Abstract
Background and Aim
A pre-specified country analysis of subjects from the Philippines in the CHInese Medicine NeuroAiD Efficacy on Stroke recovery (CHIMES) Study showed significantly improved functional and neurological outcomes on MLC601 at month (M) 3. We aimed to assess these effects on long-term functional recovery in the Filipino cohort.
Methods
The CHIMES-E (extension) Study evaluated subjects who completed three months of randomized placebo-controlled treatment in CHIMES up to two years. Blinding of treatment allocation was maintained and all subjects received standard stroke care and rehabilitation. Modified Rankin Score (mRS) and Barthel Index (BI) were assessed in-person at M3 and by telephone at M6, M12, M18, M24. Odds ratios (OR) with corresponding 95% confidence intervals (CI) for functional recovery using ordinal analysis of mRS and for achieving functional independence (mRS 0-1 or BI ≥ 95) at each time point were calculated, adjusting for age, sex, baseline National Institute of Health Stroke Scale (NIHSS), onset-to-treatment time (OTT) and pre-stroke mRS.
Results
The 378 subjects (MLC601 192, placebo 186) included in CHIMES-E from the Philippines (mean age 60.2 ± 11.1) had more women (p < 0.001), worse baseline NIHSS (p < 0.001) and longer onset to treatment time (p = 0.002) compared to other countries. Baseline characteristics were similar between treatment groups. The treatment effect of MLC601 seen at M3 peaked at M6 with OR for mRS shift of 1.53 (95% CI 1.05–2.22), mRS dichotomy 0–1 of 1.77 (95% CI 1.10–2.83), and BI ≥ 95 of 1.87 (95% CI 1.16–3.02). The beneficial effect persisted up to M24.
Conclusion
The beneficial effect of MLC601 seen at M3 in the Filipino cohort is durable up to two years after stroke.
Introduction
The CHInese Medicine Neuroaid Efficacy on Stroke recovery (CHIMES) study was an international, randomized, placebo-controlled, double-blind trial that compared MLC601 with placebo in 1099 subjects with acute ischemic stroke of intermediate severity in the preceding 72 h. 1 Almost half (n = 504) of the subjects were included from the Philippines. Hence, a pre-specified country analysis of subjects from the Philippines was performed which showed a statistically significant improvement in functional and neurological outcomes with MLC601 at month (M) 3. 