Abstract
Background
The risk of recurrence following ischemic stroke or transient ischemic attack is highest immediately after the event. Antiplatelet agents are effective in reducing the risk of recurrence and two agents are superior to one in the early phase after ictus.
Design
The triple antiplatelets for reducing dependency after ischemic stroke trial was an international multicenter prospective randomized open-label blinded-endpoint trial that assessed the safety and efficacy of short-term intensive antiplatelet therapy with three agents (combined aspirin, clopidogrel and dipyridamole) as compared with guideline treatment in acute ischemic stroke or transient ischemic attack. The primary outcome was stroke recurrence and its severity, measured using the modified Rankin Scale at 90 days. Secondary outcomes included recurrent vascular events, functional measures (cognition, disability, mood, quality of life), and safety (bleeding, death, serious adverse events). Data are number (%) or mean (standard deviation, SD).
Results
Recruitment ran from April 2009 to March 2016; 3096 patients were recruited from 106 sites in four countries (Denmark 1.6%, Georgia 2.7%, New Zealand 0.2%, UK 95.4%). Randomization characteristics included: age 69.0 (10.1) years; male 1945 (62.8%); time onset to randomization 29.4 (11.9) h; stroke severity (National Institutes for Health Stroke Scale) 2.8 (3.6); blood pressure 143.5 (18.2)/79.5 (11.4) mmHg; IS 2143 (69.2%), transient ischemic attack 953 (30.8%).
Conclusion
Triple antiplatelets for reducing dependency after ischemic stroke was a large trial of intensive/triple antiplatelet therapy in acute ischemic stroke and transient ischemic attack, and included participants from four predominantly Caucasian countries who were representative of patients in many western stroke services.
Keywords
Introduction
The risk of recurrence following an ischemic stroke (IS) or transient ischemic attack (TIA) is very high over the first hours and days after the event,1,2 reaching up to ∼10% by 90 days and then declining, with a total of about 40% by five years. Typically, recurrent events are more severe than index events and thus increased dependency, disability, cognitive impairment and dementia, poor quality of life, and institutionalization are all more likely.3,4
The long-term risk of recurrence can be reduced through lifestyle changes (smoking cessation and reducing weight, salt, saturated fat, and high alcohol intake) and proven, cost-effective pharmacological (antihypertensives and statins) and surgical interventions (carotid endarterectomy or stenting in large vessel disease).1–8 Oral anticoagulation is established for patients following cardioembolic stroke9–12 while the majority of patients with non-cardioembolic ischemic events (stroke and TIA) require antiplatelet therapy.13–20 In long-term secondary stroke prevention, dual antiplatelet therapy with aspirin and dipyridamole reduced recurrent events by 23% as compared with either aspirin or dipyridamole in isolation, and without increasing bleeding risk.17,19 However, the combination of aspirin and clopidogrel given chronically was not superior to either drug alone, and dual therapy resulted in increased bleeding as compared with clopidogrel alone.21–23
In contrast, more intensive antiplatelet therapy may give improved prophylaxis in the acute phase of stroke or TIA. While early antiplatelet therapy with aspirin following acute IS is effective at reducing early recurrence,15,16,24 dual antiplatelet therapy started within 72 h of onset reduced early recurrence risk more than monotherapy in a meta-analysis of small studies. 25 Moreover, the type of agents used was less important than using dual rather than monotherapy. This was supported by the findings of the large CHANCE trial; dual antiplatelet therapy with aspirin and clopidogrel was superior to aspirin alone in preventing stroke recurrence by 90 days when commenced within 24 h of minor IS or TIA.26,27 One potential benefit of using multi-antiplatelet agents is the mitigation of treatment resistance seen individually with aspirin and clopidogrel monotherapy.28–30
If dual antiplatelet therapy reduces stroke recurrence following acute IS or TIA as compared to monotherapy, then triple/intensive antiplatelet therapy with combined aspirin, clopidogrel, and dipyridamole might be even more effective, providing the risk of recurrence is high and bleeding is not excessive. Several laboratory studies and clinical trials have assessed this concept.31–35 Triple therapy in vitro was most effective in inhibiting leucocyte activation, platelet–leucocyte conjugation, and platelet aggregation.31–33 Phase I and II multiway crossover trials compared short-term administration of single, dual, and triple antiplatelets and found that aspirin and clopidogrel, with or without dipyridamole, were most effective at inhibiting platelet function in healthy volunteers and those with previous stroke or TIA.34,35 A parallel group trial comparing long-term triple antiplatelet therapy with aspirin alone in participants with stroke found that intensive treatment was feasible to administer for up to 24 months, but there was a non-significant trend to increased bleeding. 36 In addition, chronic use of triple antiplatelet therapy in patients with a very high risk of recurrence following events on dual therapy appeared to be effective in minimizing further events without increasing bleeding. 37
Based on these preclinical and clinical data showing feasibility, tolerability, apparent safety and potential efficacy for triple antiplatelet therapy, the triple antiplatelets for reducing dependency after ischemic stroke (TARDIS) trial tested the overall safety and efficacy of short-term intensive antiplatelet therapy in comparison with guideline treatment. The protocol and statistical analysis plan have been published previously.38,39
Methods
Eligibility and consent
The full inclusion and exclusion criteria are published
38
and listed in the latest version of the protocol (online Appendix A, B). Adults aged >50 years with non-cardioembolic TIA or stroke within 48 h of onset were eligible. Those whose index event was a TIA had:
Resolved limb weakness and/or dysphasia. Symptom duration between 10 min and 24 h. ABCD2 score
40
>4; and/or crescendo TIA; and/or already on dual antiplatelet therapy. IS participants had: Ongoing limb weakness, or ongoing facial weakness with resolved limb weakness; and/or dysphasia; and/or ongoing isolated hemianopia (with positive neuroimaging of ischemia affecting the corresponding occipital lobe); and duration >1 h. Resolved limb weakness; and/or dysphasia; with duration >24 h after onset (i.e. resolution between 24 h and randomization). Note: Any severity of stroke could be recruited.
