Abstract
Background and purpose
Although poor prognosis after intracerebral hemorrhage relates to risk factors and hematoma characteristics, there is limited evidence for the effect of race–ethnicity.
Methods
Data from 1011 patients with intracerebral hemorrhage enrolled into hyperacute trials and randomized to control were obtained from the Virtual International Stroke Trials Archive and Efficacy of Nitric Oxide in Stroke Trial. Clinical characteristics and functional outcome were compared among three racial groups – Asians, Blacks, and Caucasians.
Results
The majority of patients were Caucasian (78.1%) followed by Asians (14.5%) and Blacks (5.5%). At baseline, Caucasians were older and had larger hematoma volumes; Blacks had lower Glasgow Coma Scale and higher systolic blood pressure (all p < 0.05). Although the primary outcome of modified Rankin Scale did not differ at 90 days (p = 0.14), there were significant differences in mortality (p < 0.0001) and quality of life (EQ-5D p < 0.0001; EQ-VAS p 0.015). In test of multiple comparisons, Caucasians were more likely to die (p = 0.0003) and had worse quality of life (EQ-5D p = 0.003; EQ-VAS p < 0.0001) as compared to Asians.
Conclusion
Race–ethnicity appears to explain some of the variation in clinical characteristics and outcomes after acute intracerebral hemorrhage. Factors that explain this variation need to be identified.
Keywords
Introduction
Spontaneous intracerebral hemorrhage (ICH) is a severe cause of stroke associated with significant morbidity and mortality. 1 In contrast to ischemic stroke where incidence has declined in recent decades, the number of admissions for ICH continues to increase.2,3 Variations in ICH incidence and outcome occur between different race–ethnicity groups. A higher incidence of ICH is observed among Black and Hispanic populations when compared with Caucasians.4–6 The SINO-MONICA-Beijing study reported greater mortality due to ICH in China compared to other countries. 7 Death caused by stroke was reported to be higher in Asians compared to Caucasian patients in the UK and Canada.8,9 The causes for these observations are unclear and might be explained by increased prevalence of risk factors such as diabetes and hypertension among Asians.10–13 Additional explanatory factors also appear to include blood pressure (BP) at presentation.5,14–16 However, studies assessing ICH in different race–ethnicity groups have mostly involved small centers with limited numbers of patients and have restricted comparisons to differences between two racial populations.5,6,17,18
In this study, we compared baseline clinical characteristics and functional outcome in three racial groups in patients with ICH. The data came from the Virtual International Stroke Trials Archive (VISTA) collaboration 19 and the Efficacy of Nitric Oxide in Stroke (ENOS) trial. 20
Methods
Data sources
The included patients came from the VISTA-ICH archive, which includes data from a number of completed randomized controlled trials (RCTs) 19 and the acute ENOS BP trial.20,21 Patients were those with spontaneous ICH, as diagnosed locally using routine neuroimaging, and who were treated within 6 h of onset. We included patients allocated to the control group in ENOS, as patients assigned to intervention are not included in VISTA. Patients were excluded if: age < 18, ICH attributed to trauma, planned surgical evacuation, previously dependent (modified Rankin Scale, mRS ≥ 2), concurrent illness with life expectancy <6 months, pregnant or breast feeding women, and those already participating in a study involving another drug or device.
Baseline data
Data at baseline were obtained for age, sex, time from ictus to treatment, medical history (previous stroke, hypertension, ischemic heart disease, diabetes mellitus, hypercholesterolemia, atrial fibrillation, and prior antithrombotic use), level of consciousness (Glasgow Coma Scale, GCS), stroke severity (National Institutes of Health Stroke Scale, NIHSS), systolic blood pressure (SBP), and heart rate. Race (and ethnicity) was defined as Asian (Filipino, Oriental, Asian, Indian, Palestinian, and Arabic), Black (Black, African/American, Caribbean), Caucasian (White or Caucasian), and Others (American Indian, mixed-colored, mixed, Hispanic, colored, Hawaiian and Pacific Islanders).
Outcome data
Outcome at day 90 was assessed using the mRS and all-cause mortality; where available, data on disability (Barthel Index, BI) and quality of life (European quality of Life-5 dimensions, EQ-5D) were also assessed.
Neuroimaging
Depending on trial protocol, CT scans were performed following treatment and then again at 72 h. Information from CT scans was collected for hemorrhage location, lesion volume, edema volume, presence of mass effect, subarachnoid bleeding, intraventricular hemorrhage, brain atrophy, and leukoaraiosis.
