Abstract
Background and aims
Individual MRI markers of cerebral small vessel disease are associated with gait impairment. The impact of total cerebral small vessel disease-related brain damage, expressed by a cerebral small vessel disease MRI burden score, on mobility after stroke, has not been considered, although this score gives a better representation of the overall effect of cerebral small vessel disease on the brain. We determined if the total cerebral small vessel disease burden is associated with gait impairment three years after minor stroke.
Methods
In total, 200 patients with minor lacunar or non-lacunar stroke (NIHSS ≤ 7) underwent a brain MRI at presentation. Presence of lacunes, white matter hyperintensities, cerebral microbleeds, and perivascular spaces were summed in a total cerebral small vessel disease MRI burden score (range 0–4). Gait disturbances, measured by timed-up-and-go test and self-reported stroke impact scale mobility domain were assessed three years after stroke. We tested associations adjusted for key variables by linear regression analysis.
Results
Total cerebral small vessel disease burden was not associated with gait impairment after minor stroke in all patients, nor in lacunar stroke patients (n = 87). In non-lacunar stroke patients (n = 113), total cerebral small vessel disease burden was associated with lower stroke impact scale mobility domain scores, independent of age, vascular risk factors, and stroke severity (unstandardized B −4.61; 95% CI −8.42; −0.79, p < 0.05).
Conclusion
Patients with non-lacunar stroke and a higher total cerebral small vessel disease burden have more subjective mobility impairment three years after stroke. The total cerebral small vessel disease MRI burden score is a possible marker to identify patients at risk for subjective gait impairment. These findings should be confirmed in larger studies.
Keywords
Introduction
Cerebral small vessel disease (cSVD) causes MRI visible brain damage, including white matter hyperintensities (WMH), lacunes, perivascular spaces (PVS), and cerebral microbleeds (CMB).1,2 The burden of cSVD on public health is substantial: cSVD is a leading cause of cognitive impairment, dementia, and stroke. 3
Less recognized, cSVD is also related to gait disturbances4,5 and worse functional outcome after stroke.6–8 Several studies have demonstrated that individual MRI markers of cSVD, including WMH and CMB, are related to gait impairment.4,5,9,10 However, most of these studies have focused on individual MRI features of cSVD rather than recognizing the impact of total cSVD-related brain damage. A total cSVD burden score, based on visual MRI feature recognition, might better capture the relation between risk factors and brain injury11,12 or the overall effect of cSVD on cognitive function.13,14 Whether the total cSVD MRI burden score is associated with gait impairment after stroke is unknown.
Aims
We aimed to determine if overall cSVD-related brain damage, measured by a total cSVD MRI burden score, is associated with gait disturbances, objectively measured by the timed-up-and-go (TUG) test and subjectively assessed by self-reported stroke impact scale (SIS) mobility domain score, three years after minor ischemic stroke. In a secondary analysis, we also tested the association between the total cSVD burden scale and functional stroke outcome, measured by the modified Rankin Scale (mRS).
Methods
For a detailed description, see the online-only Supplemental Data. In short, we used data from a prospective observational study in patients with minor ischemic stroke (Mild Stroke Study-2).15–17 The study was approved by the Scotland and Lothian Research Ethics Committee (ref 09/S1101/54) and all patients gave written informed consent.
All patients underwent a 1.5 Tesla brain MRI scan, which was assessed for presence of an acute symptomatic lacunar or non-lacunar infarct. Baseline MRI was also rated for presence of lacunes, WMH, CMB, and basal ganglia PVS, all according to the Standards for Reporting Vascular Changes on Neuroimaging (STRIVE) for reporting studies in cSVD. 2 These MRI markers were summed in an ordinal “total cSVD burden score” (with a range of 0–4) by counting presence of each of these four MRI features.12,14 Definition of MRI markers and details on cSVD burden score is in the online-only Supplemental Data.
Three years after stroke, gait disturbances were objectively measured by TUG test and subjectively assessed by self-reported SIS mobility domain score. The TUG test measures time (in seconds) to get up from a chair, walk 3 m, turn around, walk back, and sit back down. 18 The SIS is a stroke-specific, self-reported assessment questionnaire with eight different domains, including mobility. The SIS mobility domain score (range 0–100) was calculated from nine mobility items by using a validated logarithm. 19 A low SIS mobility domain score reflects a high impact on health-related quality of life. Functional disability outcome was assessed by mRS at three years after stroke.
