Abstract
Background and aim
Whether cerebral microbleeds cause cognitive impairment remains uncertain. We analyzed whether cerebral microbleeds are associated with cognitive dysfunction in patients with symptomatic cerebral small vessel disease, and whether this association is independent of other neuroimaging markers of cerebral small vessel disease.
Methods
We analyzed consecutive patients with MRI-confirmed lacunar stroke included in DNA-Lacunar-2 multicenter study. Cerebral microbleeds were graded using the Brain Observer Microbleed Rating Scale (BOMBS). Neuropsychological assessment was performed using the Brief Memory and Executive Test (BMET). We analyzed the association between cerebral microbleeds, BMET, and the following subdomains: executive function/processing speed and orientation/memory. We also searched for an independent association between cerebral microbleeds and vascular cognitive impairment, defined as BMET ≤ 13.
Results
Out of 688 included patients, cerebral microbleeds were detected in 192 (27.9%). After adjusting for white matter hyperintensities severity, lacune count, and other confounders, both the presence and the number of cerebral microbleeds were significantly associated with impaired cognitive performance [β = −13.0; 95% CI = (−25.3, −0.6) and β = −13.1; 95% CI = (−19.8, −6.4), respectively]. On analysis of specific cognitive domains, associations were present for executive function/processing speed [β = −5.8; 95% CI = (−9.3, −2.2) and β = −4.3; 95% CI = (−6.2, −2.4), respectively] but not for orientation/memory [β = −0.4; 95% CI = (−4.0, 3.2) and β = −2.1; 95% CI = (−4.0, 0.1), respectively]. We also found an independent association between the presence and the number of cerebral microbleeds and vascular cognitive impairment [adjusted OR = 1.48; 95% CI = (1.01, 2.18) and OR = 1.43; 95% CI = (1.15, 1.79), respectively].
Conclusion
In a large cohort of symptomatic cerebral small vessel disease patients, after controlling for other neuroimaging markers of cerebral small vessel disease severity, cerebral microbleeds were associated with cognitive dysfunction. Executive function and processing speed were predominantly impaired. This might suggest a causal role of cerebral microbleeds in determining vascular cognitive impairment.
Introduction
Cerebral microbleeds (CMBs) are perivascular focal deposits of haemosiderin-laden macrophages, ranging in diameter between 0.80 and 1.5 mm, visible as signal voids on T2*-weighted gradient echo (GE) and susceptibility weighted imaging (SWI) magnetic resonance imaging (MRI).1,2 Together with lacunar infarcts, white matter hyperintensities (WMH) and enlarged perivascular spaces, CMBs are a recognized neuroimaging marker of cerebral small vessel disease (cSVD). 3 CMB frequency increases with age; 4 also, they are common in patients with stroke, cognitive impairment, and dementia.5,6
Whether CMBs are independently associated with cognitive impairment in patients with stroke and cSVD is uncertain, with studies to date giving inconsistent results. In patients with previous stroke or TIA, those with CMBs showed impaired executive function and this was particularly associated with CMBs in the frontal region and basal ganglia. 7 However, in another study of stroke patients, CMBs were not associated with cognitive performance during a four-year follow up. 8 In patients with cSVD and radiological lacunar stroke and confluent white matter hyperintensities, CMBs were only associated with cognition when very frequent. 9 Furthermore, the relationship was weakened after controlling for other markers of cSVD.
A key question when evaluating any association between CMB and cognition is whether they are independent markers of cognition, or merely acting as markers for other features of cSVD such as lacunar strokes and WMH which themselves are known to be associated with cognition.10,11 Previous studies have shown strong associations between CMB and both lacunar infarcts and WMH in patients with cSVD.3,12 It is also uncertain whether CMBs associate particularly with specific cognitive domains. For example, CMB in cSVD patients in whom the predominant pathology is hypertensive arteriopathy might be expected to associate more strongly with cognitive domains dependent on subcortical integrity, such as executive function and processing speed.
To further investigate these relationships, we determined associations between CMB and cognition in a large cohort of MRI confirmed lacunar stroke patients. Furthermore, we evaluated whether any associations were independent of other radiological markers of cSVD.