Statistical analysis
The primary outcome was the mRS at day 90. Baseline characteristics and outcomes were compared by Pearson χ 2 test for categorical variables and analysis of variance for continuous variables. The relationship between baseline hematoma volume and mRS between ethnic groups was analyzed using multiple regression. Individual comparisons between ethnic groups were made using Bonferroni correction. Mortality was assessed using Kaplan–Meier curves and Cox Regression. Analyses were adjusted for age, NIHSS, ICH volume, and time to treatment. Confidentiality agreements precluded identification of the individual trials in VISTA-ICH and so analyses were not adjusted for trial. Since death is present in outcome scores such as mRS (6), HUS (0), and BI (−5), death was scored for European quality of life-visual analogue scale (EQ-VAS) as −1. Statistical significance was set at p ≤ 0.05. All analyses were performed using SPSS (version 21) running on an Apple Mac computer.
Results
Baseline clinical and neuroimaging characteristics
Baseline characteristics of 1011 patients with intracerebral hemorrhage by ethnicity
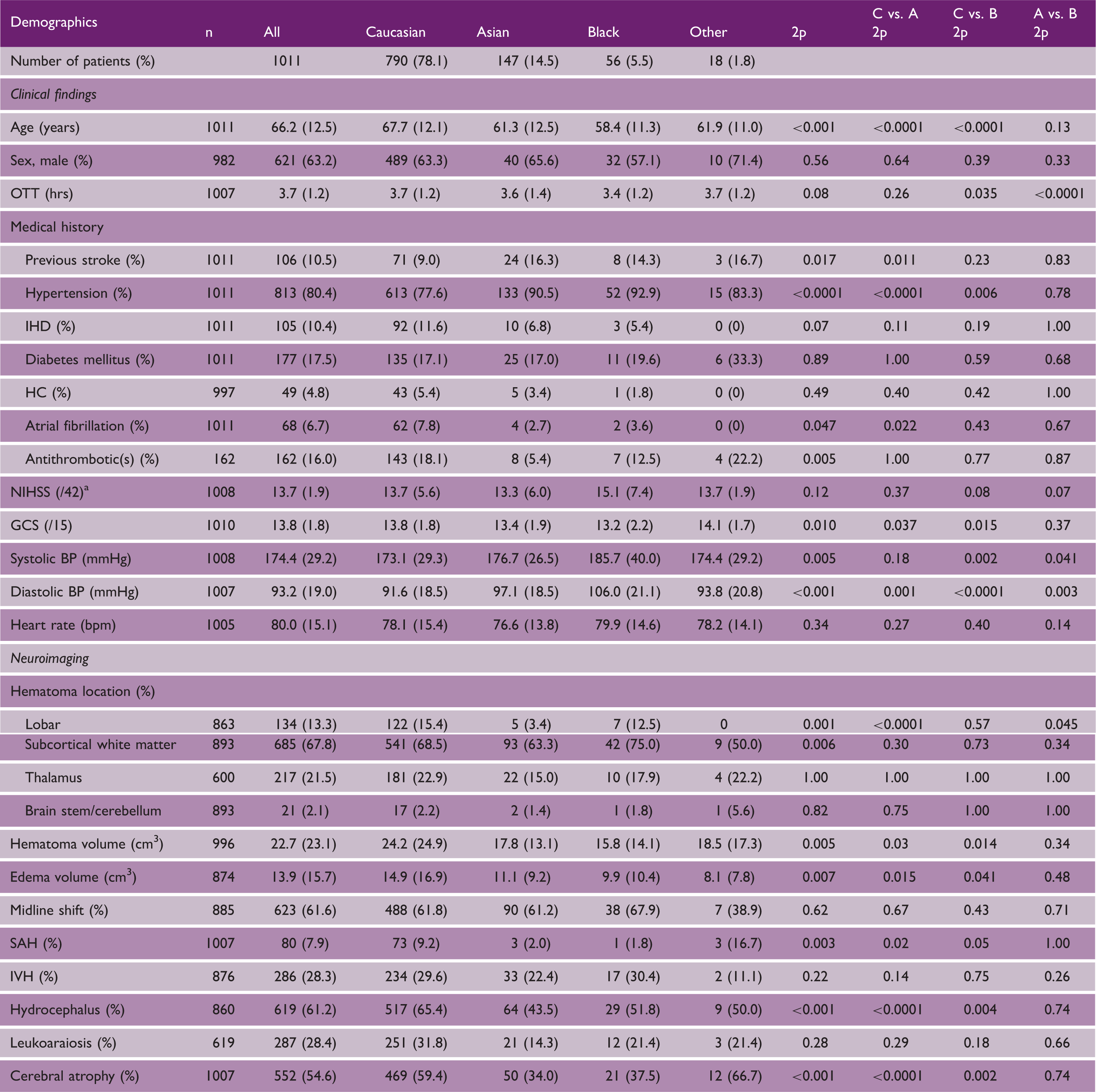
BP: blood pressure; GCS: Glasgow Coma Scale; HC: hypercholesterolemia; IHD: ischemic heart disease; IVH: intraventricular hemorrhage; OTT: onset to treatment; SAH: subarachnoid hemorrhage. Data are number (%) or mean (standard deviation). Comparisons by chi-square test or one-way analysis of variance.