Patients gave written informed consent prior to enrolment; if they lacked capacity (e.g. due to dysphasia, confusion, or reduced conscious level) then proxy consent could be obtained from a relative, carer, or legal representative, and if not available then independent physician proxy consent was permitted. Neuroimaging was essential for IS to exclude intracranial hemorrhage and a non-stroke diagnosis. Patients who were thrombolyzed for stroke were eligible for inclusion following a delay of 24 h and providing neuroimaging excluded secondary intracranial hemorrhage. Neuroimaging was performed in patients presenting with a TIA at the discretion of local clinicians.
Randomization and data collection
After obtaining informed consent, the local investigator used a secure password-protected and data-encrypted web-based randomization system to enter a participant into the trial (a system derived from that used in the ENOS trial 41 ). The system recorded baseline characteristics following validation of data using range and consistency checks, and then allocated the participant to a treatment group. To achieve optimal baseline matching between treatment groups and ensure concealment of allocation, randomization incorporated stratification (country, index event: stroke or TIA) and minimization (on baseline prognostic factors and additional criteria for TIA and stroke respectively, as highlighted in Tables 2 and 3); the minimization algorithm included a random element (simple randomization) in 5% of patients. Stratification and minimization allow for improved matching at baseline, stratification allows variable categories to be treated as trials in their own right, minimization increases statistical power, 42 and simple randomization reduces predictability. Following randomization, the local investigator was informed of the patient’s treatment allocation to triple/intensive or guideline antiplatelet therapy; randomized treatment was taken for one month, after which participants on intensive antiplatelet therapy reverted to guideline therapy. The minimization variables will be used for adjustment of the primary and secondary analyses in the main publication, 39 as recommended. 42
Investigational medicinal products
Trial interventions were given open label for one month (28 or 30 days depending on treatment pack size), a period chosen to cover the period of maximum risk of recurrence but minimize bleeding, and comprised:
Aspirin: loading dose 300 mg then 50–150 mg daily; by oral, nasogastric tube (NGT) or rectal route. Clopidogrel: loading dose 300 mg then 75 mg daily; by oral or NGT route. Dipyridamole: 225–450 mg in divided doses; by oral (as 200 mg extended release capsules twice daily, or as 25–100 mg tablets three to four times daily) or NGT (as suspension or crushed tablets three to four times daily) route. Participants were randomized to either: Intensive/triple antiplatelets (active): Combined aspirin, clopidogrel, and dipyridamole. Guideline/dual or mono antiplatelet(s): Combined aspirin and dipyridamole, or clopidogrel alone.
Throughout the trial, five protocol amendments were made covering multiple issues. The main change concerned an update of guidelines relating to antiplatelet agents after stroke. When the trial commenced, the National Institute for Health and Clinical Excellence (NICE) recommended aspirin and dipyridamole for secondary prevention, 43 which the initial protocol defined as guideline therapy. However, significant randomized evidence supporting the use of clopidogrel for secondary stroke prophylaxis, and widespread availability of generic inexpensive clopidogrel, resulted in NICE updating their guidance in 2010, this recommending that clopidogrel should be used first-line after non-cardioembolic IS. 44 Therefore, the trial protocol was updated allowing for use of either aspirin and dipyridamole, or clopidogrel monotherapy, as the guideline comparator (online Appendix C). Local investigators were able to choose whether randomization included either or both comparators, and this decision was made separately for stroke and TIA.