Materials and methods
Study population
Patients were studied who had been recruited to the prospective DNA-Lacunar-2 study, an ongoing multicenter study recruiting patients with lacunar stroke from 51 hospitals across the UK. Participants recruited between 10 August 2016 and 1 April 2020 were included in the analysis for this paper. The inclusion criteria were a clinical lacunar syndrome with an anatomically corresponding lacunar infarction confirmed on brain MRI, and the MRI performed within one year of stroke. Study participants were recruited within two years since the stroke onset; those showing any other potential cause of stroke other than cSVD (including large artery disease causing a stenosis > 50%, or cardio-embolic source) or with any other potential cause of white matter disease (such as multiple sclerosis) were excluded. Further inclusion criteria for this analysis were: the availability of hemosiderin-sensitive sequences on MRI; and the availability of a complete assessment of cognitive functions through the Brief Memory and Executive Test (BMET).
At the time of inclusion, a large number of clinical and laboratory variables were prospectively collected for each participant using prespecified forms. Information about pre-stroke functional status, pre-existing vascular risk factors, and medication at the time of the stroke were also recorded. The definitions of these variables are available in the “e-Methods” section in the Supplementary Material.
Neuroimaging variables
Clinical MRI scans were used. After recruitment, all original MR images were centrally reviewed with case histories by a neurologist to confirm eligibility. The severity of cSVD was determined radiologically at the time of the eligibility review. The number of total lacunar infarcts, including both acute lacunar lesions hyperintense on DWI and cavitated lacunar lesions on FLAIR/T1 sequences, was determined. A lacune on FLAIR/T1 was defined as a cerebrospinal fluid-filled cavity, 3–15 mm in diameter, with a surrounding rim of high signal intensity following a perforator artery distribution. 13 The Fazekas scale was used to grade the extent of WMH on T2-FLAIR. 14 This was determined separately for both for the periventricular (PV) and deep white matter (DWM) regions, each of them giving a 0–2 subscore; an overall WMH score (0–3) was also determined.
The presence, total count, and location of CMBs were graded by a single neurologist (SN) using the Brain Observer Microbleed Rating Scale (BOMBS). 15 CMBs were defined as focal area of very low intensity, <10 mm of diameter, on SWI/GE sequences. Hypointense lesions were excluded if they appeared to be vascular flow voids (based on sulcal location or linear shape), basal ganglia mineralization, or artifacts from an adjacent bone or sinus. All uncertain cases were reviewed with a consultant neuroradiologist (AM) to reach a consensus. Only CMBs meeting the BOMBS certain criteria were analyzed. The CMBs location was described according to the seven anatomical brain regions proposed by the scale, and also grouped into the following three sub-categories: cortical (for patients having CMB(s) in the cortex only), subcortical (for patients having CMB(s) in the subcortical white matter, basal ganglia, internal/external capsule, thalamus, brainstem, and/or cerebellum), and mixed (cortical and subcortical CMBs) distribution. The identification of CMB lesions was blinded to the assessment of cognitive function.
Neuropsychological assessment
All included patients were assessed with the BMET. This is a short cognitive screening tool, designed and validated to be sensitive for the detection of vascular cognitive impairment (VCI) in cSVD patients.16–18 The test is freely available at www.bmet.info. It includes a number of subscores, each ranked on an age normative scale from 0 to 2, which is used to form a total score (ranged on a scale from 0 to 16). A BMET total score of ≤13 has been previously validated to define the presence of VCI. 16 The test performance can be subdivided into two major cognitive domains clusters, executive functioning/ processing speed (EF/PS) and orientation/ memory (O/M), each with a score ranging from 0 to 8. The EF/PS subscore is calculated from the tasks: (i) letter-number matching, (ii) motor sequencing, (iii) letter sequencing, and (iv) number-letter sequencing, while the O/M subscore from the tasks: (i) orientation, (ii) five‐item repetition, (iii) five‐item recall, and (iv) five‐item recognition memory.
Statistical analysis
In the study population, ordinal and continuous variables were summarized with median and interquartile range (IQR), categorical ones with absolute counts and percentage. Demographics, clinical, and radiological variables in patients with and without CMBs, and in patients with and without VCI, were compared with appropriate tests: we used the Mann–Whitney test to compare continuous data, the chi-square test for categorical data, and the chi-square test for trend for ordinal data.
