supplementary sTable 2), the intervention was IAT-tPA in 206 (28.3%), IAT-UK in 331 (45.5%), IVT-tPA in 171 (23.5%), and IVT-UK in 20 (2.7%). Figure 2 summarizes the visual outcomes among patients who received thrombolysis within 24 h (Supplementary sFigure 1, UK and Supplementary sFigure 2, ptPA and UK).
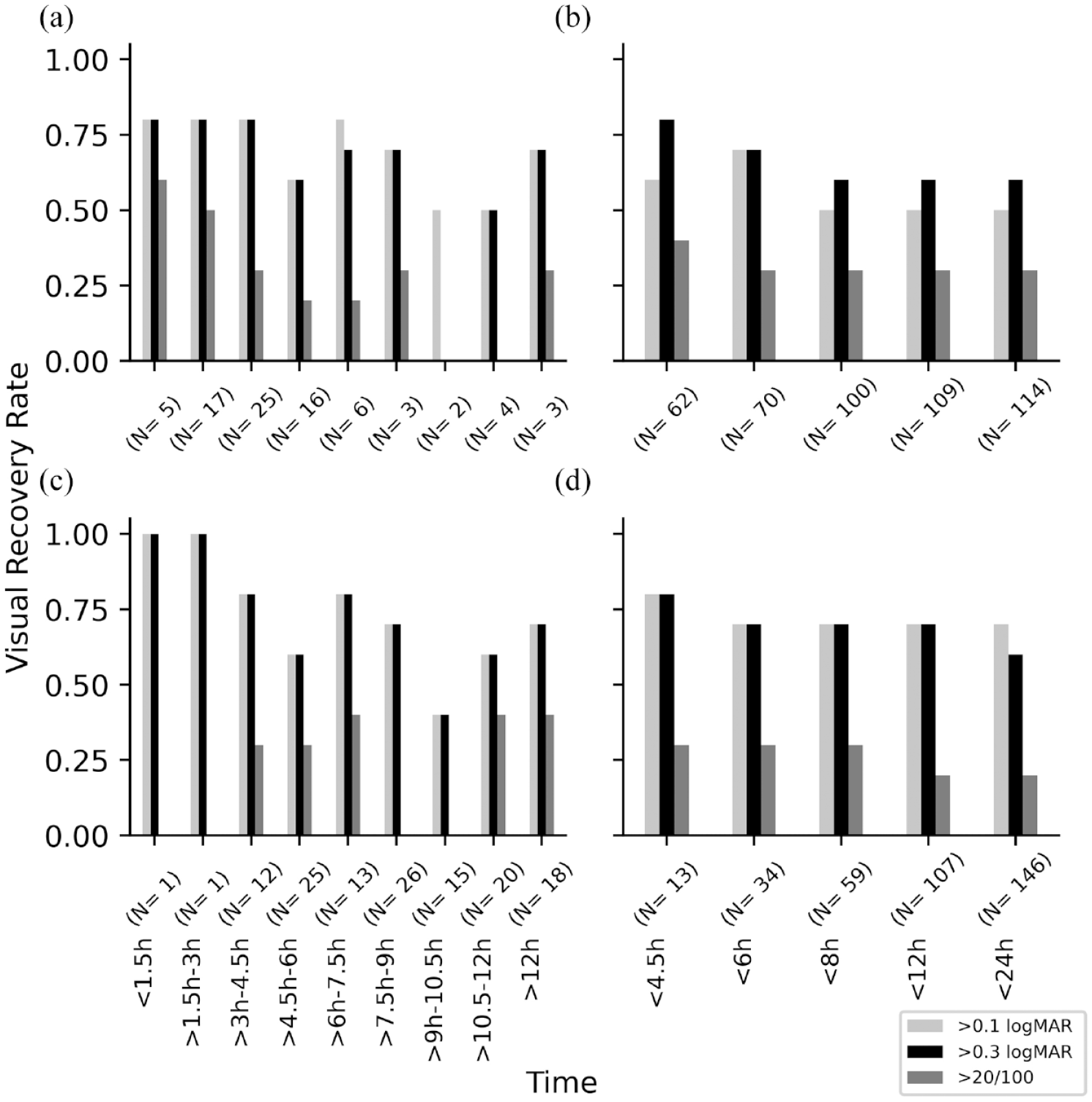
Visual recovery rate among patients with CRAO who presented with a VA of 20/200 or less and received tPA within 24 h of visual loss. Plots A and B are limited to individual participants data with available exact minutes between symptom onset and intervention. Plots C and D include all available data presenting the interval between visual loss to intervention. (a, c) Intra-venous and (b, d) intra-arterial.
