Abstract
Background
Effectiveness of early intensive aphasia rehabilitation after stroke is unknown. The Very Early Rehabilitation for SpEech trial (VERSE) aimed to determine whether intensive aphasia therapy, beginning within 14 days after stroke, improved communication recovery compared to usual care.
Methods
Prospective, randomized, single-blinded trial conducted at 17 acute-care hospitals across Australia/New Zealand from 2014 to 2018. Participants with aphasia following acute stroke were randomized to receive usual care (direct usual care aphasia therapy), or one of two higher intensity regimens (20 sessions of either non-prescribed (usual care-plus or prescribed (VERSE) direct aphasia therapy). The primary outcome was improvement of communication on the Western Aphasia Battery-Revised Aphasia Quotient (AQ) at 12 weeks after stroke. Our pre-planned intention to treat analysis combined high intensity groups for the primary outcome.
Findings
Among 13,654 acute stroke patients screened, 25% (3477) had aphasia, of whom 25% (866) were eligible and 246 randomized to usual care (n = 81; 33%), usual care-plus (n = 82; 33%) or VERSE (n = 83; 34%). At 12 weeks after stroke, the primary outcome was assessed in 217 participants (88%); 14 had died, 9 had withdrawn, and 6 were too unwell for assessment. Communication recovery was 50.3% (95% CI 45.7–54.8) in the high intensity group (n = 147) and 52.1% (95% CI 46.1–58.1) in the usual care group (n = 70; difference −1.8, 95% CI −8.7–5.0). There was no difference between groups in non-fatal or fatal adverse events (p = 0.72).
Interpretation
Early, intensive aphasia therapy did not improve communication recovery within 12 weeks post stroke compared to usual care.
Introduction
Evidence before this study
The 2016 Cochrane review 1 indicates that aphasia therapy is effective for chronic language/communication recovery. There is no Level 1 evidence for the effectiveness of aphasia treatments started within the first two weeks post stroke when the proposed ‘window of opportunity’ 2 for enhanced neuronal recovery is at its peak. The few studies which commenced treatment within the first 15 days post stroke have mixed findings;3–5 the most recent study (Rotterdam Aphasia Therapy Study-3 (RATS-3)) 3 suggested that early intervention is no better than starting therapy after the first month post stroke.
Added value of this study
VERSE 6 is the first international aphasia trial, which recruited from 17 acute hospitals. VERSE participants were followed up at 45 subacute and community healthcare centers to six months post stroke. VERSE will contribute to Level 1 evidence about the effectiveness of intensive early aphasia therapy compared to usual care in a broad population of acute aphasic stroke patients. Very early intensive, aphasia therapy during the hypothesized optimal recovery window 2 had no additional benefit to language impairment, discourse/connected speech, quality of life, or depression at 12 or 26 weeks post stroke to usual care. Critically, there was a large increase in amount of aphasia therapy in this trial (9.5 h up from a total of 14 min) compared to our earlier pilot study 5 , which looked promising.
Our findings are in line with those of the A Very Early Rehabilitation Trial (AVERT) 7 whereby an increase in the intensity of ‘usual care’ services from pilot studies to the main study was reported and the Phase III trial was subsequently negative. Taken together with the AVERT 7 results, VERSE provides a useful guide for the design and analysis of future stroke rehabilitation RCTs in early stroke recovery.
Background
Aphasia (language impairment affecting spoken language, comprehension, reading and writing) is not rare, affecting approximately one in three 8 of the 16.9 million people worldwide who experience stroke each year. 9 People with aphasia have greater mortality and morbidity, lower levels of social participation and return-to-work rates, and an almost three-fold greater risk of experiencing depression than stroke survivors without aphasia. 10 The recent global estimates of post-stroke aphasia burden in relation to annual disease costs are AUD$49.3 billion 11 ($34.4b USD).
Restorative and compensatory rehabilitation delivered by speech pathologists is the mainstay of treatment for aphasia. There are no animal equivalent basic science models to drive aphasia recovery research. The theoretical constructs that underpin aphasia research derive from an integration of cognitive and motor theoretical domains, and extrapolation is required to explain language recovery. Evidence from human motor and animal stroke recovery models suggests the first 90 days post stroke is the ‘window of opportunity’ 2 where the greatest potential to harness spontaneous recovery exists. 12 This theory suggests that high frequency repetition should strengthen neural networks and minimize independent neuronal activation, potentially reducing maladaptive behaviors.2,12 Neurorecovery therapeutic principles guide rehabilitation research and aim to strengthen interaction within and between neural networks and to promote improved accuracy and efficiency of function.2,12 Efficacy of aphasia therapy for functional communication was established in the Cochrane review (27 randomized trials including 1620 patients) 1 and supported by a recent European trial 13 which showed the benefit of intensive aphasia therapy in chronic recovery. The trial provided 10 h of direct therapy per week for three weeks for a total of 31 (median) h with an additional 15 h (total) of home practice.
Currently, there is no Level I evidence that directly addresses the timing, intensity, and type of aphasia therapy, commencing within the first two weeks post stroke. 1 Mixed results are noted from various studies3–5 comparing treatment intensity when intervention was commenced within two weeks post stroke. Laska et al. 4 (N = 123) reported no benefit in speech production and comprehension (Norsk Grunntest for Afasi 14 ) or in functional communication (Amsterdam–Nijmegen Everyday Language Test 15 ) after 15 h of a comprehension-based intervention across three weeks, when compared to no therapy. Our Phase I pilot study 5 (N = 59) indicated a statistical and clinical benefit from daily intensive therapy targeted at spoken language. Over a mean of 19 days, 7.5 h of therapy (or 2.7 h per week) was compared to standard care (on average <1 h in total). Language and communication benefits were observed at therapy completion and six months post stroke on the Western Aphasia Battery-Revised 16 and the Functional Communication Profile. 17 The more recent RATS-3 trial 3 (N = 153) demonstrated no benefit of early therapy consisting of 24.5 h over four weeks (6.1 h per week) when compared to no therapy in the first month, measured at 4, 12, and 26 weeks post stroke. However, only 29% of the intervention group complied with the prescribed treatment dose of 28 h of direct aphasia therapy. The ‘per-protocol’ analysis of the RATS-3 trial 3 identified benefit of early treatment compared to no treatment at four weeks post stroke, on measures of spoken language. Our Phase II pilot trial 18 (N = 20) that was designed to investigate the effect of treatment type received in early aphasia recovery (group therapy vs. individual therapy), for 20 h provided over four weeks (5 h per week), which is at the lower intensity range found to be effective. 19 The trial found no between-group difference.
