Abstract
Background
Idarucizumab is a monoclonal antibody fragment with high affinity for dabigatran that reverses its anticoagulant effects within minutes. It may exhibit the potential for patients under dabigatran therapy suffering ischemic stroke to regain eligibility for thrombolysis with rt-PA and may inhibit lesion growth in patients with intracerebral hemorrhage on dabigatran.
Aims
To provide insights into the clinical use of idarucizumab in patients under effective dabigatran anticoagulation presenting with signs of ischemic stroke or intracranial hemorrhage.
Methods
Retrospective data collected from German neurological/neurosurgical departments administering idarucizumab following product launch from January to August 2016 were used.
Results
Thirty-one patients presenting with signs of stroke received idarucizumab in 22 stroke centers. Nineteen patients treated with dabigatran presented with ischemic stroke and 12 patients suffered from intracranial bleeding. In patients receiving rt-PA thrombolysis following idarucizumab, 79% benefitted from i.v. thrombolysis with a median improvement of five points in NIHSS. No bleeding complications occurred. Hematoma growth was observed in 2 out of 12 patients with intracranial hemorrhage. The outcome was favorable with a median NIHSS improvement of 5.5 points and mRS 0–3 in 67%. Overall, mortality was low with 6.5% (one patient in each group).
Conclusion
Administration of rt-PA after reversing dabigatran activity with idarucizumab in case of ischemic stroke is feasible, easy to manage, effective, and appears to be safe. In dabigatran-associated intracranial hemorrhage, idarucizumab has the potential to prevent hematoma growth and improve outcome. Idarucizumab represents a new therapeutic option for patients under dabigatran treatment presenting with ischemic stroke or intracranial hemorrhage.
Introduction
Neurologists use the direct thrombin inhibitor dabigatran most frequently in secondary stroke prevention in patients with atrial fibrillation. The RE-LY study1–3 showed superiority of dabigatran 150 mg bid for the prevention of ischemic stroke compared to warfarin, and in the subgroup of patients with prior TIA or stroke, dabigatran was non-inferior to warfarin. 4 Three other non-vitamin-K-antagonizing oral anticoagulants (NOACs) are approved for stroke prevention in patients with atrial fibrillation.5–7 As a group, NOACs have lower rates of bleeding complications in comparison to warfarin, in particular when considering intracranial hemorrhage. 8 A major concern for the use of oral anticoagulation is the lack of specific and rapidly acting reversal agents for vitamin-K antagonists or NOACs.
This has changed for dabigatran with the approval of idarucizumab, a humanized Fab fragment of a monoclonal antibody which binds specifically dabigatran with very high affinity. It is indicated in dabigatran-treated patients with life-threatening or uncontrolled bleeding or those in need of urgent surgery or intervention.9,10 To date, the use of idarucizumab in patients with acute ischemic stroke who qualify for systemic thrombolysis with rt-PA is limited to single case reports.11–15
Aims
This report summarizes national experience with idarucizumab administration in dabigatran-treated patients suffering from acute ischemic stroke or intracranial hemorrhage in Germany following market launch in January 2016, for the first eight months until August 2016. We provide data about feasibility, potential efficacy and safety of idarucizumab application in these two important patient cohorts.
Methods
We retrospectively collected data from all German neurological/neurosurgical departments who used idarucizumab for the reversal of dabigatran for the first eight months from when it became available in the clinic in January 2016. The anonymized patient data collected included baseline characteristics, clinical findings, laboratory and coagulation parameters upon admission, imaging diagnostics, clinical course and status upon discharge from hospital and further co-medication. In case of intracranial hemorrhage, we collected information on hematoma growth with clinical deterioration, adverse events of idarucizumab application and outcome measured by modified Rankin score (mRS) pre- and post-treatment. For ischemic stroke, we considered NIHSS, mRS, bleeding and thrombotic complications, clinical outcome and further co-medication. All data were retrospectively collected, anonymized and transferred for analysis. A few previously reported single case reports were included in this analysis as these cases are still rare and were considered important for this multicenter collection of patients.11–13,16 The study was approved by the independent Ethics Committee (IEC), Medical Faculty of Essen University.