Calculated 22 from Scandinavian Stroke Scale in ENOS.
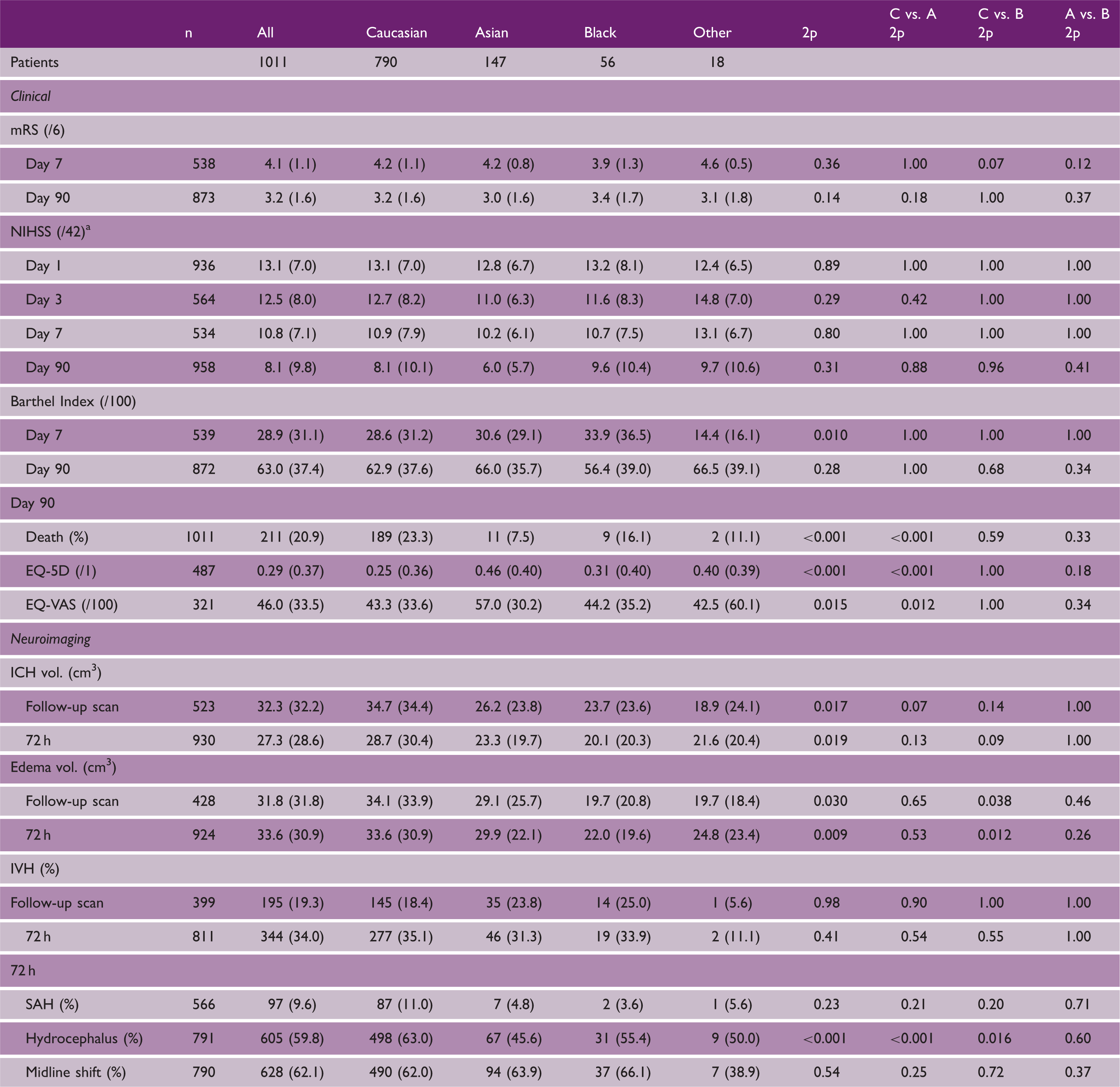
Clinical outcomes
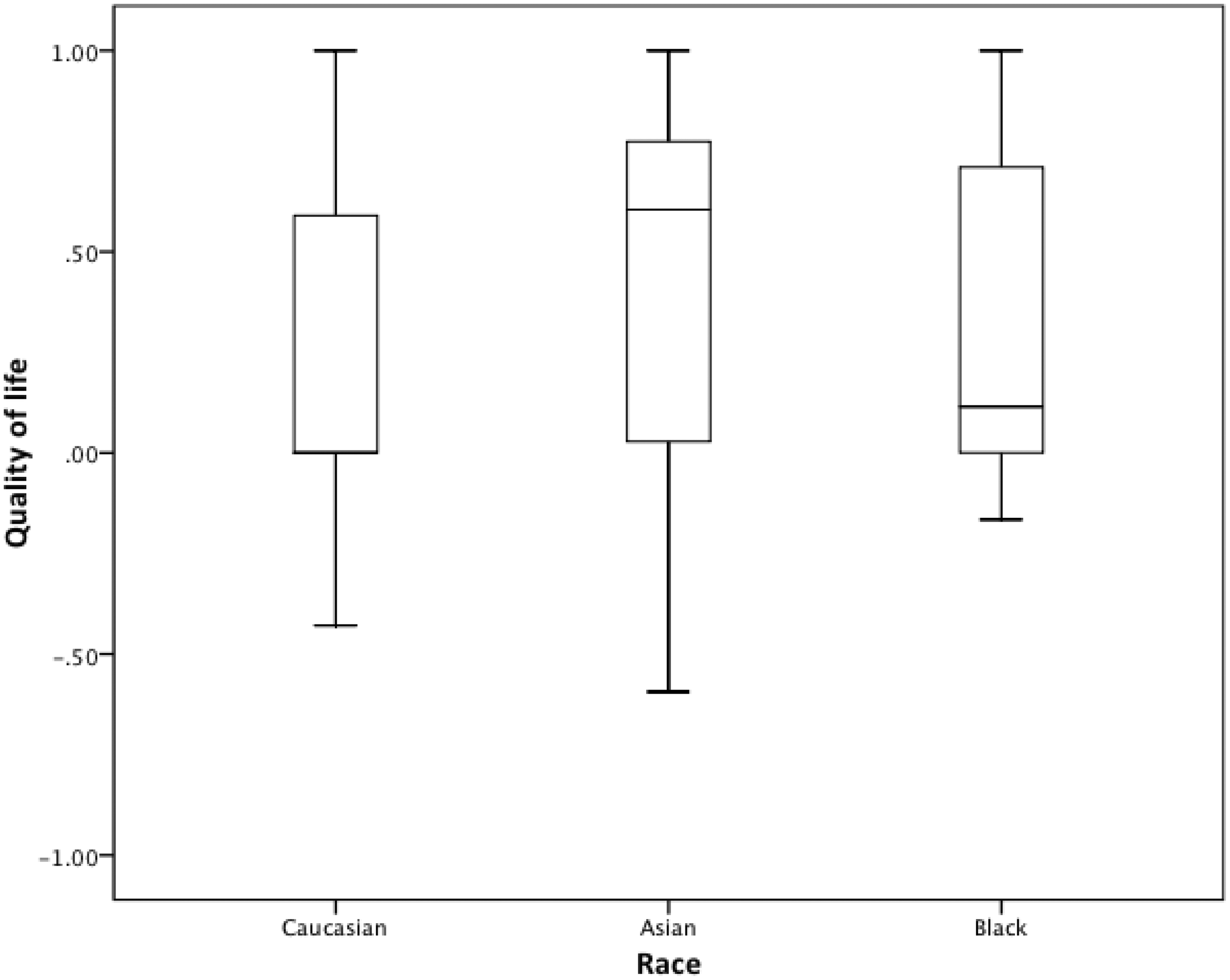
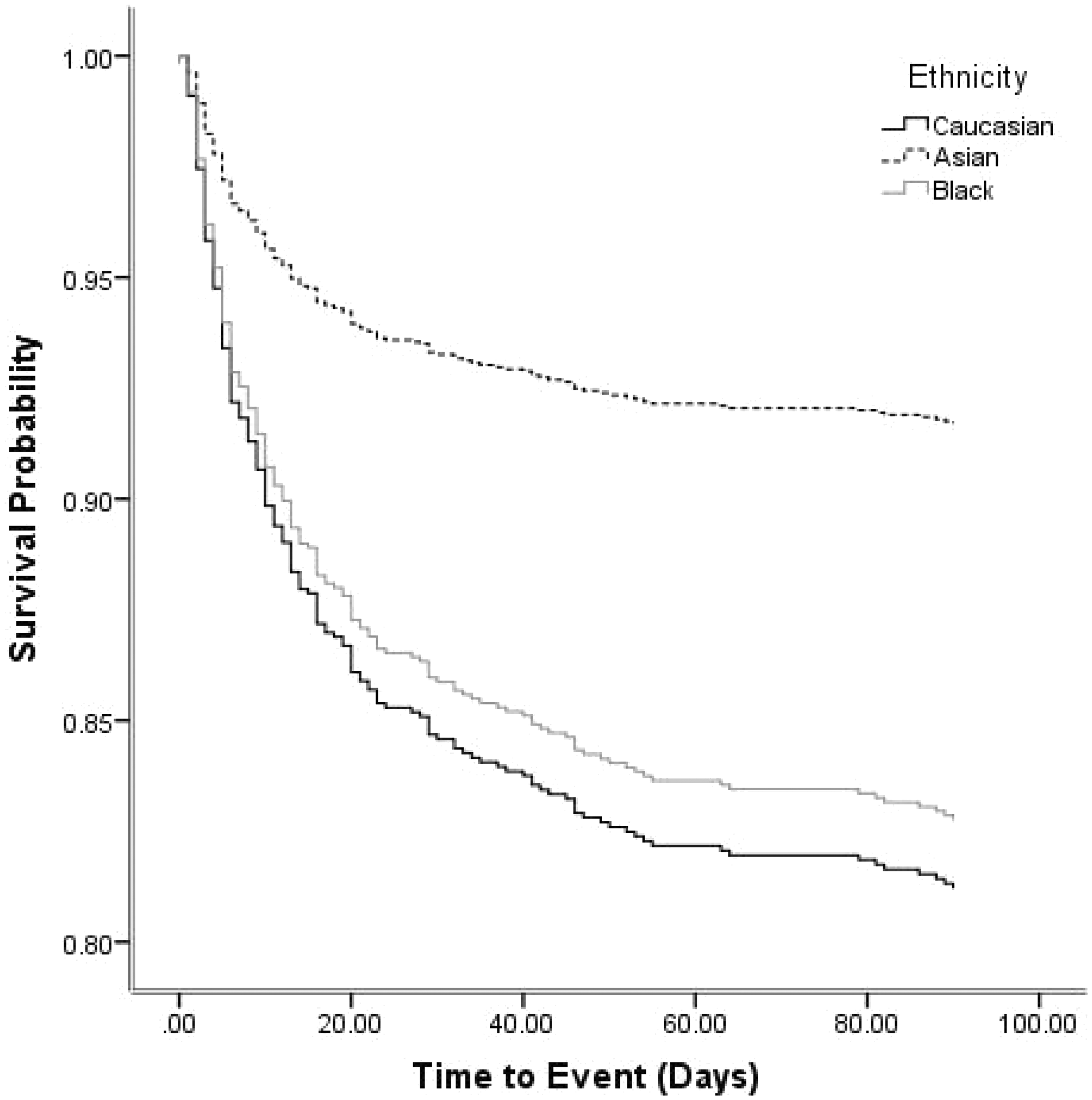
Table 2 shows clinical and radiological outcome measures by ethnicity. The primary outcome, mRS, did not differ between race groups at either day 7 or 90 (Figure 1). Similarly, there was no difference in NIHSS during the first seven days. Whilst the BI differed at day 7, it was not significant at day 90 using a test of multiple comparisons. Quality of life, assessed using two measures (health utility status derived from EQ-5D and EQ-VAS), differed between the three groups (Table 2, Figure 2); following adjustment for multiple comparisons, Caucasians had a worse quality of life scores for both EQ-5D and EQ-VAS as compared to Asians (Table 2). Adjusted survival rates differed between race–ethnicity groups (Table 2, Figure 3), explained by a significant difference between Asians and Caucasians (p = 0.005). There were no other significant differences for other outcomes.
Distribution of modified Rankin scores between the three race groups at day 90. Clinical and radiological outcomes by race groups A: Asians; B: Blacks; C: Caucasians; EQ-5d: European Quality of Life-5 dimensions; EQ-VAS: European Quality of Life-Visual Analogue Scale; ICH: intracerebral hemorrhage; IVH: intraventricular hemorrhage; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; SAH: subarachnoid hemorrhage. Data are number (%) and mean (standard deviation). Comparisons between group differences made by chi-square test or analysis of variance; multiple comparisons include Bonferroni correction. Calculated from Scandinavian Stroke Scale in the ENOS trial.