2 The demonstration of a larger treatment effect in the Filipino cohort was attributed to inclusion of more patients with poorer prognosis as compared to other countries. Subsequently, an extension study of CHIMES (CHIMES-E) wherein subjects were followed up blinded to original treatment allocation for up to two years after a stroke, showed MLC601 to be associated with significantly increased odds of attaining functional independence at 6, 12 and 18 months. 3
In this study, we aimed to assess the effect of a three-month treatment course of MLC601 on long-term functional recovery in the Filipino cohort during the 21 months of additional follow-up.
Methods
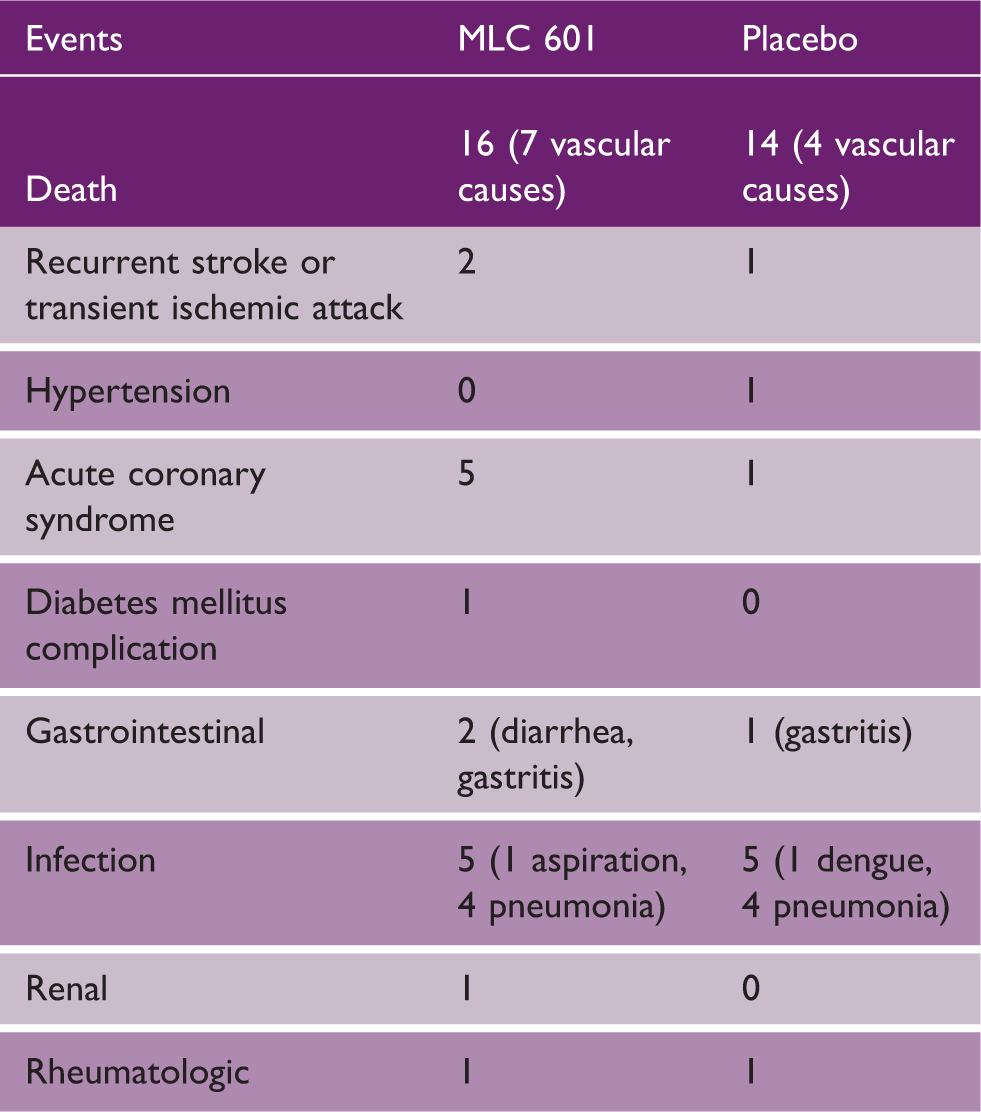
All subjects in CHIMES were eligible for CHIMES-E unless they withdrew consent. The subjects did not receive any further trial medication after the initial three months but had standard stroke care and rehabilitation as prescribed by the treating physician. Modified Rankin Score (mRS) and Barthel Index (BI) were assessed in-person at M3 (CHIMES) and by telephone at M6, M12, M18, M24 (CHIMES-E). 3 Treatment effect was estimated by calculating the odds ratios (OR) with corresponding 95% confidence intervals (CI) for functional recovery (mRS ordinal analysis) and for achieving functional independence (mRS 0–1 versus > 1 or BI ≥ 95 versus < 95 dichotomy analyses) at each time point adjusting for age, sex, baseline National Institute of Health Stroke Scale (NIHSS), stroke onset-to-treatment time (OTT) and pre-stroke mRS. For subjects whose mRS scores were not available at M3 and M24, the scores were imputed by the last observation carried forward (LOCF) method from M1 and M18, respectively. Data without LOCF were also analyzed and further sensitivity analyses were performed by assigning all missing data the worst outcome (mRS 6) and the best outcome (mRS 0) at each time point. The long-term safety of the initial treatment with MLC601 was assessed by the occurrence of any new medical condition and death.