Results
Our retrospective study retrieved 31 patients presenting with either intracranial hemorrhage or ischemic stroke from German Stroke Units, who received idarucizumab between January and August 2016.
Ischemic stroke patients
Idarucizumab for reversal of Dabigatran activity prior to iv. rt-PA treatment of acute ischemic stroke

OAC: oral anticoagulation; NVAF: non-valvular atrial fibrillation; CrCl: creatinine clearence; rt-PA: recombinant tissue type plasminogen activator (alteplase); ATT: antithrombotic treatment; VKA: vitamin-K-antagonist; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; aPTT: activated partial thromboplastin time; TT: thrombin time; dTT: diluted thrombin time;
CMP (Cardiomyopathy); ESUS (Embolic Stroke of Undetermined Source) †: off-label indication
Including mechanical thrombectomy.
The majority of patients with signs of ischemic stroke had a CrCl above 50 ml/min. Anticoagulation was restarted between 24 h and 10 days post-ischemia depending on infarct size in all surviving patients. In total, 73.7% of patients (14 out of 19) were restarted on NOAC (dabigatran n = 12, apixaban n = 1, edoxaban n = 1).
Patients with intracranial bleeding
Idarucizumab for reversal of Dabigatran activity in patients with intracranial bleeding
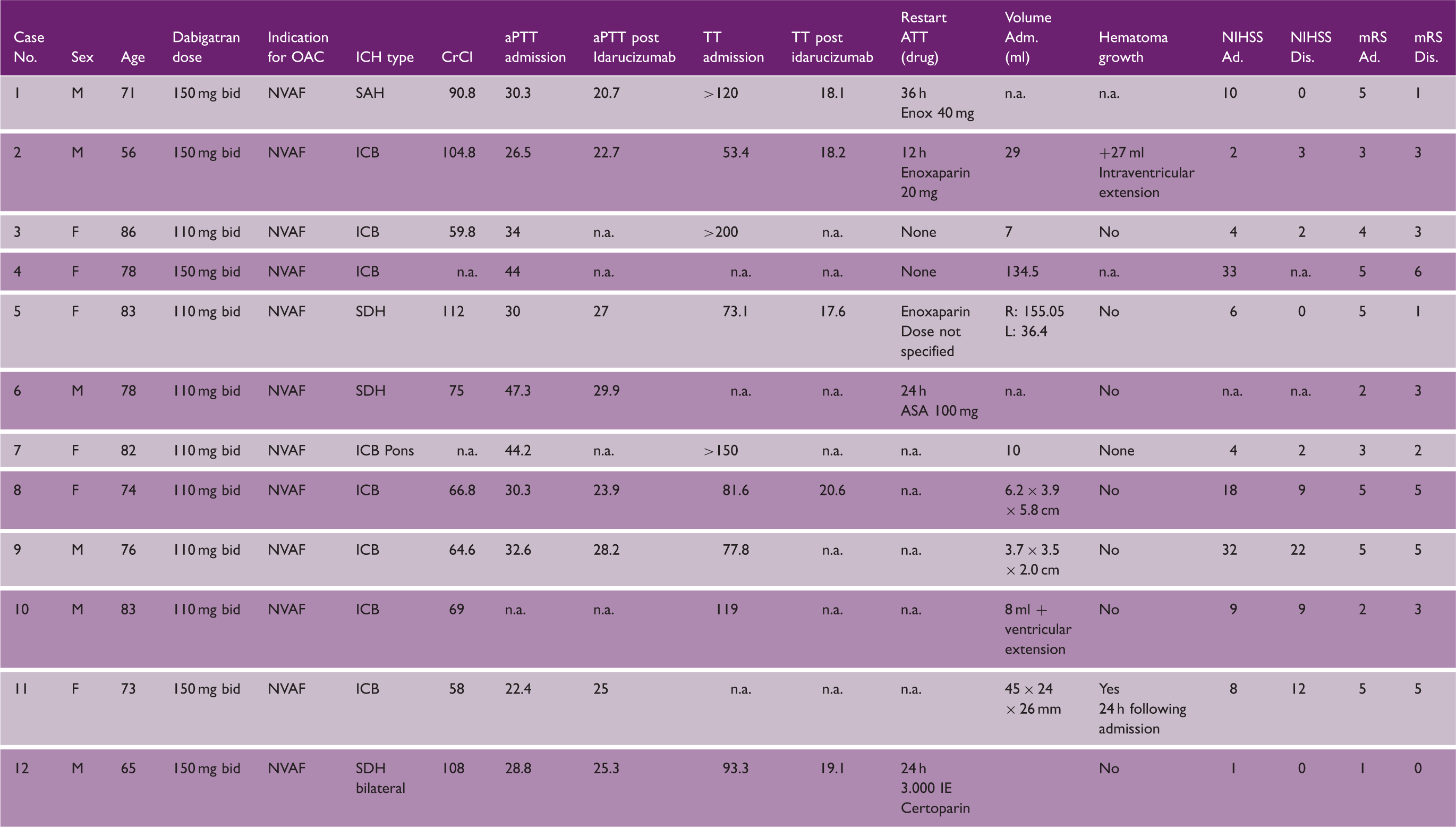
OAC: oral anticoagulation; CrCl: creatinine clearence; rt-PA: recombinant tissue type plasminogen activator (alteplase); ATT: antithrombotic treatment; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; aPTT: activated partial thromboplastin time; TT: thrombin time; dTT: diluted thrombin time.
Discussion
Thrombolysis with rt-PA is well established and the recommended therapy for acute ischemic stroke according to guidelines. Lack of antidotes for patients on effective oral anticoagulants has to date excluded patients with acute ischemic stroke, who would otherwise have been eligible for intravenous thrombolysis with rt-PA, due to the potential risk of bleeding. Prothrombin complex concentrates (PCC) are recommended by cardiology and neurology guidelines for the treatment of bleeding complications under oral anticoagulation with VKAs and NOACs; however, the efficacy