While there is no Level 1 evidence for the effectiveness of very early aphasia therapy, similar research had been completed in the area of physical rehabilitation through the AVERT trial. 7
The uncertainty around the timing, intensity, and type of aphasia therapy in the very early stroke recovery window (commencing therapy before day 15 post stroke) prompted the VERSE trial.6,20 Our primary hypothesis was that compared to usual care alone (UC), higher dose early aphasia therapy would result in improved language and communication at 12 weeks post stroke, measured by the Western Aphasia Battery-Revised 16 (primary outcome) and discourse analysis 21 (secondary outcome). To explore the question of therapy type, the higher dose therapy could be delivered in one of two ways (UC-Plus and VERSE therapy—outlined below). We further hypothesized that higher dose training would result in better quality of life, and be cost effective. This article reports primary and key secondary outcomes for the trial.
Methods
Study design
A phase three, multicenter, randomized controlled trial, with allocation 1:2 (UC: UC-Plus and VERSE) and blinded outcomes assessed at 12 (primary outcome) and 26 weeks post stroke. The trial protocol 6 and statistical analysis plan (SAP) 20 were accepted for publication before unblinding (see Supplement and publications for full trial methodology description). The trial was approved by hospital ethics committees and all participants gave written informed consent using aphasia friendly consent processes. 22
Participants
Participants required the capacity to consent to be eligible and were recruited from 17 acute-care hospitals in Australia and New Zealand and then followed up at 45 subacute and community healthcare centers.
Eligibility criteria
Participants were aged over 18 years and admitted to hospital with an acute stroke, resultant acute aphasia of any type, within 14 days of stroke onset. They required a score of less than 93.7 on the Aphasia Quotient of the Revised Western Aphasia Battery (WAB-R AQ) 16 indicating mild to severe aphasia. They were medically stable, could maintain a wakeful alert state for at least 30 min, and had normal or corrected hearing and vision. Exclusion criteria were pre-existing aphasia and dementia, a concurrent progressive neurological disorder, any head injury, neurosurgery, clinical depression at admission, inability to participate in English-based therapy, or participation in other concurrent intervention trials.
Qualified speech pathologists trained to screen and consent participants, diagnosed aphasia using the Frenchay Aphasia Screening Test, 23 and enrolled eligible participants.
Randomization and masking
Baseline assessments were completed by trained staff before randomizing participants using a computer-generated, block randomization sequence (permutated block of six, with 1:1:1 randomization to three groups (UC, UC-Plus, VERSE) to achieve an overall ratio of 1:2 (UC; high intensity therapy, combined UC-Plus and VERSE) 19 via Research Electronic Data Capture (REDCap™). 24 Randomization was performed by the baseline assessor who was not otherwise involved in the trial and participants were stratified by aphasia severity determined by the WAB-R (AQ) score. Participants, family members, ward staff, and outcome assessors were not informed of group allocation and all participants and trial staff were asked not to discuss treatment received. Only UC staff wrote in medical notes per healthcare standards and all research documentation was stored separately in a secured location to avoid unblinding. Blinded outcome assessors were not involved in participants’ stroke care and were not permitted to ask participants about treatment received during follow-up assessments. Only treating therapists (acute and subacute) were unblinded to treatment allocation.
Procedures—Treatment intensity and type
All speech pathology services received by participants up to 26 weeks post stroke were recorded for all groups. 6 Our focus was the recording of direct aphasia therapy which was defined as ‘treatment designed to restore language and communication function’ and could consist of individual or group social and or impairment-based treatment, social or communication device training. Education, counseling, goal-setting, treatment programming, documentation, and consultation were excluded from these analyses. 20
Usual care
Participants randomized to the UC arm constituted the trial control group. UC aphasia therapy was standard care at each site and was not controlled for amount, frequency of sessions, therapy type, or therapist. For data reporting purposes, we asked UC therapists to record all aspects of management and to video record aphasia therapy sessions, although videorecording was not mandatory.
High intensity therapy regimens
Participants randomized to either of the high intensity groups (UC-Plus or VERSE) received usual care therapy and had additional aphasia therapy provided by specially trained therapists. UC-Plus therapy involved any combination of therapy at the discretion of the therapist. Participants were prescribed 20 sessions of 45–60 min (15–20 h; or 4–5 h per week) of aphasia therapy, commencing before day 15 and completed within four weeks.16,20 The intensity level for these groups was chosen in-line with available evidence (≥5 h per week 19 ) and demonstrated tolerability.5,18
VERSE treatment was an impairment-based therapy program (see Supplement; Intervention Protocol), developed by an aphasia Expert Advisory Committee. The VERSE intervention prioritized error-free, verbal communication, encouraging conversation while working between 50% and 80% accuracy at each goal level to maintain a therapy challenge point. In both higher intensity groups, the amount of therapy and the timing of commencement of intervention were standardised. 6
Training, therapy, and assessment fidelity
All treating speech pathologists underwent protocol training and received procedural training manuals relevant to the arm of therapy they provided. Training details and treatment manuals are provided in Appendix 1. Clinical support was given by central research staff for UC-Plus therapists, and a VERSE Therapy Coordinator provided feedback and support for VERSE therapists. UC-Plus and VERSE therapists were allocated to a therapy group from a therapist recruitment pool, depending on trial needs. Therapists providing UC-Plus and VERSE intervention groups were mandated to video record a therapy session each week and submit this to an independent therapy integrity monitor for review and verification (see Supplement—Main and Intervention Protocols). All data discrepancies were queried by the Monitor with clinical staff and all queries resolved prior to database lock in compliance with all elements of the TIDiER 25 and SPIRIT 26 statements.