Outcomes
The primary and main secondary outcomes were collected centrally at day 90 by an assessor based in the national coordinating center who was blinded to treatment assignment. In the first instance, a check was made that the patient was still alive, either via the general practitioner or using electronic records; the patient and/or carer was then contacted by telephone. Where telephone contact could not be made, outcome information was collected using a postal questionnaire. The primary outcome was the occurrence and severity of recurrent stroke and TIA, with severity measured using a six-level ordered categorical scale incorporating the modified Rankin Scale (mRS 45 ): fatal stroke/severe non-fatal stroke (mRS 4 or 5)/moderate stroke (mRS 2 or 3)/mild stroke (mRS 0 or 1)/TIA/no stroke or TIA. Recurrent events and mRS were determined centrally by telephone by a trained assessor masked to treatment at day 90. Local investigators assessed and reported secondary, bleeding, and safety outcomes (including death) at days 7 and 35, and at hospital discharge (if admitted). Secondary outcomes were also collected at day 90, including bleeding, death, and measures of disability, cognition, mood, and quality of life. 38
Use of the internet
The TARDIS trial used the internet to randomize and collect data in real-time on line:
Trial website: www.tardistrial.org. Secure website for real-time data entry, validation and randomization: https://nottingham.ac.uk/∼nszwww/tardis/tardistrialdb/tardis_login.php Demo website for local investigators to practice data entry: https://www.nottingham.ac.uk/∼nszwww/tardis/tardistrialdb-demo/ (log-in: demoinv1, password: Nottingham; pin: 8888)
Funding and governance