of this therapy has not been established in patients with intracranial hemorrhage.17–19 In addition, PCC has partial prothrombotic effects. A specific antidote is now available for the reversal of the anticoagulant effect of dabigatran. Idarucizumab is a humanized monoclonal antibody fragment that binds dabigatran ∼350 times more avidly than dabigatran binds thrombin. The standard dose of 5 g immediately reverses the anticoagulant activity of dabigatran.9,20 Thrombolysis in ischemic stroke is considered an urgent procedure and intracranial bleeding is usually life-threatening. Idarucizumab can be applied on-label for both disease entities. Here, we report the first case collection from clinical experience with idarucizumab for two important neurological conditions: ischemic stroke and intracranial hemorrhage under effective oral anticoagulation.
Ischemic stroke patients
The administration of rt-PA in ischemic stroke requires absence of effective anticoagulation proven by appropriate laboratory tests. For patients on dabigatran, aPTT is recommended as an appropriate test to provide qualitative rather than quantitative information about the presence and anticoagulant activity of dabigatran. We suggest taking a blood sample for aPTT immediately after idarucizumab infusion to ensure an on-label application of rt-PA. Thrombolysis with rt-PA may be started without delay and only terminated if one of the coagulation parameters proves still to be prolonged when analysis of the samples has been performed (see Expert Opinion in this journal issue). Thrombolysis with rt-PA was highly effective in our patient series with a median NIH-SS improvement of 5 points and a median mRS of 1 point at discharge. This improvement was achieved without a bleeding event. One thrombotic adverse event (bilateral pulmonary embolism) leading to death occurred five days after idarucizumab/rt-PA infusion. The pulmonary embolism appeared most probably due to the fact that effective anticoagulation was not restarted in this patient. Idarucizumab infusion was generally well tolerated and anticoagulation with dabigatran could be re-established after 24 h in the majority (66.7%) of patients.