The relationship between CMBs and the cognitive performance on the BMET was investigated with multivariate linear regression models. Two models were tested: in model A we adjusted for age, sex, time from stroke onset to recruitment, years of education, and modified Rankin score (mRS) at consent time; in model B we added WMH severity score and lacune count as further variables for adjustment. We applied a logarithm transformation (base = 10; start = 1) to reduce the positively skewed distribution for CMBs total number, and the squared transformation for the negatively skewed distribution of the BMET total score and its sub-scores. A nonparametric model (Kernel regression) was also fitted to assess the sensitivity of the results, adjusting for the same covariates. As further sensitivity analyses, we studied the association between CMBs and cognition in the subgroup of patients included within the median onset to randomization time and in the subgroup of patients without previous stroke/TIA.
Lastly, a logistic regression model was used to search for independent associations between CMBs (presence, number and location) and VCI. The same adjustments as used in models A and B were applied.
Statistical analyses were performed using STATA software. p < 0.05 were regarded as significant.
Standard protocol approvals and patient consents
The study was approved by the East of England - Cambridge Central Research Ethics Committee (16/EE/0201). All study participants signed informed consent form at time of recruitment.
Data availability statement
Anonymized data will be shared by request from any qualified investigator.
Results
Patients characteristics
Demographics, vascular risk factors, clinical, and radiological characteristics of the included patients and in the subgroups of patients with and without CMBs. Ordinal and continuous variables are summarized with median and interquartile range (IQR), categorical ones with absolute counts and percentage

mRS: modified Rankin scale (at the time of consent); HTN: hypertension; DM: diabetes mellitus; MI: myocardial infarction; BMI: body mass index; TIA: transient ischemic attack; IVT: intravenous thrombolysis; WHM: white matter hyperintensity; PV: periventricular; DWM: deep white matter.
Indicates significant result.
At least one CMB was detected in 192 (27.9%) patients. Of these, 57 (29.7%) patients had 1 CMB, 60 (31.3%) had 2 to 4, 37 (19.3%) had 5 to 9, 25 (13.0%) had 10 to 19, and 13 (6.8%) had ≥ 20 CMBs. The most frequent CMBs locations were the cortex (with 55% of patients having at least one CMB in this location), the subcortical white matter (45% of patients) and the thalamus (35% of patients; Supplementary Table 2). The overall distribution was strictly subcortical in 87 (45.3%), mixed (cortical and subcortical) in 78 (40.6%) and strictly cortical in 27 (14.1%) patients.
Compared to patients without CMBs, patients with ≥ 1 CMB(s) showed more hypertension (n = 157, 82% vs. n = 352, 71%, p = 0.004) and previous history of stroke/TIA (n = 35, 18% vs. n = 38, 8%, p < 0.001), and were more frequently on antiplatelet therapy at the time of the stroke (n = 58, 30% vs. n = 110, 22%, p < 0.001; Table 1). They did not differ from patients without CMBs in age, sex, time since stroke, or vascular risk factors other than hypertension (Table 1). On MRI, the presence of CMB(s) was associated with higher lacune count score (p < 0.001) and higher total, PV, and DWM WMH scores (all p < 0.001).
In the overall population, median BMET total score was 14 (IQR = 12–16), and VCI was present in 282 (41%) patients. Characteristics of subjects with and without VCI are presented in Supplementary Table 3. Results from the univariate analysis showed that patients with VCI, compared to patients without, were predominantly male (n = 197, 70% vs. n = 244, 40%, p = 0.009), had higher frequency of diabetes (n = 80, 28% vs. n = 71, 18%, p < 0.001) and higher BMI (28 vs. 26, p = 0.020). There was no significant difference regarding age or level of education. On neuroimaging, VCI was significantly associated with WMH load, higher lacune count, and both the presence and the number of CMBs.