The TARDIS trial received two phases of funding:
Start-up phase (April 2009 – September 2012): funding from the British Heart Foundation: 902 patients recruited. Main phase (October 2012 – March 2016, with funding through to end September 2017): funding from the NIHR Health Technology Assessment Programme; 2194 patients recruited. The overall aim was to recruit a total of 4100 patients.
38
The trial was supervised by a Trial Steering Committee, with support from an International Advisory Committee; run by a Trial Management Committee (part of Nottingham Stroke Trials Unit); and monitored by an independent Data Monitoring Committee (DMC). The DMC met and assessed safety and efficacy on 13 occasions during the trial; prior to 2016, they recommended that the trial should continue with recruitment with no change to the protocol. Independent experts adjudicated brain scans and serious adverse events, blinded to treatment.
Results
Trial delivery
Recruitment progressed with a start-up phase in a limited number of sites, and then expanded to a main phase (Figure 1). During the trial’s progress, a number of logistical issues arose (online Appendix C). The DMC recommended in March 2016 that the trial should stop recruitment; the TSC reviewed the same data and agreed to stop recruitment, complete follow-ups, and close the trial once data had been validated, the database locked and analyzed, and all sites closed. An explanation to the trial’s early stopping will be provided in the main publication.
Recruitment curve by time.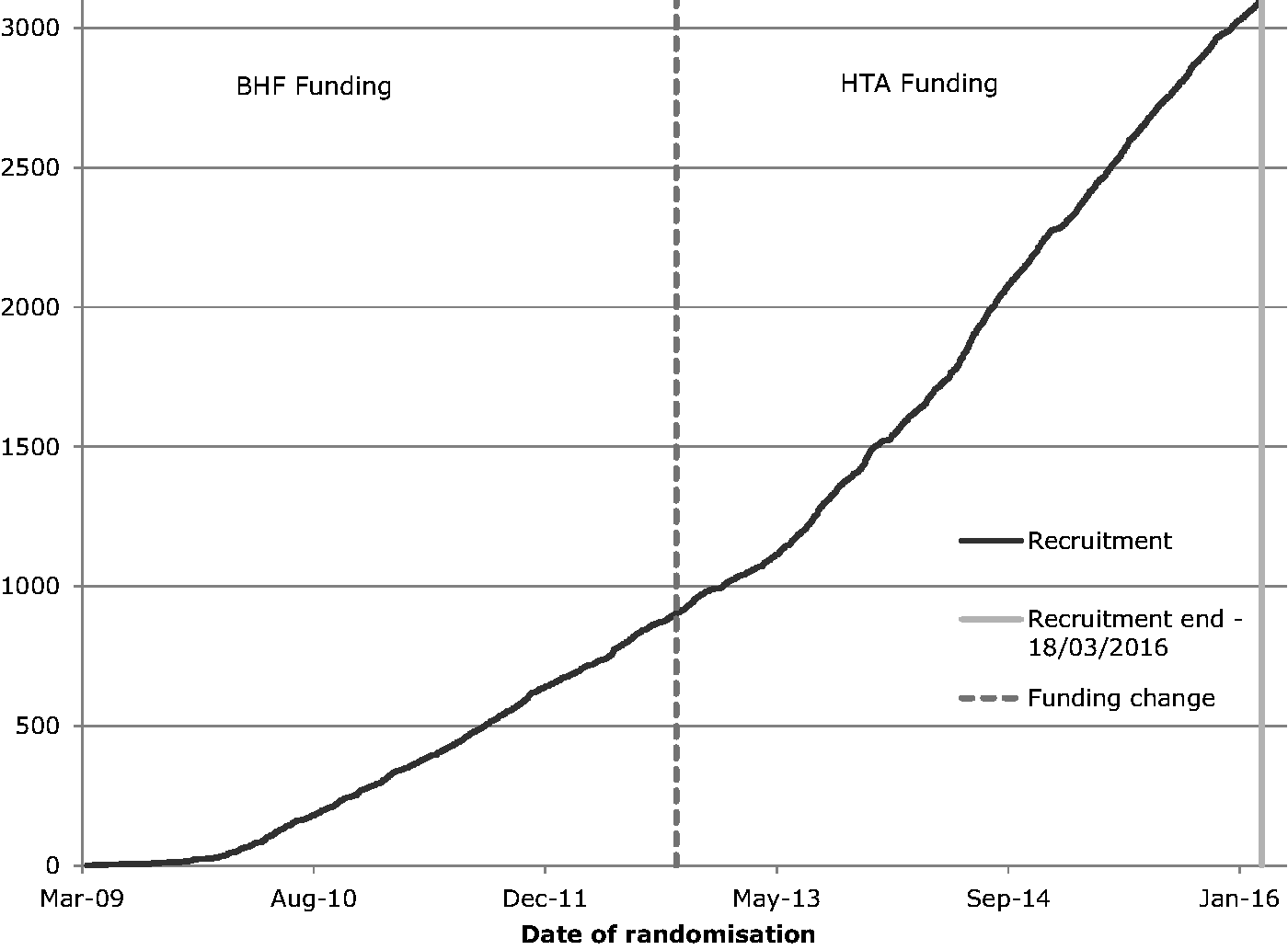
Baseline characteristics
Recruitment: Number of sites and patients by region and country
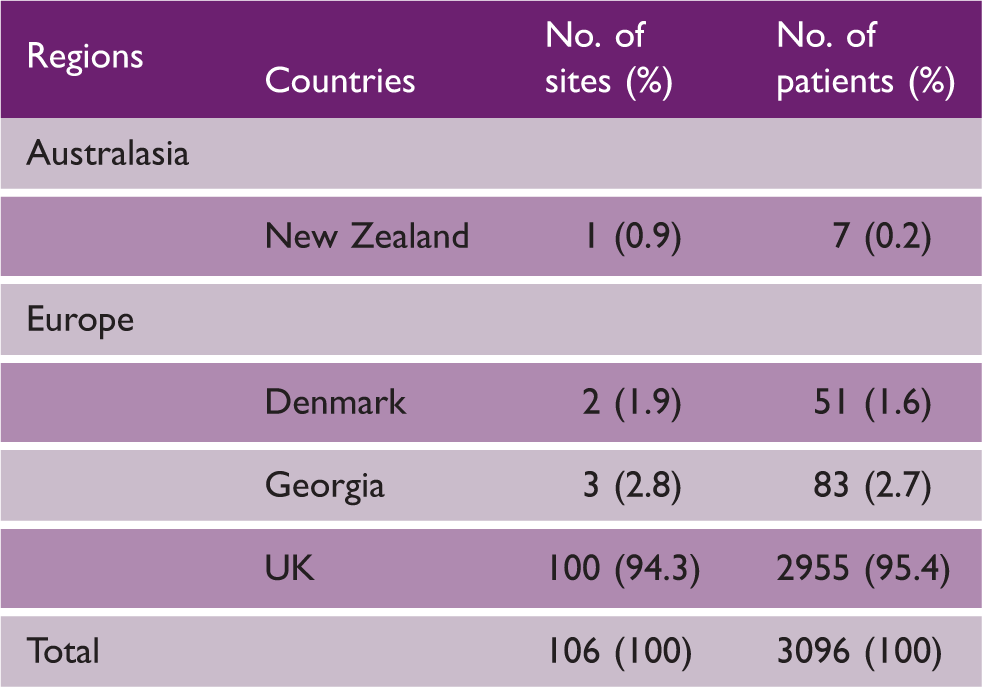
Note: Data are number and percentage.
