Abstract
Rationale:
Clinical practice guidelines support structured, progressive protocols for improving walking after stroke. Yet, practice is slow to change, evidenced by the little amount of walking activity in stroke rehabilitation units. Our recent study (n = 75) found that a structured, progressive protocol integrated with typical daily physical therapy improved walking and quality-of-life measures over usual care. Research therapists progressed the intensity of exercise by using heart rate and step counters worn by the participants with stroke during therapy. To have the greatest impact, our next step is to undertake an implementation trial to change practice across stroke units where we enable the entire unit to use the protocol as part of standard of care.
Aims:
What is the effect of introducing structured, progressive exercise (termed the Walk ’n Watch protocol) to the standard of care on the primary outcome of walking in adult participants with stroke over the hospital inpatient rehabilitation period? Secondary outcomes will be evaluated and include quality of life.
Methods and sample size estimates:
This national, multisite clinical trial will randomize 12 sites using a stepped-wedge design where each site will be randomized to deliver Usual Care initially for 4, 8, 12, or 16 months (three sites for each duration). Then, each site will switch to the Walk ’n Watch phase for the remaining duration of a total 20-month enrolment period. Each participant will be exposed to either Usual Care or Walk ’n Watch. The trial will enroll a total of 195 participants with stroke to achieve a power of 80% with a Type I error rate of 5%, allowing for 20% dropout. Participants will be medically stable adults post-stroke and able to take five steps with a maximum physical assistance from one therapist. The Walk ’n Watch protocol focuses on completing a minimum of 30 min of weight-bearing, walking-related activities (at the physical therapists’ discretion) that progressively increase in intensity informed by activity trackers measuring heart rate and step number.
Study outcome(s):
The primary outcome will be the change in walking endurance, measured by the 6-Minute Walk Test, from baseline (T1) to 4 weeks (T2). This change will be compared across Usual Care and Walk ’n Watch phases using a linear mixed-effects model. Additional physical, cognitive, and quality of life outcomes will be measured at T1, T2, and 12 months post-stroke (T3) by a blinded assessor.
Discussion:
The implementation of stepped-wedge cluster-randomized trial enables the protocol to be tested under real-world conditions, involving all clinicians on the unit. It will result in all sites and all clinicians on the unit to gain expertise in protocol delivery. Hence, a deliberate outcome of the trial is facilitating changes in best practice to improve outcomes for participants with stroke in the trial and for the many participants with stroke admitted after the trial ends.
Introduction and rationale
The ability to regain walking independence is one of the most frequently cited goals of people living with stroke. 1 Despite many trials, meta-analyses, and guidelines2–4 over the last decade which show that structured, progressive exercise is safe and effective for individuals post-stroke, activity continues to be very low.5,6 In our recent study—six sites over three provinces between 2014 and 2018 (n = 75)—participants with stroke did a small amount of walking (averaged 580 steps) in their usual daily physical therapy sessions. 7 We then found that a structured, progressive protocol integrated with typical daily physical therapy resulted in substantially greater walking activity during therapy sessions and improved walking (60 m on a 6-Minute Walk Test (6MWT)) and quality of life measures over usual care with walking gains maintained for 1 year after stroke. 7 One therapist on the unit was assigned to deliver the intervention and progressed the intensity of exercise through heart rate and step counters worn by the participants with stroke during therapy.
To have the greatest impact, our next step is to undertake an implementation trial to improve practice across stroke units where we enable the entire unit (rather than one assigned therapist) to use the protocol as part of standard of care. This implementation trial is also informed by lessons learned from our stakeholders where we formally engaged with 15 clinicians 8 and 10 participants with stroke 9 about their experience with the structured, progressive exercise protocol.
The aim is to determine what is the effect of introducing structured, progressive exercise (termed Walk ’n Watch protocol) to the standard of care on the primary outcome of walking in adult participants with stroke over the hospital inpatient rehabilitation period? We hypothesize that the Walk ’n Watch protocol, compared to Usual Care, will result in improvements of walking endurance in participants with stroke over the inpatient rehabilitation period. We also hypothesize the improvements in our secondary outcomes, which include quality of life. Changes in standard care will be undertaken through developing local expertise through hands-on workshops with physical therapists and rehabilitation assistants to integrate the Walk ’n Watch protocol within their therapy.