Results
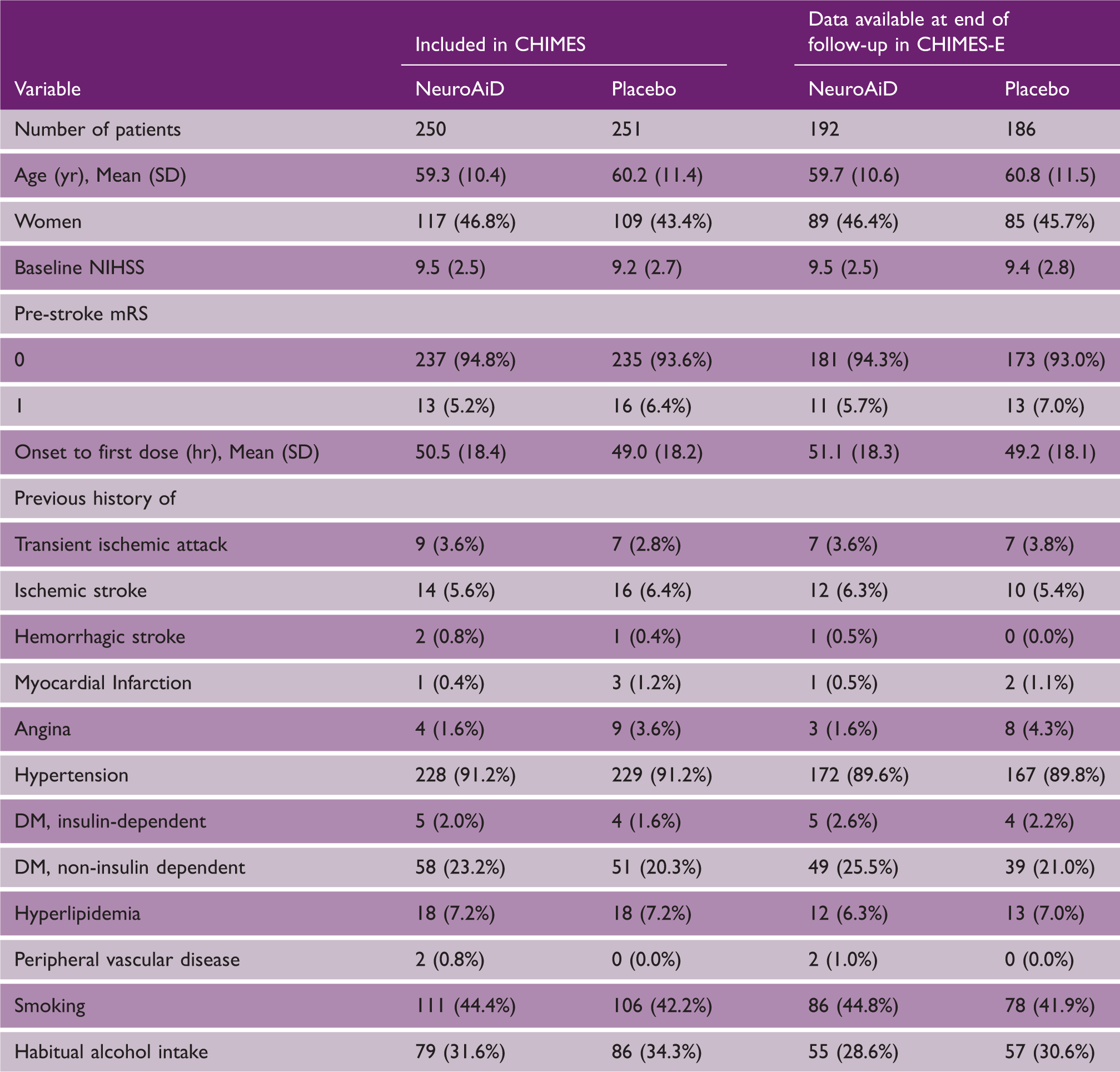
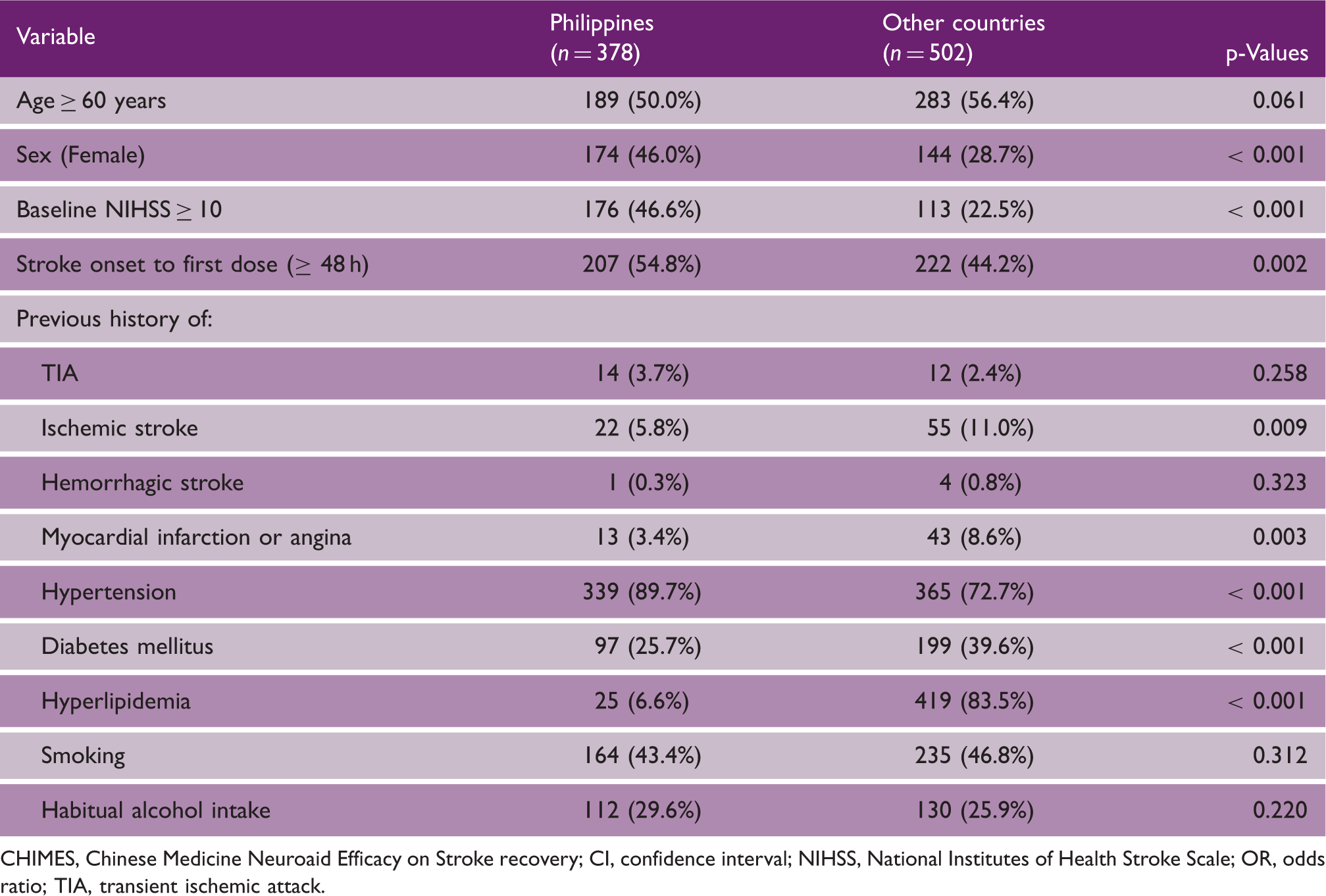
A total of 504 subjects were randomized from the Philippines in the CHIMES study among whom 378 (75%) had long-term follow-up information in CHIMES-E (Figure 1). There was no difference in baseline demographics, NIHSS, pre-stroke mRS, OTT, and risk factor profiles between Filipino subjects who did or did not have data available at final follow-up, indicating that a bias in follow-up was unlikely (Table 1). Moreover, baseline characteristics were similar between the MLC601- and placebo-treated groups. As compared to other countries, however, there were more women, (p < 0.001), worse baseline NIHSS scores (p < 0.001), and longer OTT (p = 0.002) from the Philippines compared to those included from other countries (Table 2). The risk factor profile of subjects from the Philippines was also different compared to those from other countries.