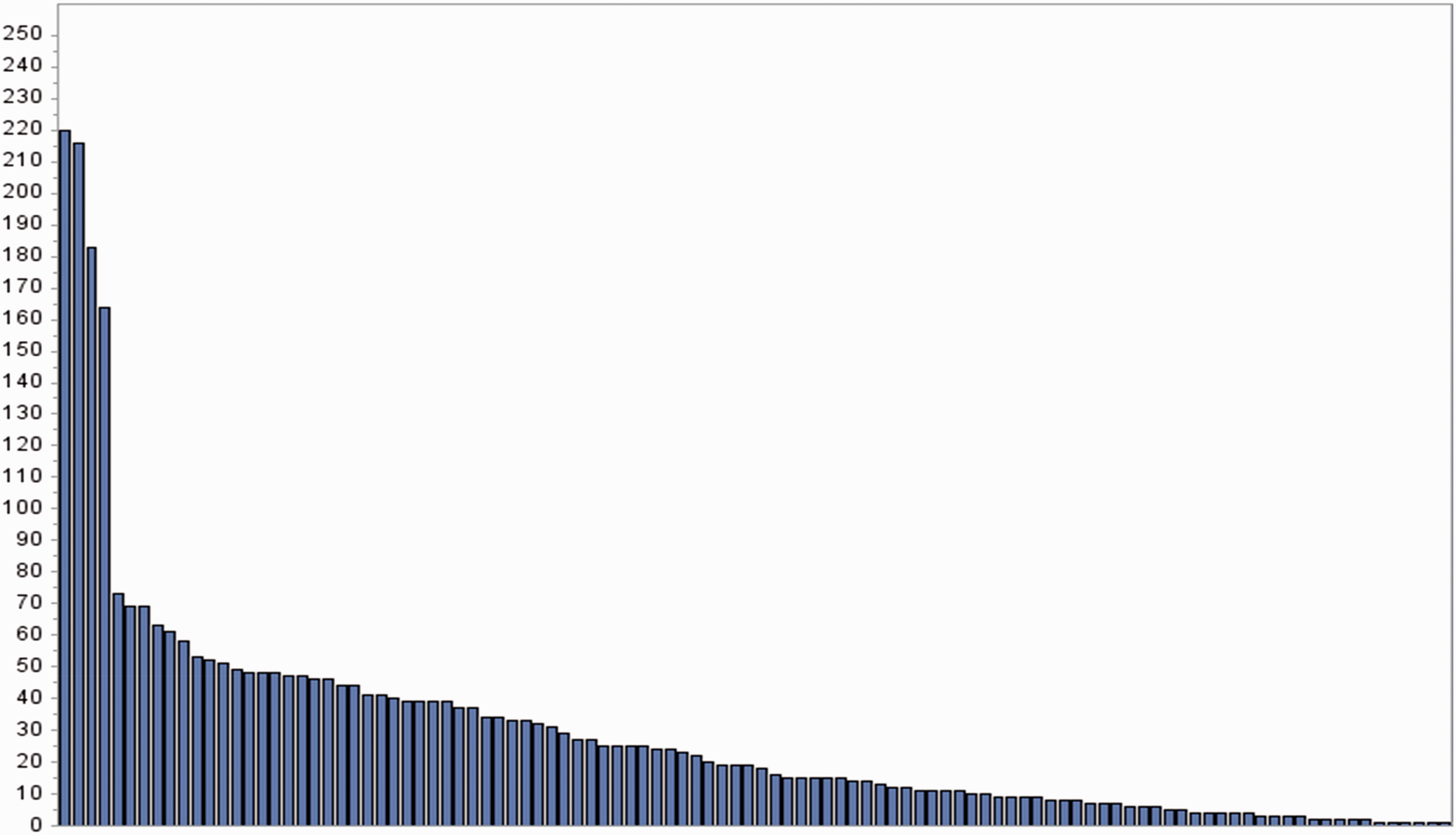
Recruitment by hospital sites.
Demographic characteristics and past medical history at time of randomization in TARDIS: Overall and by index event.

Stratification variable.
Minimisation variable.
Final hospital diagnosis may vary from diagnosis used at time of randomization
Protocol violation.
AF: atrial fibrillation; H2A: H2 antagonist; mRS: modified Rankin Scale; PPI: proton pump inhibitor; TIA: transient ischemic attack; upw: units per week.
Note: Data are number (percentage), median [interquartile range] or mean (standard deviation).
Validation is ongoing and some data may undergo minor changes prior to data lock. These data have been partially validated and may undergo minor changes prior to data lock.
Clinical characteristics at time of randomization in TARDIS: Overall and by index event

Stratification variable.
Minimisation variable.
Ischaemic patients only.
ABCD2: age, blood pressure, clinical features, duration, diabetes; BP: blood pressure; bpm: beats per minute; GCS: Glasgow coma scale; IV: intravenous; NGT: nasogastric tube; NIHSS: National Institutes of Health Stroke Scale; OCSP: Oxford community stroke project; PEG: percutaneous endoscopic gastrostomy; RIG: radiologically inserted gastrostomy; SC: subcutaneous; TIA: transient ischemic attack; TOAST: Trial of ORG 10172 in acute stroke treatment.
Note: Data are number (percentage), median [interquartile range] or mean (standard deviation). These data have been partially validated and may undergo minor changes prior to data lock.