Patients with intracranial bleeding
Five out of 12 patients with primary intracranial bleeding were taking high-dose dabigatran (150 mg twice daily). However, hematoma growth could only be documented in two patients. This is an important finding, as previous data from the RE-LY trial showed that the incidence of ICH was significantly lower in patients on dabigatran; however, mortality was similar under VKA or dabigatran treatment. 21 In addition, data from a subgroup of patients with intracranial hemorrhage were recently presented from the interim analysis of the RE-VERSE AD trial, focusing on the ability of idarucizumab to reverse the anticoagulant effects of dabigatran in this patient group. The analysis showed a rapid reversal of the anticoagulant activity and a mortality rate similar to patients with non-ICH included in the RE-VERSE AD trial. 22
The mortality after idarucizumab treatment was 6.5% in this case collection of patients with hemorrhagic stroke as compared to 35% (D150 mg) to 41% (D110 mg) mortality in the RE-LY trial. 21 A recent report by Purrucker and colleagues 23 summarizes 61 consecutive patients with non-traumatic NOAC-associated intracerebral bleeding. In this case cohort, the mortality rate was 28% and hematoma expansion was observed in 38%.
Multinational registries are necessary to quantify effects on bleeding complications, hematoma growth, thrombotic events, outcome and mortality in cases of ischemic stroke and intracranial bleeding from patients treated with dabigatran receiving idarucizumab or other NOACs and anti-factor Xa inhibitor-antidotes when they become available. In Germany, the RASUNOA prime (registry of acute stroke under novel oral anticoagulants, NCT02533960) study was started in 2015 and plans to enroll 4000 patients with atrial fibrillation receiving vitamin K antagonists, NOAC or no therapy and suffering from ischemic or hemorrhagic stroke. They will be monitored for acute treatment and hematoma characteristics. With the case series reported here, we hope to encourage activities in other countries to evaluate and further develop guidelines for the treatment of cerebrovascular complications under NOAC therapy.
In summary, our case collection illustrates:
Intravenous thrombolysis with rt-PA after antagonizing dabigatran with idarucizumab is feasible, easy to manage in an emergency room or stroke-unit setting, appears to be safe and should therefore be considered in cases where ischemic stroke occurs in patients under effective dabigatran therapy. Idarucizumab application appears to prevent hematoma growth and might reduce mortality and/or improve outcome in cases of hemorrhagic stroke or intracranial hemorrhage and should therefore be given in all patients presenting with intracranial bleeding and effective dabigatran anticoagulation.
Clearly, this study has limitations with regard to the small number of patients included in the analysis and the retrospective nature of the study. However, this case collection of administration of idarucizumab in acute neurological emergencies displays the largest real world experience available to date. Thus, this case collection adds important clinical information until further robust data from large prospective registries will be available.
Footnotes
Acknowledgement
We thank Joanne van Ryn, Boehringer Ingelheim, for critically reading the manuscript.
Authors’ contributions
PK, CCE, H-CD and MG Took part in the study design, data collection, data analysis, data interpretation, and writing. YA, KA, JB, HC, MD, SE, KG, CGH, AH, VH, AK, PK, AL, WM, MO, SP, JP, PAR, JR, RR, NS, AS, RS, RJS, KS, and RW made substantial contribution to the study by local acquisition of data and interpretation of data, critical reading of the manuscript with critical revision and approved the version to be published.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P Kermer received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbvie, Bayer Vital, Biogen, BMS, Boehringer Ingelheim, Daiichi-Sankyo, Genzyme, Novartis, Pfizer, TEVA and UCB.
CC Eschenfelder is an employee of Boehringer Ingelheim.
