Abstract
Background:
An increased risk of intracranial hemorrhage (ICH) associated with statins has been reported, but data on the relationship between statin use and cerebral microbleeds (CMBs) in patients with atrial fibrillation (AF), a population at high bleeding and cardiovascular risk, are lacking.
Aims:
To explore the association between statin use and blood lipid levels with the prevalence and progression of CMBs in patients with AF with a particular focus on anticoagulated patients.
Methods:
Data of Swiss-AF, a prospective cohort of patients with established AF, were analyzed. Statin use was assessed during baseline and throughout follow-up. Lipid values were measured at baseline. CMBs were assessed using magnetic resonance imagining (MRI) at baseline and at 2 years follow-up. Imaging data were centrally assessed by blinded investigators. Associations of statin use and low-density lipoprotein (LDL) levels with CMB prevalence at baseline or CMB progression (at least one additional or new CMB on follow-up MRI at 2 years compared with baseline) were assessed using logistic regression models; the association with ICH was assessed using flexible parametric survival models. Models were adjusted for hypertension, smoking, body mass index, diabetes, stroke/transient ischemic attack, coronary heart disease, antiplatelet use, anticoagulant use, and education.
Results:
Of the 1693 patients with CMB data at baseline MRI (mean ± SD age 72.5 ± 8.4 years, 27.6% women, 90.1% on oral anticoagulants), 802 patients (47.4%) were statin users. The multivariable adjusted odds ratio (adjOR) for CMBs prevalence at baseline for statin users was 1.10 (95% CI = 0.83–1.45). AdjOR for 1 unit increase in LDL levels was 0.95 (95% CI = 0.82–1.10). At 2 years, 1188 patients had follow-up MRI. CMBs progression was observed in 44 (8.0%) statin users and 47 (7.4%) non-statin users. Of these patients, 64 (70.3%) developed a single new CMB, 14 (15.4%) developed 2 CMBs, and 13 developed more than 3 CMBs. The multivariable adjOR for statin users was 1.09 (95% CI = 0.66–1.80). There was no association between LDL levels and CMB progression (adjOR 1.02, 95% CI = 0.79–1.32). At follow-up 14 (1.2%) statin users had ICH versus 16 (1.3%) non-users. The age and sex adjusted hazard ratio (adjHR) was 0.75 (95% CI = 0.36–1.55). The results remained robust in sensitivity analyses excluding participants without anticoagulants.
Conclusions:
In this prospective cohort of patients with AF, a population at increased hemorrhagic risk due to anticoagulation, the use of statins was not associated with an increased risk of CMBs.
Introduction
Intracranial hemorrhage (ICH) accounts for approximately 10–20% of all strokes and is associated with high mortality and disability. 1 In the Treat Stroke to Target Trial the number of ICH events was higher in patients with lower target LDL levels. 2 An increased risk of ICH associated with statins and low cholesterol levels is supported by other studies,3,4 but these associations remain controversial.5–7
Cerebral microbleeds (CMBs) are hypointense lesions on T2-weighted gradient-echo magnetic resonance imaging (MRI). They are considered to be an asymptomatic precursor of ICH, especially in patients with a high burden of CMBs,8,9 while they are also associated with an increased risk for ischemic stroke. 8 Few studies have examined the association of statin use and lipid levels with the presence of CMBs, and very few with the progression of CMBs, with conflicting results.10–13 These studies had several limitations, including small sample sizes, heterogeneous MRI protocols with lack of blinded assessment, and limitations regarding adjustment for important covariates, potentially explaining the conflicting results.
Patients with atrial fibrillation (AF) is a population at potential increased bleeding risk due to oral anticoagulation.14–16 The prevalence of CMBs may in fact be significantly higher in patients with AF than in those without AF.14,16 However, AF patients are also at increased cardiovascular risk and treatment with statins is often indicated. 16 Thus, a possible association between statins or blood lipid levels with CMBs may have therapeutic implications also in terms of identifying modifiable risk factors for CMBs in AF patients.
The aim of this prospective, observational, study was to investigate for the first time the association of statin use and lipid levels with the presence and progression of CMBs in the AF population.