Methods
Design
This national, multisite-randomized controlled clinical trial will use a cross-sectional, stepped-wedge cluster design. This design was chosen following a small efficacy trial 2 to facilitate implementation of the intervention across Canada and to prevent contamination and disappointment effects in hospitals not randomized to the intervention. The 12 inpatient hospital sites can be found on the trial registry (www.ClinicalTrials.gov ID: NCT04238260).
The coordinating center (the Rehabilitation Research Program) will prepare the protocol and study documents; organize meetings, data entry, and analysis; disseminate results through publications and presentations; and liaise with site coordinators. The coordinating center will have access to final de-identified data sets, which will be collected from each site throughout the study, inputted into master sheets and double-checked. Individual sites will retain access to site-specific data. The steering committee (authors of this article) will provide agreement of the final protocol and review any adverse events, study progress, and any changes of the study that will be updated on the trial registry. Authorship guidelines will follow the recommendations of the International Committee of Medical Journal Editors. No professional writers will be used.
Patient population
Broad inclusion criteria will be purposely implemented to capture real-world caseloads in stroke rehabilitation units. To be included, participants will be adults, will have a confirmed stroke that occurred within 12 weeks of rehab admission, admitted for inpatient stroke rehabilitation for walking, medically stable (e.g. stable cardiovascular condition, no active cancer), able to walk five steps with a maximum of one person helping, and able to understand and follow instructions. Study coordinators will be knowledgeable in supportive conversations for people with aphasia, and we will provide the option to have a caregiver present for consent. The aim is to recruit individuals as soon as they are admitted to the inpatient stroke rehabilitation unit. Participants will be excluded if they have another neurological condition, enrolled in another rehabilitation study, or expected to receive less than 2 weeks of inpatient physical therapy. If a potential participant is enrolled in a trial during acute care, we will communicate with the trialist about their study to ensure outcomes for each trial will not be affected prior to enrolling. Each site’s study coordinator will obtain consent and enroll participants. Participants will be aware they are receiving standard care for each phase of the study and will only be enrolled in one phase.
Randomization
Randomization schedule details are given in Figure 1. Prior to the start of the trial, three sites were randomly allocated to each of the four transition sequences by the trial statistician using statistical software. To mitigate the risk of loss to power due to expected large variation in enrolments across sites, randomizations were stratified on expected enrolments (four largest sites in stratum 1, remaining eight sites in stratum 2). 10 Each site will be notified of the site’s transition date after recruiting their first participant and will not be notified of the transition dates of other sites.
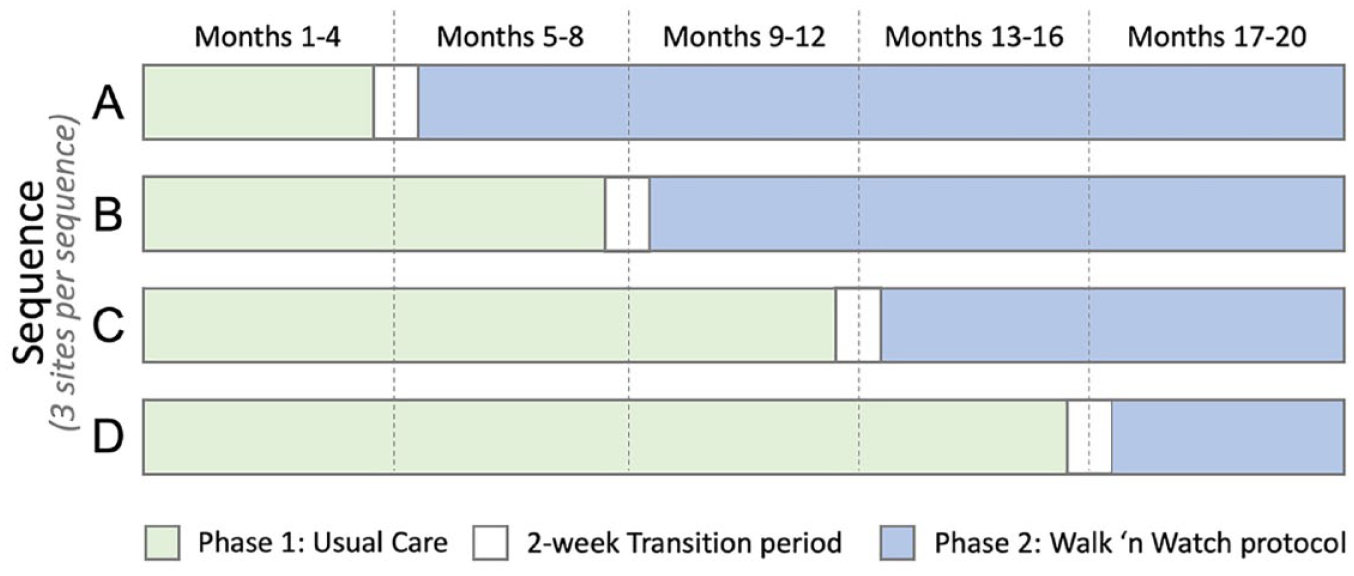
Randomization schedule.
