Abstract
Background
The endovascular treatment of acute cerebral ischemia has been proven beneficial without major safety concerns. To date, the role of endovascular treatment in patients treated with oral anticoagulants, which may be associated with periprocedural intracranial bleeding, remains uncertain.
Aims
The objective of the current analysis is to evaluate the safety of endovascular treatment in patients treated with oral anticoagulants.
Methods
The ENDOSTROKE-Registry is a commercially independent, prospective observational study in 12 stroke centers in Germany and Austria collecting pre-specified variables about endovascular stroke therapy.
Results
Data from 815 patients (median age 70 (interquartile range (IQR) 20), 57% male) undergoing endovascular treatment with known anticoagulation status were analyzed. A total of 85 (median age 76 (IQR 8), 52% male) patients (10.4%) took vitamin-K-antagonists prior to endovascular treatment. Anticoagulation status as measured with international normalized ratio was above 2.0 in 31 patients. Intracranial hemorrhage occurred in 11.8% of patients taking vitamin-K-antagonists compared to no-vitamin-K-antagonists (12.2%, p = 0.909). After adjustment for confounding factors which were unbalanced at univariate level such as NIHSS and age, anticoagulation status was not found to significantly influence clinical outcome (modified Rankin Scale 3–6) and occurrence of intracranial hemorrhage in a multivariate logistic regression analysis.
Conclusion
Prior use of vitamin-K-antagonists was not associated with a higher rate of periprocedural intracranial hemorrhage after endovascular treatment or worse outcome. Endovascular treatment should be considered as an important treatment option in patients taking vitamin-K-antagonists.
Introduction
Sufficient anticoagulation is a contraindication for intravenous thrombolysis (IVT) as it carries an additional risk of major bleeding. Current guidelines suggest starting IVT only in patients taking vitamin-K-antagonists (VKA) with INR of 1.7 or less at time of presentation with acute ischemic stroke. 1 Therefore, many patients at high risk for an embolic stroke might not be suitable for IVT. Recently, endovascular treatment (EVT) applying mostly stent-retriever devices has been proven beneficial.2,3 However, the role of EVT in acute stroke patients taking VKA – still highly prescribed compared to Non-vitamin K antagonist oral anticoagulants (NOACs) 4 – remains uncertain, because this group of patients was not considered in most randomized trials and previous interventional studies. 2
Aims
The ENDOvascular STROKE treatment (ENDOSTROKE) registry was launched to accompany the spreading use of EVT in German and Austrian stroke centers. 5 The current analysis evaluates the efficacy and safety of EVT in patients under VKA therapy.
Methods
The ENDOSTROKE-Registry represents an investigator-initiated observational, international, multicenter study. The study focuses on adult patients who underwent thrombectomy for acute ischemic stroke. The study protocol was approved by the ethics committee of the University Medical Center Frankfurt and the local ethic committees. ENDOSTROKE is registered with ClinicalTrials.gov as NCT01399762.
Data analysis
Baseline characteristics of the study population were recorded and an outcome follow-up on the basis of the modified Rankin Scale (mRS) was performed at least 90 days after intervention. The main outcome variables were the occurrence of any intracranial hemorrhage (ICH) and symptomatic ICH (sICH: according to the European Cooperative Acute Stroke Study-II (ECASS-II); online-only data supplement).
Statistical analysis
Mann–Whitney U, χ2, or Fisher exact test were used for univariate analysis. Multivariate analysis used a binary logistic regression model (details are given in the online-only data supplement). Statistical analysis was performed using SPSS, version 22 (IBM, Armonk, NY) with p values of p < 0.05 considered significant.
Results
Out of 815 patients, 85 patients (10.4%) took VKA prior to EVT. Of 83 patients with valid international normalized ratio (INR) data prior to EVT, 31 patients (37%) were sufficiently anticoagulated (INR ≥ 2), 7 of which presented with an INR > 3.0 (8%). Patients taking VKA were significantly older (median age 76 (IQR 8) vs. no-VKA 69 (20)) and more frequently had a history of atrial fibrillation, arterial hypertension, and cardiovascular diseases. Additional patient characteristics showed no significant differences between VKA and no-VKA groups at baseline (online-only data supplement Table 1).