CMBs, cognitive performance, and cognitive domains
Associations between CMBs (presence, number, and top-decile) and cognitive functions, expressed by BMET total score and O/M and EF/PS sub-scores. Results from two linear regression models are presented: model A included age, sex, stroke onset to consent time, education, and mRS at consent time as variables for adjustment; in model B we added WMH score and lacune count as further variables for adjustment. A squared transformation of cognitive scores and a logarithmic transformation of CMBs number were used. Results are expressed as β coefficient and 95% CI
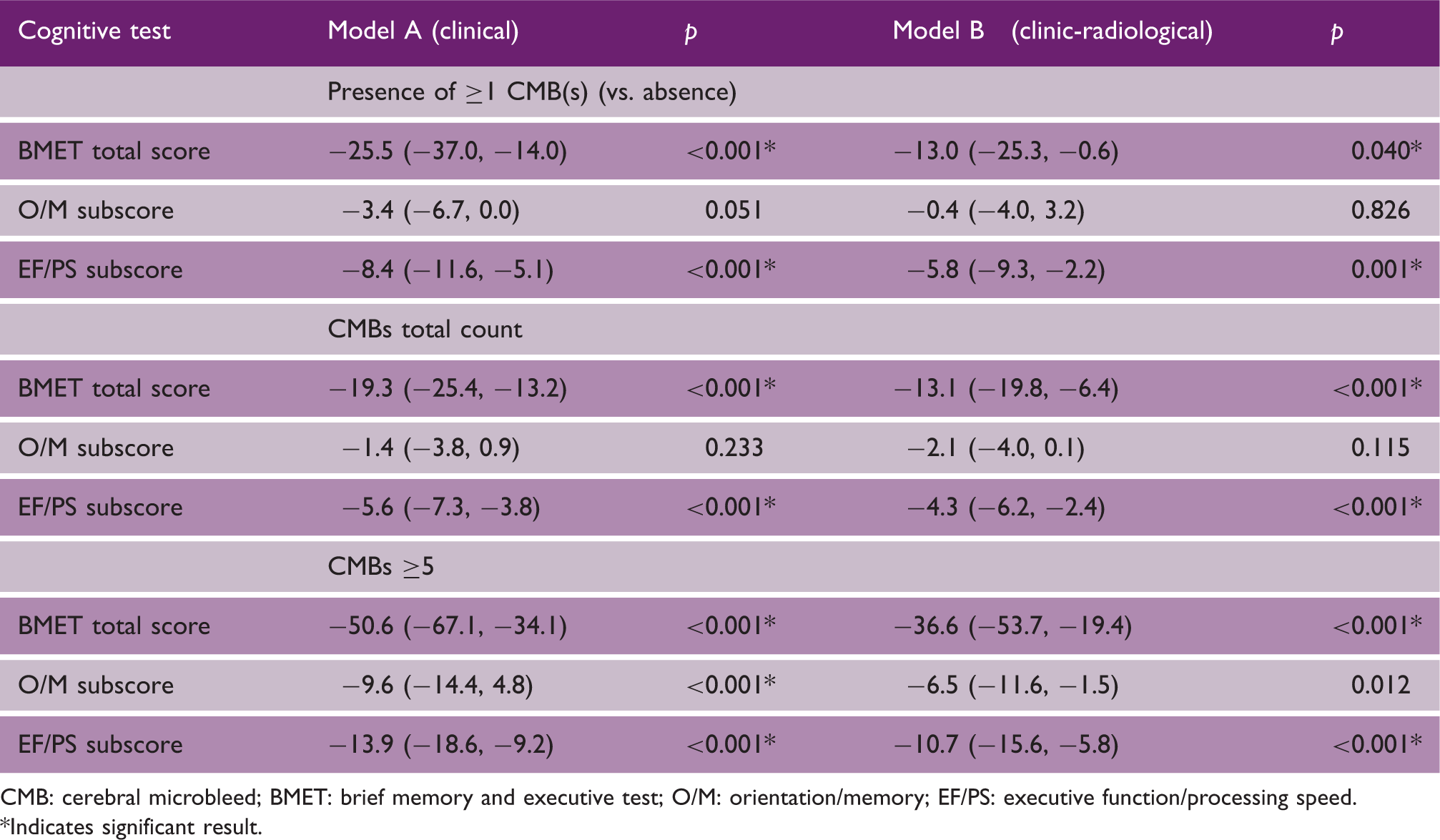
CMB: cerebral microbleed; BMET: brief memory and executive test; O/M: orientation/memory; EF/PS: executive function/processing speed.
Indicates significant result.
