Abstract
Rationale
Unruptured intracranial aneurysms are currently left untreated if the presumed complication risk of preventive endovascular or neurosurgical intervention is higher than the risk of rupture. Aneurysm wall inflammation and blood pressure are attractive modifiable risk factors of aneurysm rupture and growth.
Aim
To investigate in patients with an unruptured intracranial aneurysm who do not qualify for preventive endovascular or neurosurgical intervention whether a treatment strategy of acetylsalicylic acid 100 mg/day plus intensive blood pressure treatment (targeted systolic blood pressure < 120 mmHg, monitored with a home blood pressure measuring device) reduces the risk of aneurysm rupture or growth compared with care as usual (no acetylsalicylic acid, targeted office systolic blood pressure < 140 mmHg, no home blood pressure measuring device).
Sample size
We aim to randomize 776 patients 1:1 to the intervention arm or care as usual.
Design
Bi-national (Germany and the Netherlands) multicenter, prospective, randomized, open-label phase III trial with blinded outcome assessment.
Outcomes
The primary outcome is aneurysm rupture or growth (increase in any aneurysm diameter by ≥ 1 mm) on repeated MR or CT angiography within 36 ± 6 months after randomization.
Discussion
The Prospective Randomized Open-label Trial to Evaluate risk faCTor management in patients with Unruptured intracranial aneurysms (PROTECT-U) is the first randomized trial to investigate if a medical strategy reduces the risk of rupture or growth of intracranial aneurysms in patients not undergoing preventive endovascular or neurosurgical aneurysm treatment.
Keywords
Introduction and rationale
Preventive endovascular or neurosurgical treatment of an unruptured intracranial aneurysm (UIA) is an attractive option to reduce the risk of subarachnoid hemorrhage (SAH) and hereby increase the number of life years with high quality of life. However, both treatment modalities carry a risk of complications, with a risk of overall permanent disability or death of more than 6%.1,2 If it is decided by a multidisciplinary team of intracranial aneurysm specialists that the potential benefit of these treatment options do not outweigh the risk of treatment complications, the aneurysm often remains untreated. 3 In these patients, the aneurysm is usually followed over time with serial imaging. If substantial aneurysm growth occurs, which is a marker of aneurysm instability and a predictor of rupture, these aneurysms are treated to prevent rupture.4–6
Most patients who do not undergo endovascular or neurosurgical aneurysm treatment have a small UIA. However, since small UIAs with a low risk of rupture are much more prevalent than large UIAs with a higher risk of rupture, most instances of SAH are caused by rupture of small UIAs. 7 Therefore, it is important to establish novel, low risk treatment strategies for patients with small UIAs and for other patients who do not undergo preventive aneurysm treatment.
Smoking, hypertension and aneurysm wall inflammation are important modifiable risk factors of aneurysm rupture and growth.8–10 All patients with an UIA who smoke should be advised to quit smoking. Patients with hypertension are advised to have this treated according to current guidelines (<140/90 mmHg). Targeting on lower blood pressures (BPs), similar to the Systolic Blood Pressure Intervention Trial (SPRINT), has not been considered in patients with an UIA. 11 A small double-blind, randomized cross-over trial in 19 patients with an intracranial aneurysm (either unruptured or ruptured) showed that antihypertensive treatment was well tolerated in patients who were considered normotensive. 12 Aneurysmal wall inflammation is an attractive novel target for medical intervention. Several studies suggested that acetylsalicylic acid (ASA) decreases aneurysm wall inflammation and hereby exerts a protective effect on aneurysm rupture.9,10,13,14 Also, several studies found no more severe hemorrhage or worse outcomes if aneurysm rupture occurs under ASA treatment.15–17
The aim of this randomized trial is to investigate in patients with an UIA who do not qualify for preventive endovascular or neurosurgical intervention whether ASA in combination with intensive BP treatment decreases the risk of aneurysm rupture and growth.
Methods
Design
Prospective Randomized Open-label Trial to evaluate risk faCTor management in patients with Unruptured intracranial aneurysms (PROTECT-U) is a multicenter, prospective, randomized, open-label phase III trial with a blinded outcome assessment (PROBE design).
Patient population
Patients 18 years or older with an untreated, intradural, saccular UIA on MR- or CT-angiography are eligible for participation if the benefits of preventive neurosurgical or endovascular aneurysm treatment do not outweigh the procedural risks according to the multidisciplinary team of physicians. We will not only include patients with small aneurysms, but also those with complex, calcified, or thrombosed aneurysms, because also in the latter group of patients the risk of procedure complications from preventive endovascular or neurosurgical treatment may be higher than the risk of rupture. Therefore, those patients may also benefit from the preventive medical treatment.