Aggregated-level data analyses
AD sensitivity meta-analyses included CRAO patients with a baseline VA of 20/200 or worse, excluding two studies with a high risk of bias and substantial influence on outcomes (N = 602).52,53
In AD-Design 1, among patients who received IVT-tPA within 4.5 h (Figure 3), 80.9% (CI: 65.7–93.0%; unadjusted rate: 80.0%) had VA improvement by ⩾0.1 logMAR, 74.3% (CI: 60.9–86.0%; unadjusted rate: 73.2%) had ⩾0.3 logMAR VA improvement, and 39.0% (CI: 25.6–53.1%; unadjusted rate: 39.2%) had a final VA of ⩾20/100. SMCC of 77.1% logMAR (CI: 47.1–107.1%) was reported after receiving IVT-tPA within 4.5 h.
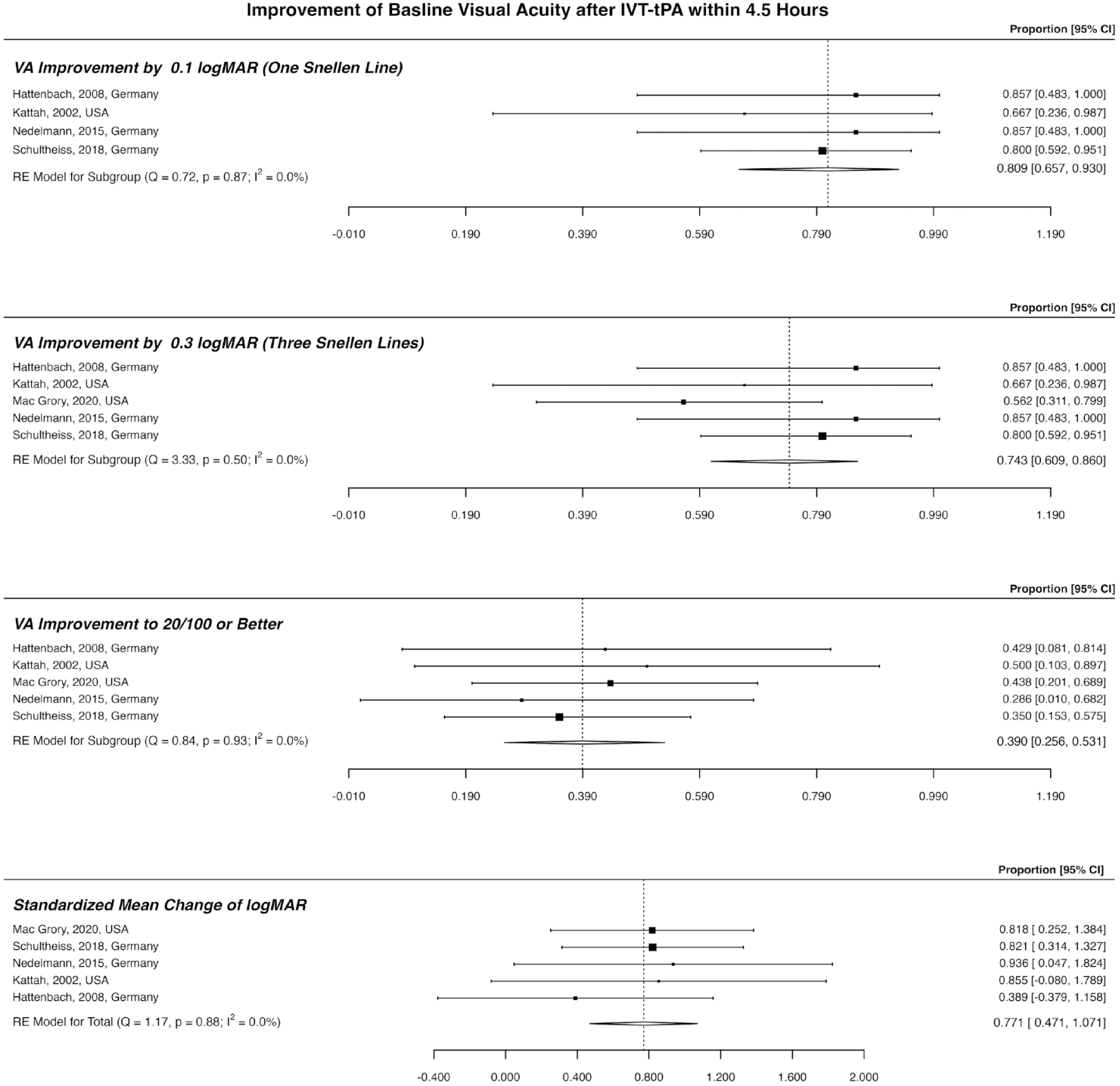
Outcome of patients who received IVT-tPA within 4.5 h of visual loss. This plot includes patients with central retinal artery occlusion and presenting a VA of 20/200 or less. IVT-tPA: intra-venous tissue plasminogen activator; VA: visual acuity.