The baseline characteristics at the time of randomization are presented in Tables 2 and 3. Key features include: age 69.0 (10.1) years; male 1945 (62.8%); time from onset to recruitment 29.4 (11.9) h (Figure 3(a)); severity (National Institutes of Health Stroke Scale, NIHSS) 2.8 (3.6); mean blood pressure 143.5 (18.2)/79.5 (11.4) mmHg; IS 2143 (69.2%), TIA 953 (30.8%). In comparison with patients whose randomizing event was stroke, patients with TIA were more likely to have pre-morbid hypertension, hyperlipidemia, and a shorter time to randomization (Figure 3(b) and (c)), but were less likely to be current smokers.
Histogram of recruitment by time from onset to randomization for: (a) all patients (median 29.3 h), (b) stroke (median 32.0 h), and (c) TIA (median 24.2 h).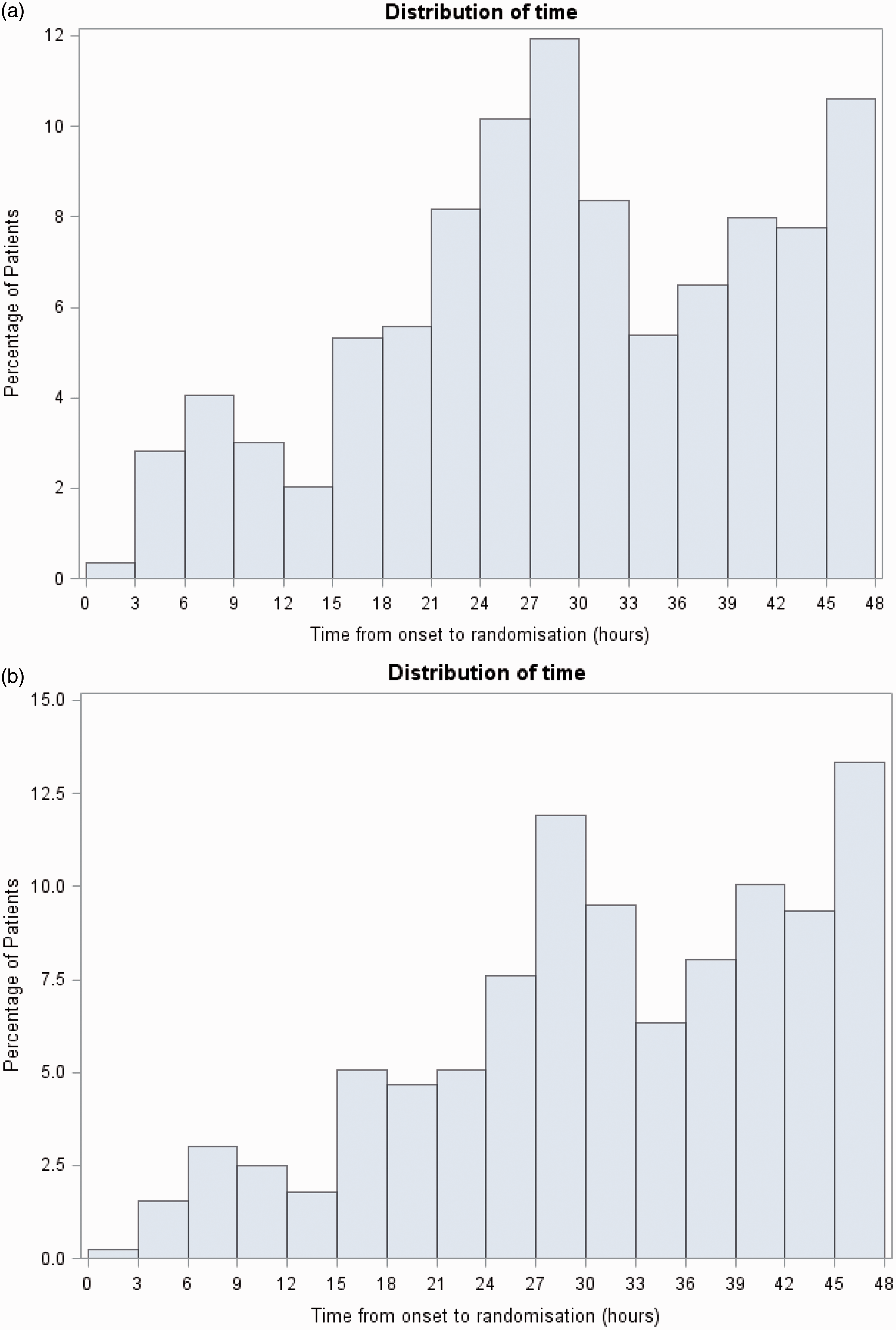
Univariate correlations between baseline characteristics for: age, sex, severity (NIHSS, ABCD2), qualifying event type (IS, TIA), systolic blood pressure (SBP), time to randomization, and year of randomization. Correlations by point biserial or Pearson’s tests; data are regression coefficient (p value)
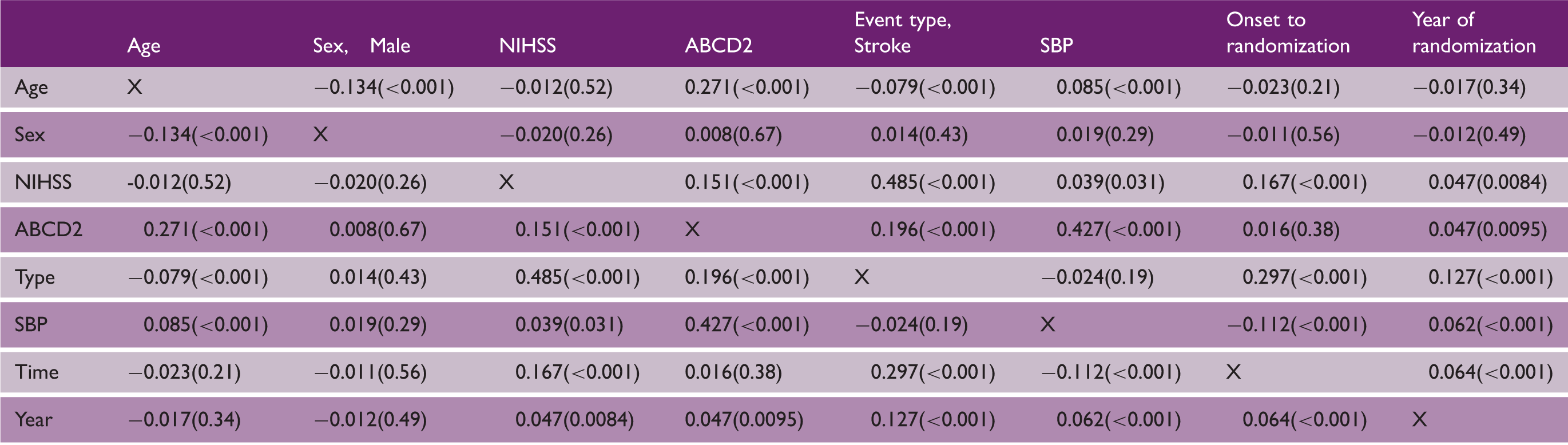
ABCD2: age, blood pressure, clinical features, duration, diabetes; IS: ischemic stroke; NIHSS: National Institutes of Health Stroke Scale; SBP: systolic blood pressure; TIA: transient ischemic attack.
Reporting of results
Following completion of data validation, the database will be locked and the trial unblinded and analyzed. It is intended to present the main results in late 2016/early 2017, with the main manuscript published in parallel. Multiple pre-specified secondary publications and analyses are described in the statistical analysis plan. 39
TARDIS data will be shared with prospective individual patient data meta-analyses:
Antiplatelet agents in acute stroke/TIA. Virtual international stroke trials archive (VISTA).
46
Ultimately, a subset of data will be made available on the internet akin to the International Stroke Trial, 47 and anonymized neuroimaging data will be published. 48
Discussion