Physical therapy
Specific exercises and treatments will be up to the discretion of the treating therapist. Any discontinuation of treatment will be determined by the site rehabilitation department as per usual rehabilitation planning. Study staff will document any circumstances leading to discontinuation.
Phase 1
Phase 1 is the care that physical therapists typically provide their participants with stroke. No changes to treatment are made during this phase.
The 2-week transition period
Training for the Walk ’n Watch protocol will be undertaken with hands-on workshops with physical therapists and rehabilitation assistants in the 2-week transition period between phase 1 and phase 2 (Figure 1). During this time, recruitment is paused to avoid contamination. Assessors are not made aware of the timing of the switch from phase 1 to phase 2.
Phase 2
To avoid contamination, a brief outline of the protocol is summarized here, and an in-depth description will be provided at the end of the trial using the TIDeiR framework. The Walk ’n Watch protocol focuses on completing a minimum of 30 min of weight-bearing, walking-related activities that progressively increase in intensity. The protocol will aim to achieve individualized, progressive step targets based on a participant’s walking distance at baseline (Table 1) and work toward 40–60% heart rate reserve for 30 min, 5 days a week.
Progression of Walk ’n Watch protocol.
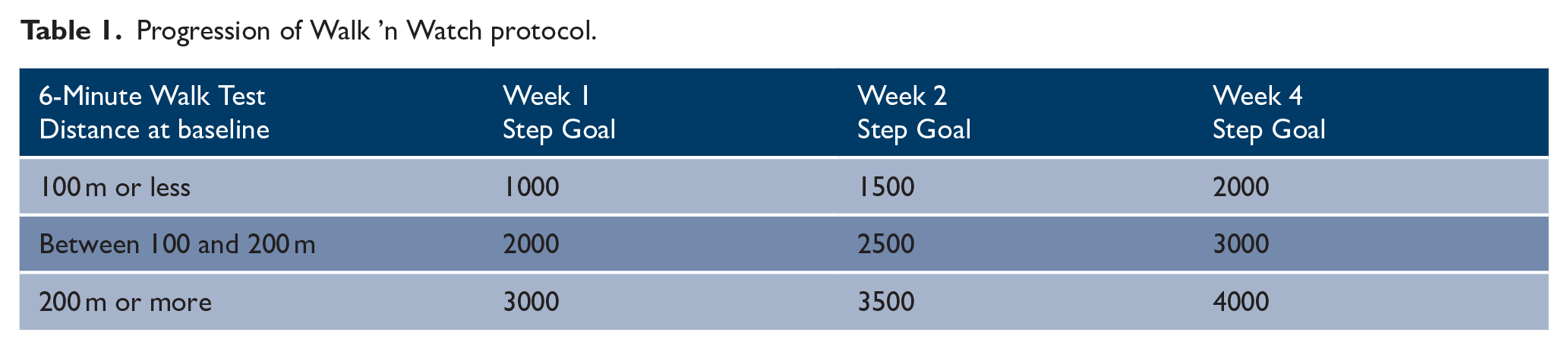
Focus will be on walking and functional weight-bearing activities, to work toward meeting national stroke guidelines.3,4 The inpatient unit will be provided with an activity tracking watch that clinicians will use to monitor heart rate (Garmin Forerunner 235, Garmin Ltd., USA) and a step counter to monitor the number of steps (Fitbit Inspire, Alphabet Inc., USA, placed on the ankle) 11 during physical therapy sessions. All eligible participants with stroke admitted to the unit will receive the Walk ’n Watch protocol as it will be considered standard care, even if they have not consented to the study. Participants with stroke who consent to the study will also receive their own activity tracking watch.