On analysis of specific cognitive domains, significant negative associations were present between both the presence and the total count of CMBs with the EF/PS subscore [β = −5.8; 95% CI = (−9.3, −2.2) and β = −4.3; 95% CI = (−6.2, −2.4), respectively, in the fully adjusted model B, Table 2]. In contrast, no association was found between CMBs (presence or total count) and performance on the O/M subscore. However, CMB count in the top decile was associated with worse performance on both EF/PS and O/M domains [β = −5.8; 95% CI = (−9.3, −2.2) and β = −4.3; 95% CI = (−6.2, −2.4), respectively, in the fully adjusted model B, Table 2].
Results from a nonparametric regression model confirmed the associations found in the linear regression model: CMBs total count was related to lower scores on total BMET and on EF/PS subscore, but was not associated with the O/M subscore (see Supplementary Table 4).
As sensitivity analyses, the analysis of patients included within 37 days from stroke onset (N = 346) and the analysis of patients without previous stroke/TIA (N = 614) confirmed the main results on the entire population, i.e. both the presence of any CMBs and the CMBs total count were independently associated with lower scores on EF/PS subscore of the BMET after full adjustment for clinic-radiological variables, but not with the O/M subscore (Supplementary Table 5).
Associations between cerebral microbleeds (CMBs) location and executive functions/processing speed (EF/PS) performance, and orientation and memory (O/M) tasks
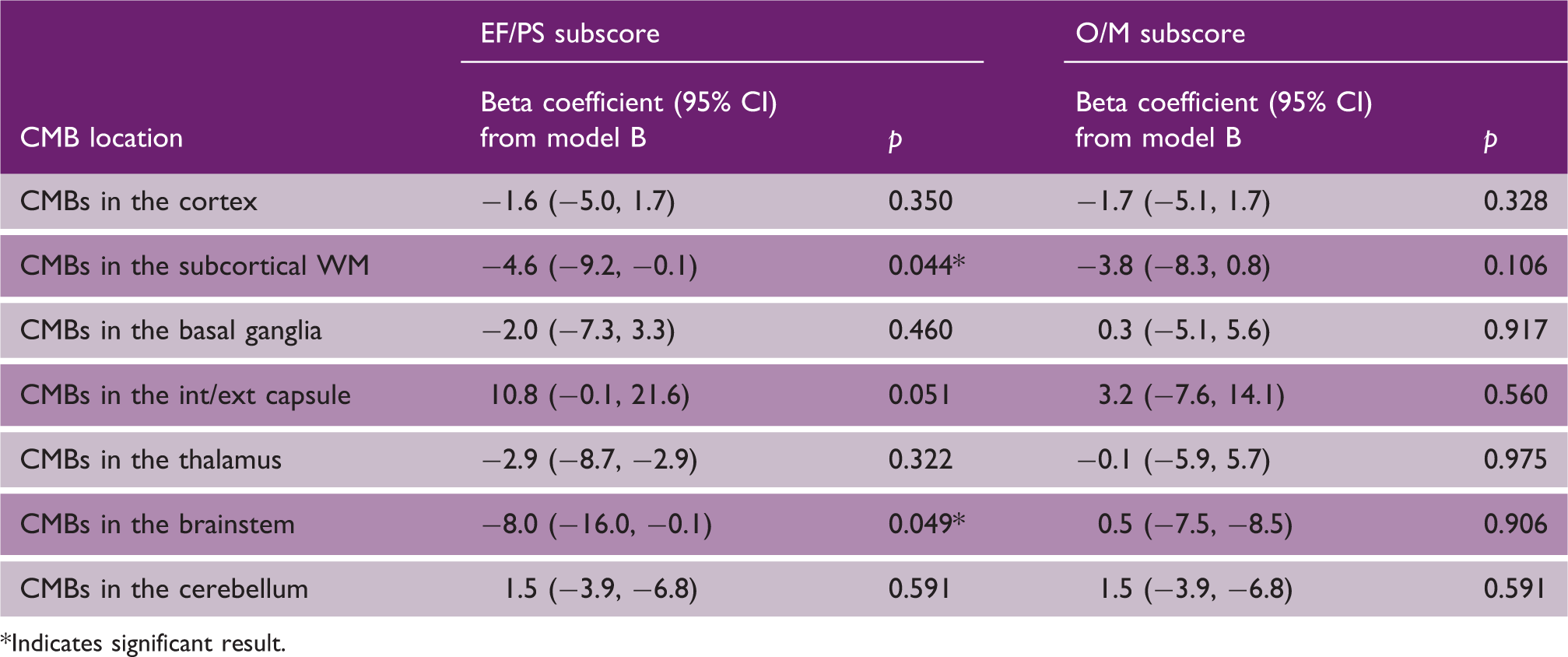
Indicates significant result.
