Abstract
Background:
Aphasia is a common consequence of stroke, and people who live with this condition experience poor outcomes. Adherence to clinical practice guidelines can promote high-quality service delivery and optimize patient outcomes. However, there are currently no high-quality guidelines specific to post-stroke aphasia management.
Aims:
To identify and evaluate recommendations from high-quality stroke guidelines that can inform aphasia management.
Summary of review:
We conducted an updated systematic review in accordance with PRISMA guidelines to identify high-quality clinical guidelines published between January 2015 and October 2022. Primary searches were performed using electronic databases: PubMed, EMBASE, CINAHL, and Web of Science. Gray literature searches were conducted using Google Scholar, guideline databases, and stroke websites. Clinical practice guidelines were evaluated using the Appraisal of Guidelines and Research and Evaluation (AGREE II) tool. Recommendations were extracted from high-quality guidelines (scored > 66.7% on Domain 3: “Rigor of Development”), classified as aphasia-specific or aphasia-related, and categorized into clinical practice areas. Evidence ratings and source citations were assessed, and similar recommendations were grouped. Twenty-three stroke clinical practice guidelines were identified and 9 (39%) met our criteria for rigor of development. From these guidelines, 82 recommendations for aphasia management were extracted: 31 were aphasia-specific, 51 aphasia-related, 67 evidence-based, and 15 consensus-based.
Conclusion:
More than half of stroke clinical practice guidelines identified did not meet our criteria for rigorous development. We identified 9 high-quality guidelines and 82 recommendations to inform aphasia management. Most recommendations were aphasia-related; aphasia-specific recommendation gaps were identified in three clinical practice areas: “accessing community supports,” “return to work, leisure, driving,” and “interprofessional practice.”
Introduction
Aphasia is an acquired language impairment that initially affects up to one-third of stroke survivors.1,2 Speech and language therapy can significantly improve communication outcomes for people with aphasia; 3 however, comprehensive clinical guidance is needed to support the delivery of high-quality services. Clinical practice guidelines are systematically developed sets of recommendations, based on the best available research evidence and expert consensus. 4 Such guidelines support clinical decision-making, optimizing service delivery and patient outcomes by linking research evidence to practice in a clinically accessible resource. 4 The routine use of clinical practice guidelines can improve health 5 and economic outcomes 6 for people living with stroke.
Currently there are no clinical practice guidelines published in English that specifically and comprehensively guide aphasia management following stroke. Clinicians can access relevant recommendations within general stroke management guidelines; however, such guidelines vary in methodological quality and comprehensiveness and quickly become outdated with changes in the evidence-base. Over the past decade, systematic reviews have been conducted7,8 to bridge this gap, identifying high-quality stroke clinical practice guidelines that contain recommendations for the clinical management of aphasia. These reviews provide a crucial resource for clinicians, given the absence of aphasia-specific guidelines; however, regular updates are required to keep pace with the rapidly evolving evidence-base. Therefore, we sought to update the most recent systematic review by Shrubsole et al. 7 with the following aims:
To identify clinical practice guidelines relevant to post-stroke aphasia management published or updated since January 2015.
To examine the methodological quality of identified clinical practice guidelines using the AGREE II instrument. 9
To describe and synthesize recommendations for post-stroke aphasia management from clinical practice guidelines of the highest methodological quality.
To describe changes in the scope of guideline recommendations and the quality of evidence since 2015.
Methods
Design
Updated systematic review conducted and reported in alignment with PRISMA guidelines. 10 Title and abstract screening and full-text review were conducted using Covidence systematic review software (available at www.covidence.org) with data extraction managed using Microsoft Office Excel. The review protocol is available at: https://osf.io/9fp47/.
Search strategy
We replicated the two-phase search strategy used by Shrubsole et al. 7 Primary searches were performed using electronic databases: PubMed, EMBASE, CINAHL, and Web of Science. Gray literature searches were conducted within: Google Scholar, guideline databases, and using stroke websites. Bibliographies of relevant articles were hand-searched. Search terms included population (aphasia OR dysphasia OR stroke OR cardiovascular) and publication type (guidelines OR recommendation OR clinical practice guideline). Refer to Supplemental Table 1 for the search strategy.