Outcome measures
Physical and cognitive abilities, physical activity, and quality of life will be measured at three time points—baseline (T1), 4 weeks later (T2), and 12 months post-stroke (T3)—by a trained assessor blinded to time of the switch-over to the Walk ’n Watch phase (Figure 2). We will also have two telephone calls at 6 and 9 months post-stroke, to ask about any changes in health.
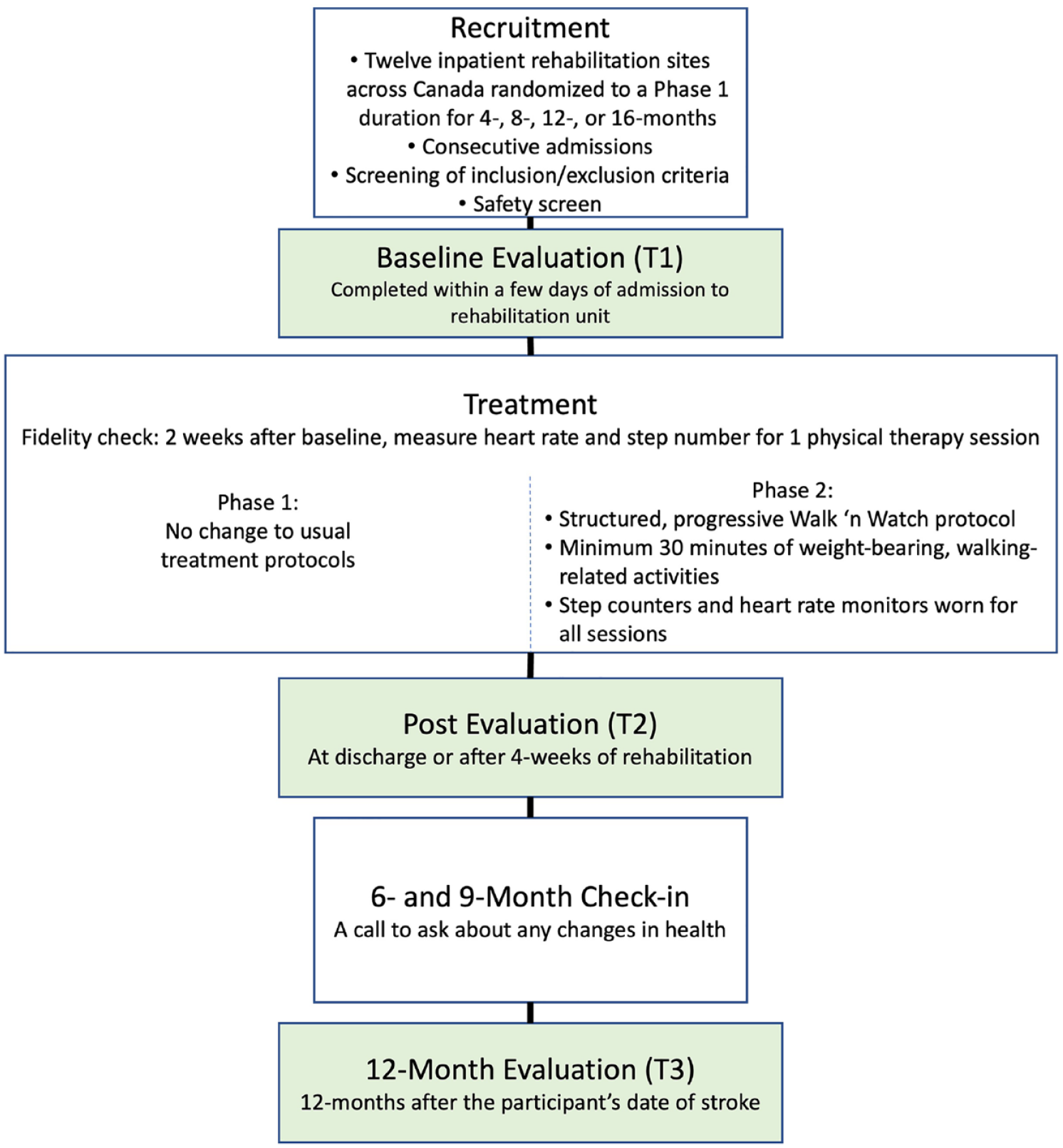
Overview of study procedures during phases 1 and 2.