Among patients who received IAT-tPA within 24 h (Figure 4), 66.8% experienced ⩾0.1 logMAR improvement (CI: 58.1–75.0%; unadjusted rate: 66.2%), 60.0% (CI: 49.1–70.5%; unadjusted rate: 59.6%) had ⩾0.3 logMAR VA improvement, 21.9% (CI: 14.1–30.7%; unadjusted rate: 23.5%) had a final VA of 20/100 or more, and SMCC was 76.9% (CI: 43.6–110.2%). Outcome of patients who received IAT-UK within 24 h is available in Supplementary sFigure 3.
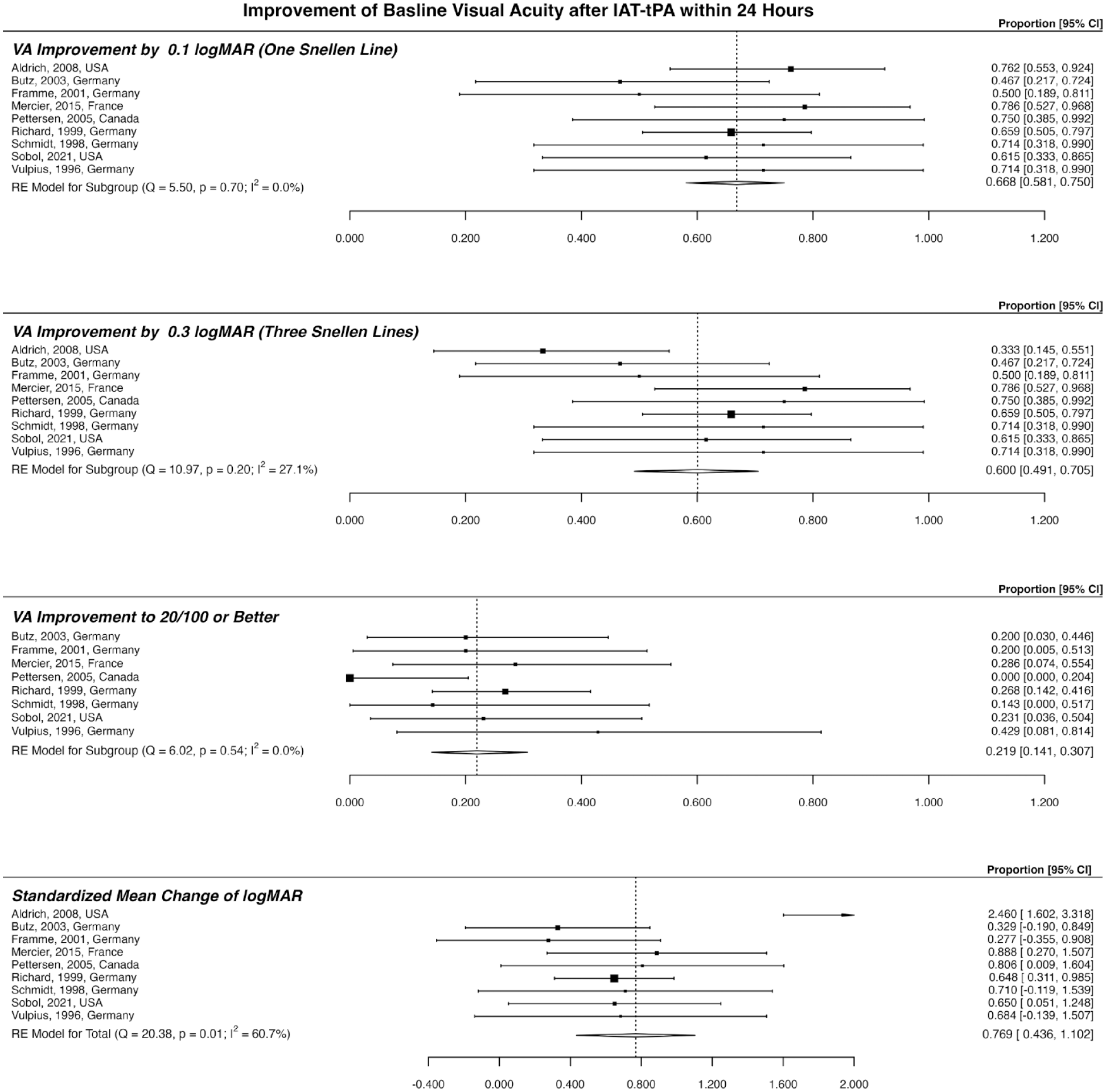
Outcome of patients who received IAT-tPA within 24 h of visual loss. This plot includes patients with central retinal artery occlusion and presenting a VA of 20/200 or less. IAT-tPA: intra-arterial tissue plasminogen activator; VA: visual acuity.