Study selection process
Electronic database searches were conducted in July 2020 and updated in October 2022. Gray literature was searched in June 2021 and October 2022. Two appraisers (M.I. and E.B.) independently screened titles and abstracts. If a discrepancy arose, a third appraiser (M.K.) determined if the article should be included.
Inclusion and exclusion criteria
Clinical practice guidelines (defined as, statements that include recommendations intended to optimize patient care that are informed by evidence or expert consensus) were included if they: (1) related to the clinical management of stroke or aphasia in adults; (2) were published between January 2015 and date of search; (3) were available in English; (4) were multi-disciplinary or speech pathology specific in nature. Clinical practice guidelines were excluded if they: (1) related selectively to sub-arachnoid hemorrhage or stroke prevention or (2) were published as conference abstracts, theses, or systematic reviews.
Evaluation of guideline quality
Guideline quality was evaluated using the rigor of development (Domain 3) score of the AGREE II instrument, as it is considered most reflective of overall guideline standard and quality.7,8,11 Domain 3 has eight items, rated on a 7-point Likert-type scale (1 = “Strongly Disagree”; 7 = “Strongly Agree”). Two reviewers (M.I. and E.B.) independently assessed guidelines following standard procedures. Domain scores were compared and where scores differed by more than two points, they were discussed to achieve a consensus. Domain scores were converted to a standardized percentage score, and as with previous reviews, the cut-off for inclusion was a rating of 66.67% to include the top third of the 7-point Likert-type scale.7,8
Evaluation of aphasia-relevant recommendations
Recommendations relating to aphasia management and their strength of evidence ratings were extracted from guidelines and categorized by clinical practice area. New clinical practice topic areas were devised where necessary. Overlapping recommendations, that is, those containing similar or the same wording, were organized together and counted as a single recommendation. Source citations for individual