The primary outcome will be the change in walking endurance (6MWT) from T1 to T2. Secondary outcome measures and assessment timing are given in Table 2.
Overview of study outcome measures.
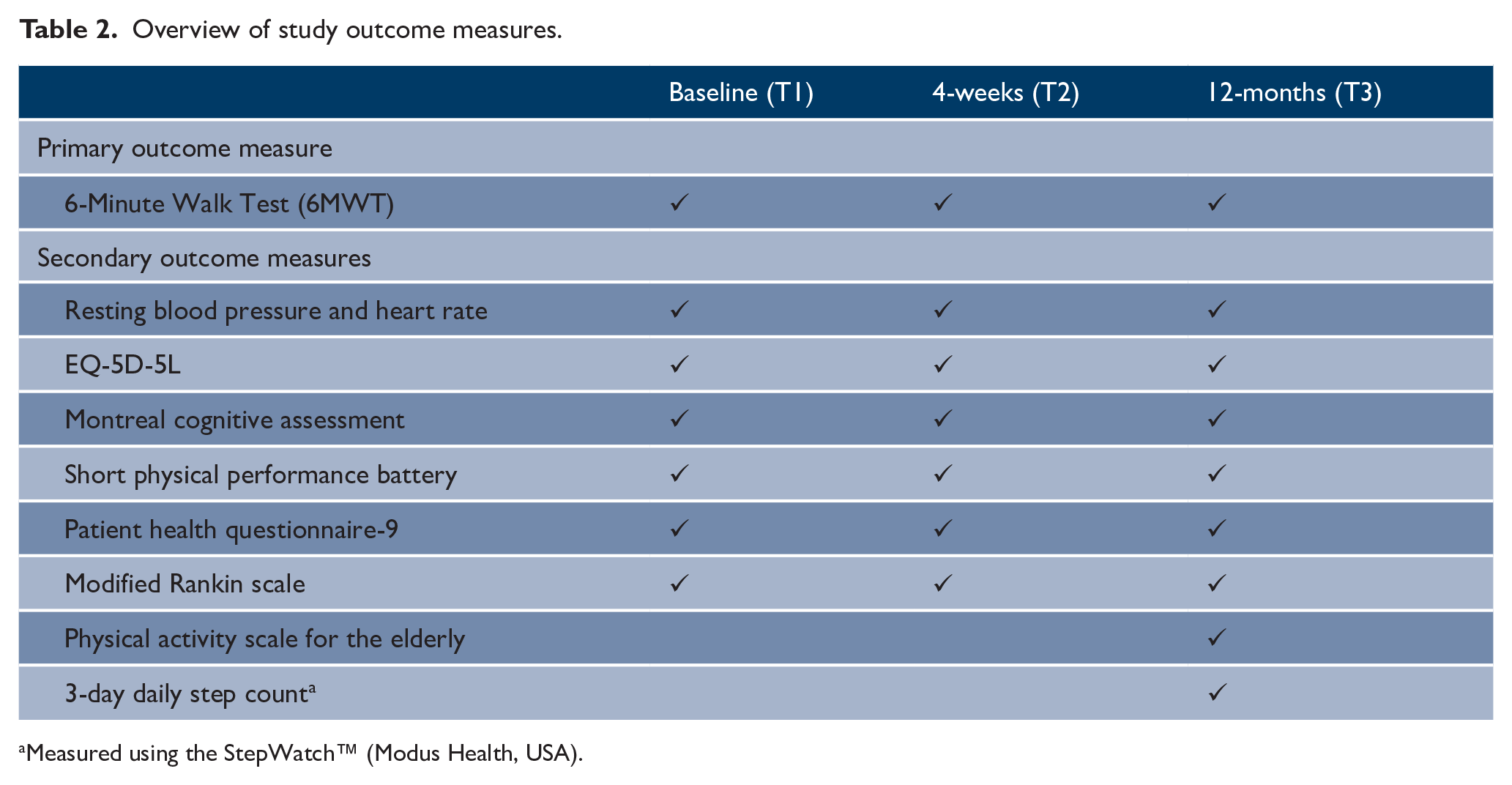
Measured using the StepWatch™ (Modus Health, USA).