22
Range of scores for patients in ENOS: Barthel Index: −5 (death) to 0 (severe disability) to 100 (no disability). Health utility status (HUS, derived from European Quality of Life-5 dimensions, EQ-5d): −0.5 (very poor quality of life, 0 (death) to 1.0 (perfect quality of life). European Quality of Life-Visual Analogue Scale (EQ-VAS): −1 (death), 0 (very poor) to 100 (excellent). Zung Depression Scale (ZDS): 0 (normal), 100 (severe depression) to 102.5 (death). Box plots of European Quality of Life-5 dimension-3 level scores at day 90. Comparison by ANOVA 2p < 0.001; Caucasian vs. Asian 2p < 0.001, Caucasian vs. Black 1.00, Asian vs. Black 2p = 0.18. Cox regression with adjustment for age, baseline systolic blood pressure, stroke severity (NIHSS), hematoma volume, and time to treatment: overall p = 0.021; Asians vs. Caucasians, hazard ratio 0.42 (95% CI 0.22, 0.77); p = 0.005.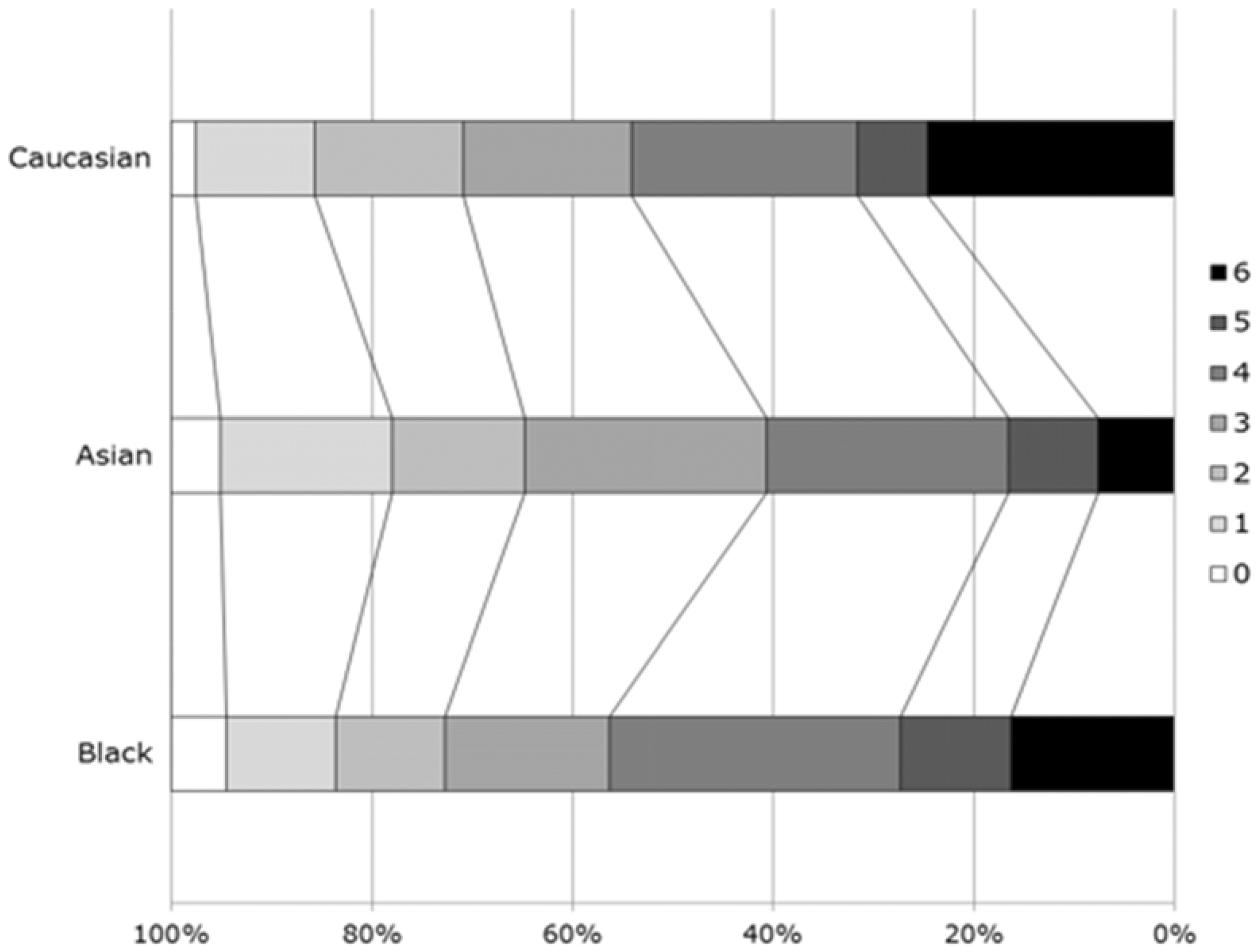
Neuroimaging outcomes
ICH volume differed across the race groups on both the first follow-up scan (Table 2, Figure 4) and at 72 h. (Table 2); in both cases, Caucasians tended to having large hematoma (Table 2). Similarly, edema volumes differed across the race groups on both the first follow-up scan and at day 7 with Caucasian patients have large edema volumes than Black patients. The presence of hydrocephalus also differed and was highest in Caucasians. The presence of intraventricular hemorrhage, subarachnoid hemorrhage and midline shift did not differ between the race groups (Table 2).