Periprocedural characteristics, safety and functional outcome
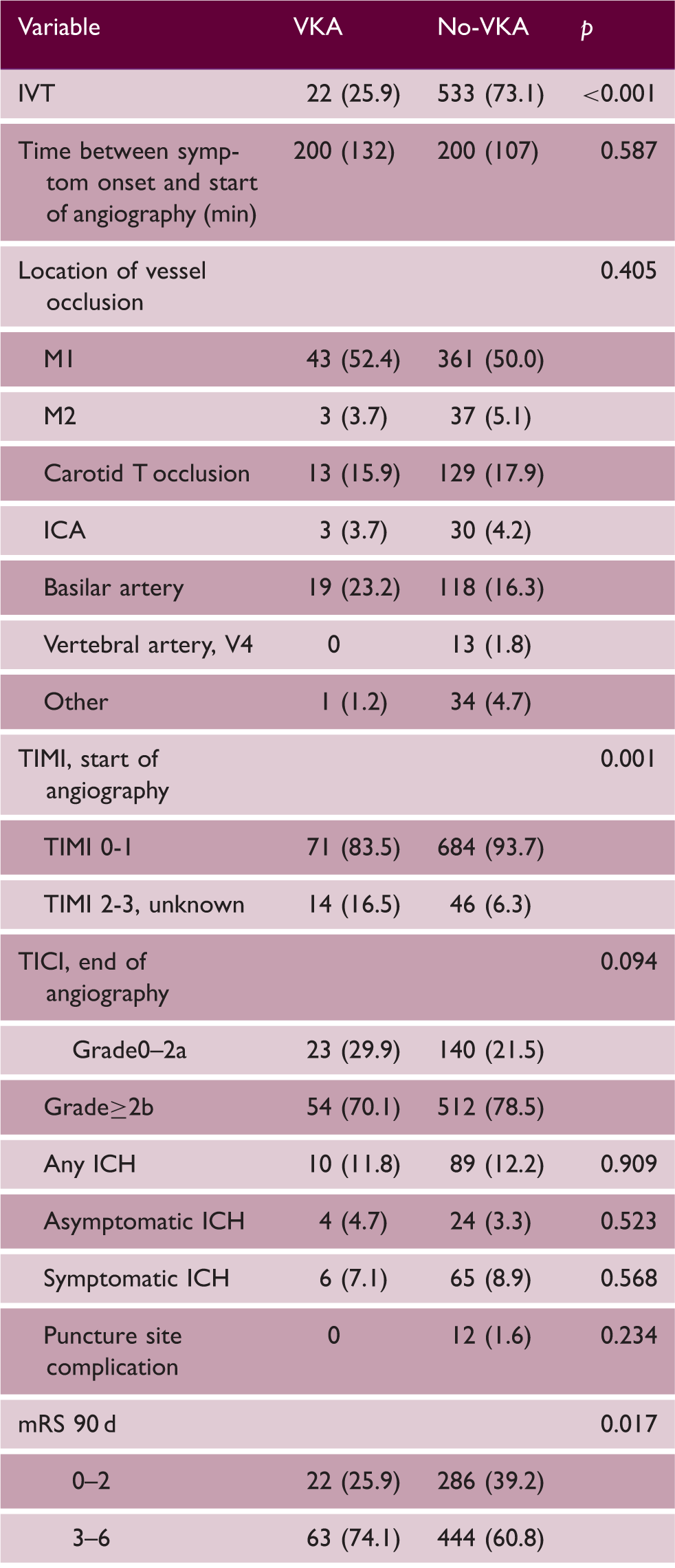
Note: Data presented as number (%) or median (IQR). p < 0.05 considered significant.
VKA: vitamin-K-antagonists; No-VKA: no vitamin-K-antagonists; IVT: intravenous thrombolysis; ICA: internal carotid artery; TIMI: thrombolysis in myocardial infarction score; TICI: Thrombolysis in cerebral infarction score; ICH: intracranial hemorrhage; mRS: modified Rankin Scale.
Patients taking VKA with INR < 2.0 received IVT more frequently than those taking VKA with an INR ≥ 2.0 (n = 20 (38.5%) vs. n = 2 (6.5%), p = 0.002). We detected no association between an increase in INR and an increased occurrence of ICH (online-only data supplement Table 2).