recommendations were retrieved and reviewed. If source citations were inconsistent with the recommendation or evidence rating, they were excluded. Where source citations were not available, guideline developers were contacted for information demonstrating recommendation-to-citation links. Recommendations not directly linked to an evidence source or expert consensus were excluded. Included recommendations were classified as aphasia-specific or aphasia-related. Aphasia-specific recommendations related to the clinical management of aphasia (e.g. language assessments or aphasia therapy). Aphasia-related recommendations included general rehabilitation considerations that were not aphasia or speech-pathology specific; however, they were relevant to clinical aphasia management (e.g. multi-disciplinary team considerations or discharge processes).
Results
Database and gray literature searches
In total, 1749 sources were retrieved through database searches and 41 sources through the gray literature search. Following the removal of duplicates, 1563 sources underwent title and abstract screening. Of these, 1456 were excluded as they did not meet the inclusion criteria. The remaining 107 sources underwent a full-text review, and 31 were evaluated using the AGREE II tool. 9 The selection process is illustrated in Figure 1.
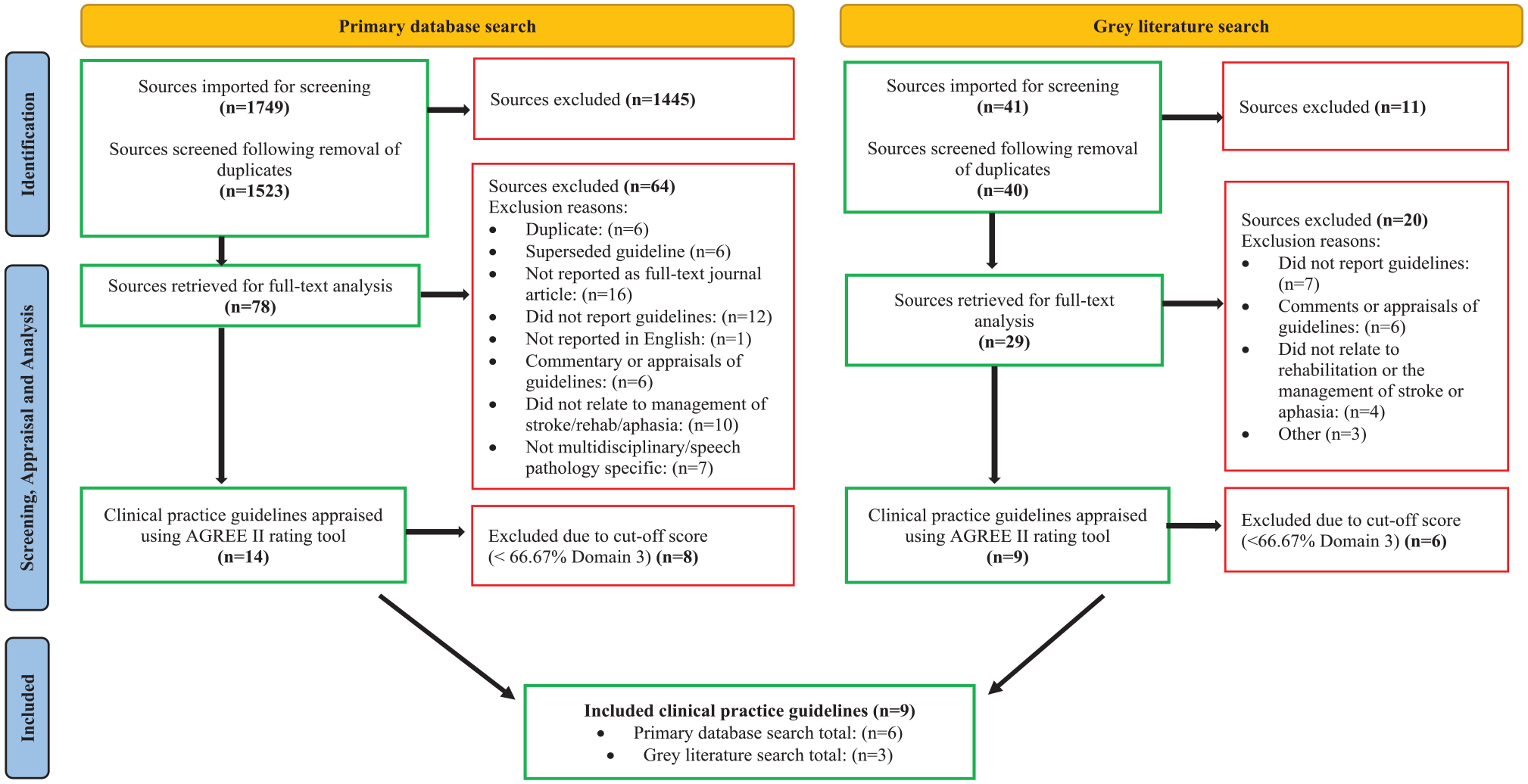
Clinical practice guideline selection process (PRISMA).
Evaluation of guidelines
We identified 23 stroke clinical practice guidelines and 9 (39%) met or exceeded our cut-off for AGREE II Domain 3: Rigor of Development. Included guidelines were from Australia, Canada, United States, and United Kingdom. Four guidelines were Canadian Stroke Best Practice Recommendations,12–15 two guidelines were from the American Heart Association and American Stroke Association,16,17 one guideline was published by each of the Australian Stroke Foundation, 18 the UK Royal College of Physicians, 19 and the US Department of Veteran’s Affairs and Department of Defense. 20 See Table 1, Supplemental Table 2 for guideline scope, and Supplemental Tables 4–9 for AGREE-II ratings.
High-quality stroke clinical practice guidelines and AGREE II ratings.