TARDIS is the first trial to assess the safety and efficacy of intensive antiplatelet therapy based on three agents in comparison with guideline treatment; with 3096 patients, TARDIS is a large international trial. The characteristics of the participants justify several comments. First, although the trial was designed to run internationally, contracting issues, largely related to national sponsorship and availability of insurance (online Appendix C), led the trial to recruit predominantly a UK-population; with a large number of recruiting sites, it is therefore representative primarily of UK stroke services and their patients; however, participants also came from three other countries with predominantly Caucasian populations. Second, the proportion of stroke patients recruited as compared with TIAs was higher than intended; the proportion of stroke increased as the trial progressed, probably related to closure of other large stroke trials such as ENOS and SO2S.41,49 Third, in addition to more stroke patients being recruited over time, their severity, SBP, and time to randomization also increased, this reflecting differences in the types of participants recruited by sites who joined the trial later. Last, TIA patients were recruited much earlier than those with stroke as their index event, which may have implications for both efficacy and risk.
Although TARDIS is large, and definitive and significant results may help drive changes in clinical practice, it will still be necessary to merge individual patient data from TARDIS with other large trials of antiplatelet agents in acute IS and TIA to fully assess their effects in different patient subgroups.
TARDIS investigators
Trial Steering Committee (UK)
Independent members: Helen Rodgers (Newcastle, TSC Chair), Ahamad Hassan (Leeds), Christine Roffe (Stoke-on-Trent), Craig Smith (Salford), William D Toff (Leicester).
Grant holders: Philip M Bath (Nottingham, Chief Investigator), Rob Dineen (Nottingham, Neuroradiology Lead), Lelia Duley (Nottingham), Stan Heptinstall (Nottingham, Platelet Expert), Marilyn James (Nottingham, Health Economic Lead), Hugh S Markus (Cambridge), Stuart Pocock (London, Statistical Lead), Thompson G Robinson (Leicester), Nikola Sprigg (Nottingham, Deputy Chief Investigator), Graham S Venables (Sheffield).