Method
Swiss-AF
Swiss-AF is an ongoing prospective multicenter observational cohort study conducted in 14 centers in Switzerland, which enrolled 2415 participants. The main protocol has been described elsewhere. 17 Details about inclusion/exclusion criteria, recruiting centers, MRI protocol, and laboratory measurements are reported in the supplemental material.
Outcomes and aims
Primary outcomes: (1a) CMBs at baseline (none or ⩾1) and (1b) CMBs progression (defined as at least one additional or new CMB on follow-up MRI at 2 years compared with baseline MRI).
Secondary outcome: ICH (excluding subarachnoid hemorrhage and subdural hematoma) during follow-up. ICH was defined according to the International Society on Thrombosis and Hemostasis criteria and was independently adjudicated according to the Swiss-AF main protocol.17,18
Primary aim: to investigate the association between statin exposure, defined as yes or no at baseline and outcomes as described above. The secondary aim focused on the association of the abovementioned primary outcomes with blood lipid levels (primarily low-density lipoprotein (LDL) but also total cholesterol (TCHOL), triglycerides (TRG), and high-density lipoprotein (HDL)) at baseline as continuous variables.
Statistical analysis
Baseline characteristics are presented separately for statin users (participants with any statin use at baseline) and non-users. Association of statin use or lipid levels (TCHOL, LDL-C, HDL-C, and TRG separately, each as a continuous variable per 1-unit increase) with the prevalence of CMBs at baseline, as well as the progression of CMBs at follow-up, was assessed using logistic regression models. The results are presented as odds ratios (OR, 95% confidence intervals (CI)). For the occurrence of ICH during follow-up, we used flexible parametric survival models. The results are presented as hazard ratios (HR) with 95% CI. 19 All analyses were adjusted for confounding factors potentially associated with both the exposure (statin and blood lipid levels) and the outcome (CMBs) using directed acyclic graph (DAG) models based on prior knowledge of association with CMBs: sex, age, history of hypertension, smoking status, body mass index (BMI), history of diabetes, history of stroke or transient ischemic attack (TIA), coronary heart disease, antiplatelet use, anticoagulant use, and education. Due to the low number of events, flexible parametric survival models for ICH were only adjusted for sex and age.
In the main analysis, participants were categorized as follows: baseline statin use versus no use. Further analyses were done for high-dose baseline use versus low- and medium-dose use versus no use (high, medium, low statin doses as defined from ACC/AHA). 20 All medications were assessed at baseline and at years 1 and 2. We performed time-updated sensitivity analyses by change in statin (stop/start) and change in statin intensity during follow-up.
Supplemental analyses were performed focusing on lobar CMBs, based on previous observations suggesting that statin and LDL levels might be primarily associated with a higher bleeding risk in the lobar brain regions. 21 Additional analyses were performed by restricting the investigation to patients under oral anticoagulants.
Power calculation is reported in the supplemental material. All data analyses were carried out using STATA version 16.0. No correction was performed for multiple testing. The study is reported following the STROBE guidelines for cohort studies. 22
Results
Patients characteristics
Of the 2415 patients enrolled in Swiss-AF, 667 did not have brain MRI at baseline. The main reason for a missing brain MRI was the presence of a cardiac device (N = 461, 69%). Other reasons included further contraindications to perform an MRI or claustrophobia of the patient. Fifty-five patients had brain MRI without CMB data. A total of 1693 (468 [27.6%] females, mean [SD] age 72.5 years [8.4], 90.1% under oral anticoagulants) having CMB data at baseline MRI were included (Figure 1). 23 Statin users (N = 802) had more hypertension (79.7% vs 59.9%), more diabetes (23.6% vs 8.6%), more often a history of stroke/TIA (29.1% vs 11.1%), and other known cardiovascular disease (44.4% vs 9.2%). The percentage of patients under oral anticoagulation was 92.1% in the statin and 88.3% in the non-statin user group. The use of vitamin K antagonist (VKA) compared with direct oral anticoagulants (DOACs) was higher under statin users (Table 1). The cohort represented an intermediate bleeding risk population. 24 A total of 72% of statin users versus 74% of non-statin users had 3.0 T scanner.