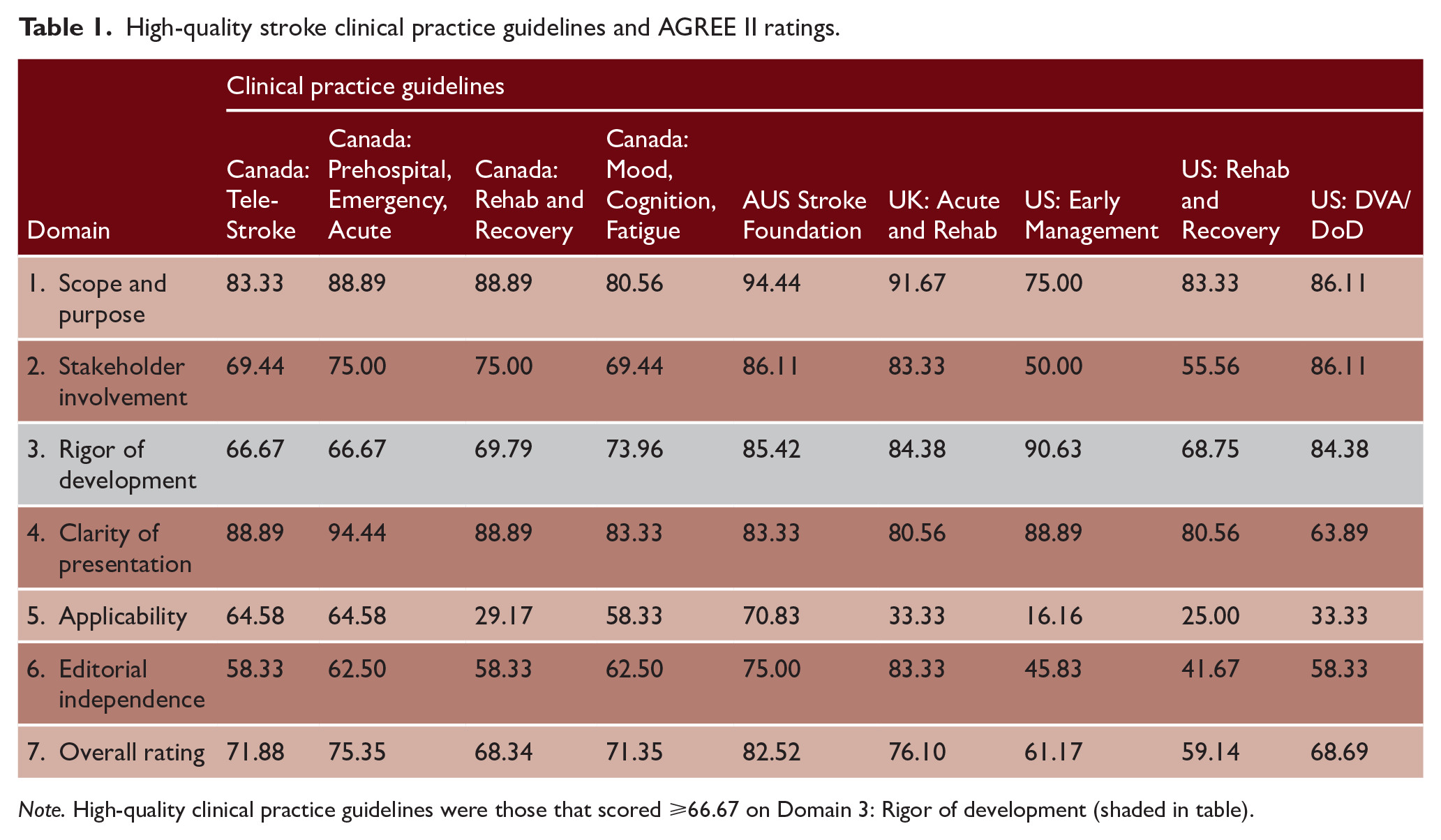
Note. High-quality clinical practice guidelines were those that scored ⩾66.67 on Domain 3: Rigor of development (shaded in table).
Identified recommendations and links to underlying literature
A total of 178 recommendations relevant to aphasia management were extracted. Following review, four were removed as they were not within the scope of this review (e.g. recommendations relating to staffing/organizational flow of stroke units). The remaining 174 recommendations were categorized across 16 topic areas (See Table 2). A further 14 recommendations were excluded due to the lack of relevancy to the clinical management of aphasia. The underlying evidence-base of each recommendation was reviewed to confirm links to the literature. A total of 36 recommendations were removed where links could not be established. The remaining 124 recommendations were reviewed within each topic area, and those with similar or the same wording were grouped together. This resulted in a total of 82 recommendations. Of the 82 included recommendations, 67 (82%) were evidence-based and 15 (18%) were consensus-based. Thirty-one (38%) were classified as aphasia-specific and 51 (62%) recommendations were classed as aphasia-related. Aphasia-specific recommendations are presented in Table 3. All recommendations relevant to aphasia management are listed in Supplemental Table 10 and are summarized in Figure 2. “Strength of Recommendation” and “Highest Level of Underpinning Evidence” ratings are provided in Supplemental Table 3.
Aphasia-relevant recommendations by clinical practice area.