We tested associations adjusted for key variables (age, gender, vascular risk factors, and stroke severity (NIHSS)) by linear regression analysis (log-transformed TUG, SIS) and by logistic regression analysis (mRS). Statistical significance was set at p < 0.05 (2-tailed).
Results
Patients and baseline characteristics
In total, 200 of 264 minor stroke patients had a three-year clinical follow-up (Figure 1). For a detailed description, see the online-only Supplemental Data. Baseline clinical and MRI characteristics are shown in Table 1.
Patient recruitment data of MSS II 3-year follow-up study. MSS II: Mild Stroke Study-2. Characteristics of 200 minor stroke patients who had a three-year clinical follow-up p < 0.05. Lacunar stroke versus non-lacunar stroke patients. SD: standard deviation; BG: basal ganglia; PVS: perivascular spaces; WMH: white matter hyperintensities; cSVD: cerebral small vessel disease.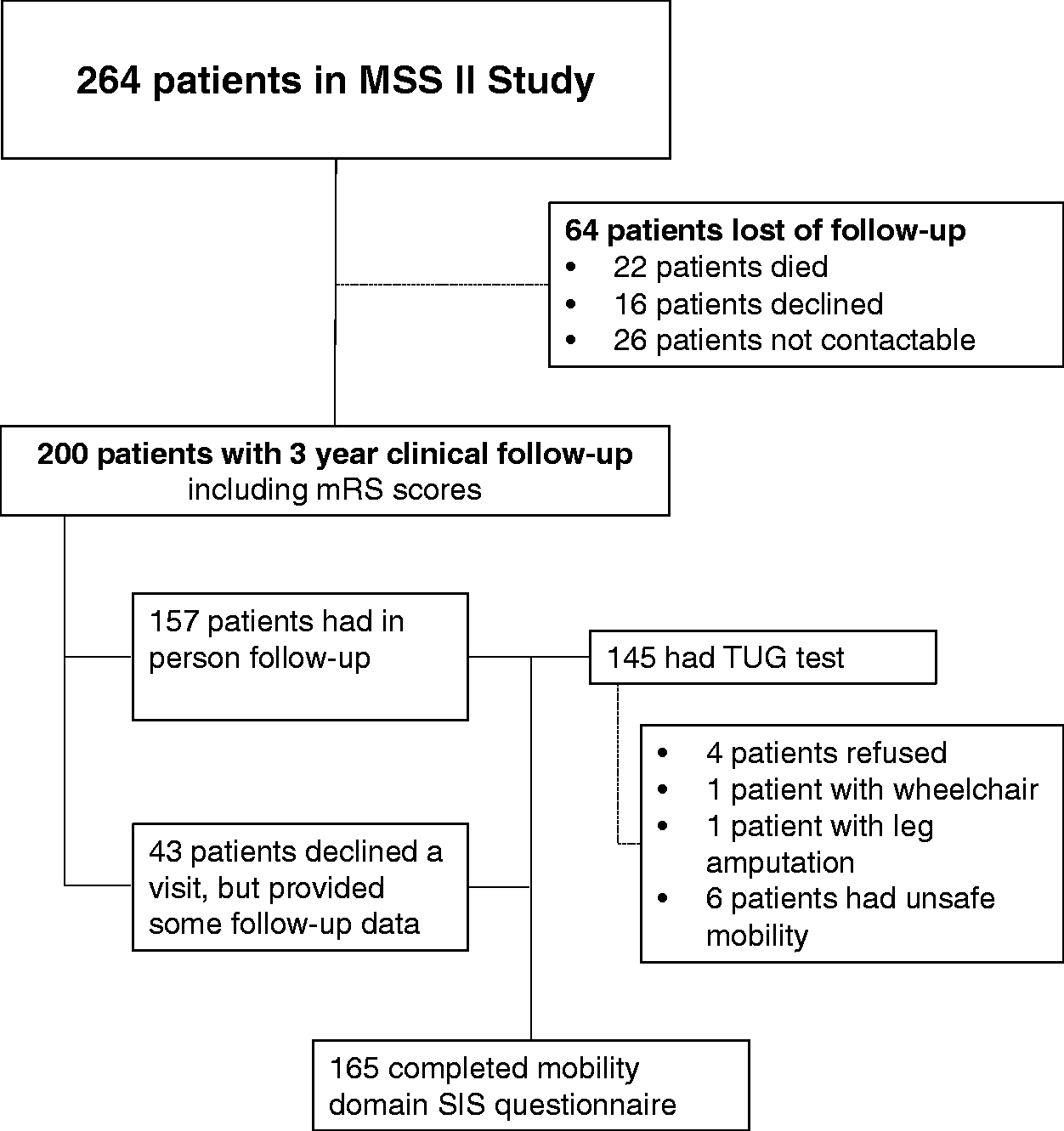
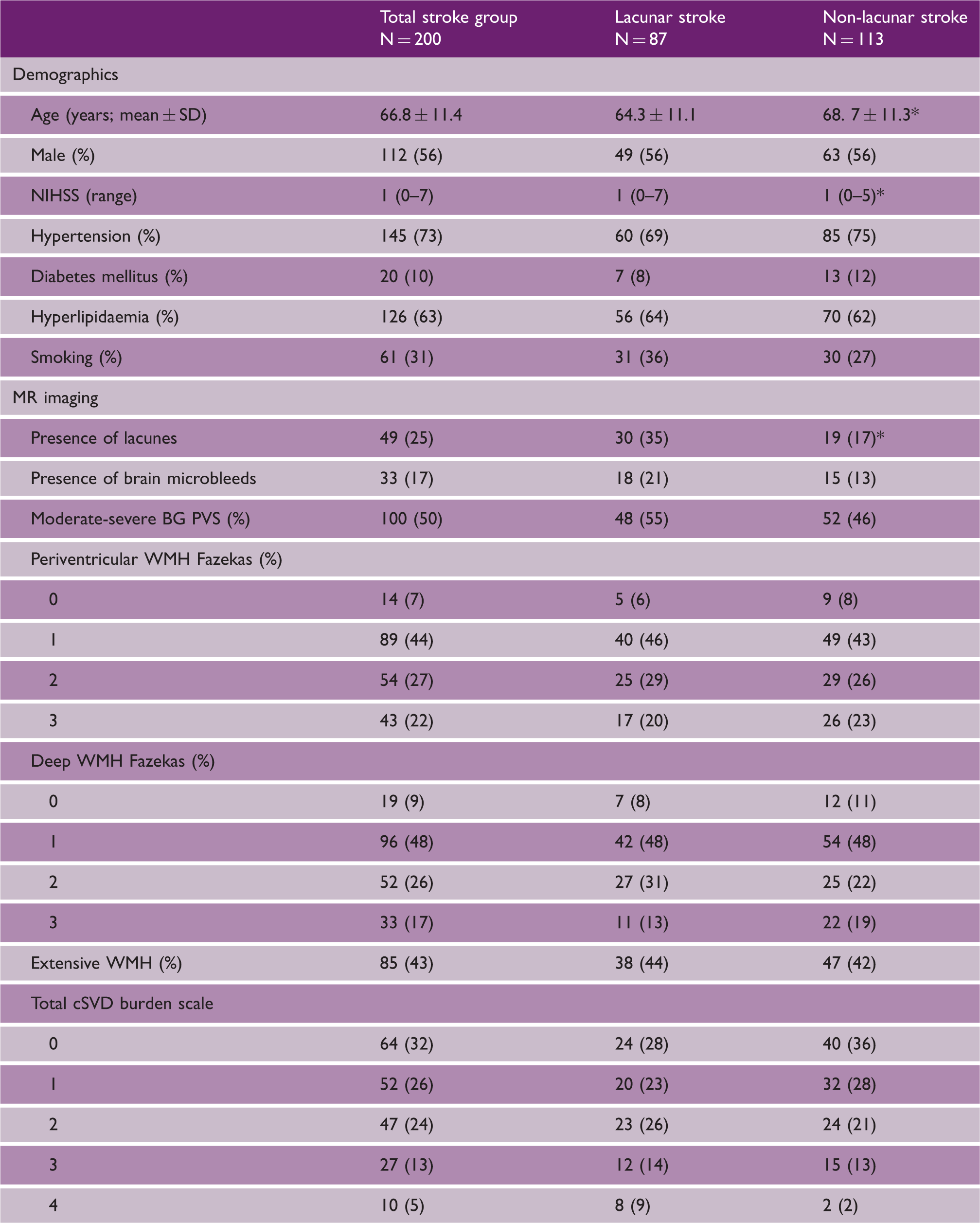
Association between total cSVD burden and (log-transformed) TUG test
Association between total cSVD burden and log-transformed timed-up-and-go test in minor stroke patients
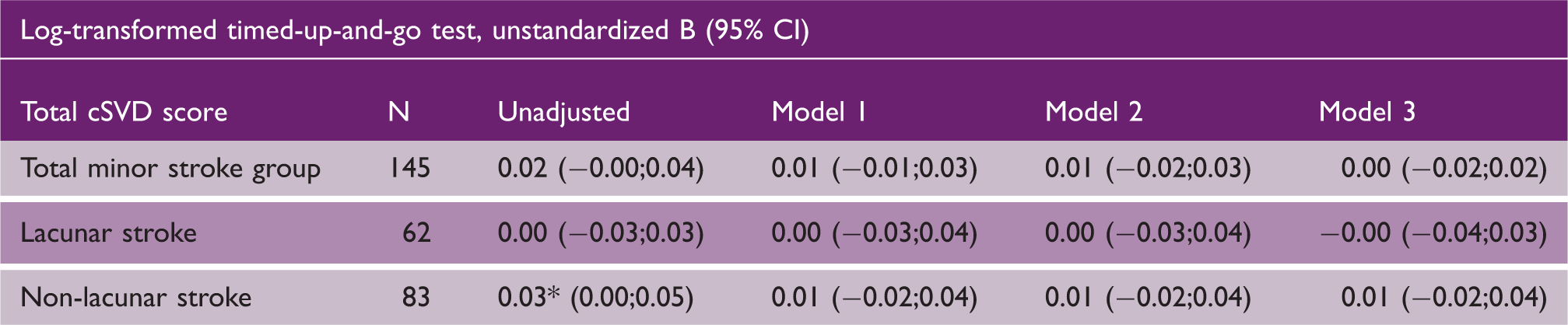
Note: Model 1 adjusted for age and gender. Model 2 adjusted for age, gender and vascular risk factors (diabetes mellitus, hypertension, hypercholesterolemia and smoking). Model 3 adjusted for age, gender, vascular risk factors and NIHSS. Multiple hierarchical linear regression analysis, *p < 0.05.