H-C Diener received honoraria in the last two years for participation in clinical trials, contribution to advisory boards or oral presentations from: Bayer Vital, BMS, Boehringer Ingelheim (significant), Covidien, Daiichi-Sankyo, Medtronic, Pfizer, Sanofi-Aventis and WebMD Global. Financial support for research projects was provided in the last two years by Boehringer Ingelheim. The Department of Neurology at the University Duisburg-Essen received research grants from the German Research Council (DFG), German Ministry of Education and Research (BMBF), European Union, NIH, Bertelsmann Foundation and Heinz-Nixdorf Foundation.
Martin Grond received honoraria for participation in clinical trials, advisory boards or speakers’ bureau for Boehringer Ingelheim, Bayer Vital, Bristol-Myers Squibb, Pfizer, Daichii-Sankyo, Medtronics and Sanofi Aventis.
Yasser Abdalla reports no conflict of interests.
Katharina Althaus reports no conflict of interests.
Jörg Berrouschot received in the last two years honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from Bayer Vital, Bristol-Myers Squibb, Boehringer Ingelheim, Daiichi-Sankyo, Medtronic, Pfizer, Sanofi-Aventis.
Hakan Cangür received honoraria for oral presentations and participation in advisory boards from Boehringer Ingelheim, Bayer and Bristol-Myers Squibb.
Michael Daffertshofer reports no conflict of interests.
Sebastian Edelbusch received travel support for participation in national medical update sessions from Boehringer Ingelheim.
Klaus Gröschel received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from Bayer Vital, Bristol-Myers Squibb, Boehringer Ingelheim, Daiichi-Sankyo and Pfizer.
Claus G Haase reports no conflict of interests.
Andreas Harloff has received honoraria for oral presentations by Bayer Health Care, Boehringer Ingelheim, Bristol-Myers Squibb, Medtronic, Pfizer and Sanofi-Aventis.
Valentin Held reports no conflict of interests.
Andreas Kauert received honoraria for oral presentations from Bayer Vital, Novartis, and Pfizer.
Peter Kraft received honoraria for oral presentations, advisory boards and travel grants from Boehringer Ingelheim, Bayer, Daiichi-Sankyo, and Bristol-Myers Squibb.
Arne Lenz received honoraria for oral presentations from Boehringer Ingelheim.
Wolfgang Müllges received honoraries for lectures and travel expenses from Boehringer Ingelheim, Bayer, KoMed GbR, diverse hospitals, German Society of Neurology, Antibiotic stewardship programme and Travel expenses from German Society of Neurocritical Care, German Interdisciplinary Society of Intensive Care, German Soc. of Neurology, German Soc. of Anesthesiology, German Soc. of Pneumology, German Soc. of Organ Transplant, German Soc of Clin. Neurophysiology.
Mark Obermann received scientific support, travel support and/or honoraria from Biogen Idec, Novartis, Sanofi-Aventis, Genzyme, Pfizer, Teva and Heel. He received research grants from Allergan, Electrocore, Heel, and the German Ministry for Education and Research (BMBF).
Someieh Partowi reports no conflict of interests.
Jan Purrucker received travel support and personal fees from Pfizer and Boehringer Ingelheim.
Peter A Ringleb received lecture-fees and travel-expenses from Bayer, Boehringer Ingelheim, Daiichi Sankyo, and Pfizer and compensation for advisory board participation from Boehringer Ingelheim, Covidien, and Daiichi Sankyo.
Joachim Röther received honoraria for participation in clinical trials from Astra Zeneca and Servier, contribution to advisory boards or oral presentations from Sanofi Aventis, Pfizer, Lundbeck, Boehringer Ingelheim. Bristol-Myers Squibb, Bayer Vital and as advisor from DAK – Deutsche Allgemeine Krankenversicherung.
Raluca Rossi reports no conflict of interests.
Niklas Schäfer received travel fees and honoraria for oral presentation from Roche.
Andreas Schneider reports no conflict of interests.
Ramona Schuppner reports no conflict of interests.
Rüdiger J Seitz reports no conflict of interests.
Kristina Szabo reports no conflict of interests.
Robert Wruck reports no conflict of interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