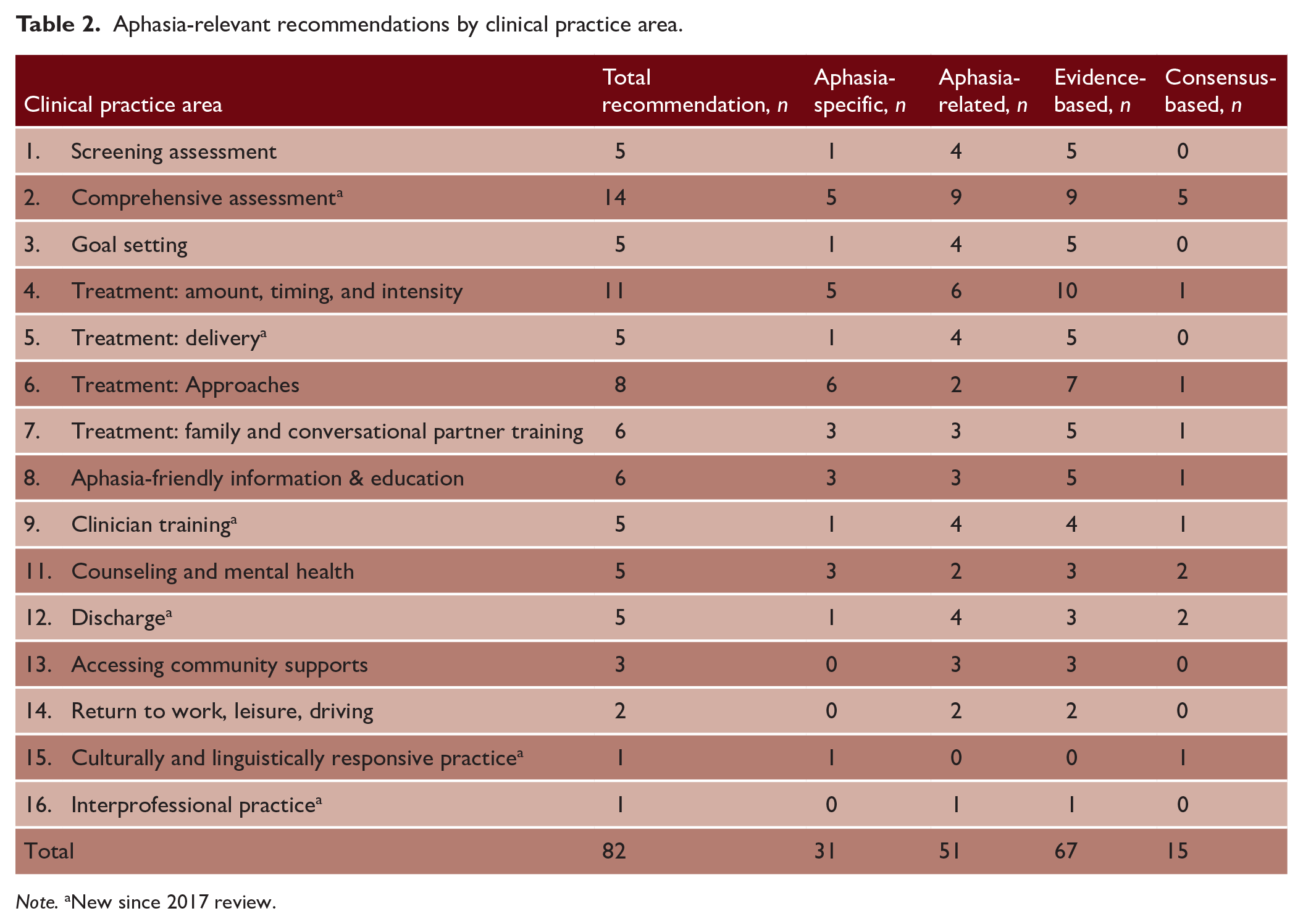
Note. aNew since 2017 review.
Aphasia-specific recommendations by clinical practice area.