Flow chart of participant selection.
Baseline characteristics of included participants (with MRI at baseline) stratified by statin use.
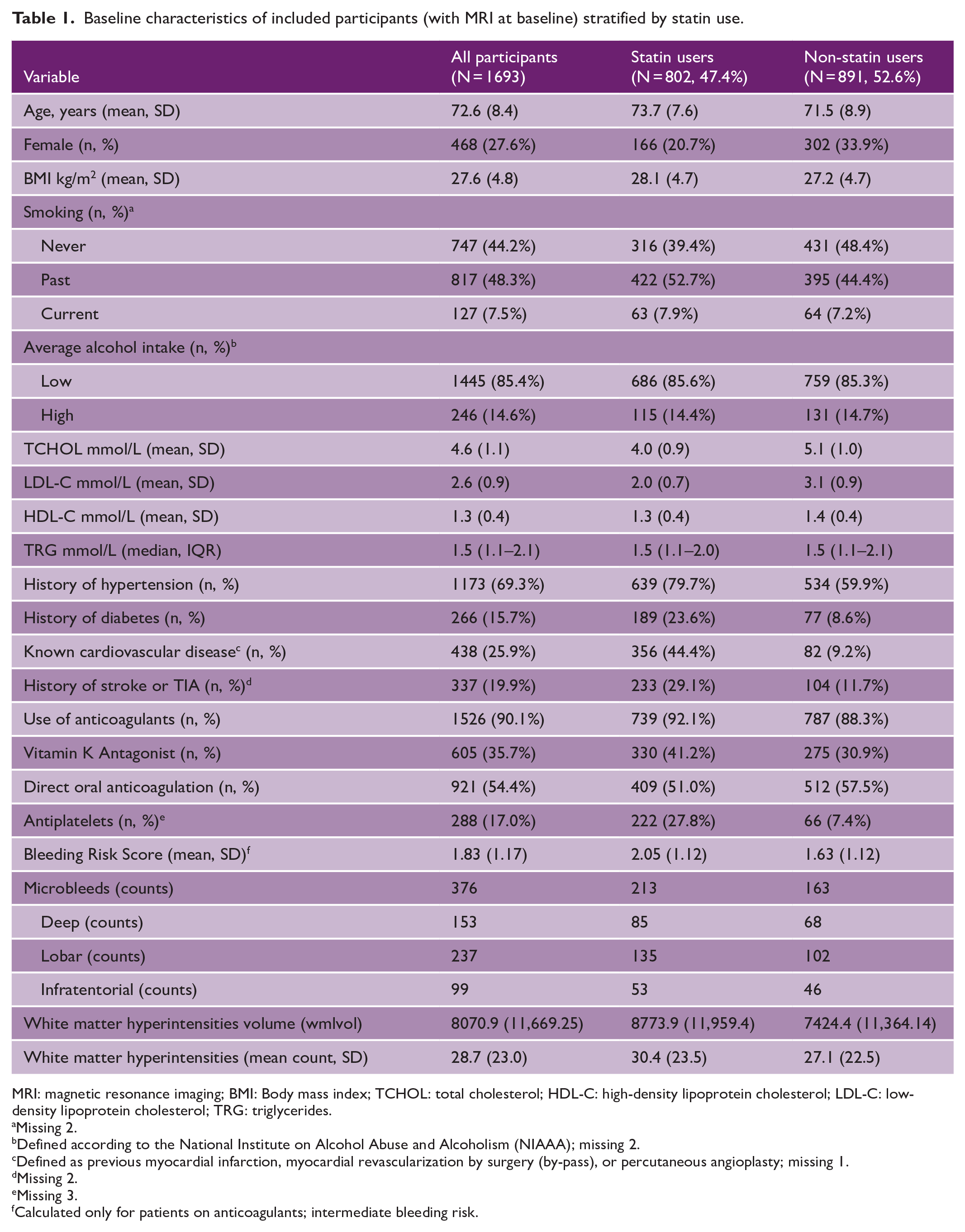
MRI: magnetic resonance imaging; BMI: Body mass index; TCHOL: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TRG: triglycerides.