Further planned evaluation of intervention fidelity and protocol adherence (developed using the Normalization Process Theory) is through: (1) weekly meetings with the unit therapists (led by the site coordinator) documenting barriers and facilitators of the implementation of Walk ’n Watch, (2) qualitative interviews with a smaller set of site therapists and rehabilitation assistants, and (3) survey of all therapists, rehabilitation assistants, and site managers who participated.
Sample size
The planned total sample size is 195, allowing for a 20% dropout. The power calculations were performed using the R package, “swCRTdesign.” 12 The input values were derived from a mixed-effects model analysis of the pilot trial data, 7 in which the treatment effect was a 60-m improvement in the 6MWT, and the effective (adjusted for covariates including baseline 6MWT) within group standard deviation was 90 m. The intracluster correlation coefficient (ICC) was essentially zero, but we used a value of 0.01 in the power calculation to be conservative. Based on these input values and an assumed cluster auto-correlation coefficient (CAC) of 0.8, a design with a total of 12 clusters, and 3 clusters transitioning at each step, requires 13 participants per cluster, or 156 participants in total, to achieve 80% power with a Type I error rate of 5%. (Note that this calculation is insensitive to the assumed CAC value due to the low ICC value.) Allowing for 20% dropout increases the sample size to 195.
Statistical analysis
The primary analysis will compare, using intent-to-treat, the change in 6MWT for the Walk ’n Watch protocol versus Usual Care using a linear mixed-effects model. A random intercept for site will be included to account for clustering. The model will adjust for the calendar time as a continuous variable (to account for the staggered start dates across sites) instead of periods of time. Other adjustment variables will include the baseline 6MWT. A detailed Statistical Analysis Plan document that identifies the full list of adjustment variables will be finalized prior to database lock. Adjustment for time effects in the stepped-wedge design induces large increases in the treatment effect standard error. As a supplementary analysis to explore how inferences might differ if expert judgments regarding the time effect are utilized, we will also conduct a Bayesian analysis to increase precision by incorporating an informative prior on the time effect that will be elicited from stroke rehabilitation experts. 13 Secondary outcomes will be analyzed using mixed-effects models similar to that used for the primary outcome. Planned exploratory subgroup analyses will assess the impact of Walk ’n Watch by sex and age.
Safety and adverse event monitoring
Three physiatrists form the Safety Monitoring Board for this implementation study. Major safety concerns have already been tested in the pilot trial, and no serious adverse events occurred related to the protocol. 7 All sites will monitor and report any serious adverse events that occur throughout the study. As recommended by the Canadian Stroke Aerobics Guidelines, the 6MWT will be used as a safety screen prior to commencing physical therapy.
Discussion and summary
Clinical practice guidelines, studies in the literature, and our own studies support structured, progressive protocols for improving walking after stroke. Implementing a stepped-wedge design enables the protocol to be tested under real-world conditions, involving all clinicians on the unit, and results in all sites and all clinicians on the unit to ultimately gain expertise in delivering the protocol to facilitate best practice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Canadian Institutes of Health Research (Operating Grant FDN 143340 (J.J.E.); Fellowship (S.P.)), Canada Research Chair Program (J.J.E.), Heart and Stroke Foundation Canadian Partnership for Stroke Recovery Operating Grant (J.J.E.), Michael Smith Foundation for Health Research Fellowship (S.P.), and Michael Smith Foundation for Health Research Scholar Award (B.M.S.). No funders contributed to any aspect of the study.
Ethical approval
Ethics approval already obtained from each site.
Availability of data and materials
The datasets will be available 12 months after publication of the trial at UBC Dataverse, a publicly accessible data repository.
