Abstract
Background
The Thrombolysis in Cerebral Infarction (TICI) scale is an important outcome measure to evaluate the quality of endovascular stroke therapy. The TICI scale is ordinal and observer-dependent, which may result in suboptimal prediction of patient outcome and inconsistent reperfusion grading.
Aims
We present a semi-automated quantitative reperfusion measure (quantified TICI (qTICI)) using image processing techniques based on the TICI methodology.
Methods
We included patients with an intracranial proximal large vessel occlusion with complete, good quality runs of anteroposterior and lateral digital subtraction angiography from the MR CLEAN Registry. For each vessel occlusion, we identified the target downstream territory and automatically segmented the reperfused area in the target downstream territory on final digital subtraction angiography. qTICI was defined as the percentage of reperfused area in target downstream territory. The value of qTICI and extended TICI (eTICI) in predicting favorable functional outcome (modified Rankin Scale 0–2) was compared using area under receiver operating characteristics curve and binary logistic regression analysis unadjusted and adjusted for known prognostic factors.
Results
In total, 408 patients with M1 or internal carotid artery occlusion were included. The median qTICI was 78 (interquartile range 58–88) and 215 patients (53%) had an eTICI of 2C or higher. qTICI was comparable to eTICI in predicting favorable outcome with area under receiver operating characteristics curve of 0.63 vs. 0.62 (P = 0.8) and 0.87 vs. 0.86 (P = 0.87), for the unadjusted and adjusted analysis, respectively. In the adjusted regression analyses, both qTICI and eTICI were independently associated with functional outcome.
Conclusion
qTICI provides a quantitative measure of reperfusion with similar prognostic value for functional outcome to eTICI score.
Introduction
Various grading scales have been introduced to assess the extent of reperfusion on digital subtraction angiography (DSA) images following endovascular treatment (EVT). 1 One of the most frequently used scales is the modified Treatment in Cerebral Ischemia (mTICI) scale. 2 This scale has been recommended because of its fair interobserver agreement and clinical prognostic value.1,3,4 mTICI assesses the proportion of brain tissue perfused in an antegrade fashion relative to the total area of the target downstream territory (TDT) distal to the target occlusion. 5 The TDT is the area of the brain that was supplied by the occluded artery prior to stroke onset. 1 The score ranges from no perfusion (mTICI = 0) to complete reperfusion (mTICI = 3). A subsequent refinement has been made to intermediate grades of reperfusion (mTICI 2B) to better define successful reperfusion.3,4,6 Recently, the extended Thrombolysis in Cerebral Ischemia (eTICI) scale has been introduced, which includes grade 2C to account for nearly complete reperfusion with slow flow in distal cortical vessels or presence of tiny distal emboli.7–9 This grade 2C has been retained in the expanded TICI scale, which added 67% perfusion threshold into the middle categories. 10 Recent findings suggest that eTICI 2C-3 has a stronger association with favorable functional outcome than eTICI 2B-3.10–15
Despite its widespread use, the eTICI score suffers from two shortcomings: it is prone to observer variance and it is an ordinal scale.1,6,7,10,16 Moreover, a recent study showed that reperfusion is generally overestimated by the interventionalist performing EVT compared to core-lab observers. 16 An automated and quantitative reperfusion grading has the potential to provide a more clinically relevant, less observer-dependent score, and a greater ease of use. In this study, we present a quantitative reperfusion measure using semantic segmentation of DSA images. We assessed the prognostic value of the proposed quantitative reperfusion grading for predicting functional outcome in comparison with eTICI.
Methods
Patients
This is a substudy of the Multicenter Clinical Registry of Endovascular treatment for acute ischemic stroke in the Netherlands (MR CLEAN Registry). Details of the registry, patient inclusion, baseline clinical and imaging characteristics, and interventional therapy have been described previously. 17 We included only patients with acute ischemic stroke attributable to an occlusion of the intracranial internal carotid artery (ICA) or proximal middle cerebral artery (MCA) (M1) as presented on DSA who were treated between March 2014 and June 2016. We also only included patients with good quality pre- and post-treatment DSA, with full arterial to venous anteroposterior and lateral runs, and who were eTICI graded by an independent core-lab of neuro/interventional radiologists. DSA images with motion artifacts and low contrast volume were excluded.
Image analysis
Similar to the eTICI grading, our quantitative reperfusion measure assesses the percentage of the reperfused area within the TDT on DSA. An overview of the proposed approach is shown in Figure 1.