Patient representative: Chibeka Kasonde (Nottingham).
Sponsor's representative: Angela Shone (University of Nottingham).
International Advisory Committee
Denmark – Hanne Christensen (Copenhagen), Georgia - Maia Beridze (Tblisi), New Zealand – Anna Ranta (Wellington).
Independent Data Monitoring Committee
Ian Ford (Glasgow, UK; Chair), Didier Leys (Lille, France), Cathie Sudlow (Edinburgh, UK), Matthew Walters (Glasgow, UK).
Independent Events (outcome, SAE) Adjudicator
Nikola Sprigg (Nottingham, UK), Marc Randall (Leeds, UK), Wayne Sunman (Nottingham, UK).
Neuroimaging Adjudicators
Rob Dineen (Nottingham, UK), Alessandro Adami (Verona, Italy), Lesley Cala (Perth, Australia), Ana Casado (Edinburgh, UK), Rebecca Gallagher (Derby, UK), David Swienton (Leicester, UK).
Platelet substudy
Stan Heptinstall, Sue Fox, Jane May (Nottingham, UK).
Trial management committee
Senior Trial Managers: Sally Utton (2012–2014), Di Havard (2015–2016).
Trial Manager: Hayley Foster (2013–2016).
UK Coordinators: Margaret Adrian (2009–2016), Tanya Payne (2009–2015), Alice Durham (2013–2014), Harriet Howard (2014–2015), Michael Stringer (2014–2016), Jamie Longmate (2015–2016).
International Coordinators: Sharon Ellender (2009–2013), Alice Durham (2013–2014), Sarah Grant (2013–2014), Joanne Keeling (2014–2016).
Outcome Coordinators: Sharon Ellender (2009–2013), Lynn Stokes (2010–2012), Patrick Cox (2011–2013), Judith Clarke (2012), Lyndsey Cobane (2012–2014), Kathy Whittamore (2013–2014), Joanne Keeling (2013–2016), Jennifer Smithson (2014–2016), James Kirby (2015–2016), Gemma Walker (2015–2016).
Statisticians: Michael Tracy (2009–2010), Cheryl Renton (2009–2012), Cyrille Correia (2011), Lydia Fox (2012–2013), Aimee Houlton (2012–2013), Katie Flaherty (2013–2016), Polly Scutt (2013–2016), Lisa Woodhouse (2013–2016).
Programming/database management: Richard Dooley, Liz Walker (Nottingham, UK).
Physicians: Tim England (2009), Chamilla Geeganage (2009–2012), Sandeep Ankolekar (2009–2011), Kailash Krishnan (2012–2015), Jason Appleton (2015–2016).
Data managers: Lida Kaur (2009–2010), Tanya Jones (2010–2013), Clare Randall (2011–2012), Dawn Hazle (2012–2016), Mark Sampson (2013–2016).
Finance: Wim Clarke (2009–2016).
Secretaries: Marilyn Stonham (2011), Susan Blencowe (2012–2013), Yvonne Smallwood (2012–2016), Lauren Dunn (2013–2014), Monika Kowalczyk (2014–2016), Patricia Robinson (2015–2016).
Administrators (temporary): Sarah Bull (2013), Georgina Phillips (2013), Harry Banks (2013–2014), Esther Akanya (2014), Sean McLoughlin (2015), Richard Barks (2015–2016).
Sites
The list of participating local investigators, sites and trial coordinating staff is given in online Appendix D.
Footnotes
Acknowledgements
We thank all the patients, and their carers, for participating in the trial. We also thank the members of the independent Data Monitoring Committee who supervised safety from the beginning to end of trial.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PMB declares advisory board/fees from Athersys, Covidien, Nestle, Phagenesis, and ReNeuron; he is an unpaid advisor to Platelet Solutions Ltd. HC declares advisory board/fees from Bayer, Boehringer-Ingelheim, BMS, Astra-Zeneca, Amgen, Covidien. SH is a founder and stockholder of Platelet Solutions Ltd. HSM declares fees from AstraZeneca. TGR declares advisory board/ fees from Bayer, Boehringer Ingelheim and Daiichi Sankyo. JPA, MB, RAD, LD, TJE, MJ, KK, SP, AR, KF, PS, GSV, LJW and NS made no declarations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The TARDIS start-up phase was funded by the British Heart Foundation (grant PG/08/083/25779, 1 April 2009 – 30 September 2012); the TARDIS main phase is funded by the National Institute of Health Research (NIHR) Heath Technology Assessment (HTA) Program (grant 10/104/24, 1 October 2012 – 30 September 2017, ![]() ). This paper presents independent research funded by the National Institute for Health Research (NIHR). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
). This paper presents independent research funded by the National Institute for Health Research (NIHR). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
PMB is Stroke Association Professor of Stroke Medicine; PMB and TGR are NIHR Senior Investigators.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