cSVD: cerebral small vessel disease: CI: confidence interval.
Association between total cSVD burden and self-reported SIS mobility domain
Association between total cSVD burden and stroke impact scale mobility domain score in minor stroke patients
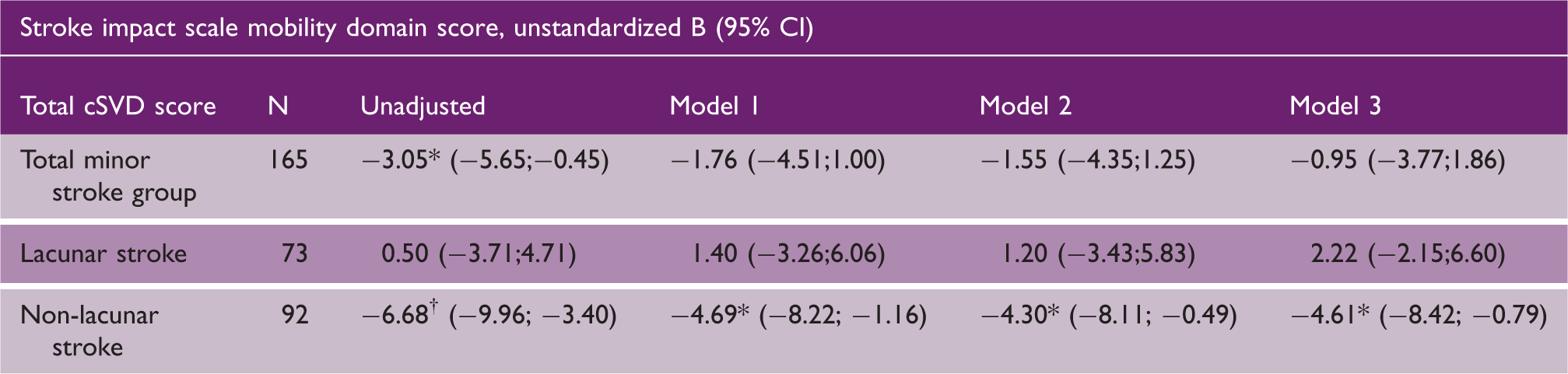
Note: Model 1 adjusted for age and gender. Model 2 adjusted for age, gender and vascular risk factors (diabetes mellitus, hypertension, hypercholesterolemia and smoking). Model 3 adjusted for age, gender, vascular risk factors and NIHSS. Multiple hierarchical linear regression analysis, *p < 0.05, †p < 0.01.
cSVD: cerebral small vessel disease; CI: confidence interval.