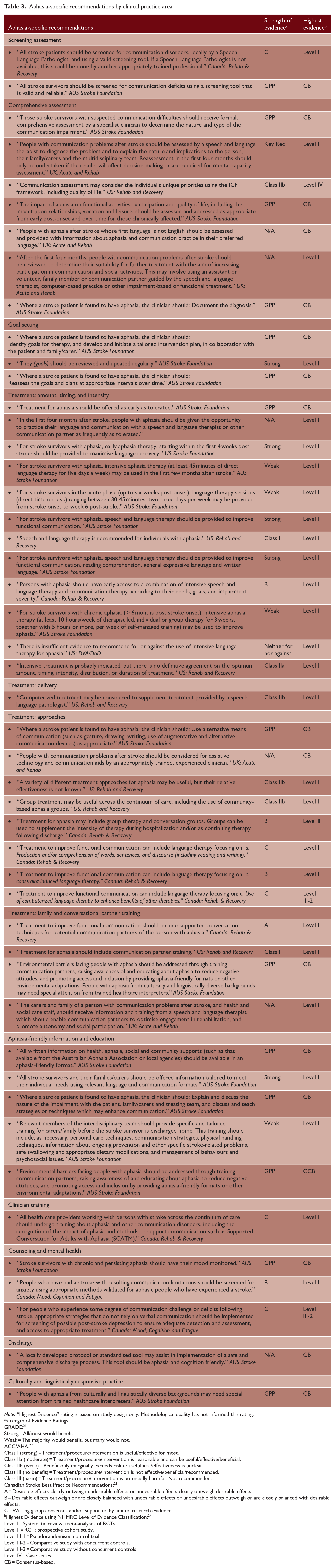
Note. “Highest Evidence” rating is based on study design only. Methodological quality has not informed this rating.
Strength of Evidence Ratings:
GRADE: 21
Strong = All/most would benefit.
Weak = The majority would benefit, but many would not.
ACC/AHA: 22
Class I (strong) = Treatment/procedure/intervention is useful/effective for most.
Class IIa (moderate) = Treatment/procedure/intervention is reasonable and can be useful/effective/beneficial.
Class IIb (weak) = Benefit only marginally exceeds risk or usefulness/effectiveness is unclear.
Class III (no benefit) = Treatment/procedure/intervention is not effective/beneficial/recommended.
Class III (harm) = Treatment/procedure/intervention is potentially harmful. Not recommended.
Canadian Stroke Best Practice Recommendations: 23
A = Desirable effects clearly outweigh undesirable effects or undesirable effects clearly outweigh desirable effects.
B = Desirable effects outweigh or are closely balanced with undesirable effects or undesirable effects outweigh or are closely balanced with desirable effects.
C = Writing group consensus and/or supported by limited research evidence.
Highest Evidence using NHMRC Level of Evidence Classification: 24
Level I = Systematic review; meta-analyses of RCTs.
Level II = RCT; prospective cohort study.
Level III-1 = Pseudorandomised control trial.
Level III-2 = Comparative study with concurrent controls.
Level III-3 = Comparative study without concurrent controls.
Level IV = Case series.
CB = Consensus-based.
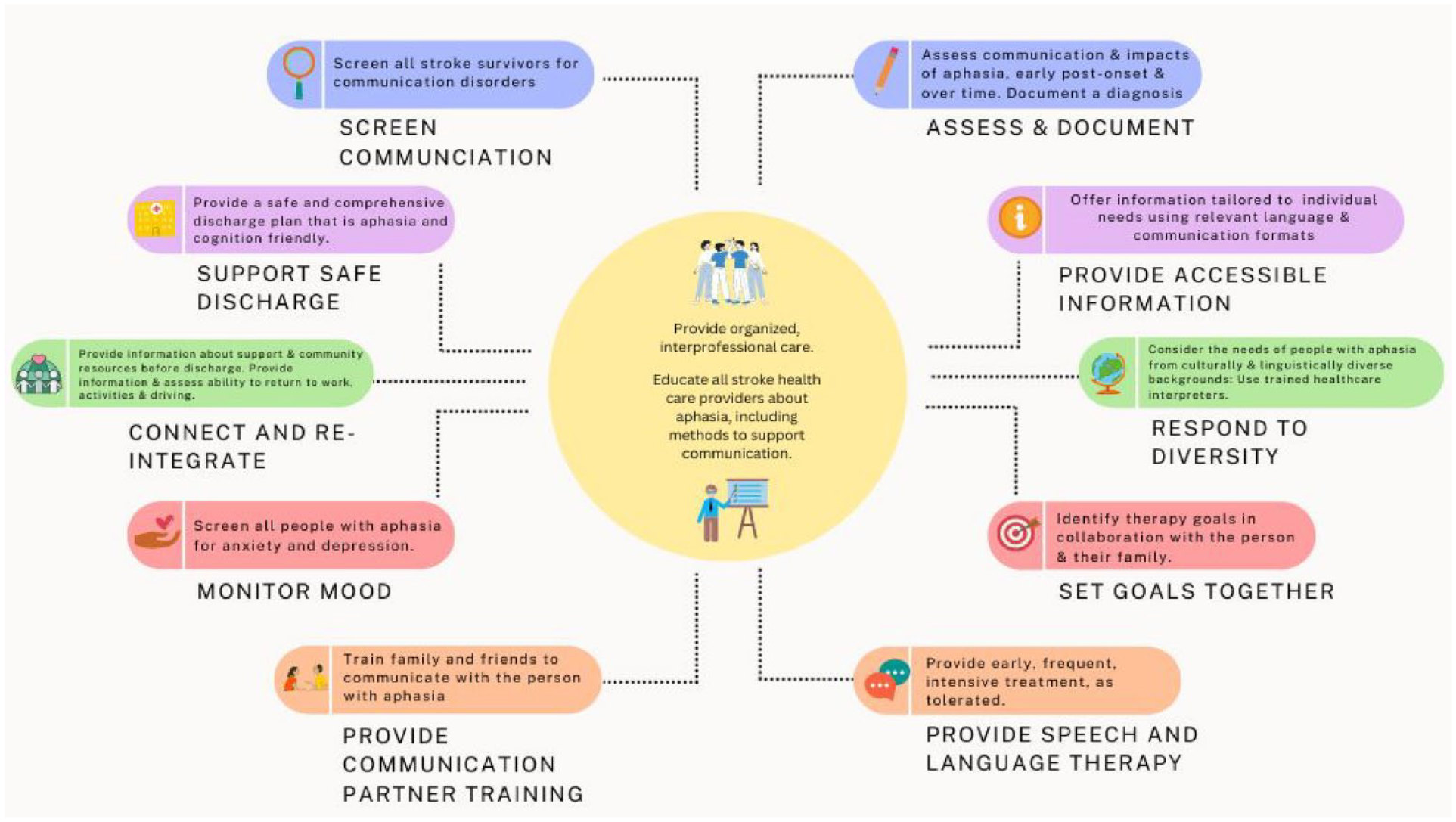
Best practice aphasia management after stroke: An overview.
Discussion
A total of 82 recommendations from nine high-quality clinical practice guidelines were identified in the current review, an increase from the 76 recommendations from five guidelines identified in 2017. 