Overview of image segmentation approach in a patient with M1 occlusion. There are two parallel segmentation tasks: segmentations of reperfused area and of TDT. The reperfused area was segmented from temporal maximum intensity projection of the images of the arterial and parenchymal phase. The TDT was defined as the middle cerebral artery territory and was manually delineated. The percentage of the reperfused area within the TDT was subsequently calculated. DSA: digital subtraction angiography; TDT: target downstream territory; TMIP, temporal maximum internsity projection.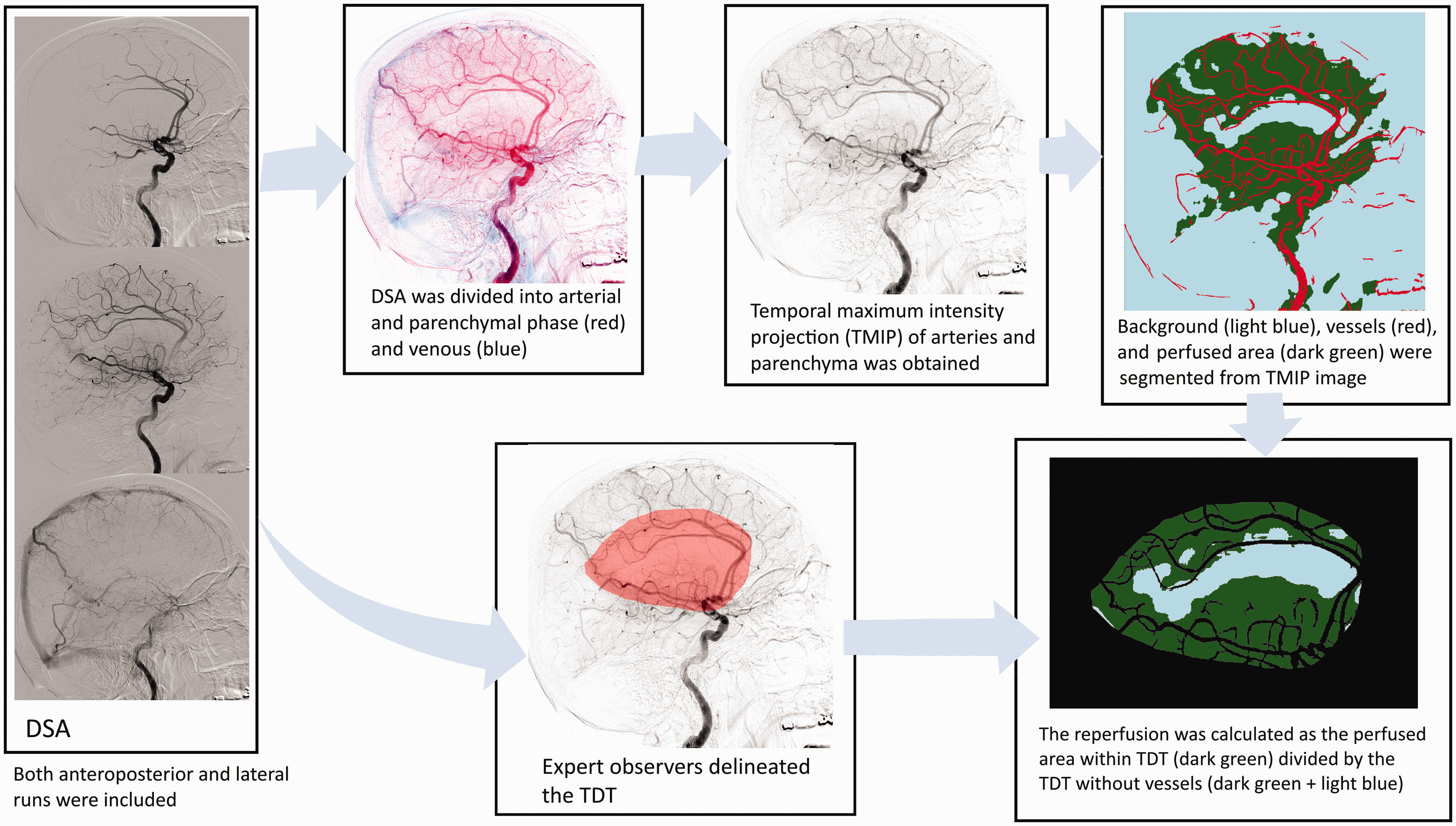
First, the reperfused area was identified by following the contrast trajectory past the initial target occlusion. The contrast trajectory can be roughly divided into three phases in the brain: the arterial, parenchymal, and venous phase. This sequence of phases occurs earlier in the proximal part of the intracranial vasculature than in the distal areas. Since perfusion is only visible during the parenchymal phase (from contrast opacification in the brain microvasculature), segmentation of the reperfused tissue can be challenging due to the lack of distinction between phases. This distinction is especially important to separate the antegrade (via recanalization) and retrograde (via pial collateral) perfusion as only the former constitutes reperfusion. Moreover, the local vessel structure, whether it is artery in earlier frames or vein in later frames, can be present during the parenchymal phase. To accurately assess the extent of reperfusion in the parenchyma, the vessel structure was removed from the images.
We quantified reperfusion on a temporal maximum intensity projection of the images starting at the arterial until the end of parenchymal phase. The last frame of parenchymal phase was manually determined as the immediate frame before the appearance of contrast in the superior sagittal sinus. We assumed that this frame marked the separation between antegrade and retrograde perfusion.
We adopted a multiscale vesselness filter to segment the arteries. 18 The vesselness filter detects tubular structures in images using local curvature information encoded in a Hessian matrix. Improvements were made by introducing a normalization factor to the vesselness classification to reduce blur surrounding the vessel. Details can be found in the Online Appendix. To obtain a parenchymal mask image that contains only the non-vessel structure, the resulting vessel mask was subtracted from the original image. This parenchymal mask comprised of reperfused area and background (which includes non-perfused areas). Finally, the reperfused area was arbitrarily determined as the area in the parenchymal mask where the pixel intensity exceeded 7% and 3% of the maximum intensity in the original image for anteroposterior and lateral runs, respectively. These numbers reflect the minimum contrast intensity in the image that distinguishes perfusion from background area.
For TDT delineation, an expert observer blinded to all clinical data except symptomatic side visually assessed the DSA images of a subpopulation and outlined the TDTs. For this study, we included three expert observers (HP, MK, and BJE) to account for three different patient subpopulations. The M1 TDT was defined as the presumed area supplied by the MCAs including part of lenticulostriate arteries and holotemporal branch. 1 In clinical practice, the ICA TDT is dependent on the patency of the anterior communicating artery. In case the anterior communicating artery is patent, the ipsilateral anterior cerebral artery (ACA) territory is usually supplied by the contralateral anterior circulation. In this case, the ICA TDT is identical to the M1 TDT. In case of an absent or severely hypoplastic anterior communicating artery, the ICA TDT also includes the ACA territory. In practice, baseline computed tomography angiography (CTA) data can be used to inspect the patency of the anterior communicating artery. However, during the core-lab evaluations of the eTICI, this information was not available and the M1 TDT was used. Therefore, we used M1 TDT for both ICA and M1 cases in this study. The effect of using different TDTs for the ICA occlusions on the quantified TICI (qTICI) score was evaluated. We did not make a distinction in TDT between a proximal and distal ICA and M1 occlusions since there is no considerable difference in the TDT. Consensus agreement between expert observers was achieved to maintain consistency in the delineation process. An example of a TDT is shown in Figure 1 in the online-only Data Supplement.