CMBs and vascular cognitive impairment
Relationship between cerebral microbleeds (CMBs; presence, total count, and top-decile) and VCI. Results are presented for model A (clinical adjustment) and model B (clinic-radiological adjustment), and are expressed as OR and 95% CI
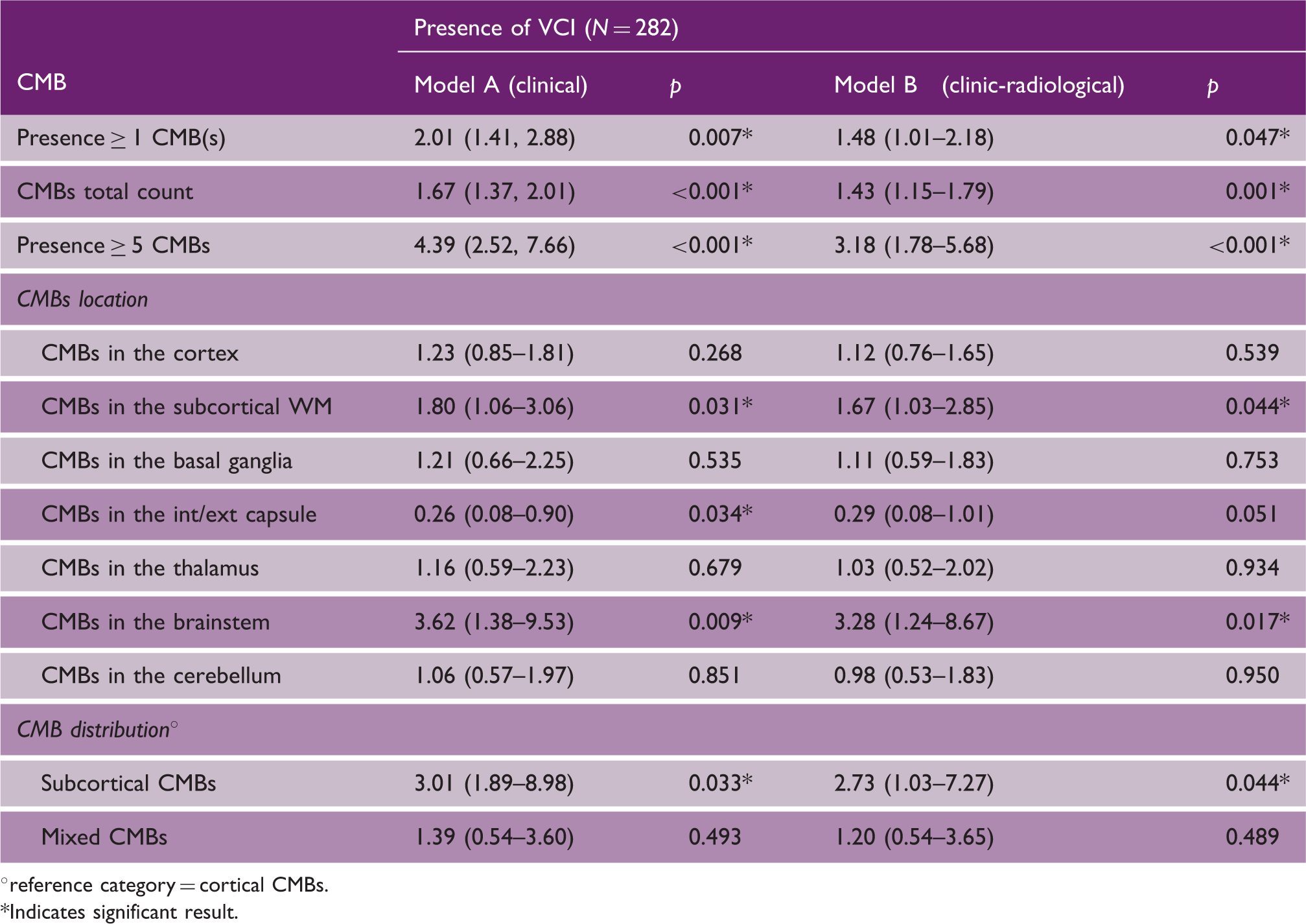
°reference category = cortical CMBs.