Missing 2.
Defined according to the National Institute on Alcohol Abuse and Alcoholism (NIAAA); missing 2.
Defined as previous myocardial infarction, myocardial revascularization by surgery (by-pass), or percutaneous angioplasty; missing 1.
Missing 2.
Missing 3.
Calculated only for patients on anticoagulants; intermediate bleeding risk.
At 2 years, 1188 of the 1693 identified patients had follow-up MRI (Figure 1). The reasons for declining 2 years MRI included the following: the patient developed contraindications for MRI (e.g. pacemaker), patient was no longer able to come to the hospital on his own (follow-up visit carried out by telephone), patient developed claustrophobia or had unpleasant experience from MRI at baseline, patient did not want to come for other reasons, and patient withdrew or was deceased. For example, at the study site of Bern (N = 368), a telephone visit was performed in 15% of the participants at 2 years follow-up. Eighty (3.3%) patients had no 2-year follow-up visit. 15 The characteristics of patients who had baseline and follow-up MRI were comparable (Supplemental Tables S1 and S2).
Cross-sectional analysis
At baseline, 213 (26.5%) statin users and 163 (18.3%) non-statin users were found to have at least one CMB. After adjustment for history of hypertension, smoking status, BMI, history of diabetes, history of stroke or TIA, coronary heart disease, antiplatelet use, anticoagulant use, and education no significant association was observed (adjOR, 1.10 (95% CI = 0.83–1.45)) (Table 2). Similar were the results for lipid levels (Supplemental Table S3). Covariates’ association with CMBs prevalence is shown in Supplemental Table S4. No association for lobar CMBs at baseline was observed, neither for statin use (Table 2) nor for LDL levels (Supplemental Table S5). Also, no association was observed by restricting the analysis to patients under oral anticoagulants (Supplemental Table S6).
Cross-sectional analyses of CMBs at baseline by statin use in patients having baseline MRI.
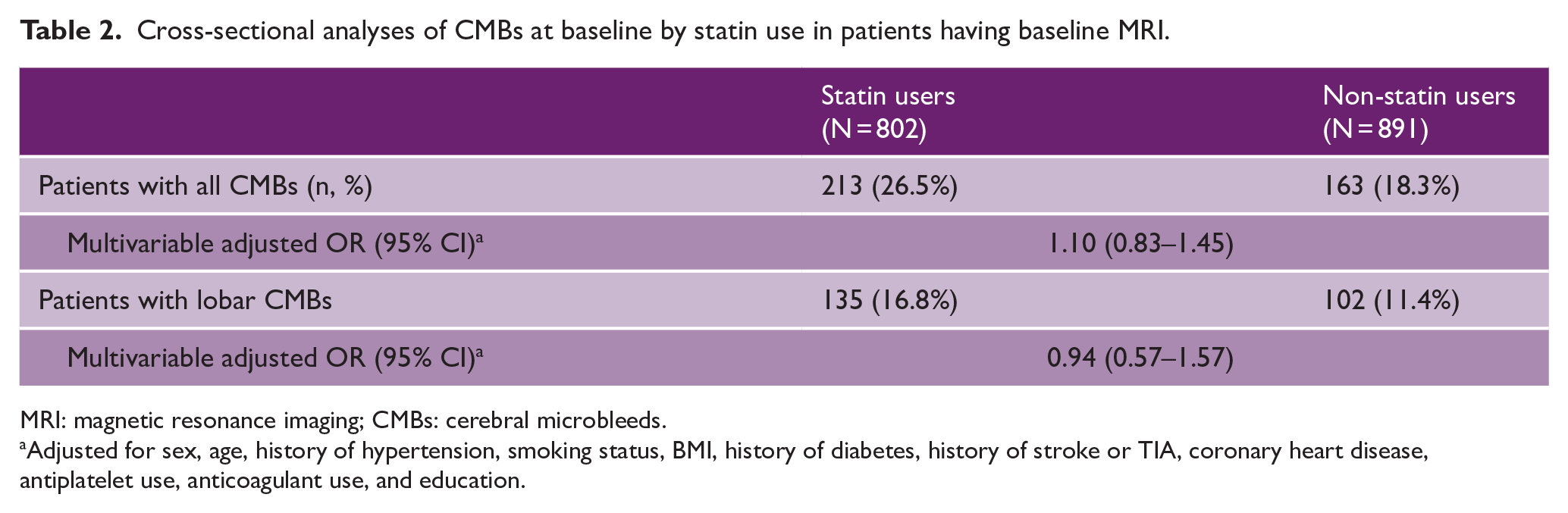
MRI: magnetic resonance imaging; CMBs: cerebral microbleeds.