Flow of subjects randomized from the Philippines in the CHIMES and CHIMES-E Studies. mRS, modified Rankin Scale; M, month; LOCF, last observation carried forward. Baseline characteristics of subjects included from the Philippines in the CHIMES and CHIMES-E studies according to treatment allocation Comparison of baseline characteristics and risk factor profiles of patients included in the CHIMES-E Study from the Philippines and other countries CHIMES, Chinese Medicine Neuroaid Efficacy on Stroke recovery; CI, confidence interval; NIHSS, National Institutes of Health Stroke Scale; OR, odds ratio; TIA, transient ischemic attack.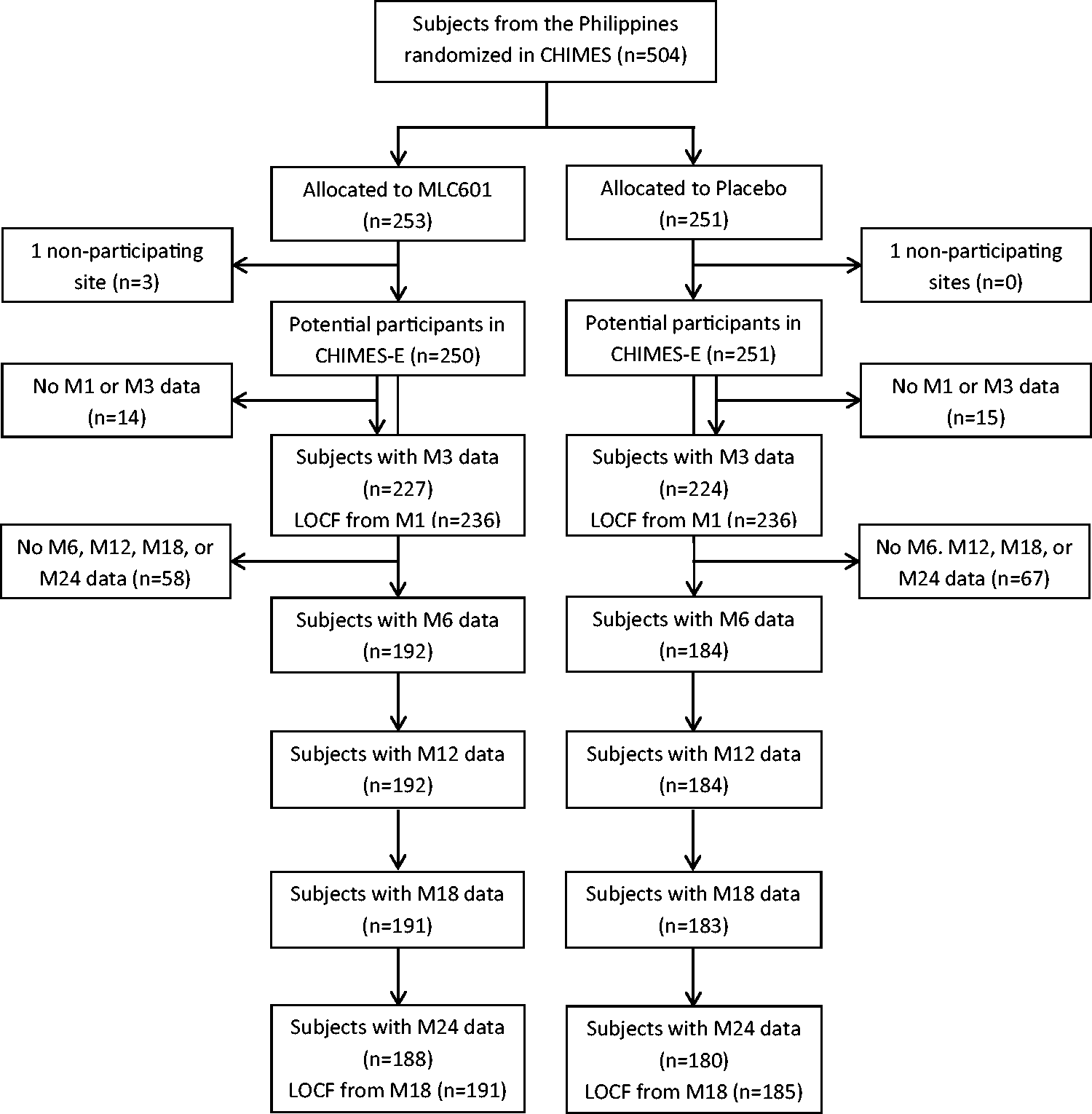
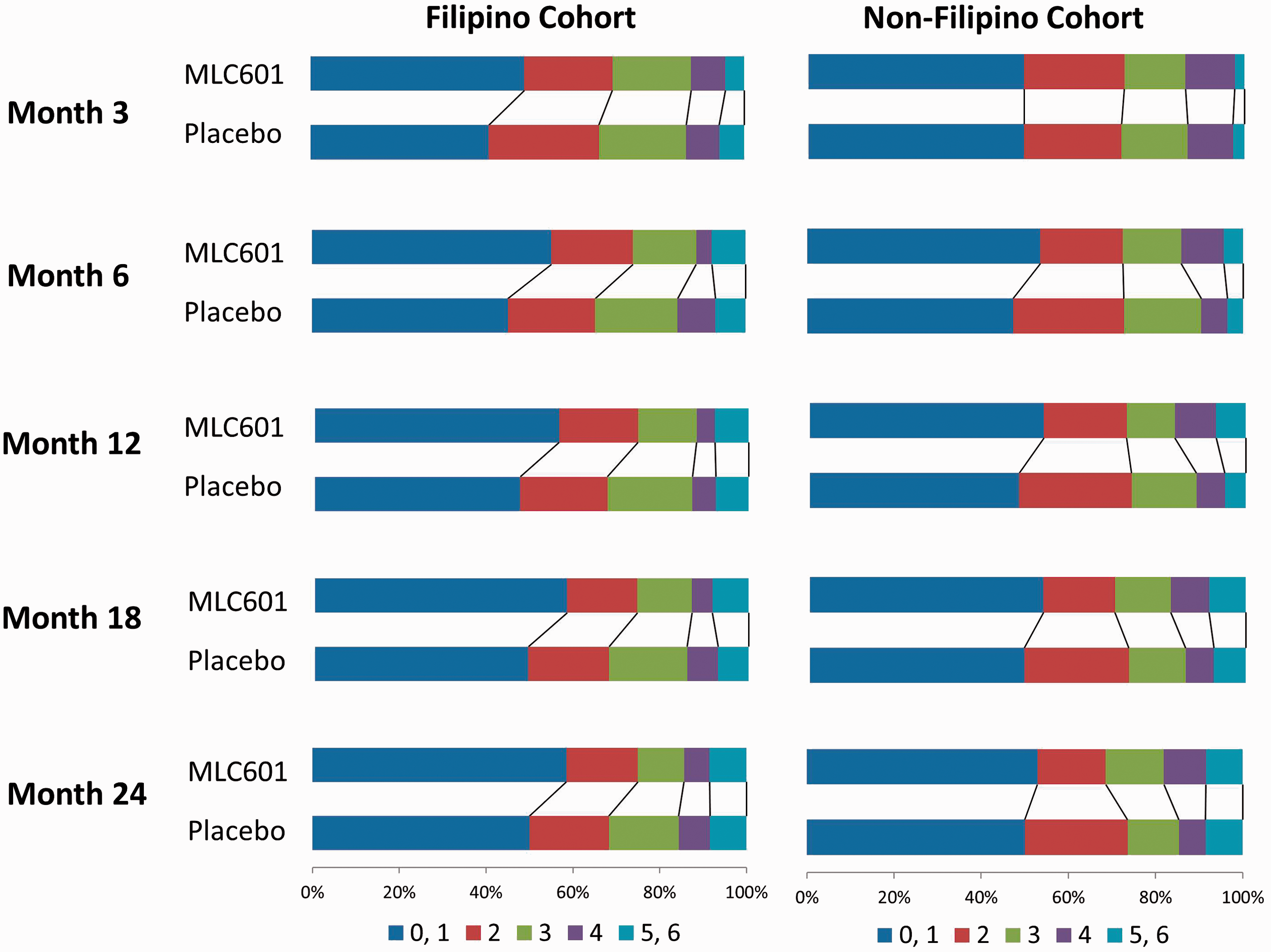
Twenty subjects died by M3, 26 by M6, 27 by M12, 28 by M16, and 30 by M24. LOCF mRS data were imputed from M1 to M3 in 21 subjects and from M18 to M24 in 8 subjects (Figure 1). The ORs for mRS and BI in favor of MLC601 were maximal at M6 with ORs of 1.53 (95% CI 1.05–2.22) for mRS ordinal analysis, 1.77 (95% CI 1.10–2.83) for mRS dichotomy 0–1, and 1.87 (95% CI 1.16–3.03) for BI ≥ 95 (Figure 2). The benefits persisted until M24. Results did not change when data were analyzed without LOCF. Sensitivity analyses assuming worst and best outcomes for missing data showed qualitatively similar trajectories for all time points with the expected variabilities in 95% CI (Figure 2). By comparison, although still in favor of MLC601, the effect sizes among non-Filipino subjects at different time points were not of the same magnitude as those of the Filipino cohort as illustrated in Figure 3.