Outcomes
The trial SAP 20 outlines the primary and secondary outcomes in detail. Our primary outcome was improvement in communication at 12 weeks after stroke, measured by the WAB-R AQ which is a sensitive, valid and reliable measure of aphasia performance with a standard error of measurement of 3 points. 27 Improvement was assessed using the percent of maximal potential recovery achieved (%MPR). 28 The %MPR represents a percentage score of the maximum potential change (i.e. the endpoint score minus the baseline score) allowing for direct meaningful comparison of amount of possible improvement on the WAB-R AQ for people with varied aphasia severity. Using the %MPR also accounts for the ceiling effects of the standardized test. All raw scores are presented in Supplemental Table S3. Based on benchmarking of aphasia intervention studies, a 5-point change on the WAB-R AQ represents a conservative, clinically meaningful effect. 29
Secondary outcomes were: treatment effectiveness (both %MPR and discourse measures 20 ) at 26 weeks, word naming (Boston Naming Test 30 ), quality of life (Stroke and Aphasia Quality of Life scale-39 31 ), depression (Aphasia Depression Rating Scale 32 ) at 12 and 26 weeks. The Aphasia Depression Rating Scale was completed by the participant with communication support from the blinded assessor. Safety (adverse and serious adverse events) was assessed at 12 and 26 weeks post stroke. Adverse event reporting followed a protocol, 20 an independent medical officer adjudicated events, and reported to the Data Safety and Monitoring Board.
Statistical analysis
Adverse events and serious adverse events
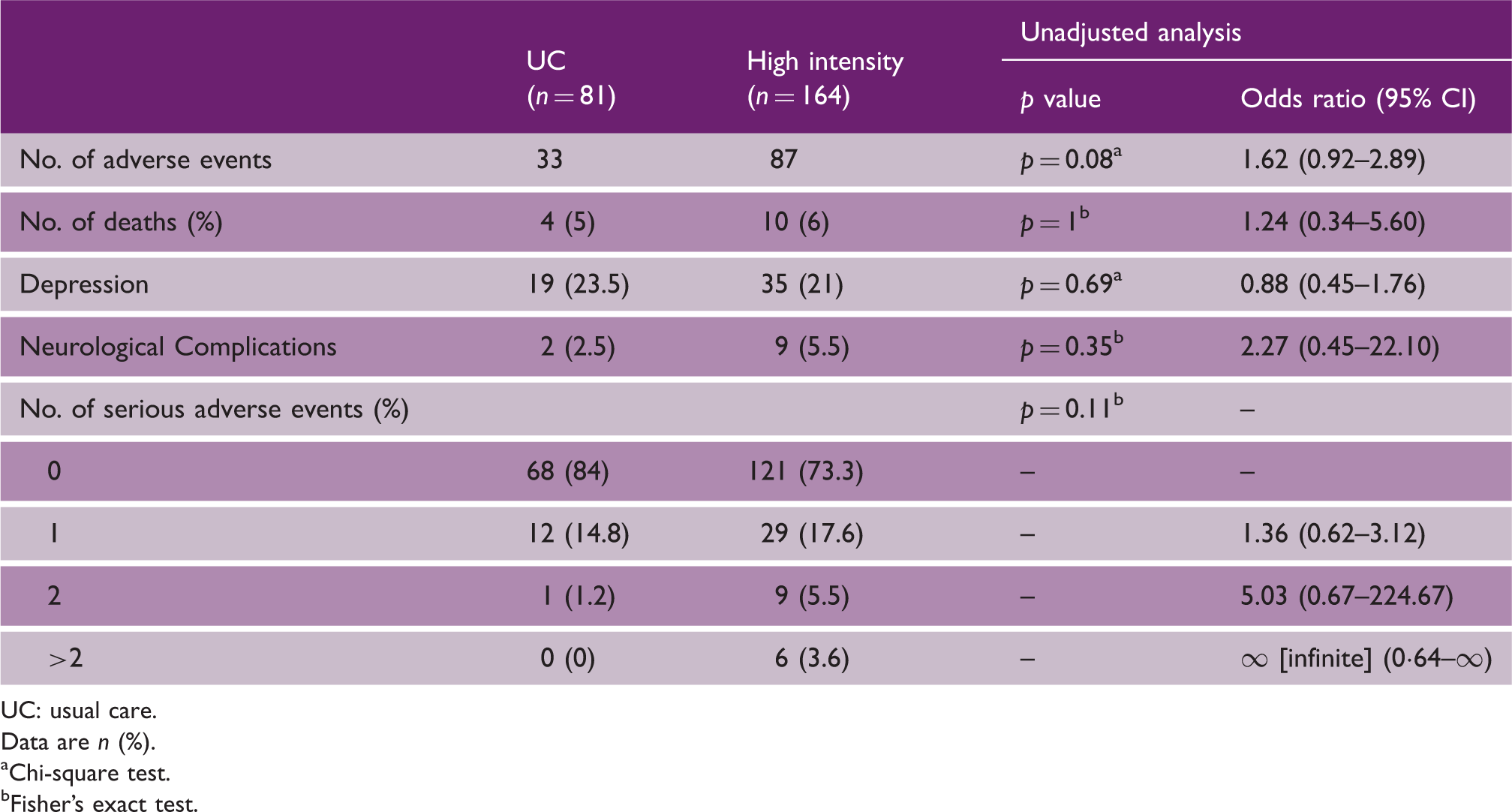
UC: usual care.
Data are n (%).
Chi-square test.
Fisher’s exact test.
The primary and secondary outcome analyses are reported without imputation of missing data, 20 supplemented by analyses with imputed data sets (see Supplement p. 7—Imputation Plot). 20 An additional efficacy subgroup analysis explored age, sex, baseline aphasia severity (AQ), and disability (mRS 34 ). Subgroup analysis of treatment type (UC-Plus compared to VERSE subgroup comparisons) used a linear mixed model (see Supplemental Table S2). No adjustments were made to p values for multiple comparisons in subgroup analyses or the analyses of secondary outcomes. All analyses were completed using R 35 and statistical analyses were performed by the trial statistician and verified by a statistician blinded to group allocation. The trial was registered with the Australian New Zealand Clinical Trials Registry (ACTRN12613000776707); and Universal Trial Number (U1111-1145-4130).