Adjusted for sex, age, history of hypertension, smoking status, BMI, history of diabetes, history of stroke or TIA, coronary heart disease, antiplatelet use, anticoagulant use, and education.
Longitudinal analysis
At 2 years, follow-up progression of CMBs was observed in 44 (8.0%) patients in the statin group and 47 (7.4%) patients in the non-statin group, respectively. Of the 91 patients who developed new CMBs, 64 (70.3%) developed a single new CMB, 14 (15.4%) developed 2 CMBs, and 13 participants developed more than 3 CMBs. In all, 42 patients had strictly lobar CMB progression, 16 had deep CMB progression, and 14 had infratentorial CMB progression. The remaining patients (N = 19) had CMB progression in mixed locations.
No association between statin use and CMB progression was observed in the multivariable logistic regression model (adjOR, 1.09 (95% CI = 0.66–1.80)) (Table 3). There was also no statistically significant association between blood lipid levels and CMB progression (Supplemental Table S3). Univariable association of covariates with CMB progression at follow-up is presented in Supplemental Table S7. The total number of patients having progression of lobar CMBs was 57 (35 statin users and 22 non-statin users). In multivariable adjusted models, no association could be seen for statin use (Table 3) or LDL levels (Supplemental Table S5) with lobar CMBs progression.
Longitudinal analyses of CMBs progression at follow-up by statin use in patients having baseline and follow-up MRI.
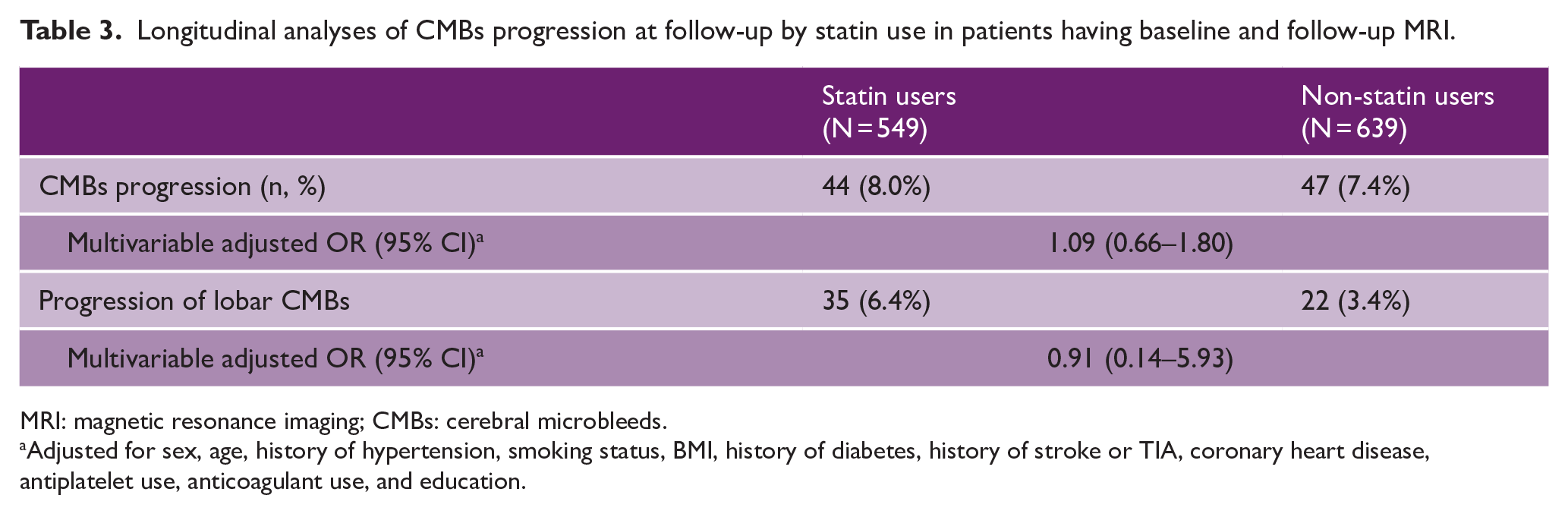
MRI: magnetic resonance imaging; CMBs: cerebral microbleeds.