There was a strong correlation between objectively measured TUG test and subjectively assessed SIS mobility domain score: correlation coefficient −0.58 (n = 142; p < 0.00).
Association between total cSVD burden and mRS
At three years of follow-up, 200 patients had mRS measurement with a median mRS of 1 (range 0–5). One hundred seventy-four patients (87%) were functionally independent (mRS ≤ 2) and 26 patients (13%) were functionally dependent (mRS 3–5). The total cSVD burden was associated with mRS > 2 in the total stroke group (OR 1.47; 95% CI: 1.05–2.06; p < 0.05). However, this association lost significance after adjusting for age, gender, vascular risk factors, and baseline stroke severity (OR 1.21; 95% CI 0.79–1.87, p = 0.38). No association was found between total cSVD burden and mRS in non-lacunar stroke patients (n = 113), nor in the lacunar stroke group (n = 87).
Discussion
Our study did not find an association between total cSVD burden and objectively measured gait disturbances, nor with subjectively assessed gait impairment in the total minor stroke group three years after stroke. However, in non-lacunar stroke patients, the total cSVD burden score was associated with lower self-reported SIS mobility domain scores three years after stroke, independent of stroke severity.
Previous neuroimaging studies have consistently demonstrated that individual MRI markers of cSVD are related to gait impairment,4,5,9,10,20 mainly in community-dwelling subjects rather than stroke patients. The Radboud University Nijmegen Diffusion Tensor and Magnetic Resonance Imaging Cohort (RUNDMC) study, which included patients with clinical cSVD (including stroke and gait impairment), showed that WMH, number of lacunes, and CMB were associated with concurrent gait dysfunction, including impaired gait velocity and a prolonged TUG test.5,9 The leukoaraiosis and disability (LADIS) study showed that WMH were associated with concurrent deterioration of gait function, especially at older age.4,10
Although these studies have shown that individual MRI markers of cSVD are independently linked to concurrent gait impairment, these cSVD markers rarely occur in isolation on MRI. There is increasing evidence that a total cSVD MRI burden score, which summarizes individual cSVD markers in a compound scale, might better reflect the global overall effect of cSVD on the brain.11–14,20,21 A study 22 in older subjects with vascular risk factors showed that concurrent gait function, measured by Unified Parkinson's Disease Rating Scale, 23 was associated with total cSVD burden. This was the first study looking at concurrent gait function and overall cSVD burden (but not after stroke), although the cSVD burden score was incomplete as they did not include PVS. Recently, a study 20 in community-dwelling older subjects found that WMH and the total cSVD score were independently associated with slower gait speed assessed by a six-meter walk test.
Contrary to these positive results in larger studies, mostly in subjects without stroke, we did not find any association between total cSVD burden and gait disturbances, measured by TUG test. There are several differences between studies. Our sample size was smaller, although the direction of the effect was consistent with an association between total cSVD burden and slowing gait. Second, we used the TUG test, and did not perform functional gait analysis, which would be a more quantitative and more sensitive method in assessing (subtle) mobility impairment. However, the TUG test is a valid outcome measurement that assesses basic functional mobility in routine practice, 18 is representative of daily activities, also in stroke patients 24 and is conceptually similar to the six-meter walk. Third, our minor stroke population was somewhat younger compared to several previous studies with community-dwelling subjects.4,10,20,22 Last, the LADIS4,10 and RUNDMC5,9 study selected patients based on presence of WMH. This could have led to a higher WMH burden in these cohorts compared to ours. However, as the measurement methods were different (volumetric versus visual scales), it is difficult to compare. In our cohort, 43% had extensive WMH (represented by 1 point in cSVD burden score), while 54% had mild WMH and only 3% had no WMH at all.