Data availability
VERSE individual deidentified participant trial data (including data dictionary) will be available through the CATS 36 international aphasia data repository.
Results
Between 4 June 2014 and 10 February 2018, 13,654 patients with acute stroke were screened; 3477 (25%) had aphasia, 866 (25%) of whom were trial eligible, and 246 (28%) participants were enrolled (Figure 1). Last patient, last visit occurred on 31 July 2018. One randomized participant withdrew consent; their data were withdrawn from analysis (N = 245). At the primary endpoint (week 12), 14 (6%) participants had died, 9 (4%) had withdrawn, and 6 (2.5%) were too unwell for assessment, leaving 217 (88%; within the 20% lost to follow-up allowance
20
) participants in the primary analysis.
CONSORT study flow diagram.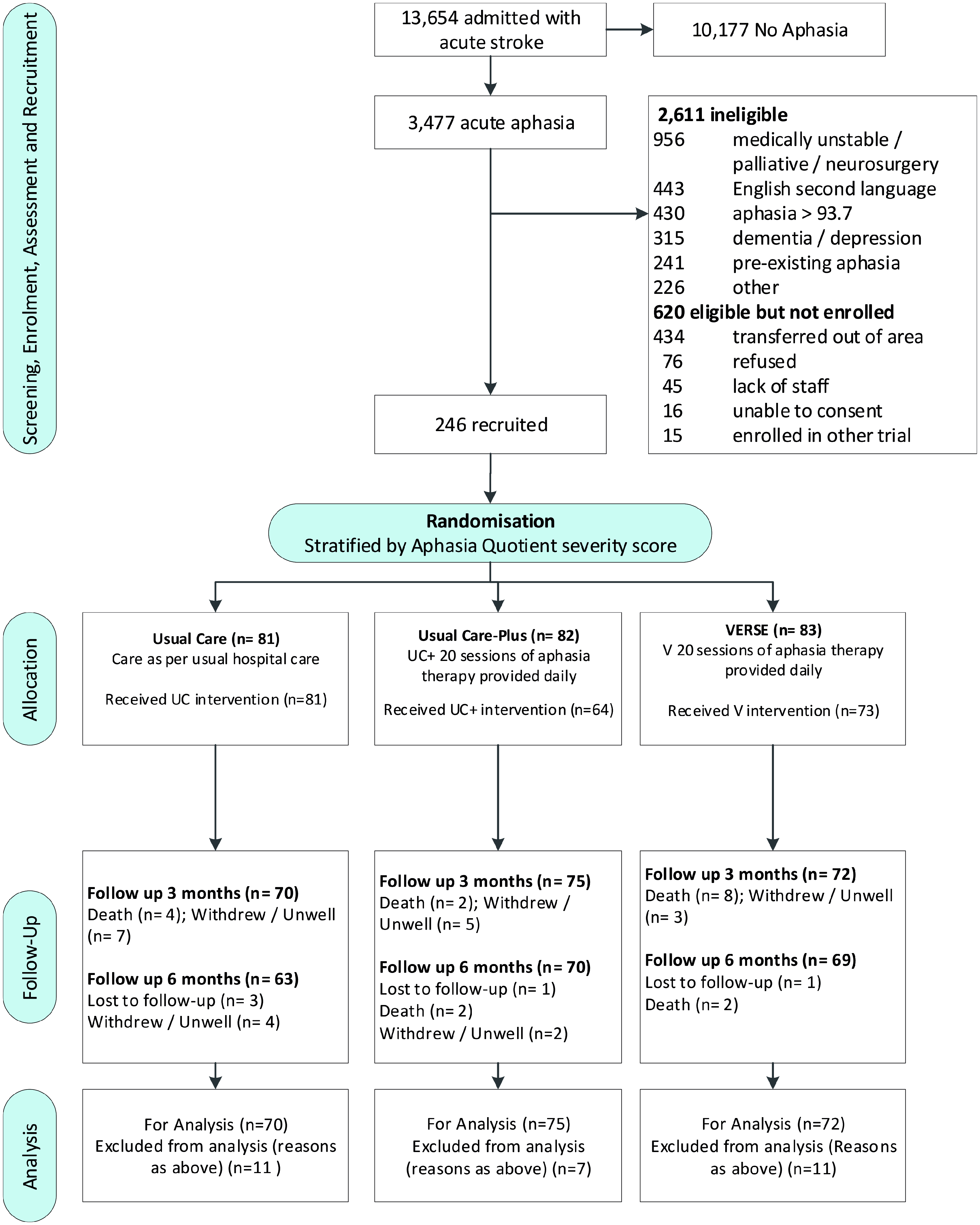
Baseline characteristics of the intention-to-treat population

NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; TACS: total anterior circulation stroke; PACS: partial anterior circulation stroke; POCS: posterior circulation stroke; LACS: lacunar stroke; AusTOMS: Australian Therapy Outcome Measures.
Data are mean (SD), n (%), or median (IQR).