Inclusion- and exclusion criteria
Inclusion- and exclusion criteria

UIA: unruptured intracranial aneurysm; ASA: acetylsalicylic acid.
Randomization
The randomization plan was developed using permuted-block randomization (random number tables). As soon as the inclusion criteria are fulfilled and written informed consent is obtained from the patient, the patient will be randomized via a centralized secured website. The trial has two arms: (1) the interventional arm and (2) care as usual.
Intervention
The intervention consists of a strategy with ASA 100 mg once daily in combination with intensive BP treatment (targeted mean systolic BP below 120 mmHg with a home BP measuring device). Patients randomized to care as usual will receive no ASA as a study drug, will have their BP treated according to current guidelines (targeted office systolic BP below 140 mmHg), and will not receive a home BP measuring device. We decided on a low dosage of ASA (100 mg) instead of a higher dosage (300 mg) since in a histological study a dosage of 81 mg already decreased aneurysm wall inflammation. 9 Irrespective of the trial arm, BP control will be performed by the participants’ own general practitioners, who will be instructed to reduce BP according to the assigned target. We do not recommend specific types of antihypertensive drugs or treatment schemes.
Patients randomized to the intervention arm will receive an Omron BP monitor (HEM-9210T) after randomization and instructed on how to use the device. BP needs to be measured in a sitting position. All BP recordings will be saved automatically (maximum of 100 recordings).
The study period for an individual patient consists of a minimum treatment period of 36 months and a maximum treatment period of 60 months (plus a follow-up visit one month after end of treatment). All patients stay on intervention until 3 years of the last patients’ enrolment (Figure 1).
Study flowchart. BP: blood pressure; ASA: acetylsalicylic acid; m: month.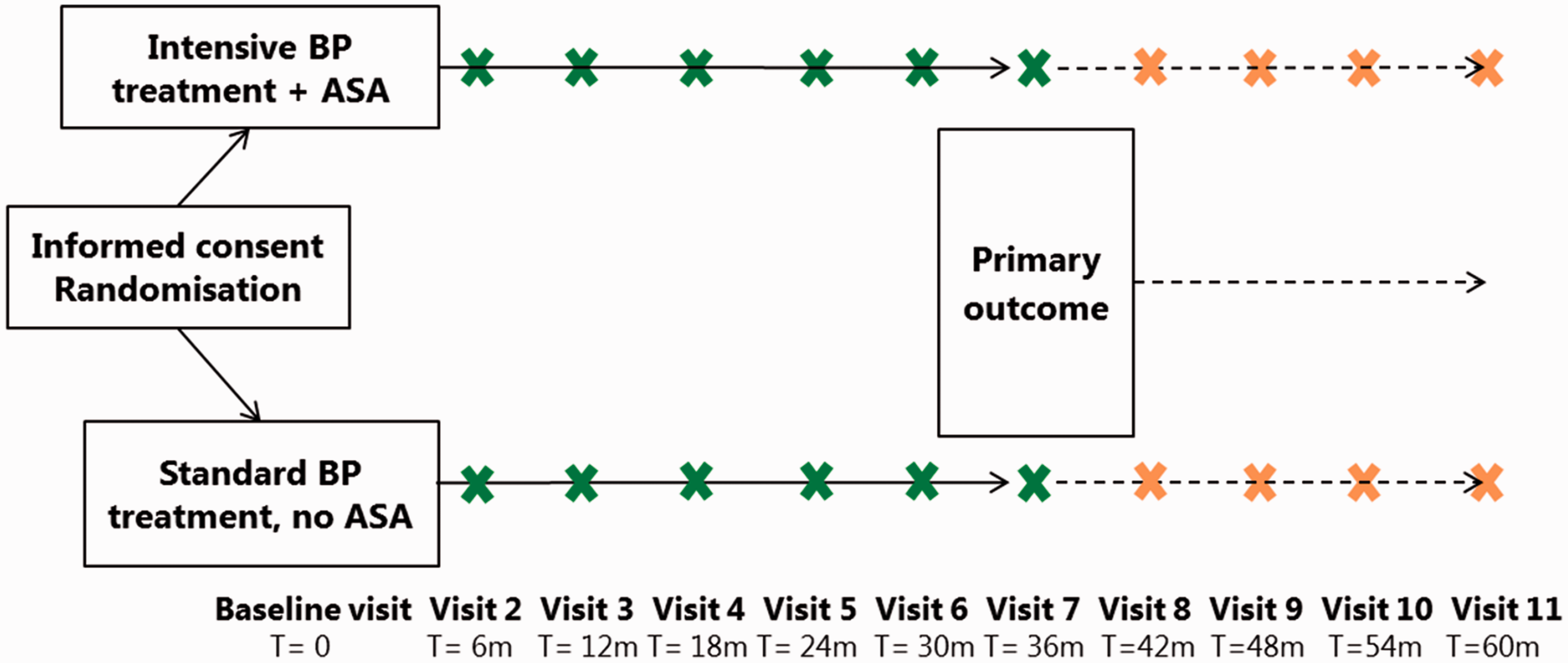
Primary outcome
The primary outcome is aneurysm rupture or growth (increase in any aneurysm diameter by ≥ 1 mm) on repeated MR- or CT-angiography within 36 ± 6 months after randomization. All aneurysm imaging made during the trial period will be uploaded to a central server in a pseudonymized manner with the patient ID number and date of imaging. The pseudonymization method will not reveal the treatment allocation to permit a blinded imaging assessment. Two independent radiologists will centrally assess aneurysm rupture or growth. The adjudication committee (one neurosurgeon, two neurologists, one cardiologist) will centrally review events suspicious for SAH but negative CT-imaging, and instances of sudden death of participants.