The parenchymal mask within the resulting TDT was subsequently determined. qTICI was calculated as the number of pixels of reperfused area in the TDT divided by the number of pixels in the parenchymal mask in the TDT and expressed as percentage. The average qTICI of both anteroposterior and lateral DSA was used as the final qTICI score.
Angiography evaluation
An independent core-lab assessed all the imaging in the MR CLEAN Registry. 19 The pre- and post-treatment DSA images were used for reperfusion evaluation with target occlusion location determined from pre-treatment DSA images. The six-point eTICI grading was used to adjudicate the reperfusion of the initial target occlusion. We should clarify that we used the eTICI and not the expanded TICI definition. The eTICI is the extension of the modified TICI in which grade 2C has been added. 9 The expanded TICI further divides the scale 2B into 2B50 and 2B67. 10 At the time of the prospective patient recruitment for the registry, the expanded TICI was not yet introduced and therefore not used in the current study. In summary, eTICI 0 describes no reperfusion in TDT; eTICI 1 indicates antegrade reperfusion past the initial occlusion but minimal reperfusion of TDT, eTICI 2A is reperfusion of <50% of TDT, eTICI 2B is a reperfusion of ≥50% of TDT, eTICI 2C reflects a near-complete perfusion with distal slow flow or presence of small cortical emboli, and eTICI 3 is a complete reperfusion. 7
Statistical analyses
Continuous and categorical variables were expressed as median (interquartile range (IQR)) and frequency (percentage), respectively. Kruskal–Wallis tests were performed to compare differences in qTICI scores between eTICI grades. Functional outcome was determined by assessing the modified Rankin Scale (mRS) at day 90 and dichotomized into favorable (mRS 0–2) and unfavorable outcome (mRS 3–6). Stacked bar plots were used to visually compare the ordinal mRS distribution with respect to qTICI and eTICI grades. A classification table for grouped qTICI based on the definition of the corresponding eTICI grades was provided.
The unadjusted and adjusted odds ratios for favorable outcome were estimated for qTICI and eTICI using logistic regression. Adjustments were made by including major prespecified baseline prognostic factors: age, sex, baseline stroke severity (National Institutes of Health Stroke Scale (NIHSS)), Alberta Stroke Program Early CT Score, CTA collateral score, history of hypertension, diabetes mellitus, and previous stroke, pre-treatment mRS, systolic blood pressure, administration of intravenous thrombolytics prior to EVT, general anesthesia, time from onset-to-groin puncture, and duration of endovascular treatment. For adjusted analyses only, missing variables were imputed based on additive regression, bootstrapping, and predictive mean matching using the aforementioned variables with additional variables: glucose level, hypercholesterolemia, atrial fibrillation, history of myocardial infarction, occlusion segment, time from onset to reperfusion or last contrast bolus, and NIHSS score after 24 to 48 h.19,20 Unadjusted and adjusted odds ratios were reported with 95% of confidence interval (CI) for statistical precision. Nagelkerke's coefficient of determination was used to compare the unadjusted and adjusted models of qTICI and eTICI in explaining the proportion of functional outcome variation. The effect of using different TDTs for ICA occlusion cases in the estimation of the odds ratio was tested in a sensitivity analysis in which the combined ACA and MCA TDTs were used instead of the MCA TDT.
The area under the receiver operating characteristic curve (AUC) was calculated to determine the prognostic value of the logistic regression models in predicting favorable outcome. P values lower than 0.05 were considered statistically significant. All statistics were performed using IBM SPSS software (version 19.0.0) and RStudio, RStudio, Inc. (version 1.2.1335).
Results
Baseline characteristics.