Subjectively measured mobility impairment was associated with the total cSVD burden score in non-lacunar stroke patients. The self-reported SIS mobility domain score was available for more patients and therefore increased power. The SIS is a valid and reliable comprehensive stroke-specific health outcome measurement, especially in minor strokes 19 and captures the impact of stroke on multiple domains, including mobility. 25 We found a strong correlation between TUG test and SIS mobility domain score, meaning that perceived mobility disturbances are relevant and furthermore, mobility is a broader function than what is measured by the TUG test. The associations we found were present in non-lacunar stroke patients, but not in lacunar stroke patients. A possible explanation could be that stroke subtype is a moderator of the effect of cSVD on outcome; a non-lacunar ischemic stroke could make the brain more vulnerable to the impact of cSVD. Furthermore, as we did not find an association between the total cSVD burden score and mRS three years after minor stroke, it should be noted that any association between the total cSVD burden score and mobility impairment is not simply because of a co-association with worse functional stroke outcome after minor stroke. Therefore, our results indicate that minor non-lacunar stroke patients with a high total cSVD load on MRI are prone to report more often subjective mobility disturbances three years after minor stroke, independent of stroke severity.
Our study has several limitations. Although this is the first study which investigates if the total cSVD burden score could be a possible marker in predicting gait impairment in patients with minor stroke, our sample is small for a common disease like stroke. A larger study is warranted to confirm if the association between gait speed and total cSVD burden reaches significance in a larger sample. We measured gait function at three years after stroke onset, so it could be that significant disabled patients may not have participated in clinical follow-up, leading to selection bias. We did not have gait function at baseline, so we could not explore gait decline, nor correct for pre-existing gait disturbances.
A strength of our study is that it was prospective, systematic and assessments were blinded. We used standardized international consensus criteria 2 to describe cSVD imaging findings and provide predictive rather than just concurrent mobility data. Furthermore, we used an overall cSVD-related brain damage score. The total cSVD burden score is a pragmatic and simple visual score, which gives a more comprehensive view of the impact of cSVD on the brain. 12 Patients with minor (non-disabling) stroke are an ideal stroke subgroup for exploring the relationship between cSVD burden and gait disturbances, as the impact of stroke itself on outcome measurements is probably reduced.
Summary
The total cSVD burden was not associated with objectively measured gait impairment, nor with subjectively assessed gait disturbances in minor stroke patients, nor in lacunar stroke patients. As patients with non-lacunar stroke and a higher total cSVD burden report more often subjective mobility impairment three years after stroke, the total cSVD burden score is a possible marker to identify non-lacunar stroke patients at risk for subjective gait impairment. These findings should be confirmed in a larger study.
Footnotes
Acknowledgements
We thank the patients and their families, and the staff of the Brain Research Imaging Centre, Edinburgh, where MRI scanning was performed.
Authors’ contributions
CMJL designed study, performed statistical analysis, drafted and edited the article and prepared the final version. CM, VC and SDJM recruited and assessed the patients. FC helped with statistical analysis. MSD identified patients and participated in expert panel of original study. JS and RvO provided critical input and edited the article. JMW conceived and supervised the original project, provided input and direction and edited the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Mild Stroke Study-2 follow up study at three years was funded by Chest Heart Stroke Scotland. The original MSS-2 study was funded by the Wellcome Trust (ref. 088134/Z/09/A) and Row Fogo Charitable Trust. The imaging was performed at the Brain Research Imaging Centre Edinburgh, which is supported by the SINAPSE collaboration and the Chief Scientist Office of the Scottish Government (![]() ). The work was supported by European Union Horizon 2020 (EU H2020), PHC-03-15, project No 666881, 'SVDs@Target', and the Fondation Leducq Transatlantic Network of Excellence for Study of Perivascular Spaces in Small Vessel Disease, ref no. 16 CVD 05. The work reflects the views of the authors and not of the funders. CMJL was supported by the Dutch Alzheimer Foundation and VC holds a NHS Research Scotland Fellowship. The work was performed in the Edinburgh Dementia Research Centre in the UK Dementia Research Initiative.
). The work was supported by European Union Horizon 2020 (EU H2020), PHC-03-15, project No 666881, 'SVDs@Target', and the Fondation Leducq Transatlantic Network of Excellence for Study of Perivascular Spaces in Small Vessel Disease, ref no. 16 CVD 05. The work reflects the views of the authors and not of the funders. CMJL was supported by the Dutch Alzheimer Foundation and VC holds a NHS Research Scotland Fellowship. The work was performed in the Edinburgh Dementia Research Centre in the UK Dementia Research Initiative.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