Hospital stay and intervention characteristics
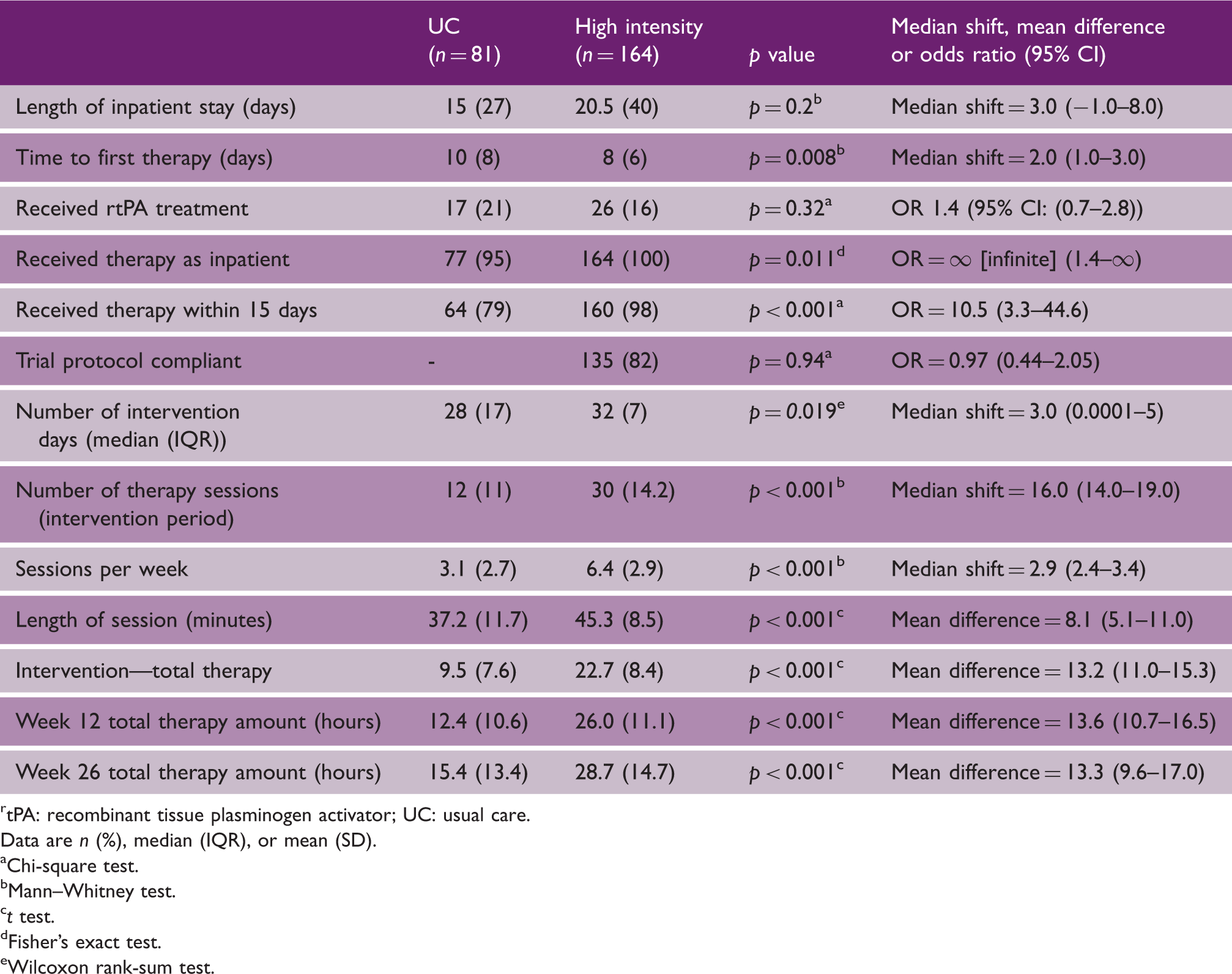
tPA: recombinant tissue plasminogen activator; UC: usual care.
Data are n (%), median (IQR), or mean (SD).
Chi-square test.
Mann–Whitney test.
t test.
Fisher’s exact test.
Wilcoxon rank-sum test.
Treatment components (type) of usual care, usual care-Plus, and VERSE intervention
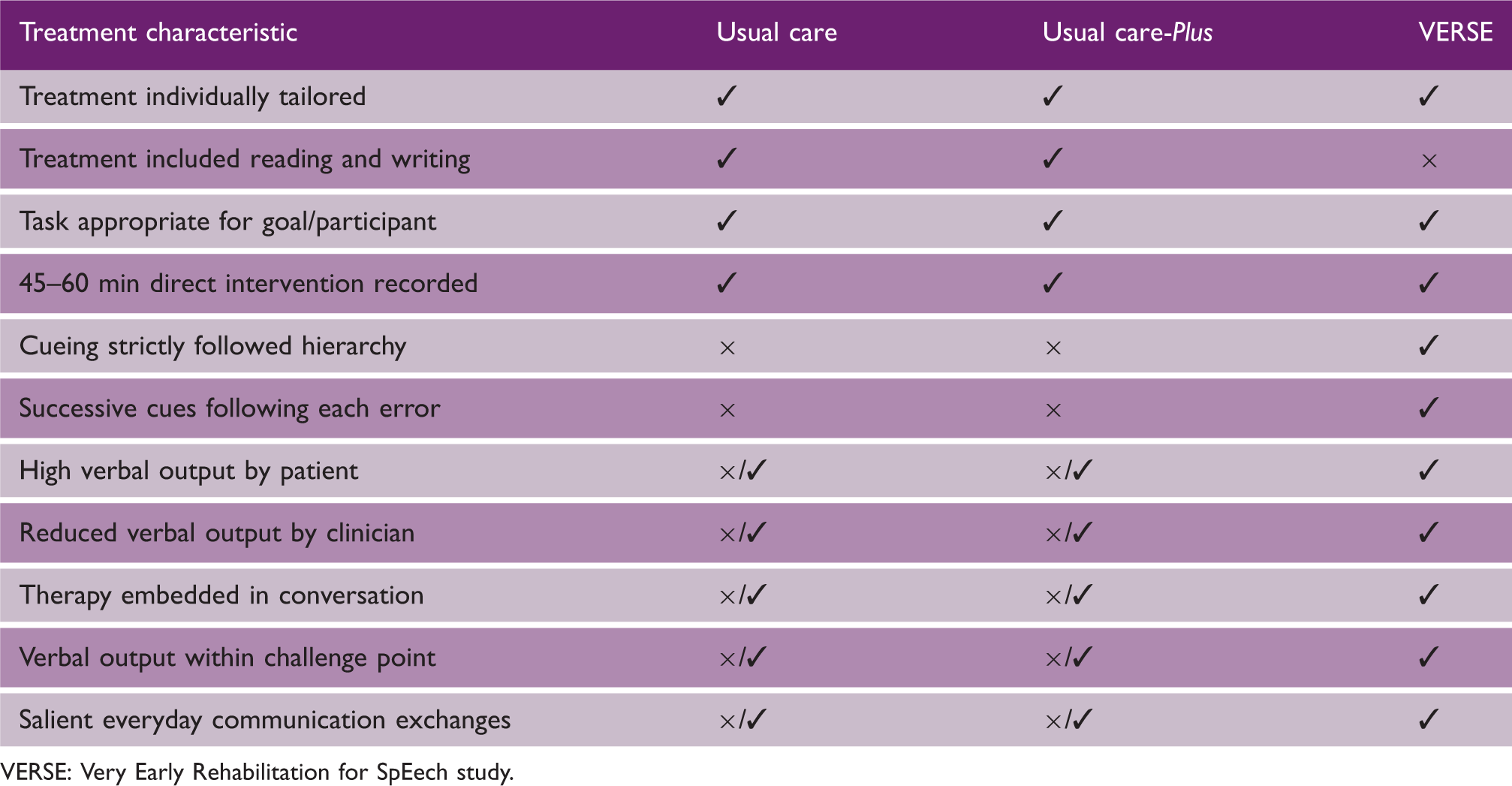
VERSE: Very Early Rehabilitation for SpEech study.