Secondary outcomes
Difference of aneurysm volume (defined as increase of aneurysm volume in computerized measurements from source images by > 10% and > 3 mm3 or aneurysm shape; e.g. development of daughter sac) Development of de novo aneurysm on serial imaging Clipping/coiling during the study period Any ischemic or hemorrhagic stroke, is defined as clinical symptoms of stroke AND a compatible lesion on imaging (either plain head CT/CT perfusion/MRI) Myocardial infarction is defined as increase of levels of troponin, CK-MB and/or presence of new significant Q waves obtained on ECG Vascular death (including fatal stroke, fatal myocardial infarction, sudden death) Major spontaneous bleeding requiring hospitalization is defined as substantially disabling bleeding, intraocular bleeding leading to the loss of vision, or bleeding necessitating the transfusion of at least 2 units of erythrocyte concentrates Blood pressure; any data on BP management will be used Safety aspects (adverse and serious adverse events, including those mentioned above) Quality-of-life (as determined by completion of the paper-based EQ-5D-5L form by patient)
Data collection
Data collection
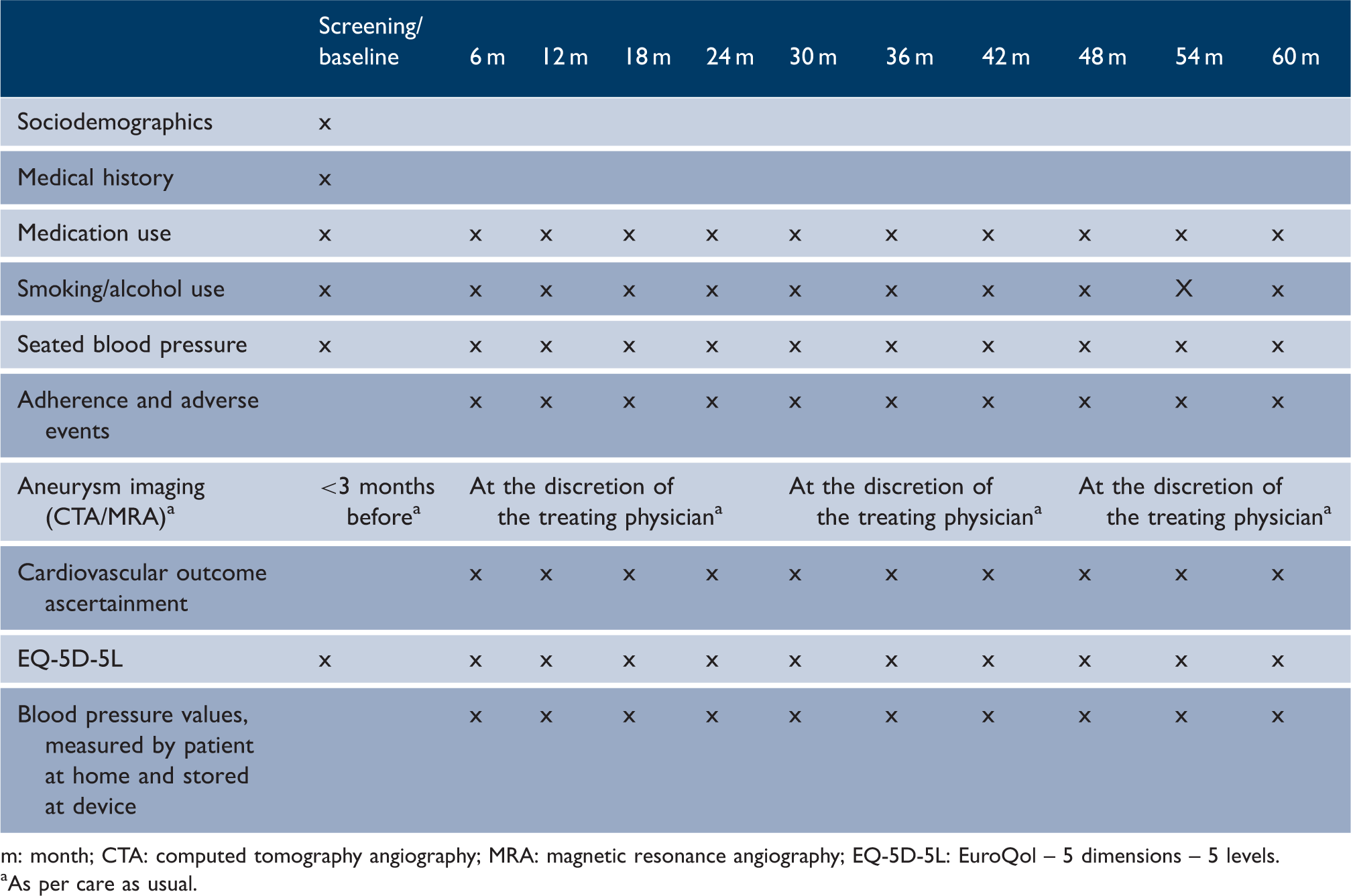
m: month; CTA: computed tomography angiography; MRA: magnetic resonance angiography; EQ-5D-5L: EuroQol – 5 dimensions – 5 levels.
As per care as usual.
Data monitoring body
The independent data safety monitoring board (DSMB) will meet once a year, with additional meetings or conference calls scheduled as needed. The DSMB can recommend the Steering Committee of the PROTECT-U trial to:
‐ Adjust the sample size ‐ Early terminate the study, when there is clear and substantial evidence of benefit, based on a significant (with alpha 0.1%) reduction in aneurysm growth and rupture (according to the Peto approach of interim analysis with alpha 5% at final analysis) ‐ Early terminate the study, when there is evidence of severe harm based on SAE reporting, outcome, and case fatality ‐ Early terminate the study, in case accrual rates are too low to provide adequate statistical power for identifying the primary outcome ‐ Early terminate the study, in case an interim analysis shows that continuation of the trial is unlikely to reveal a difference between the groups (futility analysis)