7 The number of included recommendations across reviews is not directly comparable however, as not all guidelines included in the 2017 review, were included in the current review (see Supplemental Table 2). While the total number of recommendations was relatively unchanged since the last review, there was a substantial increase in the number of clinical practice guidelines both identified and included. Of concern however, was that 61% of the clinical practice guidelines appraised did not meet our minimum standard for inclusion as a high-quality source. Among those that did, there was still considerable variation in quality ratings. There is no agreement about the AGREE-II cut-off scores that should be used to distinguish between high- and low-quality clinical practice guidelines and arbitrary changes to cut-off scores can change quality categorization. 25 As such, it is recommended that health professionals select the highest rated guidelines to inform their clinical decisions. 25 In our review, the guideline with the overall highest AGREE-II rating and the most aphasia-relevant recommendations, was the Australian Stroke Foundation Living Guideline (85.42%). 18 Living guidelines use continuous surveillance and rapid response to continually integrate new relevant evidence into clinical guidelines, addressing the perennial problem of rapid loss of currency. 26
As per the previous review, 7 there were fewer aphasia-specific, than aphasia-related recommendations. When considered by topic area, aphasia-specific recommendations in the current review predominately related to therapy and treatment approaches, comprehensive assessment, and aphasia-friendly information and education. New topic areas were created for recommendations relating to comprehensive assessment; clinician training; treatment delivery; discharge; culturally and linguistically responsive practice; and interprofessional practice. We found no aphasia-specific recommendations in the areas of: return to work, leisure and driving; accessing community supports; or interprofessional practice. Return to work and driving have been identified as priorities by stakeholders in aphasia rehabilitation. 27 Return to driving is a priority for people with aphasia and return to work for clinicians. 27 Future aphasia research should seek to bridge these gaps through targeted research.