At 12 weeks post stroke, the unadjusted mean %MPR of those in the high intensity group (UC-Plus and VERSE) was 50.5% (SD 32.4) compared to 52.9% (SD 29.5) in the UC group (Table 5). Our primary analysis (adjusted) showed recovery at 52.1% for the UC group (95% CI 46.1–58.1) and 50.3% (95% CI 45.7–54.8) for the high intensity group, after controlling for baseline AQ, NIHSS, and hospital site. The between-group difference of −1.8 (favoring control; (95% CI −8.7–5.0) was not significant (p = 0.59; Figure 2; Supplemental Table S3 for all group comparisons). Multiple imputations for missing data, assuming the data were missing at random, showed similar results. Deviations from this assumption were assessed through a sensitivity analysis (Supplemental Figure S1).
WAB-R (AQ) Baseline, 12-, and 26-week outcomes with 95% confidence intervals. Outcomes at 12 and 26 weeks post stroke UC:: usual care. Chi-square test. t test. Analysis conducted on >200 words. Data are n (%) or mean (SD). Adjusted analysis accounts for baseline Western Aphasia Battery-Revised Aphasia Quotient, National Institutes of Health Stroke Scale scores as fixed factors and randomizing sites as a random factor.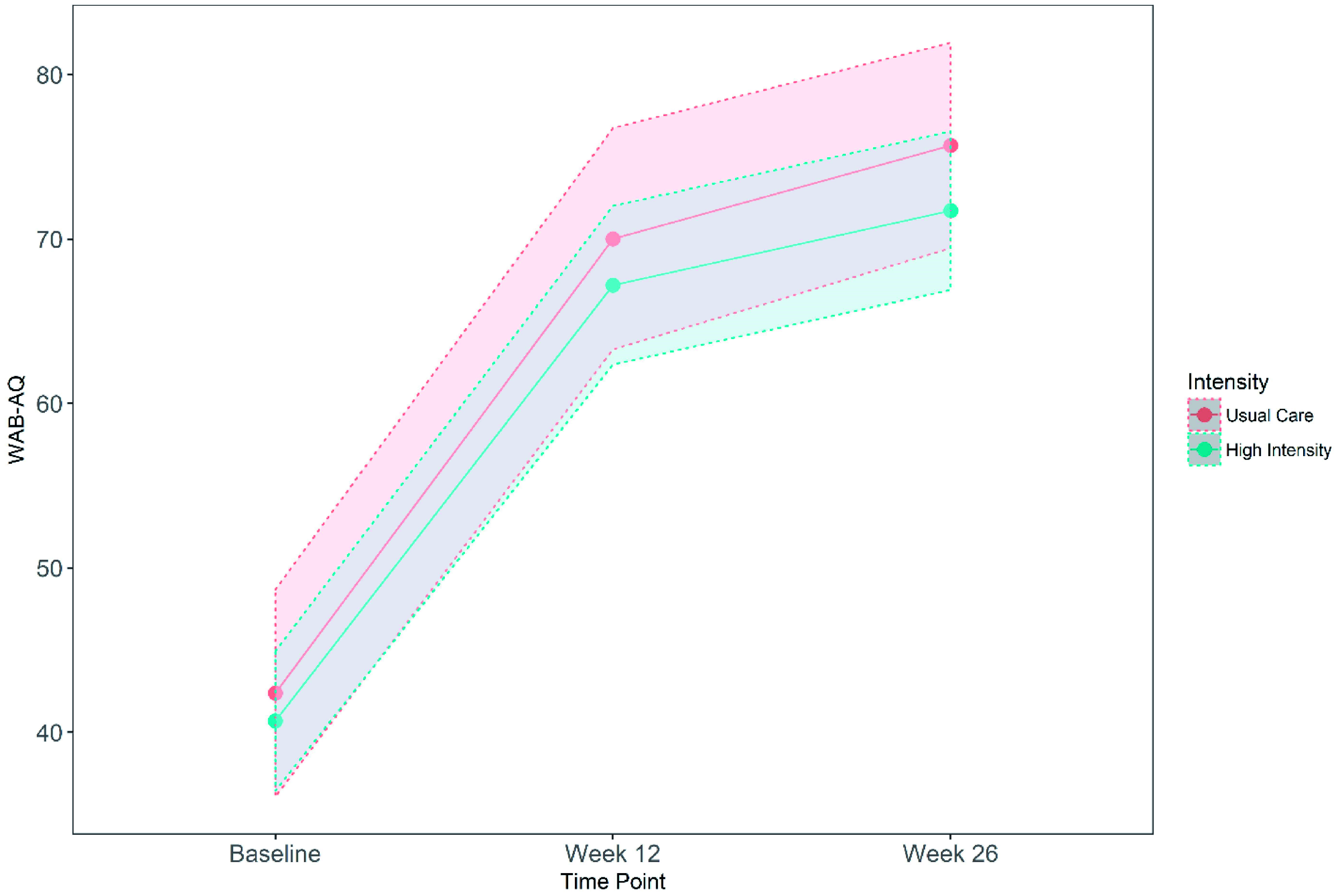

The results were similar across all secondary outcomes with no statistically significant differences noted (Table 5). The pre-specified exploratory analysis of subgroups 20 found no statistically significant differences for any variables (Supplemental Figure S3). The subgroup comparisons for %MPR at 12 weeks was 52.2% for the VERSE group (95% CI 46.3–58.2) and 48.4% for the UC-Plus group (95% CI 42.5–54.3; Supplemental Table S3).