Indicates significant result.
Regarding CMB location, the presence of strictly subcortical CMB was an independent predictor of VCI after covarying for risk factors and other MRI markers of cSVD (OR = 2.73, p = 0.044 in model B). In contrast, there was no similar association seen for strictly cortical or mixed distribution CMB. Looking at associations with specific brain regions, significant associations between CMB and VCI were seen for the deep white matter and brainstem, after controlling for risk factors and MRI markers of cSVD (model B; Table 4).
Discussion
In a large cohort of symptomatic cSVD patients, CMBs were associated with both impaired cognition and VCI. Importantly, this association persisted after controlling for other MRI markers of cSVD, suggesting that CMB are independently related to cognitive impairment and therefore may play a causal role. The association between CMBs and cognitive dysfunction was strongest in those with greater number of CMBs, was particularly related to subcortical CMBs, and strongest with the executive function and processing speed cognitive domain.
Whether CMBs are independently related to cognition in cohorts with cerebrovascular disease and cSVD has been uncertain, with previous smaller studies reporting inconsistent results.7–9,19 Importantly, associations reported in a number of studies could merely reflect CMBs acting as a marker of more severe disease rather than an independent association. In our homogenous cohort of patients with cSVD who presented with a lacunar stroke, we confirmed an independent relationship between the presence of CMBs and cognitive impairment, which remained significant after adjusting for the number of lacunar infarcts and the severity of WMH. This finding provides support that CMBs are independent contributors to cognitive impairment.
Our study showed that cognition was most impaired in patients with large numbers of CMBs. In the subgroup of patients having at least five CMBs (corresponding to the top decile distribution) we found the lowest BMET scores, lowest EF/PS and O/M subscores, and a higher frequency of VCI. These findings are in line with previous studies showing that the presence of a high CMB count was associated with an increased risk of cognitive deterioration, both in cerebrovascular patients9,19,20 and in the general population.21–23 This result may suggest a threshold effect of CMBs burden on cognition, with higher number of lesions being associated with more severe and multi-domains cognitive decline.
Our analysis also suggested that the effect of CMBs on cognition may depend on CMB location, with VCI being mostly associated with subcortical lesions. In particular, CMBs located in the subcortical white matter and in the brainstem showed the stronger relationship with the impairment of executive function and processing speed. These associations are consistent with the hypothesis that cerebral microvascular damage preferentially affects white matter and subcortical gray matter, ultimately leading to disruption of integrity of fronto-subcortical circuits and interfering with cognition.23,24 Lesions in the brainstem have been implicated in executive dysfunction. Previous reports suggested that patients with pontine brain infarcts frequently had executive dysfunction, 25 and that CMBs in the pons were associated with worse cognitive performance in CADASIL patients. 26 These findings are in line with our results, lending support to the hypothesis that disturbance of the frontal‐subcortical circuits, including projections into the brainstem, could be responsible for the executive dysfunction in patients with lacunar stroke.
The cognitive impact of CMBs according to their topographic distribution may be dependent on the underlying vascular pathology: while in population-based studies higher number of strictly lobar CMBs, presumably resulting from cerebral amyloid angiopathy, were related to cognitive decline and dementia,27,28 in our cohort of patients with stroke and cSVD, the presence of CMBs in deep regions, resulting primarily from hypertensive arteriopathy, was associated with greater decline in the executive functions, with relative sparing of memory and orientation tasks.