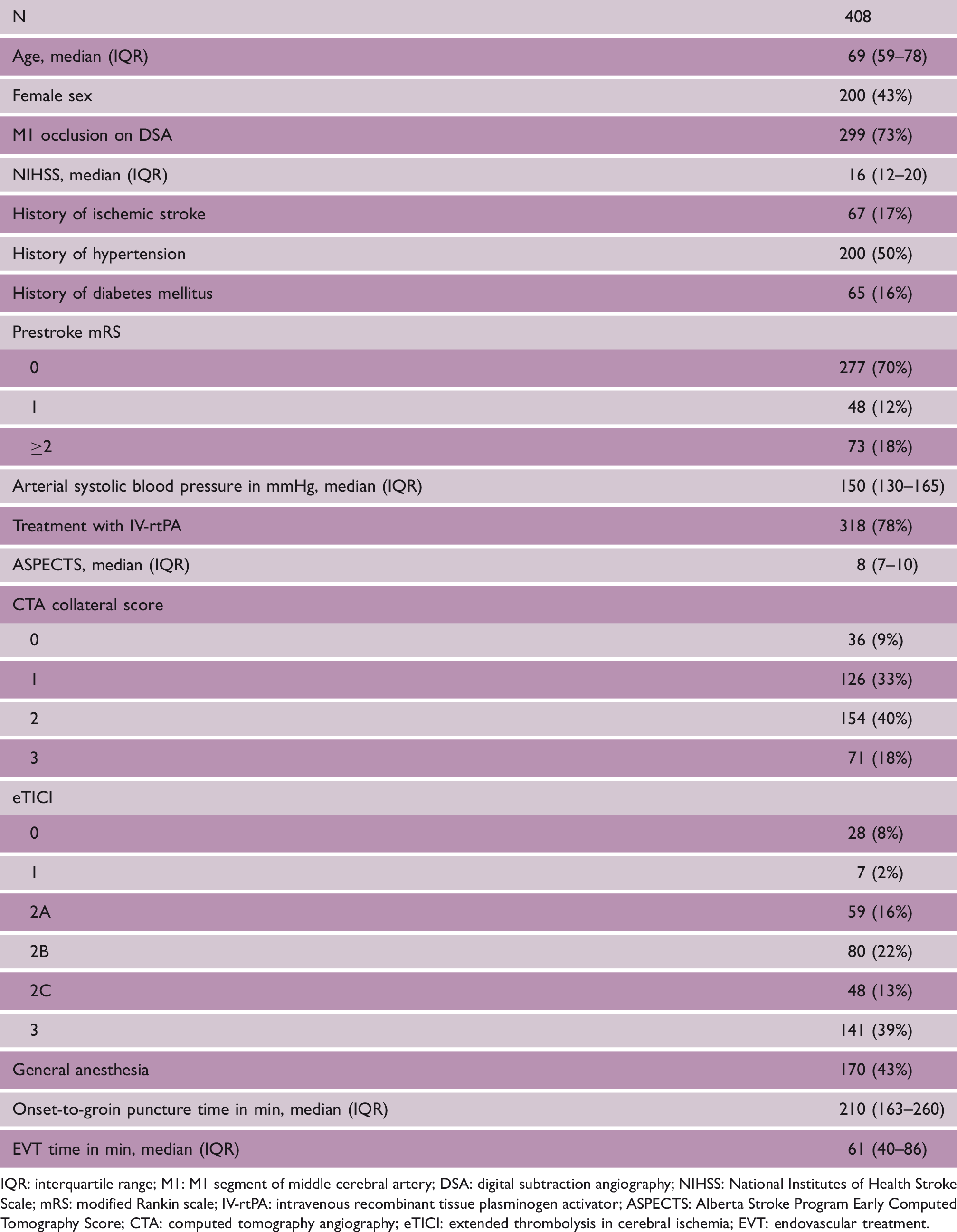
IQR: interquartile range; M1: M1 segment of middle cerebral artery; DSA: digital subtraction angiography; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin scale; IV-rtPA: intravenous recombinant tissue plasminogen activator; ASPECTS: Alberta Stroke Program Early Computed Tomography Score; CTA: computed tomography angiography; eTICI: extended thrombolysis in cerebral ischemia; EVT: endovascular treatment.
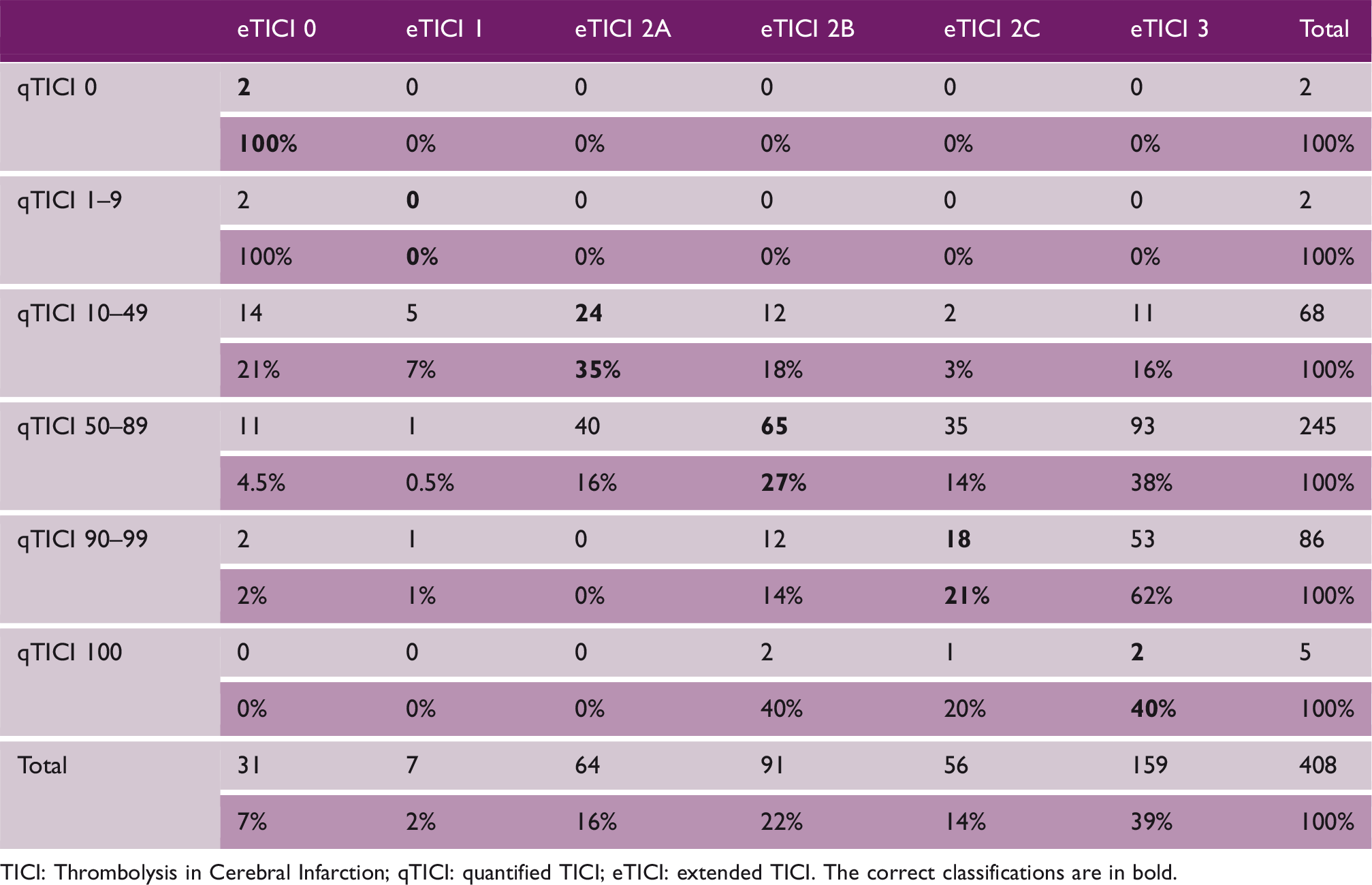
Figure 2(a) shows the distribution of qTICI values for different eTICI grades. The qTICI was significantly different between all eTICI grades (P < 0.001). For ICA occlusion cases only, the qTICI values were not significantly different for eTICI classifications when using the combined ACA and MCA territories (P = 0.28). The full result of this analysis is shown in Figure 3 in the online-only Data Supplement. Figure 2(b) shows the distribution of mRS for different qTICI groups and eTICI grades. The group-wise comparison between qTICI and eTICI is shown in Table 2.