Adjusted for sex, age, history of hypertension, smoking status, BMI, history of diabetes, history of stroke or TIA, coronary heart disease, antiplatelet use, anticoagulant use, and education.
By restricting the analysis to patients under oral anticoagulants, a total of 84 patients having CMBs progression was observed. No association could be seen either for statin use, or for LDL levels (Supplemental Table S6).
Our results remained robust in sensitivity analyses, adjusting for new statin introduction or increase in intensity or by classifying statins as low, medium or high intensity (Supplemental Table S8).
The number of ICH observed during follow-up was low in both groups, with a total of 14 events in the statin users and 16 in the non-statin users, showing no hazard difference between the two groups in the age and sex adjusted model (adjHR, 0.75, 95% CI = 0.36–1.55; Supplemental Table S9).
Discussion
In this large prospective cohort study of older persons with AF, we found no evidence that statin use or lipid levels are associated with CMBs prevalence or with CMBs progression at 2 years follow-up. To our knowledge, this is the largest and longest prospective cohort study reporting on these outcomes. This study is particularly relevant since >90% of participants were on anticoagulation, with robust results even after excluding 9.6% of patients, who were not anticoagulated.
A recent meta-analysis also observed a positive association of statin use with the prevalence of all and lobar CMBs in unadjusted, but not in fully adjusted models. 25 However, the same meta-analysis found all CMBs (4 studies, 7223 patients, I 2 = 77%) and lobar CMBs (3 studies, 7051 patients, I 2 = 0%) to be inversely associated with TCHOL levels, which we did not observe. 25 This apparent discrepancy may be explained by the fact that three of the included studies did not take into account antiplatelet and anticoagulation drug use and none of them assessed prevalent coronary artery disease.
Very few prospective studies have investigated the association of statin use or lipid levels on CMBs progression. A subgroup analysis of a randomized controlled trial conducted in 668 geriatric stroke-free patients did not find any significant difference in the risk of new-incident CMBs, between patients treated with statins compared with placebo. 12 Statins also did not show any association with CMBs progression in patients with a history of stroke, as shown by two small prospective cohort studies.26,27 Our longitudinal analysis confirms these observations in a larger population. Ding et al. 28 conducted a prospective cohort study investigating the associations of lipid levels with CMBs progression over a 5-year follow-up in 2635 patients. A direct investigation on the association between statins and CMBs was not undertaken. The authors reported that low TRG and high HDL values were associated with a higher progression of exclusively lobar CMBs. In the present study, we explored progression of lobar CMBs in relation to statin exposure and LDL levels, not finding any significant association, even though the absolute number of cases with CMBs progression was higher under statin users.
The proposed explanations for an increased bleeding risk under statins focus on pleiotropic side effects influencing negative platelet function and coagulation mechanisms. 29 Considering our population, it might be that we failed to see an association between statin use and CMBs because of altered coagulation mechanisms in patients under oral anticoagulation, though pharmacological studies have also failed to show an interaction between statins and anticoagulation in respect of bleeding tendency. 30
In specific contexts, CMBs have proven to serve as a marker of cerebrovascular disease that is known to increase the risk of ICH.31,32 The CROMIS-2 study recently showed that the presence of CMBs at baseline, after ischemic stroke due to AF, is independently associated with a higher symptomatic ICH risk in patients who start anticoagulation. 33 Other studies also identified an association between a higher burden of intracranial microhemorrhages on MRI with an increased risk for ischemic stroke, cognitive decline, and death.8,34 At the same time, prior investigations suggested that statin therapy might improve cardiovascular outcome in patients with AF and acute ischemic stroke. 35 Investigations on the overall effects of statins on cerebrovascular small vessel disease and cognitive functions in elderly patients also tended to show a benefit of statin therapy.12,27 Until now, no study has been conducted investigating the associations between statin use or lipid levels with CMBs or ICH in patients with AF, a population at elevated hemorrhagic risk. Together with the current available evidence our results suggest that if indicated statin therapy should not be withheld in these patients because of fear of bleeding complications.