Factors determining functional outcome after 90 d and occurrence of any ICH
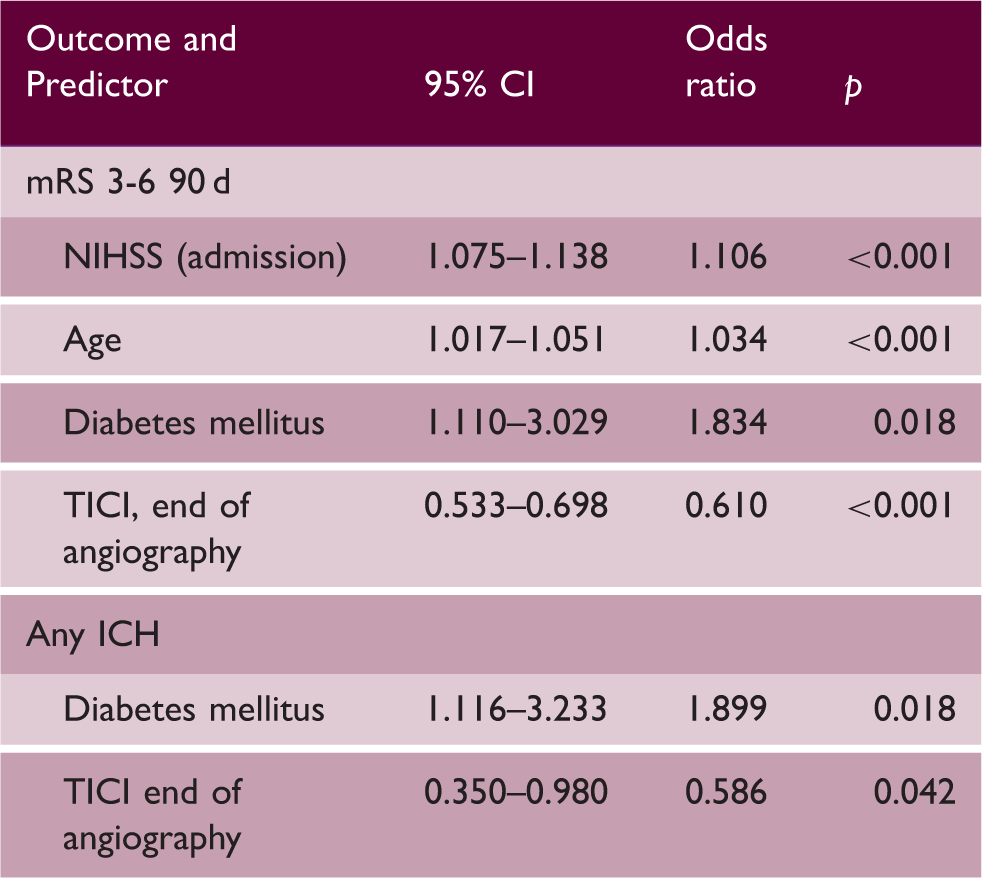
mRS: modified Rankin Scale; VKA: vitamin-K-antagonist; NIHSS: National Institute of Health Stroke Scale; TICI: thrombolysis in cerebral infarction score.
Note: p < 0.05 considered significant.
Discussion
Our analysis of the prospective ENDOSTROKE Registry detected no increased risk for periprocedural ICH after EVT in patients with prior VKA treatment. The observed rates of sICH (7.1%) and any ICH (11.8%) are comparable to the bleeding rates reported in a meta-analysis of recent thrombectomy trials. 2 Moreover, the proportion of sICH is consistent with bleeding rates reported by retrospective studies in VKA (6%) 6 and NOAC (4%) 7 patients, although these studies report higher rates of any ICH in VKA (26%) 6 and NOAC (46%) 7 patients. These results make a case for performing an EVT even if the patient is taking VKA and argue against a priori exclusion of VKA-treated patients. However, one should keep in mind that 63% of our VKA-patients had an INR < 2.0 at admission, which may not be representative of patients on VKA in other regions in Europe and North America 8 ; thus, the lack of difference in ICH in VKA patients should be interpreted with caution. Nonetheless, we detected no association between an increase in INR and an increased occurrence of ICH.
The prior use of VKA was not associated with an unfavorable outcome in multivariate analysis. In univariate comparison, an overall unfavorable outcome (mRS 3-6) was reported in 74.1% of all VKA-treated patients after EVT (no-VKA 60.8%). After adjustment for confounding factors, this difference was no longer apparent and can be mainly attributed to the non-equivalent distribution of patient age.2,5
This study faces the limitations of a non-randomized, observational registry. However, EVT in patients taking VKA appears to be safe and should therefore be considered as a treatment option in acute ischemic stroke. Further investigations are still needed to clarify the adjunctive use of IVT and the role of NOACs.
Footnotes
Acknowledgments
We thank Cheryl Ernest for proofreading the manuscript.
Authors’ contribution
Manuscript preparation and data analysis: TU, OCS, SG, KG. Study design: OCS, JB, CHN, TN. Data collection: all authors. Blinded angiographic rating DSL.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Speaker honoria from Boehringer Ingelheim, Bayer Healthcare, BMS (SJ, CW, KG); Daiichi Asubio, Penumbra (CW); Daichii Sankyo (CW, KG); Pfizer (SJ, KG). BT Scientific advisor for Acandis. The other authors report no conflicts.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