By the 26-week follow-up, 202 participants (82%), consisting of 139 (85%) participants from the high intensity group (70 (85%) UC-Plus and 69 (83%) from VERSE) and 63 (78%) participants from the UC group, were assessed. At this time point, the overall group mean %MPR was 56.9% (SD 32.2).
The high intensity group %MPR was 54.7% (SD 34.6) and the UC %MPR was 61.7% (SD 25.8) indicating greater recovery for usual care participants, though not statistically significant (p = 0.12). There were no significant differences in non-fatal or fatal adverse events between groups (Table 1). New cases of depression were diagnosed in 35 (21%) participants in the high intensity group compared with 19 (23.5%) in UC (p = 0.67).
Discussion
We conducted the largest, multicenter aphasia clinical trial in stroke to date, assessing the effectiveness of aphasia therapy in very early post-stroke recovery. Designed to determine if ‘more intensive aphasia therapy is better’ in the acute post-stroke phase, our early intensive therapy regimen commenced on average two days earlier and was delivered more frequently and in a greater amount than in UC. We found that 22 h (or an average 5 h per week) of aphasia therapy (regardless of type) delivered over 32 days, showed no benefit over 9.5 h delivered over 28 days (2.3 h per week) for the primary outcome WAB-R AQ at 12 weeks post stroke. That is, more therapy started on or before day eight post stroke, did not significantly enhance language or communication recovery, or global outcomes such as quality of life or depression at 12 or 26 weeks. Provision of more intensive therapy did not result in greater adverse outcomes and was safe and feasible to deliver. The majority of participants in the VERSE trial, regardless of group allocation, achieved significant, clinically meaningful gains in language recovery. We believe the trial sample was representative of a typical population with post-stroke aphasia evidenced by the severity of stroke and aphasia recruited to this trial. 8 Our 7% recruitment rate is consistent with recruitment for acute stroke trials. 7
Our current findings were unexpected, based on our pilot studies5,18 which showed significant benefit of early, intensive aphasia treatment. In addition, we found a considerable increase in the amount of aphasia therapy provided as usual care, but not in the type of therapy provided, 37 compared with the results of our pilot trial. 5 Our Phase I trial 5 was conducted over 10 years ago, and at this time, among the 15% of participants who received aphasia therapy, only 14 min was provided in a single session over three weeks. The shift in usual care service delivery in this trial was evidenced by 81% of UC participants receiving: (i) early aphasia therapy within the first 15 days post stroke; (ii) more frequent sessions (3 per week), and (iii) 9.5 h of aphasia therapy over 28 days. These changes may reflect a greater focus on national clinical guidelines 38 with recommendations to increase the amount of aphasia therapy based on expert consensus influenced by early, yet underpowered trials. They may also reflect the fact that the sites in this trial were self-selected, hence it may be that these sites have an increased number of therapists compared to other sites who did not participate in the trial. The increase in UC aphasia services may also be the result of an increased will to ‘do more’ as part of the trial, a phenomenon known as the ‘Hawthorn Effect’, secondary to recording and monitoring of these services. For the Usual Care therapy sessions, fewer than 1% of sessions were video recorded despite the larger than expected number of hours of aphasia therapy that were provided to this group in the trial. We believe the low number of recorded sessions in this group reflects standard videorecording practice at these sites.
Results presented here are suggestive of the economic ‘law of diminishing returns’ applied within a medical service delivery model which specifies that “beyond a certain point, additional inputs produce smaller and smaller outputs” 39 (p. 371). Our sufficiently powered trial shows diminishing marginal returns from the additional aphasia therapy provided in early stroke recovery. The small difference observed in %MPR (1.8%) between the UC and high intensity cohorts at 12 weeks post stroke, means a 1.8 point difference for a profoundly impaired participant with a baseline WAB-R AQ score of 0 (no verbal output). For a participant with a baseline AQ of 40 (average for our trial and indicative of a participant speaking in two-word phrases with frequent errors and word-finding difficulties), the between group difference equates to 1.14 points at 12 weeks. These small group differences were not considered clinically relevant (defined as 5 points 29 on the WAB-R AQ).
The within group differences (over 25 points) were between 50% and 52% of MPR. From a clinical perspective, it was reassuring to show the majority of VERSE participants achieved a large and clinically meaningful gain in language recovery, regardless of group allocation, and this was consistent with our earlier trials.5,18 Clinically, the average participant progressed from speaking in two-word phrases with multiple word, sound and grammatical errors and requiring assistance with all communication interactions, to speaking in coherent and appropriate sentences. Prior research to predict maximal potential aphasia recovery in acute stroke has been limited to a sample of 21 patients whereby the predicted improvement within the first 12 weeks was estimated at 70%. 28 Our findings suggest that average predicted recovery from aphasia at 12 weeks may be somewhat lower than that. However, our findings reflect considerably more change than expected in people with chronic aphasia (>6 months post stroke) where interventions are considered to be potent if they deliver change of 5-points 28 or more on the WAB-R AQ.
The contribution of spontaneous recovery to outcome remains elusive. Inclusion of a no treatment arm was not considered ethical in this pragmatic trial, given that patients in Australia and New Zealand expect to receive aphasia therapy in the acute phase of stroke care. It is likely that the early, rapid recovery in the first six months after stroke seen in this trial is due to a combination of spontaneous recovery and a treatment effect. We set about to determine if additional therapy to standard care enhanced spontaneous recovery rather than attempting to discern the individual value of each component. In the few studies where there has been an attempt to measure effects of spontaneous recovery, varied study designs, small participant numbers, different measurement time points, or different outcome measures have been used, making the drawing of conclusions problematic. In a meta-analysis 40 of 5928 individual patient data, there are 23 cases 5 of documented spontaneous recovery as part of usual care, that have comparable outcome measures (completed in English) collected within 14 days (baseline) and 26 weeks post stroke. The amount of spontaneous language and communication recovery of this historical control cohort is presented in the Supplement (Supplemental Figure S4) and indicates a medium effect size well below that seen in the VERSE trial.