Intracerebral hematoma volume on first follow-up imaging by race. Comparison by ANOVA 2p = 0.017; Caucasian vs. Asian 2p = 0.07, Caucasian vs. Black 0.14, Asian vs. Black 2p = 1.0.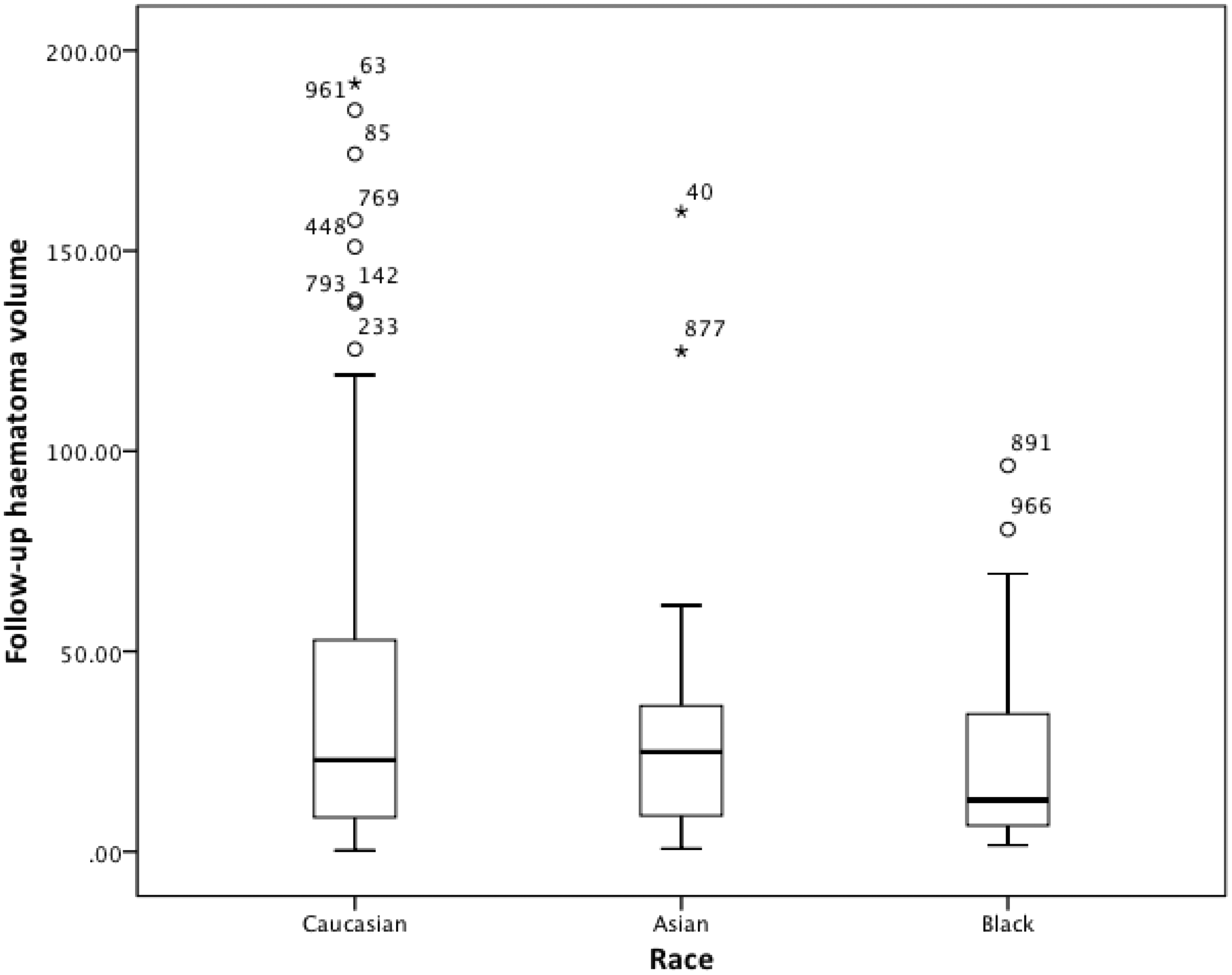
When assessing the relationship between baseline hematoma volume and mRS, there was a significant difference in the slope of the regression lines between the race–ethnicity groups; Blacks and Asians appeared to have a worse mRS for a given hematoma volume (p = 0.047; online Supplementary Figure 1). There were no other significant differences between any of the other comparison groups.
Discussion
To our knowledge, this is the first study to examine clinical and neuroimaging features and outcomes in ICH patients across three race groups. Significant differences in age, the frequency or previous stroke and hypertension, GCS, BP, and ICH volume were present between Caucasian, Asian, and Black patients. Similarly, differences in neuroimaging features were present for hematoma location and volume, edema volume, and the presence of subarachnoid hemorrhage, hydrocephalus, leukoaraiosis, and cerebral atrophy. Although there was no difference in the primary outcome of mRS at day 90, significant differences in case fatality and quality of life were present. Similarly, differences in ICH and edema volume, and the presence of hydrocephalus, were present between the race groups.