The major strengths of our study include the prospective part of the analysis, detailed assessment of statin use (with time-updated data), uniform MRI protocols for CMBs, MRI assessment at 2 years follow-up in a large subgroup of patients, blinded central assessment of brain imaging at baseline and follow-up, as well as assessment of CMBs location. The strengths in the analytical design include adjustment for important covariates as well as very few missing data.
The study limitations include a possible selection bias in terms of a healthy volunteer bias, since the main reason for denying participation was due to complicated study design, age/health-related issues, and lack of interest; being in accordance with the underrepresentation of older people in clinical research.15,36 Furthermore, in our study, the female population represented a minority (27.6%), which is compatible with more male patients suffering from AF and, therefore, reflects an AF population.36,37 Even if the cumulative use of anticoagulants between both groups was similar, statin users tended to have a higher use of VKA compared with DOAC. Based on prior observations suggesting a higher incidence of CMBs and ICH with VKA compared with DOAC,33,38 this difference might have disfavored the statin group, which, however, would still be in line with our main observation. In all, 70% of the participants with baseline MRI had also MRI at 2 years, which may imply possible bias related to availability for follow-up MRI. However, the baseline data between participants having both MRIs versus only baseline MRI were comparable. Furthermore, one-third of MRIs were performed using a 1.5 T scanner, which has a lower sensitivity to detect CMBs. However, follow-up MRI was performed using the same scanner as baseline and type of scanner was equally distributed between statin and non-statin users. In any observational study, there is a danger of residual confounding: we did not adjust for genetic factors like apoE genotype. 39 Finally, the low number of events accounts for low power for the outcomes of incidence of lobar CMBs and ICH. Studies with longer follow-up time and data on lobar CMBs may be necessary.
Conclusion
Statin use and lipid levels are not associated with an increased risk of CMBs in this population of patients with AF. The incidental finding of CMBs on brain imaging in AF patients, without recent stroke or TIA, should not influence clinical decisions about statin use.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231181010 – Supplemental material for Association of statin use and lipid levels with cerebral microbleeds and intracranial hemorrhage in patients with atrial fibrillation: A prospective cohort study
Supplemental material, sj-docx-1-wso-10.1177_17474930231181010 for Association of statin use and lipid levels with cerebral microbleeds and intracranial hemorrhage in patients with atrial fibrillation: A prospective cohort study by Elisavet Moutzouri, Matthias Glutz, Nazanin Abolhassani, Martin Feller, Luise Adam, Baris Gencer, Cinzia Del Giovane, Sylvain Bétrisey, Rebecca E Paladini, Elisa Hennings, Stefanie Aeschbacher, Jürg H Beer, Giorgio Moschovitis, David Seiffge, Gian Marco De Marchis, Michael Coslovsky, Tobias Reichlin, Giulio Conte, Tim Sinnecker, Matthias Schwenkglenks, Leo H Bonati, Peter Kastner, Drahomir Aujesky, Michael Kühne, Stefan Osswald, Urs Fischer, David Conen and Nicolas Rodondi in International Journal of Stroke
Footnotes
Acknowledgements
The authors thank all the participants from the Swiss-AF cohort.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosures
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Swiss National Science Foundation (grant nos. 33CS30_148474, 33CS30_177520, 32473B_176178, 32003B_197524, and IICT 33IC30-193052) to N.R., grant of Clinical Trial Unit (CTU) of the University of Bern (grant no. 84801740) to E.M., and McMaster University Department of Medicine Mid-Career Research Award to D.C. Lipid measurements were obtained free of charge from Roche.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