Comparison of qTICI and eTICI. (a) Distribution of the qTICI score for the various eTICI grades. (b) mRS at 90 days based on qTICI and eTICI. mRS: modified Rankin Scale; TICI: Thrombolysis in Cerebral Infarction; qTICI: quantified TICI; eTICI: extended TICI. Classification table of the grouped qTICI and the corresponding eTICI grades. TICI: Thrombolysis in Cerebral Infarction; qTICI: quantified TICI; eTICI: extended TICI. The correct classifications are in bold.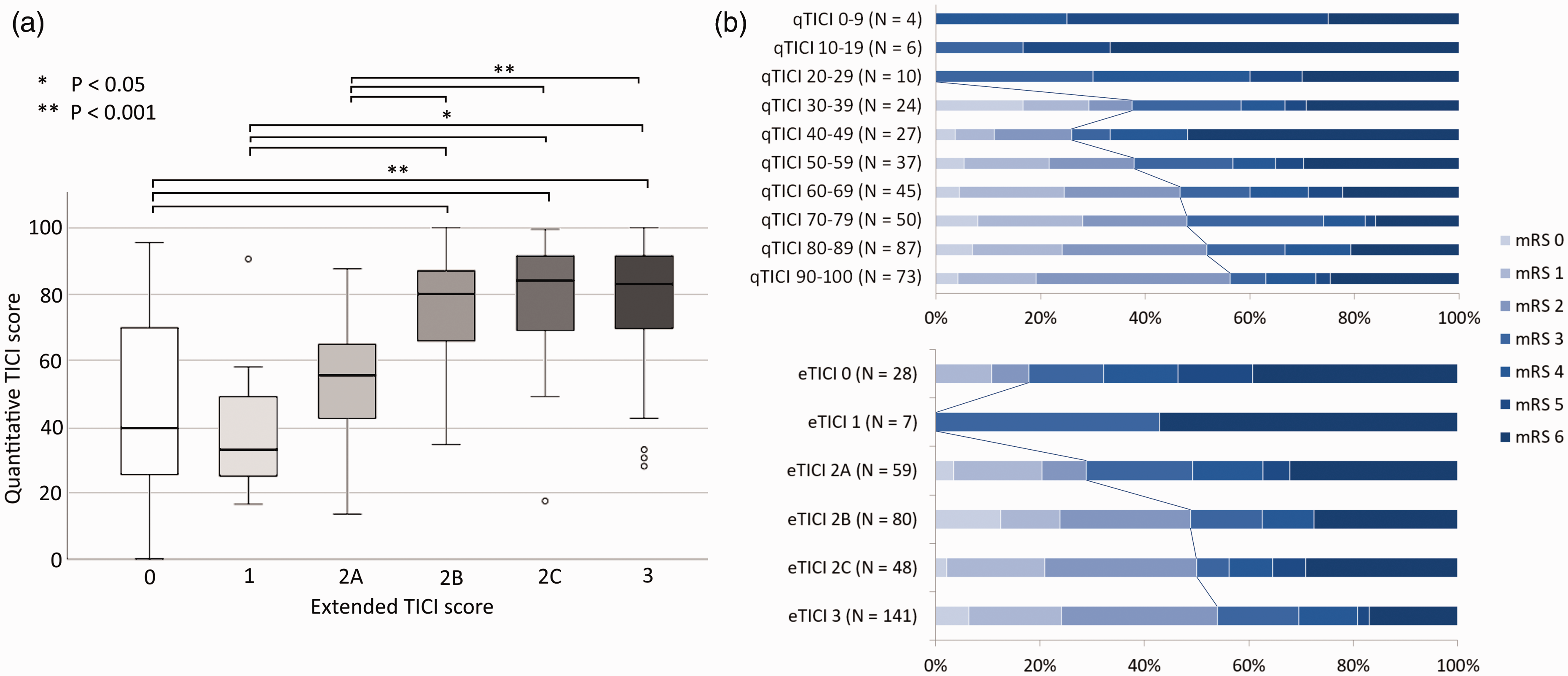
Ninety-five patients had missing baseline prognostic variables which were imputed for the subsequent predictive analytics.
Results of unadjusted and adjusted regression analysis for the effect of reperfusion score on the dichotomized functional outcome.