Each 6 months, the DSMB will be informed on the rate of SAH to monitor the safety of the study. Monitoring may be more frequent at later phases in the trial as the number of person-years increases non-linearly. If the lower limit of the 95% confidence interval is higher than 2% rupture rate, the DSMB will recommend stopping the study.
The DSMB will perform two interim analyses on the primary outcome to assess the strength of the efficacy data. The first interim analysis will be carried out after 235 patients have had their 3-year outcome assessment; the second one after 470 patients have these outcome data. The DSMB will also verify the assumptions for sample size calculations.
After the inclusion of 100, 300 and 600 patients, and if needed after later time points, the proportion of patients with aneurysms < 3 mm will be analyzed. Patients in this subgroup have the lowest risk of aneurysm growth and rupture. Care will be taken that the proportion of patients with aneurysms < 3 mm does not exceed 30% of the total study cohort, so that the power calculation is not jeopardized.
Because the first formal interim analysis will take place only after about four years and four months into the trial (because outcomes are available only after three years of follow-up), two additional preliminary interim analyses will be done based on the first 235 and 470 patients with all one-year routinely obtained follow-up data available. To this end, local reports on aneurysm growth will be used.
Sample size estimates
Previously, an odds ratio of 0.27 (95%CI: 0.27–0.67) for risk of rupture in UIA patients under ASA treatment was reported. 10 Since observational studies generally overestimate risk reduction, we conservatively assumed a risk reduction of 45% from the combined intervention (ASA plus intensive BP treatment) resulting in a risk of having a primary outcome within three years of 15% in the care as usual group and 8.25% in the intervention group. With α = 0.05 and β = 0.20, 353 patients are needed in each group.8,11,18 Assuming a dropout rate of 9%, we will need to assign 776 patients to the trial so that 706 patients can be analyzed.