Black patients were younger and a larger proportion had a history of hypertension which could be associated with increased smoking, alcohol, and drug abuse.17,18 It is noteworthy that BP in Blacks was higher on admission and probably reflects inadequate control, poor compliance with treatment or treatment resistance.17,23 Higher BP in the acute phase of ICH is common 24 and associated with neurological deterioration through hematoma expansion and re-bleeding.24–26 The treatment implications of racial difference in BP lowering need investigation as evidence suggests that early treatment is both safe and feasible20,27 and is now recommended in guidelines.28,29 In contrast to a previous report of greater mortality in Blacks for every stroke subtype, 30 VISTA–ENOS found reduced survival in Caucasians with ICH. This finding is difficult to explain and may relate to hemorrhage location, inherent susceptibility to ICH or quality of care or simply that race–ethnic differences persist even after controlling for such characteristics.
As would be expected, ICH survivors had a quality of life worse than that reported in the general population.31,32 The difference between Caucasians and Asians has been reported previously14,33 and may relate to family values, cultural attitudes, and care preferences.33,34 In one study, Asians were more likely to be living at home, whilst Caucasians were less likely to be discharged home despite better functional improvement. 35 Apart from cultural and family factors, age, female sex, stroke severity, persistent neglect, and socioeconomic status have also been shown to affect quality of life after stroke.36,37
The strengths of the study include a relatively large sample size for a study population with detailed clinical, radiological information, and multiple functional endpoints. The included patients were from neutral RCTs or control group and therefore assessment of the race–ethnic differences avoided the confounding effects of active treatment. However, there are some limitations. First, the analysis on observational data from RCTs has the disadvantage that patients were included on the basis of pre-specified selection criteria. It is possible that those with more severe stroke with reduced level of consciousness, older patients with multiple comorbidities and pre-existing do not resuscitate orders, may have been excluded. Therefore, the results may not be applicable to a population of unselected ICH patients. Second, the VISTA collaboration does not reveal the identities of the individual trials and so it is uncertain as to what inclusion criteria were used to select patients. Additionally, analyses could not be adjusted for the source trials, which is important since trials themselves are a determinant of outcome. Nevertheless, the data were subject to strict verification and so the results are externally valid. Third, ethnicity relates to culture, geography, language and it is not clear from the data if this was self-reported or defined by the investigator. Fourth, to compensate for limited numbers, distinct subpopulations were grouped into one ethnic group (for example Asians included people from China, Philippines, the Indian subcontinent, and Arabian peninsula), whilst small groups present in the ‘others’ category were excluded. This may have not been appropriate as stroke risk and functional outcome vary within subpopulations living in the same country and between countries.38,39 Last, the analysis is limited to trials sharing information within the collaboration and therefore those studies that are not represented in VISTA would not have been included.
In conclusion, this analysis from hyperacute trials in hemorrhagic stroke found significant race differences in baseline clinical characteristics and hematoma measures. There were no difference in the primary outcome of death or dependency but differences in death rates and quality of life were apparent. Further studies are needed to which factors explain race–ethnic differences and whether these differences become larger over time. Our results suggest that future trials should undertake measures to ensure adequate representation of all major race–ethnic groups. This might include standard definitions for race–ethnicity that reflect common features but not limited to biological, cultural, and social differences. Further research is also needed to understand where inadequacies in health care systems exist and what interventions can be made to reduce such disparities. This could include allowing more resources to developing countries or those without access to specialist stroke care teams.
Footnotes
Acknowledgments
The authors thank the VISTA collaboration for sharing the relevant data and investigators and patients who participated in ENOS.
VISTA-ICH Steering Committee: DF Hanley (Chair), K Butcher, S Davis, B Gregson, KR Lees, P Lyden, S Mayer, K Muir, and T Steiner.
ENOS Steering Committee: P Amarenco, PM Bath, E Berge, KR Lees, K Muir, S Pocock, G Venables, JM Wardlaw, and D Whynes.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VISTA is a non-profit collaboration of academics and commercial organizations. Each VISTA steering committee member has contributed to organization of clinical trials and where industry support was involved have acknowledged this in the individual trial publications. Philip M Bath is Stroke Association Professor of Stroke Medicine, Chief Investigator of ENOS, and an NIHR Senior Investigator. Joanna M Wardlaw was the neuroimaging lead of ENOS. No author involved in this work has any additional interest to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Research Council [grant number G0501797]; and the University of Nottingham.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