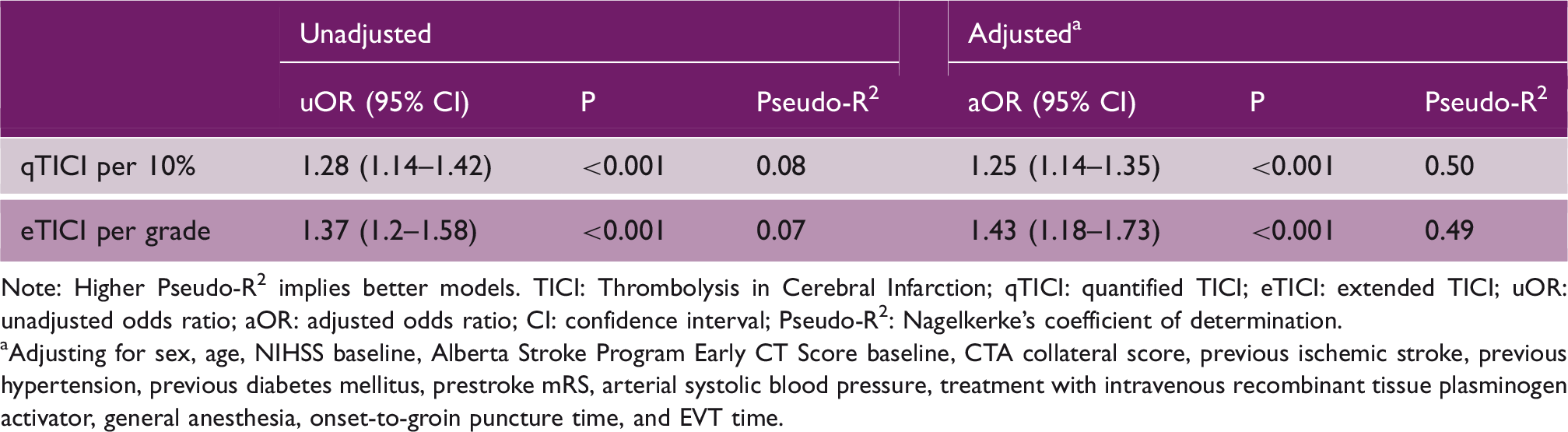
Note: Higher Pseudo-R2 implies better models. TICI: Thrombolysis in Cerebral Infarction; qTICI: quantified TICI; eTICI: extended TICI; uOR: unadjusted odds ratio; aOR: adjusted odds ratio; CI: confidence interval; Pseudo-R2: Nagelkerke's coefficient of determination.
Adjusting for sex, age, NIHSS baseline, Alberta Stroke Program Early CT Score baseline, CTA collateral score, previous ischemic stroke, previous hypertension, previous diabetes mellitus, prestroke mRS, arterial systolic blood pressure, treatment with intravenous recombinant tissue plasminogen activator, general anesthesia, onset-to-groin puncture time, and EVT time.
As shown in Figure 3, receiver operator characteristics analysis showed that qTICI and eTICI were moderate predictors of favorable outcome with an AUC of 0.63 (95% CI, 0.58–0.68) vs. 0.62 (95% CI, 0.57–0.67). For the adjusted model, qTICI and eTICI were comparable with AUC 0.87 (95% CI, 0.83–0.90) and 0.86 (95% CI, 0.83–0.90), respectively. For both the adjusted and unadjusted models, the difference in AUCs of qTICI and eTICI was not significant (P = 0.87 for the adjusted model, P = 0.80 for the unadjusted model).
Receiver operating characteristic curves for qTICI and eTICI for predicting good functional outcome (mRS 0–2). Left: The area under the curve for qTICI and eTICI as a single predictor were 0.63 and 0.62, respectively. Right: The area under the curve for adjusted predictive model with either qTICI or eTICI were 0.87 and 0.86, respectively, a minor improvement from the area under the curve 0.85 of the model with the other prognostic factors. TICI: Thrombolysis in Cerebral Infarction; qTICI: quantified TICI; eTICI: extended TICI.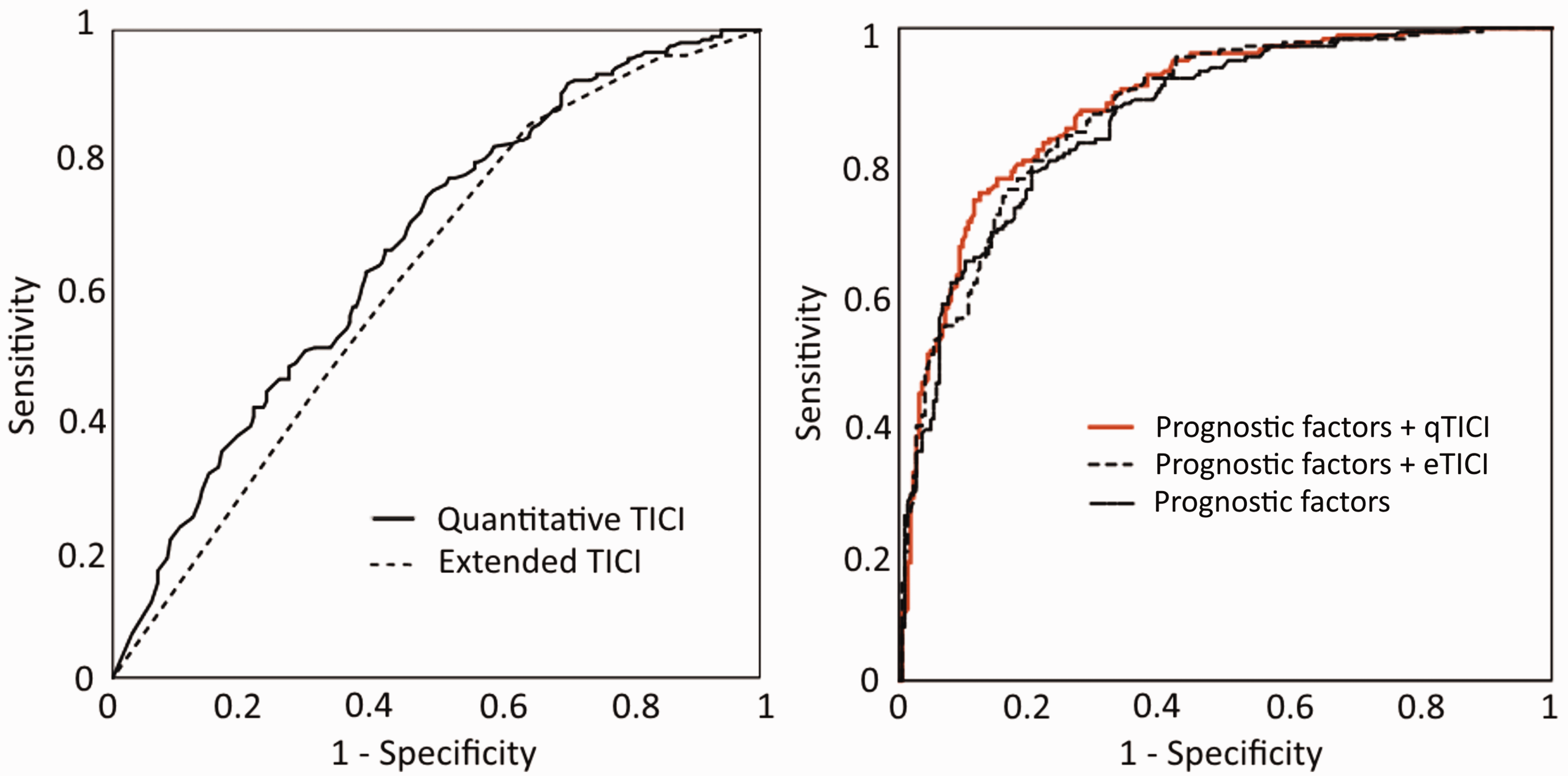
The sensitivity analysis showed that the unadjusted and adjusted odds ratio for 10% qTICI increment were 1.28 (95% CI, 1.16–1.4, P < 0.001) and 1.33 (95% CI, 1.18–1.49, P < 0.001) when the combination of ACA and MCA territory was used as ICA TDT. These odds ratios were higher compared to the odds ratios of qTICI using only MCA territory as the ICA TDT.
Discussion
We have presented a semi-automated quantitative method to assess reperfusion on DSA images after endovascular treatment of patients with acute ischemic stroke due to large vessel occlusion. Our study shows that in our population, the qTICI is independently associated with functional outcome. We demonstrated that qTICI has a similar prognostic value compared with eTICI. This result remains consistent after the adjustment for other prognostic factors, indicating that qTICI could be a potential alternative for manual reperfusion assessment with a potentially reduced bias and interobserver variation.
Our method is the first fully quantitative scale of reperfusion assessment. There have been many iterations of angiographic reperfusion scales: starting from adopting the Thrombolysis in Myocardial Infarction scale for the brain, renaming and adjusting the score to TICI, modifying the threshold for scale 2A and 2B (from two-thirds to half of the TDT), adding scale 2C, redefining 2C (90%–99% reperfusion), and expanding scale 2B with the introduction of 2b50 and 2b67.1,7,9,10,21 Each iteration claimed to give a