Forest plot of odds ratios for mRS ordinal, mRS dichotomy 0–1 and BI dichotomy ≥ 95 analyses at months 3, 6, 12, 18, and 24 adjusted for age, sex, baseline National Institute of Health Stroke Scale, stroke onset to treatment time and pre-stroke mRS. Paired horizontal stacked bar graphs (“Grotta bars”) of modified Rankin scores at different time points among Filipino and non-Filipino study subjects.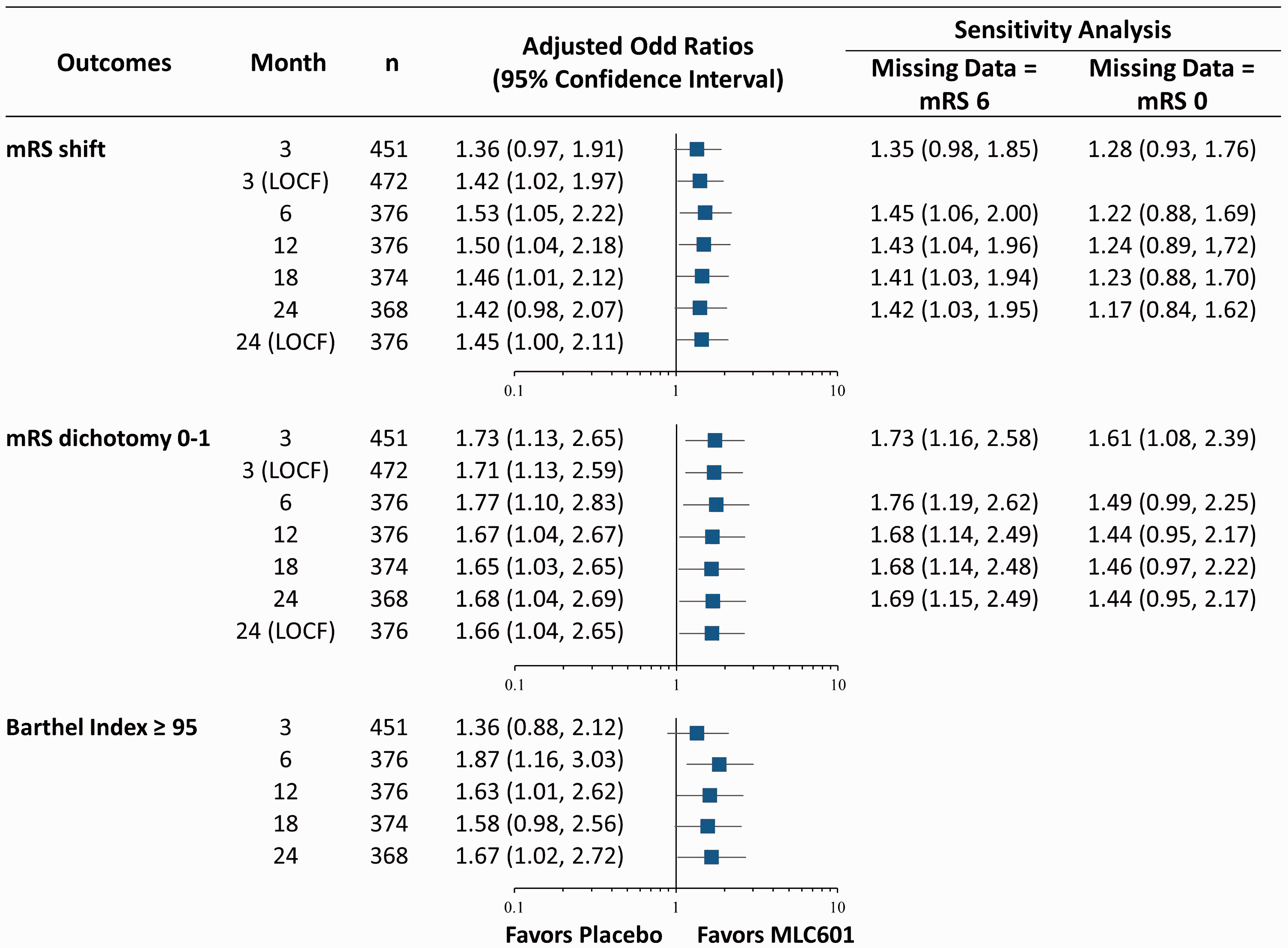
Cumulative medical events reported by month 24 among subjects from the Philippines
Discussion
Our study shows the durability of the beneficial effect in the Filipino cohort of MLC601 on post-stroke recovery for up to 2 years after stroke onset despite only three months of treatment. The results were consistent between the outcome assessments used, i.e. mRS and BI and at all time points up to 24 months after stroke. The relatively larger and more durable effects of MLC601 seen in the Filipino cohort as compared to that seen in the overall population in the CHIMES-E study and in subjects from non-Filipino sites are likely due to the difference in the characteristics of subjects recruited. We have previously demonstrated that subjects having more prognostic factors for poor outcome showed better treatment effects with MLC601 at three months.4,5 Patients in CHIMES had relatively mild stroke severity many of whom recovered well by three months even in the placebo group, 1 which likely affected the power of detecting the treatment effect.6–8 The study population from the Philippines, on the other hand, included more subjects with prognostic factors for poorer outcome, such as female sex, stroke severity, and treatment delay.