Mechanisms by which CMBs influence cognitive function remain speculative. The strong association we found between CMBs and cognition, which was independent of other neuroimaging markers of cSVD, suggest that CMBs are not simply a marker for the severity of small vessel disease, but might have a causal impact on cognitive function. Previous studies have suggested that CMBs may cause a direct damage to white matter tracts, with subsequent disconnection of functionally important cortical–subcortical circuits.7,19 Recent studies using diffusion tensor imaging and tractography have suggested the disruption of complex brain networks dependent on white matter connections plays a central role in the cognitive impairment seen in cSVD.29,30 The disruption of brain networks involving the frontal lobe and periventricular white matter was important to determine executive dysfunction and cognitive impairment seen in cSVD, and this may also be the case for CMBs.31,32
Another possible pathophysiological mechanism by which CMBs could impair cognition is suggested by studies from animal models, which reported that CMBs induced functional and structural disturbances in the surrounding brain environment: experimentally induced microbleeds disrupted the function of nearby neurons and astrocytes, 33 and promoted an inflammatory response via microglia proliferation and migration, 34 although most of these effects were transient. Alternatively, CMBs might relate to cognition via an indirect effect, through an arteriolar narrowing leading to cerebral hypoperfusion and microischemic damage. 35 In this latter scenario, higher number of CMBs may thus indicate more extensive and severe subclinical microvascular damage.
Our study has a number of strengths. These include the large number of prospectively collected patients, the homogenous characteristics of the included population (with all patients showing at least a recent lacunar stroke confirmed on MRI), and the systematic assessment of all included patients with the same neuropsychological test (BMET). The BMET has been shown to be more sensitive to the cognitive deficit in patients with cSVD than the Mini–Mental State Examination and the Montreal Cognitive Assessment test, and also allows the analysis of different cognitive subdomains, in which we showed stronger associations with the EF/PS domain. 24
There are also some limitations. First, the cross-sectional design of our study limits the capability to validate a causal relationship between CMBs and cognition, and longitudinal studies in large cohort of cSVD are required. Second, the number of included patients varied greatly across the participating centers, which might have introduced some center-effects in the analysis; we tried to minimize this by introducing a BMET training for each site members and by performing a central revision of all brain MRIs. Third, we used clinical scans performed on different scanners which included both GE and SWI sequences. However, this is likely to have reduced to the power to detect associations and despite this we were able to detect strong associations between CMB and cognition. Lastly, we used a visual rating of WMH, which could be less sensitive to the pathology burden compared to the volumetric measures of WMH.
Conclusion
We demonstrated that CMBs exhibit a strong association with VCI, especially in the domains of executive functions and processing speed. This association was independent of other neuroimaging markers of cSVD, suggesting a potential role of CMBs in the pathophysiology of cognitive deterioration. A direct tissue damage on white matter tracts, rather than or in addition to underlying cSVD, may account for the detrimental effects of CMBs on cognition. Further large longitudinal studies are needed to confirm our conclusions.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930211012837 - Supplemental material for Cognitive impact of cerebral microbleeds in patients with symptomatic small vessel disease
Supplemental material, sj-pdf-1-wso-10.1177_17474930211012837 for Cognitive impact of cerebral microbleeds in patients with symptomatic small vessel disease by Stefania Nannoni, Laura Ohlmeier, Robin B Brown, Robin G Morris, Andrew D MacKinnon, Hugh S Markus and for the DNA Lacunar 2 investigators in International Journal of Stroke
Footnotes
Acknowledgements
All DNA-Lacunar 2 Centers and Collaborators are listed in the Supplementary Material.
Authors’ contribution
S.N. was involved in study concept and design, screened papers for the literature review, performed the statistical analysis, and prepared the article with input from H.S.M. L.O. and R.B. helped in data acquisition and in critical revision of the article for important intellectual content. RGM helped in cognitive data interpretation and critical revision of the article. A.D.M. helped in neuroradiological data interpretation and critical revision of the article. H.S.M. was involved in study concept and design, supervised analysis and interpretation of data, and revised the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a British Heart Foundation programme grant (RG/4/32218). Recruitment was supported by the NIHR Clinical Research Network. S.N.’s salary is funded by an MRC experimental medicine grant (MR/N026896/1). R.B.B. is the recipient of an ABN Clinical Research Training Fellowship funded by Guarantors of Brain. H.S.M. is supported by an NIHR Senior Investigator award. Infrastructural support was provided by the Cambridge University Hospitals NIHR Biomedical Research Center. The views expressed are those of the authors and not necessarily the views of the NHS, the NIHR, or the Department of Health and Social Care.
Disclosure statement
The authors (S.N., L.O., R.B.B., R.G.M., A.D.M., H.S.M.) report no disclosures relevant to the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