In AD-Design 2 (Supplementary sFigures 4 to 8), among those treated with IAT within 24 h, 61.9% (CI: 53.8–69.8%; unadjusted rate: 60.5%) had ⩾0.1 logMAR improvement, 53.8% (CI: 39.3–68.1%; unadjusted rate: 48.4%) had ⩾0.3 logMAR improvement, 25.6% (CI: 16.6–35.6%; unadjusted rate: 29.3%) had a final VA of 20/200 or better, and SMCC was 66.4% (CI: 48.8–84.1%).
Independent participant data analyses
To better explore the impact of moderators on the final VA, 16 IPD models adjusted for age were reported. As expected, a shorter interval from visual loss to thrombolysis predicted a better outcome (<12 h in most models). Except for IPD-models 9–11 (VA improvement ⩾20/100), having at least 2 weeks follow-up before reporting the final VA was predictive of outcome. In IPD-models 9–16, baseline logMAR was suggestive of outcome (i.e. VA improvement ⩾20/100 and MC of logMAR). Antiplatelet therapy was an independent predictor of ⩾0.1 logMAR improvement (IPD-models 1–4), with marginal significance in ⩾0.3 logMAR visual recovery.
Complications of thrombolytic therapy
Intra-ocular hemorrhage was observed in 4 (0.5%) patients (all IAT).54 –56 Ischemic stroke was reported in 5 (0.6%)56 –58 and transient ischemic attacks in 10 (1.3%) patients (all IAT).48,49,56,57,59,60 Symptomatic intracerebral hemorrhage was reported in 8 (1.0%) patients following the interventions (IAT-tPA: 4 23 ,56,61, IAT-UK: 1 36 , combined IAT and IVT-UK: 1, 56 and IVT-UK: 2 62 , 63 ). One (0.1%) patient passed following large intracerebral hematoma after receiving IAT-UK. 36
Results of quality assessments, mixed model, and moderator analyses
All results were produced under minimal publication bias (Egger test p-values > 0.2 throughout, Supplementary sTable 3 and Supplementary sFigures 9 and 10). Two studies were identified with a high risk of bias (Supplementary sDocument 8 and Supplementary sFigures 11 to 15).52,53 Supplementary sTables 4 to 7 illustrate the mixed model and moderator analyses.
Discussion
We systematically reviewed the outcomes of patients with retinal artery occlusion who received IVT or IAT and the factors predictive of improved visual outcomes. We demonstrated favorable VA improvement for ⩾0.1 logMAR, ⩾0.3 logMAR, and MC from admission logMAR among patients who receive thrombolysis within currently recommended time frames for ischemic stroke. 7 We avoided comparing the visual outcome of patients who received thrombolytics to those with non-thrombolytic therapies because of the large diversity of conservative management and the high heterogeneity among these studies.11,14 The result of meta-regression models suggested that multiple factors can influence the outcome of patients. To better explore this effect, we studied the IPD, and we observed the predictive value of admission VA, having at least 2 weeks intervals before the final VA assessment, and a shorter time frame between the visual loss to thrombolysis for an improved outcome. IPD analyses also suggested that patients who receive antiplatelets following thrombolysis may have a better outcome (significant for ⩾0.1 logMAR and marginally significant for ⩾0.3 logMAR VA improvement). These results were adjusted for age, the interaction between IVT in < 4.5 h window and presenting VA.