The durability of the benefits derived from treatment with MLC601 is clinically important. Spontaneous recovery to a certain extent in the initial three months after stroke is well described, especially among strokes of milder severity. 9 Thereafter, neurological and functional recovery occurs at a much slower rate. However, a recent study from the Swedish Stroke Register showed a high proportion of patients transitioning from independence at three months to dependence in activities of daily living at 12 months after a stroke. 10 This deterioration was more common in older patients and women. While demonstrating benefit at three months would already translate to improved quality of lives of patients and caregivers, a durable benefit of up to two years would translate to significantly higher quality-adjusted life years. MLC601 has been shown to have neuro-restorative properties, in addition to its neuroprotective properties, in various animal and cellular stroke models by enhancing the natural neuro-repair processes of the brain after an injury.11,12 The gradual but long-lasting beneficial effects of such treatment strategy can, therefore, be expected in strokes of any severity, but logically can clinically be demonstrated better in patients with worse prognosis among whom improvements are likely to be more apparent with treatment.
There are some limitations in this study. Variables such as quality of medical care, nutrition, and economic factors were not collected which could have allowed us to assess their potential as confounders. There were no data on the use of medications such as anti-thrombotics, statins, and anti-hypertensives that could have affected recurrent vascular events.
The main strength of the study, nonetheless, is the long duration of follow-up of subjects randomized in an international multi-centre clinical trial with double-blinding of treatment allocation being maintained till the end of the study period. The end-points used were those employed in most stroke outcome trials. Performing the functional assessments by telephone ensured subject retention and minimized the loss of data in our study. They have been shown to be as reliable as face-to-face evaluations.13,14
Conclusion
To conclude, the Filipino cohort in the CHIMES and CHIMES-E study had different prognostic and vascular risk factor profiles as compared to other countries. The larger and more durable beneficial effects with MLC601 observed in the Filipino population are most likely due to the presence of prognostic factors for poorer outcome. This study supports our hypothesis that treatment effects are seen earlier and are sustained in stroke patients who have the potential to benefit from treatment. A longer follow-up study design is preferred in assessing the efficacy of treatment in post-stroke recovery trials. Our data further support the rationale for selecting subjects with potentially poorer outcomes in stroke recovery trials to improve the ability of the study to detect meaningful treatment effects.
Footnotes
Acknowledgments
CHIMES and/or CHIMES-E Study Investigators: Philippines—Jose C Navarro, Herminigildo H Gan, Annabelle Lao, Alejandro Baroque II, Johnny Lokin, John Harold B Hiyadan, M Socorro Sarfati, Randolph John Fangonillo, Neil Ambasing, Carlos Chua, M Cristina San Jose, Joel Advincula, Eli John Berame, Maria Teresa Canete. Singapore—Narayanaswamy Venketasubramanian, Sherry HY Young, Marlie Jane Mamauag, San SanTay, Shrikant Pande, Umapathi Thirugnanam, Rajinder Singh, Hui Meng Chang, Deidre Anne De Silva, Bernard PL Chan, Vijay Sharma, Teoh Hock Luen. Thailand—Niphon Poungvarin, Sombat Muengtaweepongsa, Somchai Towanabut, Nijasri Suwanwela, Songkram Chotickanuchit, Siwaporn Chankrachang, Samart Nitinun. Sri Lanka—H Asita de Silva, Udaya Ranawake, Nirmala Wijekoon. Hong Kong—KS Lawrence Wong. Malaysia—Gaik Bee Eow.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JCN, NV, and CLHC received funding from the CHIMES Society for conferences and meetings for presentation of CHIMES data. The other authors received funding for the trial and accommodation and transportation support for meetings from the CHIMES Society. Moleac (Singapore) provided grants to the CHIMES Society of which the society had sole discretion on use. JCN, HHG, AYL, and ACB have minor shares in E*Chimes, the Philippine distributor of NeuroAiD.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The CHIMES-E study was supported by the CHIMES Society, a nonprofit organization, and grants were received by CLHC from the National Medical Research Council of Singapore (grant numbers NMRC/1288/2011 and NMRC/1096/2006).