Our results challenge the previously held neuro-recovery theoretical perception that ‘more intensive therapy is better’ when commenced in the first two weeks of stroke onset. The UC therapy regimen of a mean 9.5 h (SD 7) h, comprising 25- to 45-min sessions provided three times per week within 38 days (10 days to randomization and 28 days of intervention) post stroke may be a sufficient therapy regimen to support recovery in the first six months post stroke. However, definitive thresholds regarding timing, intensity and type of therapy are difficult to describe without stronger data on natural recovery in the acute phase. Similarly, while this study provided a higher, more intense dose of aphasia therapy than is common in early usual care without significant benefit, it remains unclear whether a much higher dose, for example, 100 h over 12 weeks, would be more efficacious.
This study adds strong additional evidence to the RATS-33 trial findings from an internationally diverse, English speaking population. RATS-33 showed no significant difference between communication recovery at 4, 12 or 26 weeks post stroke after intensive aphasia therapy in the first six weeks of recovery when compared with a delayed intervention control group. The VERSE results demonstrate no benefit of intensive aphasia therapy compared to therapy at a lesser intensity in the first six weeks of language and communication recovery at 12 and 26 weeks. Together, these trials3–6 provide compelling evidence to challenge the ‘intensive’ mindset in early stroke language and communication recovery.
The question of ‘what is enough?’ therapy is paramount here. Some may propose that the total intervention regimen in this and other trials1,3,4,5,18,40 was insufficient (below the theoretical threshold of at least 98 h in total 19 ) to demonstrate a benefit of treatment intensity in this recovery period. We suggest that the intensity and total hours of intervention in the UC participants presented here demonstrate clinically meaningful change that can be attributed to a combination of therapy effect and spontaneous recovery.
Directions for future early aphasia therapy research
There is a risk that a ‘non-aphasia expert’ may misinterpret the results from this study to indicate that early aphasia therapy or intensive aphasia therapy does not lead to meaningful benefits post stroke. This study addresses the specific issue of ‘more intensive therapy’ in the early recovery period; it did not evaluate chronic recovery or the effect of ‘treatment’ versus ‘no treatment’. There are several important research areas that must be addressed in order to deliver improved recovery for people with aphasia. Aphasia research needs Level 1 evidence to address: (i) the dose response in early and chronic aphasia recovery; (ii) the optimal type of therapy to provide to whom and at what time post stroke; and (iii) what other factors contribute to early aphasia recovery (e.g. the role of stroke unit care in clinical improvement). The ability to monitor other therapies available (i.e. physiotherapy, occupational therapy) and social interactions was outside the funding scope of this trial. Within the RCT design, we expect that the input of other health professionals is similar across groups. The potential for physical training provided by other therapists, which may offer additional language practice, to interact with or support language recovery is currently unexplored, and may be an area for future study.
Our results do not support increasing aphasia therapy intensity in the first 38 days post stroke above what is reported as UC in this trial, nor support the modification of existing services related to type and amount of aphasia therapy provided in current practice in Australia and New Zealand for post stroke early aphasia recovery.
In aphasia research fruitful next important areas of enquiry should include: (i) strong Phase I studies determining the effect of a dose range of identified active ingredients; and (ii) Phase II studies establishing feasibility of dose specific treatment regimens in early aphasia recovery.
Supplemental Material
sj-pdf-1-wso-10.1177_1747493020961926 - Supplemental material for A randomized control trial of intensive aphasia therapy after acute stroke: The Very Early Rehabilitation for SpEech (VERSE) study
Supplemental material, sj-pdf-1-wso-10.1177_1747493020961926 for A randomized control trial of intensive aphasia therapy after acute stroke: The Very Early Rehabilitation for SpEech (VERSE) study by Erin Godecke, Elizabeth Armstrong, Tapan Rai, Natalie Ciccone, Miranda L Rose, Sandy Middleton, Anne Whitworth, Audrey Holland, Fiona Ellery, Graeme J Hankey, Dominique A Cadilhac, Julie Bernhardt and on behalf of the VERSE Collaborative Group in International Journal of Stroke
Footnotes
Acknowledgements
The authors sincerely thank the VERSE Collaboration investigators for their commitment to this trial and the participants and their families who have helped us to add valuable information and help guide best practice for the management of early aphasia recovery. Thank you to our data management team for their hard work—Sanita Kratina (Trial Operations Manager), Edith Cowan University, Perth Australia; Crystal Ladzinski (Therapy Data monitor), Perth Australia; Oriana Borschmann (Assessment Data monitor), Melbourne Australia; Leonid Churilov and Li Chun Quang for REDCap™ assistance and to Jan Chamberlain for her assistance with preparation of this article. The content of this publication is solely the responsibility of the authors and the funding bodies played no part in the trial or interpretation of the findings. Australian New Zealand Clinical Trials Registry number: ACTRN12613000776707. The authors sincerely thank the members of our DSMB (appendix 2) for their oversight of the trial.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Erin Godecke—NHMRC Funding: App1083010, APP1132468, App1153236, NIH (UK) HS&DR Program funding; Elizabeth Armstrong—NHMRC Funding: APP1132468; Tapan Rai reports no disclosures; Miranda L Rose—NHMRC Funding: App1083010, App1153236; Fiona Ellery FE reports personal fees from Florey Institute of Neurosciences and Mental Health, The University of Melbourne during the conduct of the study; Graham J Hankey has received honoraria from Bayer for lecturing at sponsored scientific symposia and consulting on advisory boards about stroke prevention in atrial fibrillation; Dominique A Cadilhac—NHMRC Funding App1063761, App1154273; Julie Bernhardt—NHMRC Funding JB—App1154904, App1058635. This study was funded by National Health and Medical Research Council (APP1044973), The Tavistock Trust for Aphasia (UK), Edith Cowan University, Australia.
Authors’ contributions
Statistical analysis was conducted by Associate Professor Tapan Rai, PhD, University of Technology Sydney, Australia.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