Statistical analyses
Primary analysis
We will compare the occurrence of outcomes in the two groups in absolute terms and as risk ratio (RR), which will be obtained by means of a Poisson regression model. Precision of the RR estimates will be described with 95% confidence intervals. Our primary analysis will be based on the intention-to-treat principle. We plan the following subgroup analyses for the primary outcome: age (≤55 versus > 55 years), sex, aneurysm type (with or without previous rupture of another aneurysm), aneurysm size (1.0–4.0 mm versus ≥ 4.1 mm), aneurysm location (posterior communicating artery/anterior communicating artery/posterior circulation versus all other aneurysms), and aneurysm morphology (regular versus irregular shape).
Additional analyses
In addition to our main research question, we aim to separately evaluate the effect of the ASA component and the BP component. To this end, we will accurately record who is using ASA and/or BP pressure lowering therapy in either arm. For ‘on protocol analysis’, we will relate either the ASA or intensive BP lowering therapy usage percentage to the primary outcome. As these analyses pertain to non-randomized comparisons, we will adjust for potential confounding by other determinants of the primary outcome, including the other component of the combined treatment. Analyses will be done with multivariable Poisson regression.
We will do an additional analysis restricted to patients in the intervention group, because within this group detailed data on achieved BP will be available as obtained with a device that transmits BP measurements to a central post. For each patient, we will calculate the proportion of time the systolic BP is below 120 mmHg. Then, we will do an analysis similar as described above in which we will relate the proportion of time that systolic BP is below 120 mmHg to the primary outcome.
Study organization and monitoring
The trial will be bi-national with patient inclusion in Germany and the Netherlands, and will be carried out according to Good Clinical Practice E6(R2) guidelines and the current version of the Declaration of Helsinki. The sponsor of the trial is the Medical Faculty Mannheim, University of Heidelberg, Germany. The Koordinierungszentrum für Klinische Studien (KKS), Ruprecht-Karls-University, Heidelberg, Germany, will coordinate and monitor the trial. The first patient was recruited in September 2017 and the study is currently actively recruiting.
Discussion
PROTECT-U is the first randomized trial to investigate if a medical strategy (ASA plus intensive BP treatment) reduces aneurysm rupture or growth in UIA patients not undergoing preventive neurosurgical or endovascular aneurysm treatment.
We decided on a two-arm trial (i.e. concomitant ASA treatment plus intensive BP treatment versus care as usual) instead of a three-arm trial (i.e. ASA treatment versus intensive BP management versus care as usual) or even a four-arm trial (i.e. ASA treatment versus intensive BP management versus combined treatment versus care as usual) for the following three reasons:
Clinical perspective: Since ASA and antihypertensive drugs have different modes of action, it is plausible that the protective effect of each treatment component is smaller. A combined effect is to be expected when the two treatments are given simultaneously. Pragmatic/feasibility perspective: To achieve the same power in the study while including an additional arm (ASA versus intensive BP treatment versus care as usual), we would need 4791 patients to be included in this investigator-initiated study. A 2 × 2 factorial design (ASA versus no ASA and intensive versus regular BP control) would need 1728 participants. We consider an investigator-initiated trial with over 1000 participants who need close monitoring unfeasible. Cost-effectiveness perspective: Combined treatment is likely to be the treatment of choice: both treatments are relatively inexpensive, and cost-effectiveness calculations showed that the overall cost of combined treatment is significantly lower than care as usual.
Supplemental Material
Appendix -Supplemental material for Prospective Randomized Open-label Trial to evaluate risk faCTor management in patients with Unruptured intracranial aneurysms: Study protocol
Supplemental material, Appendix for Prospective Randomized Open-label Trial to evaluate risk faCTor management in patients with Unruptured intracranial aneurysms: Study protocol by Mervyn DI Vergouwen, Gabriel JE Rinkel, Ale Algra, Jens Fiehler, Helmuth Steinmetz, Peter Vajkoczy, Frans H Rutten, Steffen Luntz, Daniel Hänggi and Nima Etminan; on behalf of the PROTECT-U investigators in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by the Dr. Rolf M. Schwiete Foundation from Mannheim, Germany (project number 19/2016). BP measuring devices are provided by Omron Healthcare Europe BV. The blood pressure data will be analyzed and centrally stored using the Careonline Software, Humediq, Munich, Germany.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