better definition of successful reperfusion with a more precise threshold to determine the reperfusion result.4–6,22,23 The inclusion of finer scales was based on evidence that this provides a better reperfusion assessment and clinical utility compared to that of coarser scales. This underpins the potential value of our continuous quantitative measure. Additionally, qTICI has minimum source of bias as the only dependency to the observer is during the delineation of the TDT. It must be pointed out that qTICI uses a TDT estimate, which is an actual TDT with a slight extension to both cranial and caudal direction. This crude TDT may provide a less accurate assessment since the actual TDT could differ significantly per patient and per occlusion location. However, qTICI remains significantly associated with functional outcome. This is likely due to the low sensitivity of qTICI for small TDT differences. Since the TDT of ICA and M1 occlusion is large, it is expected that minor variation in TDT delineation results in small effects in the qTICI score. In addition, the crude TDT increases the ease of use as meticulous delineation of TDT is not necessary. This could be beneficial in providing a robust reperfusion assessment especially on a large data set of clinical trials and registries. It also allows for faster assessment of difficult cases because of anatomical variation or DSA overprojection.
qTICI and eTICI behave differently in cases with failure of recanalization but retrograde reperfusion via collaterals. Here, some patients have high qTICI, whereas their reperfusion were classified as eTICI 0. For these cases, the high qTICI score may have included a substantial amount of retrograde filling from collaterals in the TDT, which is not included in the eTICI grading. While this may subvert qTICI value in assessing poor reperfusion, the true utility of qTICI is at assessing moderate to high reperfusion. It has been postulated that a fast retrograde perfusion may provide sufficient sustenance to the oligemic region and thus its inclusion may be of added value, though eventual watershed infarcts have also been described in these cases.24,25 The actual benefit of the inclusion of retrograde perfusion in the assessment was not studied. Another discrepancy between qTICI and eTICI stems from the different concern regarding vessel patency. In recanalized patients with severe stroke or a damaged blood–brain barrier, qTICI may confuse the extravasation of contrast near the artery as parenchymal blush thus exaggerating the rate of reperfusion. Further study is required to confirm the extent of this effect. Finally, Table 2 shows that the grouped qTICI and eTICI only have a modest agreement. This could be attributed to different factors according to the degree of the reperfusion: for moderate reperfusion, a large mismatch between the crude TDT and the actual TDT and for zero or complete reperfusion, the hypersensitivity of qTICI to TDT. Overall, grouping of qTICI based on the corresponding eTICI grades, especially qTICI 1 (eTICI 1) and qTICI 99 (eTICI 2C), was somewhat arbitrary and has minimum parallel to the clinical practice of eTICI grading. Hence, the group-wise comparison should be interpreted with caution.