Most aphasia-specific recommendations identified in our review were formulated through consensus, listed as good practice points, or had limited/weak evidence supporting them. Consensus-based recommendations serve an important role, as they provide advice for areas with an emerging evidence-base. However, only the Australian Stroke Foundation guidelines provided explicit information on the nature of group consensus processes, making it difficult to ascertain how consensus was achieved for many recommendations. 18 Greater transparency in future guideline iterations would support the trustworthiness of consensus-based recommendations. 28
Our review process was complicated by the different strength of evidence grading systems used across the clinical practice guidelines. Both the Australian Stroke Foundation and the Department of Veterans Affairs and USA Department of Defense adopted the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) method in the development of their guidelines.18,20 The GRADE method has only two strength of recommendation classifications (“strong” and “weak”), or alternatively classifies a recommendation as a “good practice point” (consensus-based recommendation). 21 In contrast, the American Heart Association adopted the American College of Cardiology/American Heart Association (ACC/AHA) Clinical Practice Guideline Recommendation Classification System, which assigns Classes I-III. 22 The Canadian Stroke Best Practice Recommendations used a modified GRADE method with an alphabetical ranking system to describe strength of recommendations (A, B, C). 23 The UK Royal College of Physicians did not adopt a hierarchal grading system for their guidelines, and instead formulated “key recommendations” identified through a consensus process. 19 Just as these disparate systems were a challenge to the comparison of evidence in our review process, they likely pose similar challenges for guidelines users. Use of a universally recognized, unambiguous grading system may promote transparency when reporting and describing the evidence-base, while simultaneously reducing inconsistencies in evidence interpretation. 21
Limitations
It is a limitation of our research that where links to the underlying evidence-base could not be established, recommendations were excluded. While best efforts were made to establish recommendation-to-citation links (i.e. by contacting guideline developers), some could not be established, and clinically relevant recommendations were excluded. Other limitations include the exclusion of non-invasive brain stimulation and pharmacological interventions, guidelines in languages other than English, and care standards. Care standards (non-governing quality-of-care statements published by best-practice groups) were not reviewed in this study as their development methods can vary widely, compromising reliability and quality. 29 However, care standards play an important role in supplementing clinical practice guidelines, and clinicians are encouraged to refer to high-quality care standards such as the Australian Aphasia Rehabilitation Pathway. 30 In this review, we did not use the PICO framework 24 to organize treatment-related recommendations by area of outcome (e.g. language), but rather instead grouped recommendation by area of practice, in line with the previous review. 7 Also of note, is that our “Highest Evidence” ratings are based on study design alone. Consideration of risk of bias/methodological quality may alter these ratings.
Conclusion
We identified 82 recommendations from nine high-quality clinical practice guidelines that can inform clinical practice for people with post-stroke aphasia. There is a need for aphasia-specific research in “accessing community supports,” “return to work, leisure, driving,” and “interprofessional practice.” Rigorous research in these areas may strengthen the existing evidence-base and improve the clinical services delivered to people with aphasia.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231161454 – Supplemental material for An updated systematic review of stroke clinical practice guidelines to inform aphasia management
Supplemental material, sj-docx-1-wso-10.1177_17474930231161454 for An updated systematic review of stroke clinical practice guidelines to inform aphasia management by Bridget Burton, Megan Isaacs, Emily Brogan, Kirstine Shrubsole, Monique F Kilkenny, Emma Power, Erin Godecke, Dominique A Cadilhac, David Copland and Sarah J Wallace in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.G., E.B., K.S., and E.P. are current members of the speech pathology working group for the Australian Stroke Foundation—Clinical Guidelines for Stroke Management. D.C. is a current member of the Australian Stroke Foundation—Clinical Guidelines for Stroke Management Steering Committee. During this research, E.P. was on the Research Advisory Committee of the Stroke Foundation. E.G. was an expert panel member in development of the Australian Aphasia Rehabilitation Pathway Best Practice Statements. E.P. led the expert panel on the Australian Aphasia Rehabilitation Pathway Best Practice Statements.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a National Health and Medical Research Council (NHMRC) Investigator Grant (grant no. 1175821) awarded to S.J.W. and The Center for Research Excellence in Aphasia Rehabilitation and Recovery (Aphasia CRE; grant no. 1153236, La Trobe University (ABN: 64 804 735 113).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