Our findings are comparable with previous studies. Mac Grory et al. 15 reported a visual recovery rate of about 37% (our study: 39%) to a VA of ⩾20/100 after IVT-tPA within 4.5 h compared with a spontaneous recovery rate of 17.7%. They reported 56.3% recovery rate to ⩾0.3 logMAR (vs 62.8% among all IVT-tPA < 4.5 h patients in our study). We recognized the substantial influence of one study 52 on estimated recovery rates in this time frame. The authors of this retrospective observational study limited enrollment to patients with severe vision loss (i.e. Snellen ⩽ 6/120) and reported visual recovery among 3 out of 16 patients who received IVT-tPA < 4.5 h, not significantly different from patients in the conservative therapy cohort (0 out of 21). By reproducing meta-analyses of CRAO patients with a broader baseline visual range (Snellen ⩽20/200), ⩾0.3 logMAR VA improvement was observed among 74.3% of patients in our study.
A more recent systematic review 17 reported 47% rate of VA improvement in CRAO patients treated with IVT, compared with 12% in the group of patients treated conservatively (odds ratio (OR) 5.97). These results are confounded by a high degree of heterogeneity. In addition, the authors considered any degree of visual improvement as a favorable outcome. In another review by this team, 16 the outcome of CRAO patients who received any IAT (i.e. streptokinase, UK, or tPA) was significantly better than the non-IAT cohort (OR 3.55). The authors stratified the VA improvement among patients who received IAT < 6 h (39%) and > 6 h (60%, I2 = 75%).
So far, many studies have tried to recommend an optimum window for thrombolytic therapy. 64 But not all studies confirmed any correlation between time to thrombolysis and rate of visual recovery.56,65 Similar to Pielen et al., 66 we observed a trend for better outcomes of patients when they were treated within 12 h of the visual change (multiple IPD models, Table 1). However, our results can be affected by the low number of patients in each category. Moreover, as current AHA recommendations suggest IVT as the preferred route of thrombolysis within 4.5 h, and IAT only for patients not candidates for IVT and presenting within 6 h from visual loss, 6 future prospective studies are required to shed light on the possible superior effectiveness of IAT and its safety within 12 h or beyond from visual loss.
In addition to the time passed from the retinal artery occlusion, the involved artery (central vs branch), the location of the arterial occlusion, collateral circulation, the stage of the occlusion, and patients’ age are other factors affecting the rate of visual recovery.23,57,67 We also observed the value of baseline VA on predicting visual outcomes, possibly reflecting the severity of the initial insult and the potential for recovery.
Our results suggest a potential role for antiplatelets in addition to thrombolysis for managing CRAO. Antiplatelet therapy has been presented in multiple trials for secondary prevention and better outcome in patients with minor ischemic stroke.68 –71 AHA/ASA recommends that dual antiplatelet therapy be started within 24 h of minor non-cardioembolic ischemic stroke and be continued for 21 days to reduce the 3-month recurrence of ischemic stroke. 7 In the presence of intravenous tPA, AHA/ASA recommends aspirin administration to be delayed until 24 h after IVT. 7 There is increasing evidence suggesting the low risk of hemorrhagic events following early aspirin administration (<24 h) after IVT.72,73 Despite the increasing trend of antiplatelet use among patients with minor and non-minor ischemic strokes over the years, 74 also as part of CRAO management in some institutes,23,62,75 initiation of antiplatelets is not addressed in detail in current naCRAO acute management guidelines. 6
Although our study was attempted to be inclusive and explore multiple influential factors on the outcome of naCRAO patients, we could not control for all variability among studies such as degree of carotid stenosis and additional ischemic cerebral changes.22,52,58,76 –78 In addition, the detail of heparin56,77 and antiplatelet therapy varies among the studies and limits the conclusion about timing and the appropriate regiment of antiplatelets after thrombolysis.
Conclusion
This systematic review and IPD meta-analysis demonstrated improved visual outcomes among naCRAO patients who received thrombolysis. About one-third of patients had VA improvement to 20/100 or more. In addition, its importance of time of onset to thrombolysis administration. Large-scale prospective controlled trials should further refine our findings and provide recommendations for the optimal timelines of administration of thrombolytic agents in acute naCRAO.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231189352 – Supplemental material for Thrombolysis for central retinal artery occlusion: An individual participant-level meta-analysis
Supplemental material, sj-docx-1-wso-10.1177_17474930231189352 for Thrombolysis for central retinal artery occlusion: An individual participant-level meta-analysis by Shima Shahjouei, Reza Bavarsad Shahripour and Oana M Dumitrascu in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