It was demonstrated that the reperfusion scores by themselves have limited value in predicting functional outcome. This is illustrated by the modest predictive value of both qTICI and eTICI. Similar analyses performed on the adjusted models with either qTICI or eTICI show stronger predictive value, where these models explain about half of the variance. This suggests that in a population where most of the patients achieve successful reperfusion, the reperfusion measure becomes a non-variable and has limited effect on functional outcome prediction. However, it should be noted that in both these models, the perfusion scores are independently associated with outcome. The huge effect of endovascular treatment in recent trials has shown that the procedural outcome of endovascular treatment is important.17,19,26,27 This confirms that although reperfusion outcome may play a substantial role, functional outcome is multifactorial.
The limitations of our study include the availability of adequate images. qTICI is sensitive to noise; motion artifacts could result in false-positive identification of reperfused tissue. A full frontal and lateral view of the affected hemisphere were required, and this was not available for all patients. Another requirement for images includes the first passage of contrast as later passages may be detrimental to the assessment accuracy. We also limited our patient inclusion to only ICA and M1 occlusions as the TDT variation was expected to be minimal.
Our method may benefit from an automatic delineation of TDT, entirely eliminating observer dependency. To complete the full spectrum of perfusion assessment, cases with more distal occlusion could be included. The high variability of TDT for distal occlusions, however, is a challenge for automated methods. Another potential improvement would be to incorporate a region-weighted score. It has been reported that infarct location, especially in the pre-central sulcus and central sulcus, is associated with functional outcome. 28 Therefore, if implemented, qTICI may have an improved prognostic value as the perfusion is weighted based on the importance of the territory.
Conclusion
We developed a quantitative and semi-automated reperfusion score for endovascular treatment evaluation of patients with acute ischemic stroke due to a large vessel occlusion. We have shown that qTICI is an independent predictor of functional outcome and has similar prognostic value as the standard eTICI. The use of crude TDT in qTICI potentially minimizes observer variations and allows more robust assessment across large imaging data set, making qTICI valuable for reperfusion assessment in clinical trial or registry.
Supplemental Material
WSO909632 Supplemental Material1 - Supplemental material for qTICI: Quantitative assessment of brain tissue reperfusion on digital subtraction angiograms of acute ischemic stroke patients
Supplemental material, WSO909632 Supplemental Material1 for qTICI: Quantitative assessment of brain tissue reperfusion on digital subtraction angiograms of acute ischemic stroke patients by Haryadi Prasetya, Lucas A Ramos, Thabiso Epema, Kilian M Treurniet, Bart J Emmer, Ido R van den Wijngaard, Guang Zhang, Manon Kappelhof, Olvert A Berkhemer, Albert J Yoo, Yvo BEWM Roos, Robert J van Oostenbrugge, Diederik WJ Dippel, Wim H van Zwam, Aad van der Lugt, Bas AJM de Mol, Charles BLM Majoie, Ed van Bavel, Henk A Marquering and on behalf of the MR CLEAN Registry Investigators in International Journal of Stroke
Supplemental Material
WSO909632 Supplemental Material2 - Supplemental material for qTICI: Quantitative assessment of brain tissue reperfusion on digital subtraction angiograms of acute ischemic stroke patients
Supplemental material, WSO909632 Supplemental Material2 for qTICI: Quantitative assessment of brain tissue reperfusion on digital subtraction angiograms of acute ischemic stroke patients by Haryadi Prasetya, Lucas A Ramos, Thabiso Epema, Kilian M Treurniet, Bart J Emmer, Ido R van den Wijngaard, Guang Zhang, Manon Kappelhof, Olvert A Berkhemer, Albert J Yoo, Yvo BEWM Roos, Robert J van Oostenbrugge, Diederik WJ Dippel, Wim H van Zwam, Aad van der Lugt, Bas AJM de Mol, Charles BLM Majoie, Ed van Bavel, Henk A Marquering and on behalf of the MR CLEAN Registry Investigators in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HP reports personal fees from Indonesia Endowment Fund for Education (LPDP), during the conduct of the study. DWJD reports grants from Dutch Heart Foundation, Brain Foundation Netherlands, The Netherlands Organisation for Health Research and Development and unrestricted grants from AngioCare BV, Medtronic/Covidien/EV3®, MEDAC Gmbh/LAMEPRO, Penumbra Inc., Top Medical/Concentric, Stryker, all outside the submitted work. WHvZ reports modest consulting activities for Cerenovus. AvdL reports grants from Dutch Heart Foundation, Brain Foundation Netherlands, H2020 and unrestricted grants from AngioCare BV, Medtronic/Covidien/EV3®, MEDAC Gmbh/LAMEPRO, Penumbra Inc., Top Medical/Concentric, Stryker, all are outside the submitted work. AJY reports research grants from Cerenovus, Medtronic, Penumbra, Stryker, Genentech, and funding for consultancy from Cerenovus. CBLMM is a shareholder of Nico.lab. HAM is a co-founder and a shareholder of Nico.lab. The other authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Indonesia Endowment Fund for Education (LPDP), Ministry of Finance, Republic of Indonesia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
